
Title: Scientific American Supplement, No. 508, September 26, 1885
Author: Various
Release date: October 3, 2005 [eBook #16792]
Most recently updated: December 12, 2020
Language: English
Other information and formats: www.gutenberg.org/ebooks/16792
Credits: Produced by Juliet Sutherland, Josephine Paolucci and the
Online Distributed Proofreading Team at www.pgdp.net
Among oil producers, there has been much discussion as to whether the sand rock in which petroleum occurs is of necessity fissured or is still in its original unbroken condition.
The earliest and most natural theory, which for years was indisputed, and is still given by some textbooks, was, that oil wells reached a cavity filled with petroleum.
Within the past few years, however, the opinion has been gaining ground that the oil is stored in the sandrock itself in the minute spaces between the small grains of sand, not entirely filled by cementing material, and that crevices holding and conducting oil are rare, all fissures as a rule being confined to the upper fresh-water bearing rocks of the well. Mr. Carll, in III. Pennsylvania Second Geological Survey, has discussed this subject very fully, and has made estimates of the quantity of oil that the sand rock can hold and deliver into a well; also, T. Sterry Hunt, in his Chemical and Geological Essays, has made deductions as to the petroleum contained in the Niagara limestone that outcrops about Chicago.
While the experiments and conclusions of these geologists go to prove that these rocks are capable of holding the oil, there are on record no facts as to the phenomena of its flow, other than by capillarity, through the rock. To obtain some data of the flow of liquids under pressure through certain oil-bearing stones, series of tests on small pieces were made. These tests were carried on during this spring, and many results quite unlooked for were obtained. When crude oil, kerosene, or water (river or distilled) was forced through the specimens, the pressure being constant, the rate of flow was variable. At first, the amount flowing through was large, then fell off rapidly, and when the flow had diminished to about one-quarter of its original rate, the decrease was very slight, but still continued as long as measurements were made, in some cases for three weeks.
When using crude oil, this result was not surprising, for, as the oil men say, crude oil "paraffines up" a rock, that is, clogs the minute pores by depositing solid paraffine (?); but this so-called paraffining took place, not only with crude oil, but with refined oil, and even with distilled water.
The only explanation as yet is, that liquids flowing under pressure through rock on which they exert little or no dissolving effect, instead of washing out fine particles, tend to dislodge any minute grains of the stone that may not be firmly held by cement, and these block up extremely fine and crooked pores in which the fluid is passing.
Several tests indicated that this blocking up was largely near the surface into which the fluid was passing. When this surface was ground off, even 1/50 of an inch, the flow increased immediately nearly to the original rate.
Reversing the flow also had the effect of increasing the rate, even above that of any time previous.
With the moderate pressures used—from 2" to 80" of mercury—the results show that the rate of flow, other things being equal, is directly proportional to the pressure.
The porosity of rock is not always a criterion of its permeability; a very fine grained marble, containing about 0.6 per cent. cell space, transmitted water and oil more freely than a shale that would hold 4 per cent. of its bulk of water.
If the above conclusions hold on a large scale as on the small, they may aid in explaining the diminished flow of oil wells. Not only will the flow lessen from reduced gas pressure, but the passages in the rock become less able to allow the oil to flow through.
The increase in flow following the explosion of large shots in a sand rock may be due not only to fissuring of the rock, but to temporary reversal of the pressure, the force of the explosive tending to drive the oil back for an instant.
The large shots now used (up to 200 quarts, or say 660 pounds of nitroglycerine) must exert some influence of this kind, especially when held down by 500± feet of liquid tamping. In the course of these tests, it was noticed that fresh water has a more energetic disintegrating action on the shales and clay than on salt water.
This may furnish a reason for the fact, noticed by the oil men, that fresh water has a much more injurious effect than salt in clogging a well. No oil-bearing sand rock is free from laminæ of shale, and when fresh water gets down into the sand, the water must, as the experiments show, rapidly break up the shale, setting free fine particles, which soon are driven along into the minute interstices of the sand rock, plastering it up and injuring the well.—Engineering and Mining Journal.
The grotto of Gargas is located in Mount Tibiran about three hundred yards above the level of the valley, and about two miles southeast of the village of Aventignan. Access to it is easy, since a road made by Mr. Borderes in 1884 allows carriages to reach its entrance.
This grotto is one of the most beautiful in the Pyrenees, and presents to the visitor a succession of vast halls with roofs that are curved like a dome, or are in the form of an ogive, or are as flat as a ceiling. It is easy to explore these halls, for the floor is covered with a thick stalagmitic stratum, and is not irregular as in the majority of large caves.
Upon entering through the iron gate at the mouth of the grotto, one finds himself in Bear Hall, wherein a strange calcareous concretion offers the form of the carnivorous animal after which the room is named. This chamber is about 80 feet in width by 98 in length. We first descend a slope formed of earth and debris mostly derived from the outside. This slope, in which are cut several steps, rests upon a hard, compact, and crystalline stalagmitic floor. Upon turning to the right, we come to the Hall of Columns, the most beautiful of all. Here the floor bristles with stalagmites, which in several places are connected with the stalactites that depend from the ceiling. This room is about 50 feet square. After this we reach the Hall of Crevices, 80 feet square, and this leads to the great Hall of Gargas, which is about 328 feet in length by 80, 98, and 105 in width. In certain places enormous fissures in the vault rise to a great height. Some of these, shaped like great inverted funnels, are more than 60 yards in length. The grotto terminates in the Creeping Hall. As its name indicates, this part of the cave can only be traversed by lying flat upon the belly. It gives access to the upper grotto through a narrow and difficult passage that it would be possible to widen, and which would then allow visitors to make their exit by traversing the beautiful upper grotto, whose natural entrance is situated 150 yards above the present one. This latter was blasted out about thirty years ago.
Upon following the direction of the great crevices, we reach a small chamber, wherein are found the Oubliettes of Gargas—a vertical well 65 feet feet in depth. The aperture that gives access to this strange well (rendered important through the paleontological remains collected in it) is no more than two feet in diameter. Such is the general configuration of the grotto.
In 1865 Dr. Garrigou and Mr. De Chastaignier visited the grotto, and were the first to make excavations therein. These latter allowed these scientists to ascertain that the great chamber contained the remains of a quaternary fauna, and, near the declivity, a deposit of the reindeer age.
As soon as it was possible to obtain a permit from the Municipal Council of Aventignan to do so, I began the work of excavation, and the persistence with which I continued my explorations led me to discover one of the most important deposits that we possess in the chain of the Pyrenees. My first excavations in Bear Hall were made in 1873, and were particularly fruitful in an opening 29 feet long by 10 wide that terminates the hall, to the left. I have remarked that these sorts of retreats in grottoes are generally rich in bones. Currents of water rushing through the entrance to the grotto carry along the bones—entire, broken, or gnawed—that lie upon the ground. These remains are transported to the depths of the cave, and are often stopped along the walls, and lie buried in the chambers in argillaceous mud. Rounded flint stones are constantly associated with the bones, and the latter are always in great disorder. The species that I met with were as follows: the great cave bear, the little bear, the hyena, the great cat, the rhinoceros, the ox, the horse, and the stag.
The stalagmitic floor is 1½, 2, and 2¼ inches thick. The bones were either scattered or accumulated at certain points. They were generally broken, and often worn and rounded. They appeared to have been rolled with violence by the waters. The clay that contained them was from 3 to 6 feet in thickness, and rested upon a stratum of water-worn pebbles whose dimensions varied from the size of the fist to a grain of sand. A thick layer of very hard, crystalline stalagmite covers the Hall of Columns, and it was very difficult to excavate without destroying this part of the grotto.
I found that there anciently existed several apertures that are now sealed up, either by calcareous concretions or by earthy rubbish from the mountain. One of these was situated in the vicinity of the present mouth, and permitted of the access to Bear Hall of a host of carnivora that found therein a vast and convenient place of shelter.
These excavations revealed to me at this entrance, at the bottom of the declivity, a thick stratum of remains brought thither by primitive man. This deposit, which was formed of black earth mixed with charcoal and numerous remains of bones, calcined and broken longitudinally for the most part, contained rudely worked flint stones. I collected a few implements, one surface of which offered a clean fracture, while the other represented the cutting edge. According to Mr. De Mortillet, such instruments were not intended to have a handle. They were capable of serving as paring knives and saws, but they were especially designed for scraping bones and skins. The deposit was from 26 to 32 feet square and from 2 inches to 5 feet deep, and rested upon a bed of broken stones above the stalagmite. The animals found in it were the modern bear (rare), the aurochs, the ox, the horse, and the stag—the last four in abundance.
At the extremity of the grotto there is a well with vertical sides which is no less than 65 feet in depth. It is called the Gargas Oubilettes. Its mouth is from 15 to 24 inches in diameter, and scarcely gives passage to a man (Fig. 1). Mr. Borderes, in the hope of discovering a new grotto, was the first to descend into this well, which he did by means of a rope ladder, and collected a few bones that were a revelation to me. Despite the great difficulty and danger of excavating at this point, I proceeded, and found at the first blow of the pick that there was here a deposit of the highest importance, since all the bones that I met with were intact. The first thing collected was an entire skull of the great cave bear, with its maxillaries in place. From this moment I began a series of excavations that lasted two years.
The descent is effected through a narrow vertical passage 6½ feet in length. The cavity afterward imperceptibly widens, and, at a depth of 12 yards, reaches 6½ feet in diameter, and at 15 yards 10 feet. Finally, in the widest part (at a depth of 62 feet) it measures about 16 feet (Fig. 1).
A glance at the section of the well, which I have drawn as accurately as possible (not an easy thing to do when one is standing upon a rope ladder), will give an idea of the form of this strange pocket formed in the limestone of the mountain through the most complex dislocations and erosions. Two lateral pockets attracted my attention because of the enormous quantity of clay and bones that obstructed them. The first, to the left, was about 15 feet from the orifice. When we had entirely emptied it, we found that it communicated with the bottom of the well by a narrow passage. An entire skeleton of the great cave bear had stopped up this narrow passage, and of this, by the aid of a small ladder, we gathered the greater part of the skeleton, the state of preservation of which was remarkable.
The second pocket, which was almost completely filled with clay, and situated a little lower than the other, likewise communicated with a third cavity that reached the bottom of the well. The clay of these different pockets contained so large a quantity of bones that we could hardly use our picks, and the excavation had to be performed with very short hooks, and often by hand. In this way I was enabled to remove the bones without accident. The lower pocket was dug out first, and with extreme care, the bones being hoisted out by means of a basket attached to a rope. Three or four candles sufficed to give us light. The air was heavy and very warm, and, after staying in it for two hours, it was necessary to come to the surface to breathe. After extracting the bones from the lower pocket, and when no more clay remained, we successively dug out the upper ones and threw the earth to the bottom of the well.
On the 20th of December, 1884, my excavating was finished. To-day the Oubliettes of Gargas are obstructed with the clay that it was impossible to carry elsewhere. The animals that I thus collected in the well were the following: The great bear (in abundance), the little bear (a variety of the preceding), the hyena, and the wolf. The pockets contained nearly entire skeletons of these species. How had the animals been able to penetrate this well? It is difficult to admit that it was through the aperture that I have mentioned. I endeavored to ascertain whether there was not another communication with the Gargas grotto, and had the satisfaction of finding a fissure that ended in the cave, and that probably was wider at the epoch at which the place served as a lair for the bear and hyena.
Very old individuals and other adults, and very young animals, were living in the grotto, and, being surprised, without power to save themselves, by a sudden inundation, reached the bottom of the well that we have described. The entire remains of these animals were carried along by the water and deposited in the pockets in the rock. Once buried in the argillaceous mud, the bones no longer underwent the action of the running water, and their preservation was thence secured.—F. Regnault, in La Nature.
A correspondent of the New York Sun, writing from Virginia City, Nevada, describes the progress of the work there on the Combination shaft of the Comstock lode, the deepest vertical shaft in America, and the second deepest in the world. It is being sunk by the Chollar Potosi, Hale & Norcross, and Savage mining companies; hence its name of the Combination shaft. This shaft has now reached a perpendicular depth of a little over 3,100 feet. There is only one deeper vertical shaft in the world—the Adalbent shaft of the silver-lead mines of Przibram, Bohemia, which at last accounts had reached a depth of 3,280 feet. The attainment of that depth was made the occasion of a festival, which continued three days, and was still further honored by the striking off of commemorative medals of the value of a florin each. There is no record of the beginning of work on this mine at Przibram, although its written history goes back to 1527.
Twenty years ago very few mining shafts in the world had reached a depth of 2,000 feet. The very deepest at that time was in a metalliferous mine in Hanover, which had been carried down 2,900 feet; but this was probably not a single perpendicular shaft. Two vertical shafts near Gilly, in Belgium, are sunk to the depth of 2,847 feet. At this point they are connected by a drift, from which an exploring shaft or winze is sunk to a further depth of 666 feet, and from that again was put down a bore hole 49 feet in depth, making the total depth reached 3,562 feet. As the bore hole did not reach the seam of coal sought for, they returned and resumed operations at the 2,847 level. In Europe it is thought worthy of particular note that there are vertical shafts of the following depths:
| Feet. | |
| Eimkert's shaft of the Luganer Coal Mining Company, Saxony | 2,653 |
| Sampson shaft of the Oberhartz silver mine, near St. Andreasberg, Hanover. | 2,437 |
| The hoisting shaft of the Rosebridge Colliery, near Wigan, Lancashire, England. | 2,458 |
| Shaft of the coal mines of St. Luke, near St. Chaumont, France. | 2,253 |
| Amelia shaft, Shemnitz, Hungary. | 1,782 |
| The No. 1 Camphausen shaft, near Fishbach, in the department of the Saarbruck Collieries, Prussia. | 1,650 |
Now, taking the mines of the Comstock for a distance of over a mile—from the Utah on the north to the Alto on the south—there is hardly a mine that is not down over 2,500 feet, and most of the shafts are deeper than those mentioned above; while the Union Consolidated shaft has a vertical depth of 2,900 feet, and the Yellow Jacket a depth of 3,030 feet. In his closing argument before the Congressional Committee on Mines and Mining in 1872, Adolph Sutro of the Sutro tunnel said: "The deepest hole dug by man since the world has existed is only 2,700 feet deep, and it remains for the youngest nation on earth to contribute more to science and geology by giving opportunities of studying the formation of mineral veins at a greater depth than has ever been accomplished by any other nation in the world." Mr. Sutro was of the opinion that the completion of his tunnel would enable our leading mining companies to reach a vertical depth of 5,000 feet.
This great depth has never yet been attained except in a bore hole or artesian well. The deepest points to which the crust of the earth has ever been penetrated have been by means of such borings in quest of salt, coal, or water. A bore hole for salt at Probst Jesar, near Lubtheen, for the Government of Mecklenberg-Schwerin, is down 3,315 feet, the size of which bore is twelve inches at the top and three inches at the bottom. A bore hole was put down for the Prussian Government to the depth of 4,183 feet. But in these bore holes the United States leads the world, as there is one near St. Louis, Mo., that is 5,500 feet in depth. Here on the Comstock, in the Union Consolidated mine, a depth of 3,300 feet has been attained, but not by means of a single vertical shaft. The vertical depth of the shaft is 2,900 feet; the remainder of the depth has been attained by means of winzes sunk from drifts. Several long drifts were run at this great depth without difficulty as regards ventilation or heat.
The combination shaft is situated much further east (in which direction the lode dips) than any other on the Comstock. It is 3,000 feet east of the point where the great vein crops out on the side of Mount Davidson; 2,200 feet east of the old Chollar-Potosi shaft, 1,800 ft. east of the old Hale & Norcross (or Fair) shaft, and 2,000 ft. east of the Savage shaft. Thus, it will be seen it is far out to the front in the country toward which the vein is going. The shaft is sunk in a very hard rock (andesite), every foot of which requires to be blasted. The opening is about thirty feet in length by ten feet in width. In timbering up this is divided into four different compartments, some for the hoisting and some for the pumping machinery, thus presenting the appearance at the top of four small shafts set in a row. Over the shaft stand several large buildings, all filled with ponderous machinery.
The Sutro drain tunnel (nearly four miles in length) connects with the shaft at a depth of 1,600 ft., up to which point all the water encountered below is pumped. The shaft was sunk to the depth of 2,200 ft. before more water was encountered than could be hoisted out in the "skips" with the dirt. At the 2,200 level two Cornish pumps, each with columns fifteen inches in diameter, were put in. At the 2,400 level the same pumps were used. On this level a drift was run that connected with the old Hale & Norcross and Savage shafts, producing a good circulation of air both in the shaft and in the mines mentioned. At this point, on account of the inflow from the mines consequent upon connecting with them by means of the drift, they had more water than the Cornish pumps could handle, and introduced the hydraulic pumps, which pumps are run by the pressure of water from the surface through a pipe running down from the top of the shaft, whereas the Cornish pumps are run by huge steam engines.
By means of the hydraulic pumps they were enabled to sink the shaft to the 2,600 level, and extended the Cornish pumps to that point, where another set of hydraulic pumps was put in. They then sunk the shaft to the 2,800 level, when they ran another drift westward, and tapped the vein. The prospects at this depth in the Hale & Norcross and Chollar mines were so encouraging that the management decided to sink the shaft to the depth of 3,000 ft. On reaching the 3,000 level, they ran a third drift through to the vein. The distance from the shaft to the east wall of the vein was found to be only 250 ft. At the depth of 3,000 ft. they put in one of the pair of hydraulic pumps that is to be set up there. The second pump is now arriving from San Francisco, and as soon as the several parts are on the ground, it will be at once put in place alongside its fellow on the 3,000 level. This additional pump will increase the capacity from 600,000 to 700,000 gallons in twenty-four hours, or about forty-five miners' inches.
Owing to the excellent showing of ore obtained on the 3,000 level by the Hale & Norcross Company, and to the continuation of the ore below that level (as shown by a winze sunk in the vein), the management determined to sink the shaft to the vertical depth of 3,200 ft. It is now 3,120 ft. deep, and it is safe to say that it will reach the depth of 3,200 ft. early in September, when it will lack but eighty feet of being as deep as the shaft at Przibram was at the time of the great festival. Although the shaft is of great size—about thirty feet by ten feet before the timbers are put in—the workmen lower it at the rate of about three feet a day, in rock as hard as flint.
The hydraulic pump now working at the 3,000 foot level of the shaft is the deepest in the world. In Europe the deepest is in a mine in the Hartz Mountains, Germany, which is working at the depth of 2,700 feet. It is, however, a small pump not half the size of the one in the Combination shaft. Although these pumps were first used in Europe, those in operation here are far superior in size, and in every other respect, to those of the Old World, several valuable improvements having been made in them by the machinists of the Pacific coast.
The capacity of the two Cornish pumps, which lift the water from the 2,900 foot level to the Sutro drain tunnel (at the 1,600 level), is about 1,000,000 gallons in twenty-four hours, and the capacity of the present hydraulic pumps is 3,500,000 gallons in the same time. They are now daily pumping, with both hydraulic and Cornish pumps, about 4,000,000 gallons, but could pump at least 500,000 gallons more in twenty-four hours than they are now doing. The daily capacity with the hydraulic pump now coming, and which will be set up as mate to that now in operation at the 3,000 foot level, will be 5,200,000 gallons.
The water which feeds the pressure pipe of the three sets of hydraulic pumps is brought from near Lake Tahoe, in the Sierra Nevada Mountains. The distance is about thirty miles, and the greater part of the way the water flows through iron pipes, which at one point cross a depression 1,720 feet in depth. The pressure pipe takes this water from a tank situated on the eastern slope of Mount Davidson, 3,500 feet west of the shaft. At the tank this pipe is twelve inches in diameter, but is only eight inches where it enters the top of the shaft. The tank whence the water is taken is 426 feet higher than the top of the shaft, therefore the vertical pressure upon the hydraulic pump at the 3,000 foot level is 3,426 feet. The pressure pipe is of ordinary galvanized iron where it receives the water at the tank, but gradually grows thicker and stronger, and at the 3,000 level it is constructed of cast iron, and is 2½ inches in thickness. The pressure at this point is 1,500 pounds to the square inch.
In the early days of hydraulic mining in California the miners thought that with a vertical pressure of 300 feet they could almost tear the world to pieces, and not a man among them could have been made to believe that any pipe could be constructed that would withstand a vertical pressure of 1,000 feet; but we now see that a thickness of two and a half inches of cast iron will sustain a vertical pressure of over 3,400 feet.
There is only one pressure pipe for all the hydraulic pumps. This extends from the tank on the side of the mountain to the 3,000 foot level. It is tapped at the points where are situated the several sets of hydraulic pumps. The water from the pressure pipe enters one part of the pump, where it moves a piston-back and forth, just as the piston of a steam engine is moved by steam. This water engine moves a pump which not only raises to the surface the water which has been used as driving power, but also a vast quantity of water from the shaft, all of which is forced up to the Sutro drain tunnel through what is called a return pipe. Each set of hydraulic pumps has its return pipe; therefore there are three return pipes—one from the 2,400, one from the 2,600, and another from the 3,000 level.
Some idea may be formed of the great size of these hydraulic engines when it is known that the stations excavated for them at the several levels where they are placed are 85 feet long, 28 feet wide, and 12 feet high. All this space is so filled with machinery that only sufficient room is left to allow of the workmen moving about it. One of these stations would, on the surface, form a hall large enough for a ball room, and to those who are unacquainted with the skill of our miners it must seem wonderful that such great openings can be made and securely supported far down in the bowels of the earth; yet it is very effectually done. These great subterranean halls are supported by timbers 14×16 inches square set along the walls three feet apart, from center to center, and the caps or joists passing overhead are timbers of the same size. The timber used is mountain spruce. Not one of these huge stations has thus far cost one dollar for repairs. The station at the 2,400 level has been in use five years, that at the 2,600 three years, and the one at the 3,000 level eight months. Room for ventilation is left behind the timbers, and all are still sound. Timbers of the same kind are used in the shaft, and all are sound. The shaft has cost nothing for repairs. Being in hard andesite rock from top to bottom, the ground does not swell and crowd upon the timbers.
If it shall be thought advisable to go to a greater depth than 3,200 feet, a station of large size will be made on the east side of the present shaft, and in this station will be sunk a shaft of smaller size. The reason why the work will be continued in this way is that in a single hoist of 3,200 feet the weight of a steel wire cable of that length is very great—so great that the loaded cage it brings up is a mere trifle in comparison. In this secondary shaft the hoisting apparatus and pumps will be run by means of compressed air. As it is very expensive to make compressed air by steam power, the pressure pipe will be tapped at the level of the Sutro tunnel, and a stream of water taken out that will be used in running a turbine wheel of sufficient capacity to drive three air compressors. As there will be a vertical pressure upon the turbine at this depth of over 2,000 feet, a large stream of water will not be required. The water used in driving the wheel will flow out through the Sutro tunnel, and give no trouble in the shaft.
By means of this great shaft and its powerful hydraulic and Cornish pumps the crust of the earth will probably yet be penetrated to far greater depth than in any other place in the world. It has been only a little over ten years since the work of sinking it was begun, whereas in the mines of the Old World they have been delving since "time whereof the memory of man runneth not to the contrary." The work on the Combination shaft has been by no means continuous. There have been long stoppages aside from those required at such times as they were engaged in running long drifts to the westward to tap the vein, and at times for many months, when the several companies interested in the shaft were engaged in prospecting the various levels it had opened up.
Yucatan is one of the most interesting States of Mexico, owing to the splendid ancient palaces and temples of once grand cities, now hidden in the forests. That country also presents great attractions for geologists and botanists, as well as naturalists, who there find rare and beautiful birds, insects, and reptiles.
There are no rivers on the surface of the land, but in many parts it is entirely undermined by extensive caverns, in which are basins of water fed by subterranean currents. The caverns are delightfully cool even at midday, and the fantastic forms of some of the stalactites and stalagmites are a never-ending source of interest. There are long winding passages and roomy chambers following one after another for great distances, with here and there some chink in the stony vault above, through which a sunbeam penetrates, enabling us to see to the right and left openings leading to untrodden places in the bowels of the earth. As few of these caves have been explored, the wildest accounts are given by the natives concerning the dark recesses where only wild beasts seek shelter. Before venturing far in, it is advisable to secure one end of a ball of twine at the entrance, and keep the ball in hand; nor is it safe to go without lanterns or torches, lest we step into some yawning chasm or deep water. The leader of one party suddenly saw a very dark spot just before him; he jumped over, instead of stepping on it, and told the others to halt. Examination proved the dark patch to be a pit that seemed bottomless.
Awe-inspiring as are the interiors of some of these caves, they are frequently most beautiful. The natural pillars are often grand in dimensions and sparkling with various hues, while stalactites and stalagmites sometimes resemble familiar objects with astonishing perfection. It is, however, not advisable to place implicit confidence in accounts of the natives, for the reality, no matter how beautiful, can hardly be equal to what the vivid imagination of the Indian has pictured. Anything bearing the least resemblance to a woman is called "a most beautiful Virgin Mary." Fantastic flutings become "an organ," and a level rock "an altar." Only once we were not disappointed, when, having been told to look for a pulpit, we found one that appeared as if man must have fashioned it, supported on a slender pyramidal base, the upper part very symmetrical, and ornamented with a perfect imitation of bunches of grapes and other fruit.
As I have already said, in these caves are sheets of water, some very large, others only a few feet in circumference, fed by subterranean currents. When the water is clear and sweet, it is peopled by a kind of bagre, a blind fish called by the natives tzau, also a species of Silurus. But there are likewise medicinal and thermal waters, by bathing in which many people claim to have been cured of most painful and obstinate diseases.
Strange stories are told of some of these waters. Of one it is said that those who approach it without holding their breath fall dead. People who live near the place swear it is so, and say the water appears to boil on such occasions. From the thermal waters, in some cases 100 feet below the soil, and without means of access except by buckets let down through an opening in the rock, warm vapors issue at early morn, but when the sun is high the water is cool and pleasant to drink.
The name senote is given to all these deposits of water, also to some immense natural circular wells from 50 to 300 feet in diameter. The walls are more or less perpendicular, generally covered with tropical vegetation. The current in some is swift, but no inlets or outlets are visible. The water is deliciously pure and sweet, much better than that of wells opened by man in the same country. These enormous deposits generally have a rugged path, sometimes very steep, leading to the water's edge, but daring natives throw themselves from the brink, afterward ascending by stout roots that hang like ropes down the walls, the trees above sucking through these roots the life-sustaining fluid more than a hundred feet below.
In the west part of Yucatan is a village called Bolonchen (nine wells), because in the public square there are nine circular openings cut through a stratum of rock. They are mouths of one immense cistern, if natural or made by hand the natives do not know, but in times of drought it is empty, which shows that it is not supplied by any subterranean spring. Then the people depend entirely on water found in a cave a mile and a half from the village; it is perhaps the most remarkable cavern in the whole country. The entrance is magnificently wild and picturesque. It is necessary to carry torches, for the way is dark and dangerous. After advancing sixty or seventy feet we descend a strong but rough ladder twenty feet long, placed against a very precipitous rock. Not the faintest glimmer of daylight reaches that spot; but after a while we stand on the brink of a perpendicular precipice, the bottom of which is strongly illuminated through a hole in the surface rock more than 200 feet above. Standing on the verge of this awful pit in the dim light, the rocks and crags seem to take on most weird shapes. We go down into the great hole by a ladder eighty feet high and twelve wide, and, reaching the bottom, are as yet but at the mouth of the cave, which, by the bye, is called Xtacunbi Xunan (the hidden lady), because, say the Indians, a lady was stolen from her mother and hidden there by her lover. Now, to our right, we find a narrow passage, and soon another ladder; the darkness is intense and the descent continuous, though irregular, like a series of hills and dales, ladders being placed against the steepest places.
After an exhausting journey we reach a vast chamber, from which crooked passages lead in various directions to wells, seven in all, each named according to the peculiar kind of water. One, always warm, is called Chocohá (hot water); another, O[c]ihá (milky water), and Akabhá (dark water). About 400 paces away from the chamber, passing through a very narrow, close passage, there is a basin of red water that ebbs and flows like the sea, receding with the south wind, increasing with the northwest.
To reach the most distant well, we go down yet one more ladder, the seventh. On one side of it there is a perpendicular wall, on the other a yawning gulf, so when one of the steps, merely round sticks tied with withes, gave way beneath our feet, we tightly grasped the stick above. Having reached the bottom of the ladder, we crawl on our hands and feet through a broken, winding passage about 800 feet long, then see before us a basin of crystalline water, and how thirsty we are! This basin is 1,400 feet from the mouth of the cave, and about 450 feet below the earth's surface. Several hundred people during five months in every year depend entirely on that source for all the water they use. With their frail pitchers and flaring torches they wend their way, gasping for breath, through the intricate passages, and reaching the water, are so profusely perspiring that they must wait before quenching their thirst. The way back is even harder, and they are tired and loaded; yet these people are such lovers of cleanliness that on their arrival at their poor huts, before tasting food, they will use some of the water that has cost them so much, to bathe their smoke-begrimed skin. As several women once fainted in the cave, men generally fetch the water now.
Yucatan is, and has been for ages past, quite free from earthquakes, while all surrounding countries are from time to time convulsed. This immunity may be due to the vast caverns and numerous great wells existing throughout the land. Pliny the Elder was of opinion that if numerous deep wells were made in the earth to serve as outlets for the gases that disturb its upper strata, the strength of the earthquakes would be diminished, and if we may judge by Yucatan, Pliny was right in his conjectures. After him, other scientists who have carefully studied the subject have expressed the same opinion with regard to the efficacy of large wells.
ALICE D. LE PLONGEON.
Brooklyn, July 15, 1885.
Cholera failed to strike a single one of the 4,000 women employed in the national tobacco factory at Valencia, Spain, though the disease raged violently in that city, and the Medical World recalls that tobacco workers were also noticed to enjoy exemption from attack during an epidemic at Amsterdam.
A patch of eggs and the minute caterpillars or larvæ nearly emerged from them are seen on the leaf. These tiny eggs are at first quite white or pale yellow, and form an object for the microscope of remarkable beauty, which is worthy of the examination of all who take an interest in the garden and its insect life. An egg magnified is drawn at the bottom left-hand corner of the woodcut. When the eggs are near the hatching point they darken in color, and a magnifying glass reveals through the delicate transparent shell a sight which fills the observer with amazement; the embryo caterpillar is seen in gradual course of formation, and if patience and warmth have permitted it, the observer will witness slight movements within the life-case, and presently the shell will break and a black head with moving jaws will be thrust out; the little caterpillar unfolds and slowly crawls away from the egg-shell, and inserts its jaws into the green leaf. It is curious to witness how judiciously the little creatures avoid crowding together, but strike out in different directions, and thus they make sure of a plentiful supply of food, and distribute the effects of their depredations. These caterpillars eat continually, and hence rapidly increase in size, until they present the appearance shown in our drawing at the bottom of the illustration, which is a full grown caterpillar.
It will be observed that this insect is composed of thirteen segments from head to tail, which is a distinctive characteristic of all insects both in the larval and perfect states; but in the case of this and most other caterpillars these segments are sharply defined and readily recognized. It will also be noticed that the three segments or "joints" nearest the head bear a pair of legs each; these are the real feet, or claspers, as they are sometimes termed, which develop into the feet of the future butterfly. There are four pairs of false feet or suckers, which adhere to the ground by suction, and which disappear in the butterfly. On the last or tail end is a fifth pair of suckers also, which can attach themselves to a surface with considerable force, as any one can attest who has noticed the wrigglings of one of these caterpillars when feeling for new feeding ground.
The caterpillar now ceases to eat, and quietly betakes itself to a secluded corner, where in peace it spins a web around its body, and wrapt therein remains quiescent, awaiting its change into the butterfly. Although so dormant outwardly, activity reigns inside; processes are going on within that chrysalis-case which are the amazement and the puzzle of all naturalists. In course of time the worm is changed into the beautiful winged butterfly, which breaks its case and emerges soft and wet; but it quickly dries and spreads its wings to commence its life in the air and sunshine. The chrysalis is represented in the figure on the left. The butterfly, it will be recognized, is one of the common insects so familiar to all, with strongly veined white wings, bearing three black spots, two on the upper and one on the lower wing, and dark coloring on the corner of the upper wings. The antennæ, as with all butterflies, are clubbed at the extremity—unlike moths', which are tapering—and the large black staring eyes are the optical apparatus, containing, we are told, thousands of lenses, each a perfect, simple eye.
The wings derive their chief coloring from the covering of scales, which lie on like slates on a roof, and are attached in a similar manner. A small portion of the wing magnified is represented at the bottom right hand corner, and detached scales more highly magnified next to it, exhibiting somewhat the form of battledoors.
Another well known insect is illustrated in the figure in the upper portion—the peacock butterfly (Vanessa Io). The curious spiked and spotted caterpillar feeds upon the common nettle. This beautiful butterfly—common in most districts—is brilliantly colored and figured on the upper side of the wings, but only of a mottled brown on the under surface, somewhat resembling a dried and brown leaf, so that it is no easy matter to detect the conspicuous, brightly-decked insect when it alights from flight upon foliage, and brings its wings together over its back after the manner of butterflies. At the left-hand corner is seen the head of the insect, magnified, showing the long spiral tongue.
This is a curious structure, and one that will repay the trouble of microscopic examination. In the figure the profile is seen, the large compound eye at the side and the long curved tongue, so elephantine-looking in form, though of minute size, is seen unrolled as it is when about to be inserted into flowers to pump up the honey-juice. This little piece of insect apparatus is a mass of muscles and sensitive nerves comprising a machine of greater complexity and of no less precision in its action than the modern printing machine. When not in use, the tongue rolls into a spiral and disappears under the head. A butterfly's tongue may readily be unrolled by carefully inserting a pin within the first spiral and gently drawing it out.—The Gardeners' Chronicle.
This cypress, apart from its elegant growth, is interesting as being the only species of Cupressus indigenous to India. It is a native of the Himalayas in the Bhotan district, and it also occurs on the borders of Chinese Tartary. It forms, therefore, a connecting link, as it were, between the true cypresses of the extreme east and those that are natives of Europe. It is singular to note that this genus of conifers extends throughout the entire breadth of the northern hemisphere, Cupressus funebris representing the extreme east in China, and C. macrocarpa the extreme west on the Californian seacoast. The northerly and southerly limits, it is interesting to mark, are, on the contrary, singularly restricted, the most southerly being found in Mexico; the most northerly (C. nutkaensis) in Nootka Sound, and the subject of these remarks (C. torulosa) in Bhotan. The whole of the regions intervening between these extreme lateral points have their cypresses. The European species are C. lusitanica (the cedar of Goa), which inhabits Spain and Portugal; C. sempervirens (the Roman cypress), which is centered chiefly in the southeasterly parts of Europe, extending into Asia Minor. Farther eastward C. torulosa is met with, and the chain is extended eastward by C. funebris, also known as C. pendula. The headquarters of the cypresses are undoubtedly in the extreme west, for here may be found some four or five distinct species, including the well-known C. Lawsoniana, probably the most popular of all coniferæ in gardens, C. Goveniana, C. Macnabiana, C. macrocarpa, and C. nutkaensis (spelt C. nutkanus by the Californian botanists). The eastern representative of the cypresses in the United States of North America is C. thyoides, popularly known as the white cedar. In Mexico three or four species occur, so that the genus in round numbers only contains about a dozen species. The Californian botanist Mr. Sereno Watson takes away Lawson's cypress from Cupressus and puts it in the genus Chamæcyparis, the chief points of distinction being the flattened two-ranked branchlets and the small globose cones maturing the first year.
All the cypresses are undoubtedly valuable from a garden point of view, but the various species vary in degree as regards their utility as ornamental subjects. I should rank them in the following order in point of merit: C. Lawsoniana, C. nutkaensis, C. macrocarpa, C. sempervirens, C. thyoides, C. Macnabiana, and C. Goveniana; then would follow C. torulosa, C. funebris, C. Knightiana, and other Mexican species. These are placed last, not because they are less elegant than the others, but on account of their tenderness, all being liable to succumb to our damp and cold winters. The species which concerns us at present, C. torulosa, is an old introduction, seeds of it having been sent to this country by Wallich so long back as 1824, and previous to this date it was found by Royle on the Himalayas, growing at elevations of some 11,500 feet above sea level. Coming from such a height, one would suppose it to be hardier than it really is, but its tenderness may probably be accounted for by the wood not getting thoroughly ripened during our summers. It is a very handsome tree, said to reach from 20 feet to 125 feet in height in its native habitat. It has a perfectly straight stem; the growth is pyramidal or rather conical, and the old wood is of a warm purplish-brown. The foliage is a glaucous gray-green, and the branches have a twisted and tufted appearance.
There are several varieties of it which are, or have been, in cultivation. Of these one of the best is corneyana, which Gordon ranked as a distinct species. It was supposed to be Chinese, and was introduced to cultivation by Messrs. Knight & Perry, the predecessors of Messrs. Veitch at the Chelsea Nurseries. It differs from C. torulosa proper, its habit being of low stature, and has slender pendulous branches; hence, it has been known in gardens by the names of C. gracilis, C. cernua, and C. pendula. Other varieties of C. torulosa are those named in gardens and nurseries—viridis, a kind devoid of the glaucous foliage of the original; majestica, a robust variety; and nana, a very dwarf and compact-growing sort. There is also a so-called variegated form, but it is not worthy of mention. The synonyms of C. torulosa itself are C. cashmeriana, C. nepalensis, and C. pendula. Having regard to the tenderness of this Bhotan cypress, it should only be planted in the warmest localities, and in dry sheltered positions; upland districts, too, provided they are sheltered, are undoubtedly suitable for it, inasmuch as growth is retarded in spring, and, therefore, the young shoots escape injury from late spring frosts.—W.G., in The Garden.
The variety of the pitcher plant (Sarracenia variolaris) found in North America is carnivorous, being a feeder on various animal substances.
Mrs. Mary Treat, an American naturalist, made, a few years ago, several experiments upon the plants of this species to be found in Florida; and to the labors of this lady the writer has been indebted, in some measure, in the preparation of this paper.
The Sarracenia derives its name of "pitcher plant" from the fact of its possessing the following curious characteristics: The median nerve is prolonged beyond the leaves in the manner of a tendril, and terminates in a species of cup or urn. This cup is ordinarily three or four inches in depth, and one to one and a half inches in width. The orifice of the cup is covered with a lid, which opens and shuts at certain periods. At sunrise the cup is found filled with sweet, limpid water, at which time the lid is down. In the course of the day the lid opens, when nearly half the water is evaporated; but during the night this loss is made up, and the next morning the cup is again quite full, and the lid is shut.
About the middle of March the plants put forth their leaves, which are from six to twelve inches long, hollow, and shaped something like a trumpet, while the aperture of the apex is formed almost precisely in the same manner as those of the plants previously described. A broad wing extends along one side of the leaf, from the base to the opening at the top; this wing is bound or edged with a purple cord, which extends likewise around the cup. This cord secretes a sweet fluid, and not only flying insects, but those also that crawl upon the ground, are attracted by it to the plants. Ants, especially, are very fond of this fluid, so that a line of aphides, extending from the base to the summit of a leaf, may frequently be observed slowly advancing toward the orifice of the cup, down which they disappear, never to return. Flying insects of every kind are equally drawn to the plant; and directly they taste the fluid, they act very curiously. After feeding upon the secretions for two or three minutes they become quite stupid, unsteady on their feet, and while trying to pass their legs over their wings to clear them, they fall down.
It is of no use to liberate any of the smaller insects; every fly, removed from the leaf upon which it had been feeding, returned immediately it was at liberty to do so, and walked down the fatal cup as though drawn to it by a species of irresistible fascination.
It is not alone that flies and other small insects are overpowered by the fluid which exudes from the cord in question. Even large insects succumb to it, although of course not so quickly. Mrs. Treat says: "A large cockroach was feeding on the secretion of a fresh leaf, which had caught but little or no prey. After feeding a short time the insect went down the tube so tight that I could not dislodge it, even when turning the leaf upside down and knocking it quite hard. It was late in the evening when I observed it enter; the next morning I cut the tube open; the cockroach was still alive, but it was covered with a secretion produced from the inner surface of the tube, and its legs fell off as I extricated it. From all appearance the terrible Sarracenia was eating its victim alive. And yet, perhaps, I should not say 'terrible,' for the plant seems to supply its victims with a Lethe-like draught before devouring them."
If only a few insects alight upon a leaf, no unpleasant smell is perceptible during or after the process of digestion; but if a large number of them be caught, which is commonly the case, a most offensive odor emanates from the cup, although the putrid matter does not appear to injure in any manner the inner surface of the tube, food, even in this condition, being readily absorbed, and going to nourish the plant. In fact, it would seem that the Sarracenia, like some animals, can feed upon carrion and thrive upon it.
In instances in which experiments have been made with fresh, raw beef or mutton, the meat has been covered in a few hours with the secretions of the leaves, and the blood extracted from it. There is, however, one difference between the digesting powers of the leaves when exercised upon insects or upon meat. Even if the bodies of insects have become putrid, the plant, as has already been stated, has no difficulty in assimilating them; but as regards meat, it is only when it is perfectly sweet that the secretions of the leaves will act upon it.
The pitcher plant undoubtedly derives its principal nourishment from the insects it eats. It, too—unlike most other carnivorous plants, which, when the quantity of food with which they have to deal is in excess of their powers of digestion, succumb to the effort and die—appears to find it easy to devour any number of insects, small or large, the operation being with it simply a question of time. Flies, beetles, or even cockroaches, at the expiration of three or four days at most, disappear, nothing being left of them save their wings and other hard, parts of their bodies.
The Sarracenia is, indeed, not only the most voracious of all known species of carnivorous plants, but the least fastidious as to the nature of the food upon which it feeds.—W.C.M., Nature.
Mr. Worsley-Benison has been discussing this question in a very interesting way, and he says in conclusion that "physiologically the most distinctive feature of plant-life is the power to manufacture protein from less complex bodies; that of animal-life, the absence of such power." He finds that in form, in the presence of starch, of chlorophyl, in power of locomotion, in the presence of circulatory organs, of the body called nitrogen, in the functions of respiration and sensation, there are no diagnostic characters. He finds, however, "fairly constant and well-marked distinctions" in the presence of a cellulose coat in the plant-cell, in digestion followed by absorption, and in the power to manufacture protein.
The morphological feature of plants is this cellulose coat; of animals, its absence; the physiological peculiarity of plants, this manufacturing power; of animals, the want of it. But after all the discussion he says: "To the question, Is this an animal or a plant? we must often reply, We do not know.—The Microscope.
Next to the rose, no flower is more beautiful or more useful than the camellia. It may readily be so managed that its natural season of blooming shall be from October to March, thus coming in at a time when roses can hardly be had without forcing. In every quality, with the single exception of scent, the camellia may be pronounced the equal of the rose. It can be used in all combinations or for all purposes for which roses can be employed. In form and color it is probably more perfect, and fully as brilliant. It is equally or more durable, either on the plant or as a cut flower. It is a little dearer to buy, and perhaps slightly more difficult to cultivate; but like most plants the camellia has crucial periods in its life, when it needs special treatment. That given, it may be grown with the utmost ease; that withheld, its culture becomes precarious, or a failure. The camellia is so hardy that it will live in the open air in many parts of Great Britain, and herein lies a danger to many cultivators. Because it is quite or almost hardy, they keep it almost cool. This is all very well if the cool treatment be not carried to extremes, and persisted in all the year round. Camellias in a dormant state will live and thrive in any temperature above the freezing point, and will take little or no hurt if subjected to from 3°-4° below it, or a temperature of 27° Fahr.
They will also bloom freely in a temperature of 40°, though 45° suits them better. Hence, during the late summer and early autumn it is hardly possible to keep camellias too cool either out of doors or in. They are also particularly sensitive to heat just before the flower-buds begin to swell in late autumn or winter; a sudden or sensible rise of temperature at that stage sends the flower-buds off in showers. This is what too often happens, in fact, to the camellias of amateurs. No sooner do the buds begin to show then a natural impatience seizes the possessor's of well-budded camellias to have the flowers opened. More warmth, a closer atmosphere, is brought to bear upon them, and down fall the buds in showers on stage or floor—the chief cause of this slip between the buds and the open flowers being a rise of temperature. A close or arid atmosphere often leads to the same results. Camellias can hardly have too free a circulation of air or too low a temperature. Another frequent cause of buds dropping arises from either too little or too much water at the roots. Either a paucity or excess of water at the roots should lead to identical results. Most amateurs overwater their camellias during their flowering stages. Seeing so many buds expanding, they naturally rush to the conclusion that a good deal of water must be used to fill them to bursting point. But the opening of camellia buds is less a manufacture than a mere development, and the strain on the plant and drain on the roots is far less during this stage than many suppose. Of course the opposite extreme of over-dry roots must be provided against, else this would also cause the plants to cast off their buds.
But our object now is less to point out how buds are to be developed into fully expanded flowers than to show how they were to be formed in plenty, and the plants preserved in robust health year after year. One of the simplest and surest modes of reaching this desirable end is to adopt a system of semi-tropical treatment for two months or so after flowering. The moment or even before the late blooms fade, the plants should be pruned if necessary. Few plants bear the knife better than camellias, though it is folly to cut them unless they are too tall or too large for their quarters or have grown out of form. As a rule healthy camellias produce sufficient or even a redundancy of shoots without cutting back; but should they need pruning, after flowering is the best time to perform the operation.
During the breaking of the tender leaves and the growth of the young shoots in their first stages, the plant should be shaded from direct sunshine, unless, indeed, they are a long way from the glass, when the diffusion and dispersion of the rays of light tone down or break their scorching force; few young leaves and shoots are more tender and easily burned than camellia, and scorching not only disfigures the plants, but also hinders the formation of fine growths and the development of flower-buds.
The atmosphere during the early season of growth may almost touch saturation. It must not fail to be genial, and this geniality of the air must be kept up by the surface-sprinkling of paths, floors, stages, walls, and the plants themselves at least twice a day.
With the pots or border well drained it is hardly possible to overwater the roots of camellias during their period of wood-making. The temperature may range from 50° to 65° during most of the period. As the flower-buds form, and become more conspicuous, the tropical treatment may become less and less tropical, until the camellias are subjected to the common treatment of greenhouse or conservatory plants in summer. Even at this early stage it is wise to attend to the thinning of the buds. Many varieties of camellias—notably that most useful of all varieties, the double white—will often set and swell five or ten times more buds than it ought to be allowed to carry. Nothing is gained, but a good deal is lost, by allowing so many embryo flower-buds to be formed or partially developed. It is in fact far wiser to take off the majority of the excess at the earliest possible point, so as to concentrate the strength of the plant into those that remain.
As it is, however, often a point of great moment to have a succession of camellia flowers for as long a period as possible on the same plants, buds of all sizes should be selected to remain. Fortunately, it is found in practice that the plants, unless overweighted with blooms, do not cast off the smaller or later buds in their efforts to open their earlier and larger ones. With the setting, thinning, and partial swelling of the flower-buds the semi-tropical treatment of camellias must close; continued longer, the result would be their blooming out of season, or more probably their not blooming at all.
The best place for camellias from the time of setting their flower-buds to their blooming season is a vexed question, which can hardly be said to have been settled as yet. They may either be left in a cool greenhouse, or placed in a shaded, sheltered position in the open air. Some of the finest camellias ever seen have been placed in the open air from June to October. These in some cases have been stood behind south, and in others behind west walls. Those facing the east in their summer quarters were, on the whole, the finest, many of them being truly magnificent plants, not a few of them having been imported direct from Florence at a time when camellias were far less grown in England than now.
In all cases where camellias are placed in the open air in summer, care will be taken to place the pots on worm proof bases, and to shield the tops from direct sunshine from 10 to 4 o'clock. If these two points are attended to, and also shelter from high winds, it matters little where they stand. In all cases it is well to place camellias under glass shelter early in October, less for fear of cold than of saturating rains causing a sodden state of the soil in the pots.
While adverting, however, to the safety and usefulness of placing camellias in the open air in summer, it must not be inferred that this is essential to the successful culture; it is, in fact, far otherwise, as the majority of the finest camellias in the country are planted out in conservatories with immovable roofs. Many such houses are, however, treated to special semi-tropical treatment as has been described, and are kept as cool and open as possible after the flower-buds are fairly set, so that the cultural and climatic conditions approximate as closely as possible to those here indicated.
Soil and seasons of potting may be described as vexed questions in camellia culture. As to the first, some affect pure loam, others peat only, yet more a half and half of both, with a liberal proportion of gritty sand, or a little smashed charcoal or bruised bones as porous or feeding agents, or both. Most growers prefer the mixture, and as good camellias are grown in each of its constituents, it follows without saying that they may also be well grown in various proportions of both.
Under rather than over potting suits the plants best, and the best time is doubtless just before they are about to start into fresh growth, though many good cultivators elect to shift their plants in the late summer or autumn, that is, soon after the growth is finishing, and the flower-buds fairly and fully set for the next season. From all which it is obvious that the camellia is not only among the most useful and showy, but likewise among the most accommodating of plants.
Under good cultivation it is also one of the cleanest, though when scab gets on it, it is difficult to get rid of it. Mealy-bugs also occasionally make a hurried visit to camellias when making their growth, as well as aphides. But the leaves once formed and advanced to semi-maturity are too hard and leathery for such insects, while they will bear scale being rubbed off them with impunity. But really well-grown camellias, as a rule, are wholly free from insect pests, and their clean, dark, glossy leaves are only of secondary beauty to their brilliant, exquisitely formed, and many sized flowers.—D.T., The Gardeners' Chronicle.
Some few years since we had occasion to figure some very remarkable Himalayan species of this genus, in which the end of the spadix was prolonged into a very long, thread-like appendage thrown over the leaves of the plant or of its neighbors, and ultimately reaching the ground, and thus, it is presumed, affording ants and other insects means of access to the flowers, and consequent fertilization. These species were grown by Mr. Elwes, and exhibited by him before the Scientific Committee. The present species is of somewhat similar character, but is, we believe, new alike to gardens and to science. We met with it in the course of the autumn in the nursery of Messrs. Sander, at St. Alban's; but learn that it has since passed into the hands of Mr. W. Bull, of Chelsea. It was imported accidentally with orchids, probably from the Philippine Islands. It belongs to Engler's section, trisecta, having two stalked leaves, each deeply divided into three ovate acute glabrous segments. The petioles are long, pale purplish, rose-colored, sprinkled with small purplish spots. The spathes are oblong acute or acuminate, convolute at the base, brownish-purple, striped longitudinally with narrow whitish bands. The spadix is cylindrical, slender, terminating in along, whip-like extremity, much longer than the spathe. The flowers have the arrangement and structure common to the genus, the females being crowded at the base of the spadix, the males immediately above them, and these passing gradually into fleshy incurved processes, which in their turn pass gradually into long, slender, purplish threads, covering the whole of the free end of the spadix.—M.T.M., in The Gardeness' Chronicle.
In the new edition of Mason's "Burma" we read that among other uses to which the bamboo is applied, not the least useful is that of producing fire by friction. For this purpose a joint of thoroughly dry bamboo is selected, about 1½ inches in diameter, and this joint is then split in halves. A ball is now prepared by scraping off shavings from a perfectly dry bamboo, and this ball being placed on some firm support, as a fallen log or piece of rock, one of the above halves is held by its ends firmly down on it, so that the ball of soft fiber is pressed with some force against its inner or concave surface. Another man now takes a piece of bamboo a foot long or less, and shaped with a blunt edge, something like a paper knife, and commences a sawing motion backward and forward across the horizontal piece of bamboo, and just over the spot where the ball of soft fiber is held. The motion is slow at first, and by degrees a groove is formed, which soon deepens as the motion increases in quickness. Soon smoke arises, and the motion is now made as rapid as possible, and by the time the bamboo is cut through not only smoke but sparks are seen, which soon ignite the materials of which the ball beneath is composed. The first tender spark is now carefully blown, and when well alight the ball is withdrawn, and leaves and other inflammable materials heaped over it, and a fire secured. This is the only method that I am aware of for procuring fire by friction in Burma, but on the hills and out of the way parts, that philosophical toy, the "pyrophorus," is still in use. This consists1 of a short joint of a thick woody bamboo, neatly cut, which forms a cylinder. At the bottom of this a bit of tinder is placed, and a tightly-fitting piston inserted composed of some hard wood. The tube being now held in one hand, or firmly supported, the piston is driven violently down on the tinder by a smart blow from the hand, with the result of igniting the tinder beneath.
Another method of obtaining fire by friction from bamboos is thus described by Captain T.H. Lewin ("Hill Tracts of Chittagong, and the Dwellers Therein", Calcutta, 1869, p. 83), as practiced in the Chittagong Hills. The Tipporahs make use of an ingenious device to obtain fire; they take a piece of dry bamboo, about a foot long, split it in half, and on its outer round surface cut a nick, or notch, about an eighth of an inch broad, circling round the semi-circumference of the bamboo, shallow toward the edges, but deepening in the center until a minute slit of about a line in breadth pierces the inner surface of the bamboo fire-stick. Then a flexible strip of bamboo is taken, about 1½ feet long and an eighth of an inch in breadth, to fit the circling notch, or groove, in the fire-stick. This slip or band is rubbed with fine dry sand, and then passed round the fire-stick, on which the operator stands, a foot on either end. Then the slip, grasped firmly, an end in each hand, is pulled steadily back and forth, increasing gradually in pressure and velocity as the smoke comes. By the time the fire-band snaps with the friction there ought to appear through the slit in the fire-stick some incandescent dust, and this placed, smouldering as it is, in a nest of dry bamboo shavings, can be gently blown into a flame.—The Gardeners' Chronicle.
It is also made of a solid cylinder of buffalo's horn, with a central hollow of three-sixteenths of an inch in diameter and three inches deep burnt into it. The piston, which fits very tightly in it, is made of iron-wood or some wood equally hard.
When we read how one mediæval saint stood erect in his cell for a week without sleep or food, merely chewing a plantain-leaf out of humility, so as not to be too perfect; how another remained all night up to his neck in a pond that was freezing over; and how others still performed for the glory of God feats no less tasking to their energies, we are inclined to think, that, with the gods of yore, the men, too, have departed, and that the earth is handed over to a race whose will has become as feeble as its faith. But we ought not to yield to these instigations, by which the evil one tempts us to disparage our own generation. The gods have somewhat changed their shape, 'tis true, and the men their minds; but both are still alive and vigorous as ever for an eye that can look under superficial disguises. The human energy no longer freezes itself in fish-ponds, and starves itself in cells; but near the north pole, in central Africa, on Alpine "couloirs," and especially in what are nowadays called "psycho-physical laboratories," it maybe found as invincible as ever, and ready for every fresh demand. To most people a north pole expedition would be an easy task compared with those ineffably tedious measurements of simple mental processes of which Ernst Heinrich Weber set the fashion some forty years ago, and the necessity of extending which in every possible direction becomes more and more apparent to students of the mind. Think of making forty thousand estimates of which is the heavier of two weights, or seventy thousand answers as to whether your skin is touched at two points or at one, and then tabulating and mathematically discussing your results! Insight is to be gained at no less price than this. The new sort of study of the mind bears the same relation to the older psychology that the microscopic anatomy of the body does to the anatomy of its visible form, and the one will undoubtedly be as fruitful and as indispensable as the other.
Dr. Ebbinghaus1 makes an original addition to heroic psychological literature in the little work whose title we have given. For more than two years he has apparently spent a considerable time each day in committing to memory sets of meaningless syllables, and trying to trace numerically the laws according to which they were retained or forgotten. Most of his results, we are sorry to say, add nothing to our gross experience of the matter. Here, as in the case of the saints, heroism seems to be its own reward. But the incidental results are usually the most pregnant in this department; and two of those which Dr. Ebbinghaus has reached seems to us to amply justify his pains. The first is, that, in forgetting such things as these lists of syllables, the loss goes on very much more rapidly at first than later on. He measured the loss by the number of seconds required to relearn the list after it had been once learned. Roughly speaking, if it took a thousand seconds to learn the list, and five hundred to relearn it, the loss between the two learnings would have been one-half. Measured in this way, full half of the forgetting seems to occur within the first half-hour, while only four-fifths is forgotten at the end of a month. The nature of this result might have been anticipated, but hardly its numerical proportions.
The other important result relates to the question whether ideas are recalled only by those that previously came immediately before them, or whether an idea can possibly recall another idea, with which it was never in immediate contact, without passing through the intermediate mental links. The question is of theoretic importance with regard to the way in which the process of "association of ideas" must be conceived; and Dr. Ebbinghaus' attempt is as successful as it is original, in bringing two views, which seem at first sight inaccessible to proof, to a direct practical test, and giving the victory to one of them. His experiments conclusively show that an idea is not only "associated" directly with the one that follows it, and with the rest through that, but that it is directly associated with all that are near it, though in unequal degrees. He first measured the time needed to impress on the memory certain lists of syllables, and then the time needed to impress lists of the same syllables with gaps between them. Thus, representing the syllables by numbers, if the first list was 1, 2, 3, 4 ... 13, 14, 15, 16, the second would be 1, 3, 5 ... 15, 2, 4, 6 ... 16, and so forth, with many variations.
Now, if 1 and 3 in the first list were learned in that order merely by 1 calling up 2, and by 2 calling up 3, leaving out the 2 ought to leave 1 and 3 with no tie in the mind; and the second list ought to take as much time in the learning as if the first list had never been heard of. If, on the other hand, 1 has a direct influence on 3 as well as on 2, that influence should be exerted even when 2 is dropped out; and a person familiar with the first list ought to learn the second one more rapidly than otherwise he could. This latter case is what actually occurs; and Dr. Ebbinghaus has found that syllables originally separated by as many as seven intermediaries still reveal, by the increased rapidity with which they are learned in order, the strength of the tie that the original learning established between them, over the heads, so to speak, of all the rest. It may be that this particular series of experiments is the entering wedge of a new method of incalculable reach in such questions. The future alone can show. Meanwhile, when we add to Dr. Ebbinghaus' "heroism" in the pursuit of true averages, his high critical acumen, his modest tone, and his polished style, it will be seen that we have a new-comer in psychology from whom the best may be expected.—W.J., Science.
"Ueber das Gedächtniss. Untersuchungen zur experimentellen Psychologie." Von Herm. Ebbinghaus. Leipzig: Duncker u. Humblot, 1885. 10+169 pp. 8vo.
The sinking of mine shafts in certain Belgian and French basins, where the coal deposit is covered with thick strata of watery earth, has from all times been considered as the most troublesome and delicate, and often the most difficult operation, of the miner's art. Of the few modern processes that have been employed for this purpose, that of Messrs. Kind and Chaudron has been found most satisfactory, although it leaves much to be desired where it is a question of traversing moving sand. An interesting modification of this well-known process has recently been described by Mr. E. Chavatte, in the Bulletin de la Societe Industrielle du Nord de la France. Two years ago the author had to sink a working shaft at Quievrechain, 111 feet of which was to traverse a mass of moving and flowing sand, inconsistent earth, gravel, and marls, and proceeded as follows:
He first put down two beams, A B (Pl. 1, Figs. 2, 3, and 9), each 82 feet in length and of 20×20 inch section in the center, and upon these placed two others, E F, of 16×16 inch section. Beneath the two first were inserted six joists, c c c c c c, about 82 feet in length and of 14 or 16 inch section in the center. Finally these were strengthened at their extremities with two others, d d, about 82 feet in length. All these timbers, having been connected by tie bands and bolts, constituted a rigid structure that covered a surface of nearly seven hundred square yards.
From the beams, A B and E F, there was suspended a red fir frame by means of thirty-four iron rods.
Upon this frame, which was entirely immersed in the moving sand, there was established brick masonry (Figs. 1, 2, and 3). As the ends of the timbers entered the latter, and were connected by 1½ inch bolts, they concurred in making the entire affair perfectly solid. The frame, K K, was provided with an oaken ring, which was affixed to it with bolts.
After this, a cast iron tubbing, having a cutting edge, and being composed of rings 3.28 feet wide and made of six segments, was lowered. This tubbing was perfectly tight, all the surfaces of the joints having been made even and provided with strips of lead one-tenth of an inch thick. It weighed 4,000 pounds to the running foot.
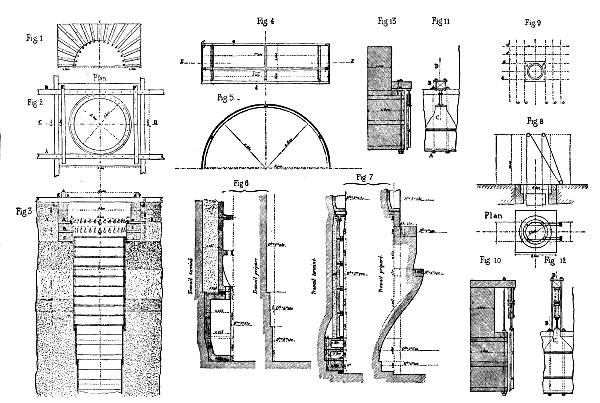
Fig. 1.—Section through A B. Fig. 2.—Plan. Fig. 3.—Section through C D. Fig. 5.—Section through E F of Fig. 4. Figs. 6 and 7.—Work Prepared and finished. Fig. 10.—Section through A B and C D of Fig. 12. Figs. 11 6 and 12.—Arrangement of jack-screw. Fig. 13.—Section through A B and C D of Fig. 11.
Plate I.—SINKING A MINE SHAFT.
It was first raised to a height of fifteen feet, so as to cause it to enter the sand by virtue of its own gravity. It thus penetrated to a depth of about twenty inches. After this the workmen were ordered to man the windlasses and hoist out some of the sand. This caused the tubbing to descend about eight inches more, when it came to a standstill. It was now loaded with 17,000 pounds of pig iron, but in vain, for it refused to budge. Mr. Chavatte therefore had recourse to a dredge with vertical axis, constructed as follows:
Upon a square axis, A B (Pl. 2, Figs. 1, 2, and 3), provided with double cross braces, C D, and strengthened by diagonals, were riveted, by their upper extremities, two cheeks, G H, whose lower extremities held the steel plates, I J I' J', which, in turn, were fastened to the axis, A B, by their other extremities. These plates were so inclined as to scrape the surface of the ground over which they were moved. They each carried two bags made of coarse canvas and strengthened by five strong leather straps (Figs. 2 and 4). To the steel plates were riveted two plates of iron containing numerous apertures, through which passed leather straps designed for fastening thereto the lower part of the mouth of the bags. That portion of the mouth of the latter that was to remain open was fastened in the same way to two other plates, X Y, X¹ Y¹ (Fig. 1), held between the lower cross-braces.
When the apparatus was revolved, the plates scraped the earth to be removed, and descended in measure as the latter entered the bags. These bags, when full, were hooked, by means of the five rings which they carried, to the device shown in Fig. 8 (Pl. 2), and raised to the surface and emptied into cars.
The dredge was set in motion by four oak levers (Figs. 5 and 6). Two of these were manned by workmen stationed upon the surface flooring, and the other two by workmen upon the flooring in the tubbing. The axis was elongated, in measure as the apparatus descended, by rods of the same dimensions fastened together by cast iron sleeves and bolts (Fig. 7).
The steel plates were not capable of acting alone, even in cases where they operated in pure moving sand containing no pebbles, for the sand was too compact to be easily scraped up by the steel, and so it had to be previously divided. For this purpose Mr. Chavatte used rakes which were in form exactly like those of the extirpators, U and V, of Figs. 1, 2, and 3, of Pl. 2, except that the dividers carried teeth that were not so strong as those of the extirpators, and that were set closer together. These rakes were let down and drawn up at will. They were maneuvered as follows:
The dredge descended with the extirpators pointing upward. When their heads reached the level of the upper floor, the tools were removed. Then the dredge was raised again. In this way the extirpators lay upon the floor, and, if the lifting was continued, they placed themselves in their working position, in which they were fixed by the bolts A" B" C" (Fig. 1). After this, the apparatus was let down and revolved. The rakes divided the earth, the scrapers collected it, and the bags pocketed it.
The great difficulty was to cause the tubbing to descend vertically, and also to overcome the enormous lateral pressure exerted upon it by the earth that was being traversed. Water put into the shaft helped somewhat, but the great stress to be exerted had to be effected by means of powerful jack screws. These were placed directly upon the tubbing, and bore against strong beams whose extremities were inserted into the masonry.
As a usual thing it is not easy to use more than four or six such jacks, since the number of beams that can be employed is limited, owing to the danger of obstructing the mouth of the shaft. Yet twelve were used by Mr. Chavatte, and this number might have been doubled had it been necessary. As we have seen, the frame, K K (Pl. 1, Fig. 3), was provided with an oak circle traversed by 32 bolts. The length of these latter was two inches and a quarter longer than they needed to have been, or they were provided with wooden collars of that thickness. Later on, these collars were replaced with iron bars that held the wood against which the jacks bore in order to press the tubbing downward (Pl. 1, Figs. 10, 11, 12, and 13).
Mr. Chavatte's great anxiety was to know whether he should succeed in causing the first section of tubbing to traverse the four feet of gravel; for in case it did not pass, he would be obliged to employ a second section of smaller diameter, thus increasing the expense. He was persuaded that the coarse gravel remaining in the side of the shaft would greatly retard the descent of the tubbing. So he had decided to remove such obstructions at the proper moment through divers or a diving bell. Then an idea occurred to him that dispensed with all that trouble, and allowed him to continue with the first section. This was to place upon the dredge two claw-bars, T (Pl. 2, Fig. 3), which effected the operation of widening with wonderful ease. To do this it was only necessary to turn up the bags, and revolve the apparatus during its descent. The claw at the extremity of the bar pulled out everything within its reach, and thus made an absolutely free passage for the tubbing.
The sands and gravels were passed by means of a single section of tubbing 31 feet in length, which was not stopped until it had penetrated a stratum of white chalk to a depth of two yards. This chalk had no consistency, although it contained thin plates of quite large dimensions. These were cut, as if with a punch, by means of the teeth of the extirpator.
It now remains to say a few words concerning the sinking of the shaft, which, after the operation of the dredge, was continued by the process called "natural level" The work was not easy until a depth of 111 feet had been reached. Up to this point it had been necessary to proceed with great prudence, and retain the shifting earth by means of four iron plate tubes weighing 54 tons. Before finding a means of widening the work already done by the dredge, Mr. Chavatte was certain that he would have to use two sections of tubbing, and so had given the first section a diameter of 16½ feet. He could then greatly reduce the diameter, and bring it to 15¾ feet as soon as the ground auger was used.
After two yards of soil had been removed from beneath the edge of the tubbing, the earth began to give way. Seeing this, Mr. Chavatte let down a tube 13 feet in length and 15.4 in diameter. The exterior of this was provided with 12 oak guides, which sliding over the surface of the tubbing had the effect of causing the tube to descend vertically. And this was necessary, because this tube had to be driven down every time an excavation of half a yard had been made.
Afterward, a diameter of 15.35 feet was proceeded with, and the small central shaft of 4¼ feet diameter was begun. This latter had not as yet been sunk, for fear of causing a fall of the earth.
Next, the earth was excavated to a depth of 8.2 feet, and a tube 16.4 feet in length was inserted; then a further excavation of 8.2 feet was made, and the tube driven home.
After this an excavation of 26¼ feet was made, and a tube of the same length and 14½ feet in diameter was driven down. Finally, the shifting soil was finished with a fourth tube 19½ feet in length and 14 feet in diameter.
A depth of 111 feet had now been reached, and the material encountered was solid white chalk. From this point the work proceeded with a diameter of 13.9 feet to a depth of 450 feet. The small shaft had been sunk directly to a depth of 475 feet. At 450 feet the diameter was diminished by three inches. Then an advance of a foot was made, and the diameter reduced by one and a half inch.
The reason for this reduction in the diameter and change in the mode of deepening was as follows:
The Chaudron moss-box, when it chances to reach its seat intact, and can consequently operate well, undoubtedly makes a good wedging. But how many times does it not happen that it gets injured before reaching its destination? Besides, as it often rests upon earth that has caved in upon its seat during the descent of the tubbing, it gets askew, and later on has to be raised on one side by means of jacks or other apparatus. Under such circumstances, Mr. Chavatte considered this moss-box as more detrimental than useful, and not at all indispensable, and so substituted beton for it, as had previously been done by Mr. Bourg, director of the Bois-du-Luc coal mines.
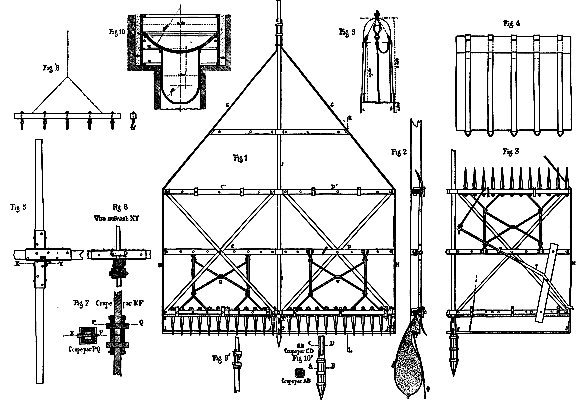
Figs. 1, 2, 3, 6 and 4.—Details of dredge. Figs. 5 6 and 6.—Details of maneuvering lever. Fig. 7.—Mode of lengthening the axis of the dredge. Fig. 8.—Hooks for lifting the dredge bags. Fig. 9.—Arrangement of valves in the beton box. Fig. 10.—Device for centering the tubbing.
Plate II.
This engineer likewise suppressed the balancing column, which is often a source of trouble in the descent of the tubbing, and forced his tubbing to center itself with the shaft through a guide with four branches riveted under the false bottom that entered the small shaft (Pl. 2, Fig. 10). Mr. Bourg so managed that there remained an empty space of ten inches to fill in with beton. Mr. Chavatte had at first intended to proceed in the same way, but the two last tubbings, upon which he had not counted, forced him to reduce the space to 5¾ inches. Under such circumstances it was not prudent to employ the same means for guiding the base of the tubbing, because, if the central shaft had not exactly the same center as the large one, there would have been danger of throwing the tubbing sideways and causing it to leak. Seeing which, Mr. Chavatte strengthened the lower part of the base ring and placed it upon another ring tapering downward, and 27½ inches in height (Pl. 1, Fig. 5). The object of this lower ring was to force the tubbing to remain concentric with the shaft, to form a tight joint with its upper conical portion, and to form a joint upon the seat with its lower flange, so as to prevent the beton from flowing into the small shaft.
After the shaft was pumped out, digging by hand was begun with a diameter of 12 feet. After descending 20 inches an 8×10 inch curb was laid, in order to consolidate the earth and prevent any movement of the tubbing. Then the excavating was continued to a depth of 31½ inches, and with a diameter of 9¾ feet. At this point another curb was put in for consolidating the earth. Finally, the bottom was widened out as shown in Fig. 7, so that three basal wedged curbs could be put in. This done, the false tubbing was put in place; and finally, when proceeding upward, the last ring composed of twelve pieces was reached, the earth was excavated and at once replaced with a collar composed of twelve pieces of oak tightened up by oak wedges. Each of these pieces was cemented separately and in measure as they were assembled.
Through motive of economy no masonry was placed under the base of the three wedged curbs. In fact, by replacing this with a wedged curb of wood traversed by six bolts designed to fix the cast iron curb immediately above, Mr. Chavatte obtained a third curb that he would have had to have made of cast iron.
Among the most useful inventions of the latter half of the nineteenth century the gas-engine holds a prominent place. While its development has not been so brilliant or so startling as that which we can note in the employment of electricity, it holds, among the applications of heat, the most important place of any invention made within that period. Even amid the contrivances by which, in recent times, the other forces of nature have been subdued to the uses of man, there are only a few which rival the gas-engine in practical importance. With regard to the steam-engine itself, it is remarkable how little that is new has really been invented since the time of Watt and Woulfe. In the specifications of the former can be shown completely delineated, or fully foreshadowed, nearly every essential condition of the economy and efficiency attained in our own days; and it is only by a gradual "survival of the fittest" of the many contrivances which were made to carry out his broad ideas that the steam-engine of the present has attained its great economy.
It is but within the last fifty years that the laws of the relation between the different physical forces were first enunciated by Justice Grove, and confirmed by the classical researches of Dr. Joule—the one a lawyer, working hard at his profession, the other a man of business engaged in manufacture. Both are still living among us; the latter having withdrawn from business, while the former is a Judge of the High Court of Justice. I always regret that the claims of his profession have weaned Justice Grove from science; for, while it may be possible to find in the ranks of the Bar many who might worthily occupy his place on the Bench, it would be hard to find among men of science any with as wide-reaching and practical philosophy as that which he owns. The chemist demonstrated long since that it was impossible for man to create or destroy a single particle of ponderable matter; but it remained for our own time to prove that it was equally impossible to create or destroy any of the energy which existed in nature as heat, mechanical power, electricity, or chemical affinity. All that it is in the power of man to do is to convert one of these forms into another. This, perhaps the greatest of all scientific discoveries since the time of Newton, was first, I believe, enunciated in 1842 by Grove, in a lecture given at the London Institution; and it was experimentally proved by the researches of Joule, described in a paper which he read at the meeting of the British Association which was held at Cork—my native city—in 1843. My friend Dr. Sullivan, now President of Queen's College, Cork, and I myself had the privilege of being two of a select audience of half a dozen people, who alone took sufficient interest in the subject to hear for the first time developed the experimental proof of the theory which welds into one coherent system the whole physical forces of the universe, and enables one of these to be measured by another. One branch of the "correlation of physical forces," as it was termed by Grove, was the relation between mechanical power and heat, and the convertibility of each into the other, which, under the name of "Thermodynamics," has become one of the most important branches of practical science.
Joule's first experiments clearly proved that each of these forms of energy was convertible into the other; but some discrepancies arose in determining the exact equivalent of each. His subsequent researches, however, clearly demonstrated the true relation between both. Taking as the unit of heat the amount which would be necessary to raise 1 lb. of water 1° of Fahrenheit's scale (now called "the English thermal unit"), he proved that this unit was equivalent to the mechanical power which would be required to raise 772 lb. 1 foot, or to raise 1 lb. 772 ft. perpendicularly against the force of gravity. The heat-unit—the pound-degree—which I will distinguish by the Greek letter θ, is a compound unit of mass and temperature; the second—the foot-pound = f.p.—a compound unit of mass and space. This equation, called "Joule's equivalent," or 1 thermal unit = 772 foot-pounds, is the foundation and the corner-stone of thermodynamics.
It is essential to understand the meaning of this equation. It expresses the maximum effect of the given cause, viz., that if all the heat were converted into power, or all the power were converted into heat, 1 thermal unit would produce 772 foot-pounds, or 772 foot-pounds would raise 1 lb. of water 1° Fahr. But there is never a complete conversion of any form of energy. Common solid coal may be partly converted into gases in a retort; but some of the carbon remains unchanged, and more is dissipated but not lost. In the same way, if I take five sovereigns to Paris and convert them into francs, and return to London and convert the francs into shillings, I shall not have 100 shillings, but only perhaps 95 shillings. But the five shillings have not been lost; three of them remain in the French change de monnaies, and two of them in the English exchange office. I may have forfeited something, but the world has forfeited nothing. There remains in it exactly the same number of sovereigns, francs, and shillings as there was before I set out on my travels. Nothing has been lost, but some of my money has been "dissipated;" and the analogous case, "the dissipation of energy," has formed the subject of more than one learned essay.
Before the invention of the steam-engine, the only powers employed in mechanics were those of wind and water mills, and animal power. In the first two, no conversion of one force into another took place; they were mere kinematic devices for employing the mechanical force already existing in the gale of wind and the head of water. With regard to the power developed by man and other animals, we had in them examples of most efficient heat-engines, converting into power a large percentage of the fuel burnt in the lungs. But animal power is small in amount, and it is expensive for two reasons—first, because the agents require long intervals of rest, during which they still burn fuel; and next, because the fuel they require is very expensive. A pound of bread or beef, or oats or beans, costs a great deal more than a pound of coal; while it does not, by its combustion, generate nearly so much heat. The steam-engine, therefore, took the place of animal power, and for a long time stood alone; and nearly all the motive power derived from heat is still produced by the mechanism which Watt brought to such great efficiency in so short a time.
Now the practical question for all designers and employers of heat-engines is to determine how the greatest quantity of motive force can be developed from the heat evolved from a given kind of fuel; and coal being the cheapest of all, we will see what are the results obtainable from it by the steam-engine. In this we have three efficiencies to consider—those of the furnace, the boiler, and the cylinder.
First, with respect to the furnace. The object is to combine the carbon and the hydrogen of the coal with a sufficient quantity of the oxygen of the air to effect complete combustion into carbonic acid and water. In order to do this, we have to use a quantity of air much larger than is theoretically necessary, and also to heat an amount of inert nitrogen five times greater than the necessary oxygen; and we are therefore obliged to create a draught which carries away to the chimney a considerable portion of the heat developed. The combustion, moreover, is never perfect; and some heat is lost by conduction and radiation. The principal loss is by hot gases escaping from the flues to the chimney. Even with well-set boilers, the temperature in the chimney varies from 400° to 600° Fahr. Taking the mean of 500°, this would represent a large proportion of the total heat, even if the combustion were perfect; for, as a general rule, the supply of air to a furnace is double that which is theoretically necessary. For our present purpose, it will be sufficient to see how much the whole loss is, without dividing it under the several heads of "imperfect combustion," "radiation," and "convection," by the heated gases passing to the chimney.
With a very good boiler and furnace each pound of coal evaporates 10 pounds of water from 62° Fahr., changing it into steam of 65 lb. pressure at a temperature of 312°, or 250° above that of the water from which it is generated. Besides these 250°, each pound of steam contains 894 units of latent heat, or 1,144 units in all. A very good condensing engine will work with 2.2 lb. of coal and 22 lb. of steam per horse power per hour. Now. 1 lb. of good coal will, by its combustion, produce 14,000 heat-units; and the 2.2 lb. of coal multiplied by 14,000 represent 30,800 θ. Of these we find in the boiler 22 × 1,144, or 25,168 units, or about 81½ per cent., of the whole heat of combustion; so that the difference (5,632 units, or 18½ per cent.) has been lost by imperfect combustion, radiation, or convection. The water required for condensing this quantity of steam is 550 lb.; and, taking the temperature in the hot well as 102°, 550 lb. have been raised 40° from 62°. Thus we account for 550 × 40 = 22,000, or (say) 71½ per cent. still remaining as heat. If we add this 71½ per cent. to 18½ per cent. we have 90 per cent., and there remain only 10 per cent. of the heat that can possibly have been converted into power. But some of this has been lost by radiation from steam-pipes, cylinder, etc. Allowing but 1 per cent. for this, we have only 9 per cent. as the efficiency of a really good condensing engine. This estimate agrees very closely with the actual result; for the 2.2 lb. of coal would develop 30,800 θ; and this, multiplied by Joule's equivalent, amounts to nearly 24 millions of foot-pounds. As 1 horse power is a little less than 2 million foot-pounds per hour, only one-twelfth, or a little more than 8 per cent. of the total heat is converted; so that whether we look at the total quantity of heat which we show unconverted, or the total heat converted, we find that each supplements and corroborates the other. If we take the efficiency of the engine alone, without considering the loss caused by the boiler, we find that the 25,168 θ which entered the boiler should have given 19,429,696 foot-pounds; so that the 2 millions given by the engine represent about 10 per cent. of the heat which has left the boiler. The foregoing figures refer to large stationary or marine engines, with first-rate boilers. When, however, we come to high-pressure engines of the best type, the consumption of coal is twice as much; and for those of any ordinary type it is usual to calculate 1 cubic foot, or 62½ lb., of water evaporated per horse power. This would reduce the efficiency to about 6 per cent. for the best, and 3 per cent. for the ordinary non-condensing engines; and if to this we add the inefficiency of some boilers, it is certain that many small engines do not convert into power more than 2 per cent. of the potential energy contained in the coal.
At one time the steam-engine was threatened with serious rivalry by the hot-air engine. About the year 1816 the Rev. Mr. Stirling, a Scotch clergyman, invented one which a member of this Institute (Mr. George Anderson) remembers to have seen still at work at Dundee. The principle of it was that a quantity of air under pressure was moved by a mass, called a "displacer," from the cold to the hot end of a large vessel which was heated by a fire beneath and cooled by a current of water above. The same air was alternately heated and cooled, expanded and contracted; and by the difference of pressure moved the piston in a working cylinder. In this arrangement the furnace was inefficient. As only a small portion of heat reached the compressed air, the loss by radiation was very great, and the wear and tear exceedingly heavy. This system, with some modifications, was revived by Rankine, Ericsson, Laubereau, Ryder, Buckett, and Bailey. Siemens employed a similar system, only substituting steam for air. Another system, originally proposed by Sir George Cayley, consisted in compressing by a pump cold air which was subsequently passed partly through a furnace, and, expanding, moved a larger piston at the same pressure; and the difference of the areas of the pistons multiplied by the pressure common to both represented the indicated power. This principle was subsequently developed by a very able mechanic, Mr. Wenham; but his engine never came much into favor. The only hot-air engines at present in use are Ryder's, Buckett's, and Bailey's, employed to a limited extent for small powers. I have not said anything of the thermal principles involved in the construction of these engines, as they are precisely the same as those affecting the subject of the present paper.
Before explaining the principle upon which the gas-engine and every other hot-air engine depends, I shall remind you of a few data with which most of you are already familiar. The volume of every gas increases with the temperature; and this increase was the basis of the air thermometer—the first ever used. It is to be regretted that it was not the foundation of all others; for it is based on a physical principle universally applicable. Although the volume increases with the temperature, it does not increase in proportion to the degrees of any ordinary scale, but much more slowly. Now, if to each of the terms of an arithmetical series we add the same number, the new series so formed increases or decreases more slowly than the original; and it was discovered that, by adding 461 to the degrees of Fahrenheit's scale, the new scale so formed represented exactly the increment of volume caused by increase of temperature. This scale, proposed by Sir W. Thomson in 1848, is called the "scale of absolute temperature." Its zero, called the "absolute zero," is 461° below the zero of Fahrenheit, or 493° below the freezing point of water; and the degree of heat measured by it is termed the "absolute temperature." It is often convenient to refer to 39° Fahr. (which happens to be the point at which water attains its maximum density), as this is the same as 500° absolute; for, counting from this datum level, a volume of air expands exactly 1 per cent. for 5°, and would be doubled at 1,000° absolute, or 539° Fahr.
Whenever any body is compressed, its specific heat is diminished; and the surplus portion is, as it were, pushed out of the body—appearing as sensible heat. And whenever any body is expanded, its specific heat is increased; and the additional quantity of heat requisite is, as it were, sucked in from surrounding bodies—so producing cold. This action may be compared to that of a wet sponge from which, when compressed, a portion of the water is forced out, and when the sponge is allowed to expand, the water is drawn back. This effect is manifested by the increase of temperature in air-compressing machines, and the cold produced by allowing or forcing air to expand in air-cooling machines. At 39° Fahr., 1 lb. of air measures 12½ cubic feet. Let us suppose that 1 lb. of air at 39° Fahr. = 500° absolute, is contained in a non-conducting cylinder of 1 foot area and 12½ feet deep under a counterpoised piston. The pressure of the atmosphere on the piston = 144 square inches × 14.7 lb., or 2,116 lb. If the air be now heated up to 539° Fahr. = 1,000° absolute, and at the same time the piston is not allowed to move, the pressure is doubled; and when the piston is released, it would rise 12½ feet, provided that the temperature remained constant, and the indicator would describe a hyperbolic curve (called an "isothermal") because the temperature would have remained equal throughout. But, in fact, the temperature is lowered, because expansion has taken place, and the indicator curve which would then be described is called an "adiabatic curve," which is more inclined to the horizontal line when the volumes are represented by horizontal and the pressures by vertical co-ordinates. In this case it is supposed that there is no conduction or transmission (diabasis) of heat through the sides of the containing vessel. If, however, an additional quantity of heat be communicated to the air, so as to maintain the temperature at 1,000° absolute, the piston will rise until it is 12½ feet above its original position, and the indicator will describe an isothermal curve. Now mark the difference. When the piston was fixed, only a heating effect resulted; but when the piston moved up 12½ feet, not only a heating but a mechanical, in fact, a thermodynamic, effect was produced, for the weight of the atmosphere (2,116 lb.) was lifted 12½ feet = 26,450 foot-pounds.
The specific heat of air at constant pressure has been proved by the experiments of Regnault to be 0.2378, or something less than one-fourth of that of water—a result arrived at by Rankine from totally different data. In the case we have taken, there have been expended 500 × 0.2378, or (say) 118.9 θ to produce 26,450 f.p. Each unit has therefore produced 26,450 / 118.9 = 222.5 f.p., instead of 772 f.p., which would have been rendered if every unit had been converted into power. We therefore conclude that 222.5 / 772 = 29 per cent. of the total heat has been converted. The residue, or 71 per cent., remains unchanged as heat, and may be partly saved by a regenerator, or applied to other purposes for which a moderate heat is required.
The quantity of heat necessary to raise the heat of air at a constant volume is only 71 per cent. of that required to raise to the same temperature the same weight of air under constant pressure. This is exactly the result which Laplace arrived at from observations on the velocity of sound, and may be stated thus—
| Specific heat. | Foot- pounds. | Per cent. | |
| Kp= 1 lb. of air at constant pressure | 0.2378 × 772 = | 183.5 = | 100 |
| Kv= 1 lb. of air at constant volume | 0.1688 × 772 = | 130.3 = | 71 |
| Difference, being heat converted into power | 0.0690 × 772 = | 53.2 = | 29 |
Or, in a hot-air engine without regeneration, the maximum effect of 1 lb. of air heated 1° Fahr. would be 53.2 f.p. The quantity of heat Ky necessary to heat air under constant volume is to Kv, or that necessary to heat it under constant pressure, as 71:100, or as 1:1.408, or very nearly as 1:√2—a result which was arrived at by Masson from theoretical considerations. The 71 per cent. escaping as heat may be utilized in place of other fuel; and with the first hot-air engine I ever saw, it was employed for drying blocks of wood. In the same way, the unconverted heat of the exhaust steam from a high-pressure engine, or the heated gases and water passing away from a gas-engine, may be employed.
We are now in a position to judge what is the practical efficiency of the gas-engine. Some years since, in a letter which I addressed to Engineering, and which also appeared in the Journal of Gas Lighting,2 I showed (I believe for the first time) that, in the Otto-Crossley engine, 18 per cent. of the total heat was converted into power, as against the 8 per cent. given by a very good steam-engine. About the end of 1883 a very elaborate essay, by M. Witz, appeared in the Annales de Chimie et de Physique, reporting experiments on a similar engine, which gave an efficiency somewhat lower. Early in 1884 there appeared in Van Nostrand's Engineering Magazine a most valuable paper, by Messrs. Brooks and Steward, with a preface by Professor Thurston,3 in which the efficiency was estimated at 17 to 18 per cent. of the total heat of combustion. Both these papers show what I had no opportunity of ascertaining, that is, what becomes of the 82 per cent. of heat which is not utilized—information of the greatest importance, as it indicates in what direction improvement may be sought for, and how loss may be avoided. But, short as is the time that has elapsed since the appearance of these papers, you will find that progress has been made, and that a still higher efficiency is now claimed.
When I first wrote on this subject, I relied upon some data which led me to suppose that the heating power of ordinary coal gas was higher than it really is. At our last meeting, Mr. Hartley proved, by experiments with his calorimeter, that gas of 16 or 17 candles gave only about 630 units of heat per cubic foot. Now, if all this heat could be converted into power, it would yield 630 × 772, or 486,360 f.p.; and it would require only 1,980,000 / 486,360 = 4.07 cubic feet to produce 1 indicated horse power. Some recent tests have shown that, with gas of similar heating power, 18 cubic feet have given 1 indicated horse power, and therefore 4.07 / 18 = 22.6 of the whole heat has been converted—a truly wonderful proportion when compared with steam-engines of a similar power, showing only an efficiency of 2 to 4 per cent.
The first gas-engine which came into practical use was Lenoir's, invented about 1866, in which the mixture of gas and air drawn in for part of the stroke at atmospheric pressure was inflamed by the spark from an induction coil. This required a couple of cells of a strong Bunsen battery, was apt to miss fire, and used about 90 cubic feet of gas per horse power. This was succeeded by Hugon's engine, in which the ignition was caused by a small gas flame, and the consumption was reduced to 80 cubic feet. In 1864 Otto's atmospheric engine was invented, in which a heavily-loaded piston was forced upward by an explosion of gas and air drawn in at atmospheric pressure. In its upward stroke the piston was free to move; but in its downward stroke it was connected with a ratchet, and the partial vacuum formed after the explosion beneath the piston, together with its own weight in falling, operated through a rack, and caused rotation of the flywheel. This engine (which, in an improved form, uses only about 20 cubic feet of gas) is still largely employed, some 1,600 having been constructed. The great objection to it was the noise it produced, and the wear and tear of the ratchet and rack arrangements. In 1876 the Otto-Crossley silent engine was introduced. As you are aware, it is a single-acting engine, in which the gas and air are drawn in by the first outward, and compressed by the first inward stroke. The compressed mixture is then ignited; and, being expanded by heat, drives the piston outward by the second outward stroke. Near the end of this stroke the exhaust-valve is opened, the products of combustion partly escape, and are partly driven out by the second inward stroke. I say partly, for a considerable clearance space, equal to 38 per cent. of the whole cylinder volume, remains unexhausted at the inner end of the cylinder. When working to full power, only one stroke out of every four is effective; but this engine works with only 18 to 22 cubic feet of gas per horse power. Up to the present time I am informed that about 18,000 of these engines have been manufactured. Several other compression engines have been introduced, of which the best known is Mr. Dugald Clerk's, using about 20 feet of Glasgow cannel gas. It gives one effective stroke for every revolution; the mixture being compressed in a separate air-pump. But this arrangement leads to additional friction; and the power measured by the brake is a smaller percentage of the indicated horse power than in the Otto-Crossley engine. A number of gas engines—such as Bisschop's (much used for very small powers), Robson's (at present undergoing transformation in the able hands of Messrs. Tangye), Korting's, and others—are in use; but, so far as I can learn, all require a larger quantity of gas than those previously referred to.
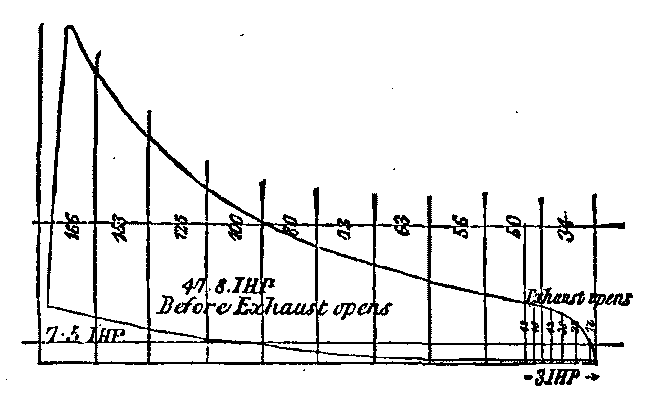
 OTTO-CROSSLEY GAS ENGINE, 16 H.P.
OTTO-CROSSLEY GAS ENGINE, 16 H.P.
I have all along spoken of efficiency as a percentage of the total quantity of heat evolved by the fuel; and this is, in the eyes of a manufacturer, the essential question. Other things being equal, that engine is the most economical which requires the smallest quantity of coal or of gas. But men of science often employ the term efficiency in another sense, which I will explain. If I wind a clock, I have spent a certain amount of energy lifting the weight. This is called "energy of position;" and it is returned by the fall of the weight to its original level. In the same way if I heat air or water, I communicate to it energy of heat, which remains potential as long as the temperature does not fall, but which can be spent again by a decrease of temperature. In every heat-engine, therefore, there must be a fall from a higher to a lower temperature; otherwise no work would be done. If the water in the condenser of a steam-engine were as hot as that in the boiler, there would be equal pressure on both sides of the piston, and consequently the engine would remain at rest. Now, the greater the fall, the greater the power developed; for a smaller proportion of the heat remains as heat. If we call the higher temperature T and the lower T' on the absolute scale, T - T' is the difference; and the ratio of this to the higher temperature is called the "efficiency." This is the foundation of the formula we meet so often: E = (T - T')/T. A perfect heat-engine would, therefore, be one in which the temperature of the absolute zero would be attained, for (T - O)/T = 1. This low temperature, however, has never been reached, and in all practical cases we are confined within much narrower limits. Taking the case of the condensing engine, the limits were 312° to 102°, or 773° and 563° absolute, respectively. The equation then becomes (773 - 563)/773 = 210 / 773 or (say) 27 per cent. With non-condensing engines, the temperatures may be taken as 312° and 212°, or 773° and 673° absolute respectively. The equation then becomes (773 - 673)/773 = 100 / 773, or nearly 13 per cent. The practical efficiencies are not nearly this, but they are in about the same ratio—27/13. If, then, we multiply the theoretical efficiencies by 0.37, we get the practical efficiencies, say 10 per cent. and 5 per cent.; and it is in the former sense that M. Witz calculated the efficiency of the steam-engine at 35 per cent.—a statement which, I own, puzzled me a little when I first met it. These efficiencies do not take any account of loss of heat before the boiler. In the case of the gas-engine, the question is much more complicated on account of the large clearance space and the early opening of the exhaust. The highest temperature has been calculated by the American observers at 3,443° absolute, and the observed temperature of the exhaust gases was 1,229°. The fraction then becomes (3443 - 1229)/3443 = 64 per cent. If we multiply this by 0.37, as we did in the case of the steam-engine, we get 23.7 per cent., or approximately the same as that arrived at by direct experience. Indeed, if the consumption is, as sometimes stated, less than 18 feet, the two percentages would be exactly the same. I do not put this forward as scientifically true; but the coincidence is at least striking.
I have spoken of the illuminating power of the gas as of importance; for the richer gases have also more calorific power, and an engine would, of course, require a smaller quantity of them. The heat-giving power does not, however, vary as the illuminating power, but at a much slower rate; and, adopting the same contrivance as that on which the absolute scale of temperature is formed, I would suggest a formula of the following type: H = C (I + K), in which H represents the number of heat-units given out by the combustion of 1 cubic foot of gas, I is the illuminating power in candles, and C and K two constants to be determined by experiment. If we take the value for motive power of the different qualities of gas as given in Mr. Charles Hunt's interesting paper in our Transactions for 1882, C might without any great error be taken as 22 and K as 7.5. With Pintsch's oil gas, however, as compared with coal gas, this formula does not hold; and C should be taken much lower, and K much higher than the figures given above. That is to say, the heating power increases in a slower progression. The data available, however, are few; but I trust that Mr. Hartley will on this, as he has done on so many other scientific subjects, come to our aid.
I will now refer to the valuable experiments of Messrs. Brooks and Steward, which were most carefully made. Everything was measured—the gas by a 60 light, and the air by a 300 light meter; the indicated horse power, by a steam-engine indicator; the useful work, by a Prony brake; the temperature of the water, by a standard thermometer; and that of the escaping gases, by a pyrometer. The gas itself was analyzed; and its heating power calculated, from its composition, as 617.5θ. Its specific gravity was .464; and the volume of air was about seven times that of the gas used (or one-eighth of the mixture), and was only 11½ per cent. by weight more than was needed for perfect combustion. The results arrived at were as follows:
| Per cent. | |
| Converted into indicated horse power, including friction, etc. | 17.0 |
| Escaped with the exhaust gas. | 15.5 |
| Escaped in radiation. | 15.5 |
| Communicated to water in the jacket. | 52.0 |
It will thus be seen that more than half of the heat is communicated to the water in the jacket. Now, this is the opposite of the steam-engine, where the jacket is used to transmit heat to the cylinder, and not from it. This cooling is rendered necessary, because without it the oil would be carbonized, and lubrication of the cylinder rendered impossible. Indeed, a similar difficulty has occurred with all hot-air engines, and is, I think, the reason they have not been more generally adopted. I felt this so strongly that, for some time after the introduction of the gas-engine, I was very cautious in recommending those who consulted me to adopt it. I was afraid that the wear and tear would be excessive. I have, however, for some time past been thoroughly satisfied that this fear was needless; as I am satisfied that a well-made gas-engine is as durable as a steam-engine, and the parts subject to wear can be replaced at moderate cost. We have no boiler, no feed pump, no stuffing-boxes to attend to—no water-gauges, pressure-gauges, safety-valve, or throttle-valve to be looked after; the governor is of a very simple construction; and the slide-valves may be removed and replaced in a few minutes. An occasional cleaning out of the cylinder at considerable intervals is all the supervision that the engine requires.
The very large percentage of heat absorbed by the water-jacket should point out to the ingenuity of inventors the first problem to be attacked, viz., how to save this heat without wasting the lubricant or making it inoperative; and in the solution of this problem, I look for the most important improvement to be expected in the engine. The most obvious contrivance would be some sort of intercepting shield, which would save the walls of the cylinder and the rings of the piston from the heat of the ignited gases. I have just learned that something of the kind is under trial. Another solution may possibly be found in the employment of a fluid piston; but here we are placed in a dilemma between the liquids that are decomposed and the metals that are oxidized at high temperatures. Next, the loss by radiation—15 per cent.—seems large; but this is to be attributed to the fact that the inside surface of the cylinder is at each inward stroke exposed to the atmosphere—an influence which contributes to the cooling necessary for lubrication. The remaining 15 per cent., which is carried away by the exhaust, is small compared with the proportion passing away with the exhaust steam of a high-pressure or the water of a condensing engine. As the water in the jacket can be safely raised to 212° Fahr., the whole of the jacket heat can be utilized where hot water is required for other purposes; and this, with the exhaust gases, has been used for drying and heating purposes.
With such advantages, it may be asked: Why does not the gas-engine everywhere supersede the steam-engine? My answer is a simple one: The gas we manufacture is a dear fuel compared with coal. Ordinary coal gas measures 30 cubic feet to the pound; and 1,000 cubic feet, therefore, weigh 33 lb. Taking the price at 2s. 9d. per 1,000 cubic feet, it costs 1d. per lb. The 30 cubic feet at 630θ give 19,000θ all available heat. Although good coal may yield 14,000 units by its combustion, only about 11,000 of these reach the boiler; so that the ratio of the useful heat is 11/19. The thermal efficiency of the best non-condensing engine to that of the gas-engine is in the ratio 4/22. Multiplying together these two ratios, we get (11 / 19)×(4 / 22½) = 44 / 4.28. That is, speaking roughly, 1 lb. of gas gives about ten times as much power as 1 lb. of coal does in a good non-condensing engine. But at 18s. 8d. a ton we get 10 lb. of coal for 1d.; so that with these figures the cheapness of the coal would just compensate for the efficiency of the gas. As to the waste heat passing away from the engine being utilized, here the gas-engine has no advantage; and, so far as this is concerned, the gas is about eight times dearer than coal. The prices of gas and coal vary so much in different places that it is hard to determine in what cases gas or coal will be the dearer fuel, considering this point alone.
But there are other kinds of non-illuminating gases—such as Wilson's, Strong's, and Dowson's—which are now coming into use; and at Messrs. Crossley's works you will have an opportunity of seeing a large engineering factory employing several hundred mechanics, and without a chimney, in which every shaft and tool is driven by gas-engines supplied by Dowson's gas, and in which the consumption of coal is only 1.2 lb. per indicated horse power. The greatest economy ever claimed for the steam-engine was a consumption of 1.6 lb.; and this with steam of very high pressure, expanded in three cylinders successively. Thus in a quarter of a century the gas-engine has beaten in the race the steam-engine; although from Watt's first idea of improvement, nearly a century and a quarter have elapsed.
As regards the steam-engine, it is the opinion of competent authorities that the limits of temperature between which it works are so restricted, and so much of the heat is expended in producing a change of state from liquid to vapor, that little further improvement can be made. With respect to gas-engines, the limits of temperature are much further apart. A change of state is not required, and so very great improvement may still be looked for. It is not impossible even that some of the younger members of our body may live to see that period foretold by one of the greatest of our civil engineers—that happy time when boiler explosions will only be matters of history; that period, not a millennium removed by a thousand years, but an era deferred perhaps by only half a dozen decades, when the use of the gas-engine will be universal, and "a steam-engine can be found only in a cabinet of antiquities."
The President said this was a very delightful paper; and nothing could be finer than Mr. Lane's description of the conversion of heat into power, and the gradual growth of theory into practical work.
Mr. W. Foulis (Glasgow) agreed that it was admirable; but it required to be read to be thoroughly appreciated. When members were able to read it, they would find Mr. Lane had given a very clear description of the elementary principles of thermo-dynamics in their relation to the gas-engine and the steam-engine. There was very little in the paper to raise discussion; but Mr. Lane had made exceedingly clear how the present loss in a gas-engine was occasioned, and had also shown how, in the future development of the engine, the loss might be saved, and the engine rendered more efficient.
Mr. H.P. Holt (of Messrs. Crossley Bros., Limited) said he could indorse everything Mr. Lane had said. He had found the paper most interesting and instructive even to himself, though he had some little practical experience of gas-engines, and was supposed to know a little about them. He did not pretend to be able to teach other people; but if he could say anything as to indicator cards, or answer any questions, he should be happy to do so. (He then described the indicator diagram of the atmospheric gas-engine.) In this engine the proportion of the charging stroke to the whole sweep of the piston was about 10 per cent.; and as the charge drawn in consisted of about 10 per cent. of gas, about 1-100 of the total sweep of the piston was composed of the gas.
Mr. Foulis asked what proportion the power indicated on the diagram bore to the power indicated on the brake in the atmospheric engine.
Mr. Holt said unfortunately he had not any figures with him which would give this information; and it was so long since he had anything practically to do with this form of engine, that he should not like to speak from memory. He might add that the largest size of gas-engine made (of about 100 horse power indicated) was at work at Messrs. Edwin Butterworth and Co.'s, of Manchester. It was now driven by ordinary coal gas; but Dowson plant was to be put up very shortly in order to reduce the cost of working, which, though not excessive, would be still more economical with the Dowson gas—probably only about 30s. per week. The present cost was about £4 per week, though it was not working always at full power.
Mr. T. Holgate (Batley) said he thought it was generally understood, by those who had studied the subject, that the adoption of compression of the gaseous mixture before ignition had, so far, more than anything else, contributed to the improved working of gas-engines. This fact had not been sufficiently brought out in the paper, although Mr. Lane had clearly indicated some of the directions in which further improvements were likely to obtain. Gas engineers were largely indebted to Mr. Dugald Clerk for the statement he had made of the theory of the gas-engine.4 Mr. Lane had given some figures, arrived at by Messrs. Brooks and Steward, from experiments made in America; but, prior to these Mr. Clerk had given others which were in the main in accordance with them. Professor Kennedy had also made experiments, the results of which agreed with them.5 The extent of the loss by the cooling water was thus well ascertained; and it was no doubt by a reduction of this loss that further improvement in the working of gas-engines would eventually be obtained.
Mr. J. Paterson (Warrington) expressed his appreciation of the paper, as one of exceptional interest and value. He said he did not rise with a view to make any observations thereon. The analysis of first principles required more matured consideration and thought than could be given to it here. The opinion, however, he had formed of the paper placed it beyond the reach of criticism. It was now many years since his attention had been drawn to the name of Denny Lane; and everything that had come from his facile pen conveyed sound scientific conclusions. The paper to which they had just listened was no exception. It was invested with great interest, and would be regarded as a valuable contribution to the Transactions of the Institute.
Mr. Lane, in reply, thanked the members for the kind expressions used with respect to his paper. His object in writing it was that any one who had not paid any attention to the subject before should be able to understand thoroughly the principles on which gas and hot-air engines operated; and he believed any one who read it with moderate care would perfectly understand all the essential conditions of the gas-engine. He might mention that not long after the thermo-dynamic theory was so far developed as to determine the amount of heat converted into power, a very eminent French Engineer—M. Hirn—conducted some experiments on steam-engines at a large factory, and thought he could account for the whole heat of combustion in the condensed water and the heat which passed away; so much so that he actually doubted altogether the theory of thermo-dynamics. However, being open to conviction, he made further experiments, and discovered that he had been in error, and ultimately became one of the most energetic supporters of the theory. This showed how necessary it was to be careful before arriving at a conclusion on such a subject. He had endeavored, as far as the nature of the case allowed, to avoid any scientific abstractions, because he knew that when practical men came to theory—x's and y's, differentials, integrals, and other mathematical formulæ—they were apt to be terrified.
The President said it was like coming down to every day life to say that it was important that gas managers should be familiar with the appliances used in the consumption of gas, and should be able, when called upon, to give an intelligent description of their method of working. A study of Mr. Lane's paper would reveal many matters of interest with regard to this wonderful motor, which was coming daily more and more into use, not only to the advantage of gas manufacturers, but of those who employed them.
A paper read before the Gas Institute, Manchester, June, 1885.
See Journal, vol. xxxv, pp. 91, 133.
Ibid., vol. xliii., pp. 703, 744.
See Journal, vol. xxxix., p. 648.
Ibid., vol. xl., p. 955.
At the recent Congress of the Societe Technique de l'Industrie du Gaz en France, M. Meizel, Chief Engineer of the St. Etienne Gas Works, described a new exhauster devised by him on the reciprocating principle, and for which he claims certain advantages over the appliances now in general use. Exhausters constructed on the above-named principle have hitherto, M. Meizel says, been costly to fit up, owing to the necessity for providing machinery and special mechanism for the transmission of motion. This has prevented the employment of cylinders of large dimensions; and, consequently, when the quantity of gas to be dealt with has been considerable, the number of exhausters has had to be increased. The result of this has been inconvenience, which has led to a preference being shown for other kinds of exhausters, notwithstanding the manifest advantages which, in M. Meizel's opinion, those of the reciprocating type possess. The improvement which he has effected in these appliances consists in the application to them of cylinders working automatically; and the general features of the arrangement are shown in the accompanying illustrations.
The principal advantages to be gained by the use of this exhauster are stated by M. Meizel to be the following: Considerably less motive force is necessary than is the case with other exhausters, which require steam engines and all the auxiliary mechanism for the transmission of power. By its quiet and regular action, it prevents oscillation and unsteadiness in the flow of gas in the hydraulic main, as well as in the pipes leading therefrom—a defect which has been found to exist with other exhausters. The bells, being of large area, serve the purpose of a condenser; and as, owing to its density, the tar falls to the bottom of the lower vessels, which are filled with water, contact between the gas and tar is avoided. Although the appliance is of substantial construction, its action is so sensitive that it readily adapts itself to the requirements of production. It may be placed in the open air; and therefore its establishment is attended with less outlay than is the case with other exhausters, which have to be placed under cover, and provided with driving machinery and, of course, a supply of steam.
The total superficial area of the exhauster above described, including the governor, is 150 square feet; and its capacity per 24 hours is 230,000 cubic feet. It works silently, with an almost entire absence of friction; and consequently there are few parts which require lubrication. Exhausters of this type (which, M. Meizel says, could be made available for ventilation purposes, in case of necessity) may be constructed of all sizes, from 500 cubic feet per hour upward.
When, at an elevated point in a meadow, there exists a spring or vein of water that cannot be utilized at a distance, either because the supply is not sufficient, or because of the permeability of the soil, it becomes very advantageous to accumulate the water in a reservoir, which may be emptied from time to time through an aperture large enough to allow the water to flow in abundance over all parts of the field.
The storing up of the water permits of irrigating a much greater area of land, and has the advantage of allowing the watering to be effected intermittingly, this being better than if it were done continuously. But this mode of irrigating requires assiduous attention. It is necessary, in fact, when the reservoir is full, to go and raise the plug, wait till the water has flowed out, and then put in the plug again as accurately as possible—a thing that it is not always easy to do. The work is a continuous piece of drudgery, and takes just as much the longer to do in proportion as the reservoir is more distant from one's dwelling. In order to do away with this inconvenience, Mr. Giral, of Langogne (Lozere), has invented a sort of movable siphon that primes itself automatically, however small be the spring that feeds the reservoir in which it is placed. The apparatus (see figure) consists of an elbowed pipe, C A B D E, of galvanized iron, whose extremity, C, communicates with the outlet, R, where it is fixed by means of a piece of rubber of peculiar form that allows the other extremity, B D E, to revolve around the axis, K, while at the same time keeping the outlet pipe hermetically closed. This rubber, whose lower extremity is bent back like the bell of a trumpet, forms a washer against which there is applied a galvanized iron ring that is fixed to the mouth of the outlet pipe by means of six small screws. This ring is provided with two studs which engage with two flexible thimbles, K and L, that are affixed to the siphon by four rivets. These studs and thimbles, as well as the screws, are likewise galvanized. Between the branches, A B D E, of the pipe there is soldered a sheet of galvanized iron, which forms isolatedly a receptacle or air-chamber, F, that contains at its upper part a small aperture, b, that remains always open, and, at its lower part, a copper screw-plug, d, and a galvanized hook, H.
In the interior of this chamber there is arranged a small leaden siphon, a b c, whose longer leg, a, passes through the bottom, where it is soldered, and whose shorter one, c, ends in close proximity to the bottom. Finally, a galvanized iron chain, G H, fixed at G to the bottom of the reservoir, and provided with a weight, P, of galvanized iron, is hooked at H to the siphon and allows it to rise more or less, according as it is given a greater or less length.
From what precedes, it will be seen that the outlet is entirely closed, so that, in order that the water may escape, it must pass into the pipe in the direction, E D B A C.
This granted, let us see how the apparatus works: In measure as the water rises in the reservoir, the siphon gradually loses weight, and its extremity, B D H, is finally lifted by the thrust, so that the entire affair revolves upon the studs, K, until the chain becomes taut. The apparatus then ceases to rise; but the water, ever continuing to rise, finally reaches the apex, b, of the smaller siphon, and, through it, enters the air chamber and fills it. The equilibrium being thus broken, the siphon descends to the bottom, becomes primed, and empties the reservoir. When the level of the water, in descending, is at the height of the small siphon, a b c, this latter, which is also primed, empties the chamber, F, in turn, so that, at the moment the large siphon loses its priming, the entire apparatus is in the same state that it was at first.
In short, when the water enters the reservoir, the siphon, movable upon its base, rises to the height at which it is desired that the flow shall take place. Being arrested at this point by the chain, it becomes primed, and sinks, and the water escapes. When the water is exhausted, the siphon rises anew in order to again sink; and this goes on as long as the period of irrigation lasts.
This apparatus, which is simple in its operation, and not very costly, is being employed with success for irrigating several meadows in the upper basin of the Allier.—Le Genie Civil.
Lead oxide melted or incompletely vitrified is still in common use in the manufacture of inferior earthenware, and sometimes leads to serious results. To detect lead in a glaze, M. Herbelin moistens a slip of white linen or cotton, free from starch, with nitric acid at 10 per cent. and rubs it for ten to fifteen seconds on the side of the utensil under examination, and then deposits a drop of a solution of potassium iodide, at 5 per cent. on the part which has been in contact. A lead glaze simply fused gives a very highly colored yellow spot of potassium iodide; a lead glaze incompletely vitrified gives spots the more decided, the less perfect the vitrification; and a glaze of good quality gives no sensible color at all.—M. Herbelin.
The application of electricity to metallurgical processes has hitherto been confined to the reduction of metals from solutions, and few attempts have been made to effect dry reductions by means of an electric current. Sir W. Siemens attempted to utilize the intense heat of an electric arc for this purpose, but accomplished little beyond fusing several pounds of steel. A short time since, Eugene H. Cowles and Alfred H. Cowles of Cleveland conceived the idea of obtaining a continuous high temperature on an extended scale by introducing into the path of an electric current some material that would afford the requisite resistance, thereby producing a corresponding increase in the temperature. After numerous experiments that need not be described in detail, coarsely pulverized carbon was selected as the best means for maintaining a variable resistance and at the same time as the most available substance for the reduction of oxides. When this material, mixed with the oxide to be reduced, was made a part of the electric circuit in a fire clay retort, and submitted to the action of a current from a powerful dynamo machine, not only was the oxide reduced, but the temperature increased to such an extent that the whole interior of the retort fused completely. In other experiments lumps of lime, sand, and corundum were fused, with indications of a reduction of the corresponding metal; on cooling, the lime formed large, well-defined crystals, the corundum beautiful red, green, and blue hexagonal crystals.
Following up these results with the assistance of Charles F. Mabery, professor of chemistry in the Case School of Applied Science, who became interested at this stage of the experiments, it was soon found that the intense heat thus produced could be utilized for the reduction of oxides in large quantities, and experiments were next tried on a large scale with a current from two dynamos driven by an equivalent of fifty horse power. For the protection of the walls of the furnace, which were made of fire brick, a mixture of the ore and coarsely pulverized gas carbon was made a central core, and it was surrounded on the sides and bottom by fine charcoal, the current following the lesser resistance of the central core from carbon electrodes which were inserted at the ends of the furnace in contact with the core. In order to protect the machines from the variable resistance within the furnace, a resistance box consisting of a coil of German silver wire placed in a large tank of water was introduced into the main circuit, and a Brush ammeter was also attached by means of a shunt circuit, to indicate the quantity of current that was being absorbed in the furnace. The latter was charged by first filling it with charcoal, making a trough in the center, and filling this central space with the ore mixture, which was covered with a layer of coarse charcoal. The furnace was closed at the top with fire brick slabs containing two or three holes for the escape of the gaseous products of the reduction, and the entire furnace made air-tight by luting with fire clay. Within a few minutes after starting the dynamo, a stream of carbonic oxide issued through the openings, burning usually with a flame eighteen inches in height. The time required for complete reduction was ordinarily about an hour.
The furnace at present in use is charged in substantially the same manner, and the current is supplied by a Brush machine of variable electromotive force driven by an equivalent of forty horse power. A Brush machine capable of utilizing 125 horse power, or two and one-half times as large as any hitherto constructed by the Brush Electric Company, is being made for the Cowles Electric Smelting and Aluminum Company, and this machine will soon be in operation. Experiments already made so that aluminum, silicon, boron, manganese, magnesium, sodium and potassium can be reduced from their oxides with ease. In fact, there is no oxide that can withstand temperatures attainable in this electrical furnace. Charcoal is changed to graphite. Does this indicate fusion or solution of carbon? As to what can be accomplished by converting enormous electrical energy into heat within a limited space, it can only be said that it opens the way into an extensive field for both pure and applied chemistry. It is not difficult to conceive of temperatures limited only by the capability of carbon to resist fusion. The results to be obtained with the large Brush machine above mentioned will be of some importance in this direction.
Since the cost of the motive power is the chief expense in accomplishing reductions by this method, its commercial success is closely connected with the cheapest form of power to be obtained. Realizing the importance of this point, the Cowles Electric Smelting and Aluminum Company has purchased an extensive and reliable water power, and works are soon to be erected for the utilization of 1,200 horse power. An important feature in the use of these furnaces, from a commercial standpoint, is the slight technical skill required in their manipulation. The four furnaces in operation in the experimental laboratory at Cleveland are in charge of two young men twenty years of age, who, six months ago, knew absolutely nothing of electricity. The products at present manufactured are the various grades of aluminum bronze made from a rich furnace product that is obtained by adding copper to the charge of ore, silicon bronze prepared in the same manner, and aluminum silver, an alloy of aluminum with several other metals. A boron bronze may be prepared by the reduction of boracic acid in contact with copper.
As commercial results may be mentioned the production in the experimental laboratory, which averages fifty pounds of 10 per cent. aluminum bronze daily, and it can be supplied to the trade in large quantities at prices based on $5 per pound for the aluminum contained, the lowest market quotation of this metal being at present $15 per pound. Silicon bronze can be furnished at prices far below those of the French manufacturers.
The alloys which the metals obtained by the methods above described form with copper have been made the subject of careful study. An alloy containing 10 per cent. of aluminum and 90 per cent. of copper forms the so-called aluminum bronze with a fine golden color, which it retains for a long time. The tensile strength of this alloy is usually given as 100,000 pounds to the square inch; but castings of our ten per cent. bronze have stood a strain of 109,000 pounds. It is a very hard, tough alloy, with a capacity to withstand wear far in excess of any other alloy in use. All grades of aluminum bronze make fine castings, taking very exact impressions, and there is no loss in remelting, as in the case of alloys containing zinc. The 5 per cent. aluminum alloy is a close approximation in color to 18 carat gold, and does not tarnish readily. Its tensile strength in the form of castings is equivalent to a strain of 68,000 pounds to the square inch. An alloy containing 2 or 3 per cent. aluminum is stronger than brass, possesses greater permanency of color, and would make an excellent substitute for that metal. When the percentage of aluminum reaches 13, an exceedingly hard, brittle alloy of a reddish color is obtained, and higher percentages increase the brittleness, and the color becomes grayish-black. Above 25 per cent. the strength again increases.
The effect of silicon in small proportions upon copper is to greatly increase its tensile strength. When more than 5 per cent. is present, the product is exceedingly brittle and grayish-black in color. It is probable that silicon acts to a certain extent as a fluxing material upon the oxides present in the copper, thereby making the metal more homogeneous. On account of its superior strength and high conductivity for electrical currents, silicon bronze is the best material known for telegraph and telephone wire.
The element boron seems to have almost as marked an effect upon copper as carbon does upon iron. A small percentage in copper increases its strength to 50,000 or 60,000 pounds per square inch without diminishing to any large extent its conductivity.
Aluminum increases very considerably the strength of all metals with which it is alloyed. An alloy of copper and nickel containing a small percentage of aluminum, called Hercules metal, withstood a strain of 105,000 pounds, and broke without elongation. Another grade of this metal broke under a strain of 111,000 pounds, with an elongation equivalent to 33 per cent. It must be remembered that these tests were all made upon castings of the alloys. The strength of common brass is doubled by the addition of 2 or 3 per cent. of aluminum. Alloys of aluminum and iron are obtained without difficulty; one product was analyzed, containing 40 per cent. of aluminum. In the furnace iron does not seem to be absorbed readily by the reduced aluminum when copper is present; but in one experiment a mixture composed of old files, 60 per cent.; nickel, 5 per cent.; and of 10 per cent. aluminum bronze 35 per cent., was melted together, and it gave a malleable product that stood a strain of 69,000 pounds.
When the reduction of aluminic oxide by carbon is conducted without the addition of copper, a brittle product is obtained that behaves in many respects like pig iron as it comes from the blast furnace. The same product is formed in considerable quantities, even when copper is present, and frequently the copper alloy is found embedded in it. Graphite is always found associated with it, even when charcoal is the reducing material, and analysis invariably shows a very high percentage of metallic aluminum. This extremely interesting substance is at present under examination.
Read at the recent meeting of the American Association, Ann Arbor, Mich.
The use of electricity in the reduction of metals from their ores is extending so rapidly, and the methods of its generation and application have been so greatly improved within a few years, that the possibility of its becoming the chief agent in the metallurgy of the future may now be admitted, even in cases where the present cost of treatment is too high to be commercially advantageous.
The refining of copper and the separation of copper, gold, and silver by electrolysis have thus far attracted the greatest amount of attention, but a commercial success has also been achieved in the dry reduction by electricity of some of the more valuable metals by the Cowles Electric Smelting and Aluminum Company, of Cleveland, Ohio. Both this method of manufacture and the qualities of the products are so interesting and important that it is with pleasure we call attention to them as steps toward that large and cheap production of aluminum that the abundance of its ores and the importance of its physical properties have for several years made the unattained goal of many skillful metallurgists.
The Messrs. Cowles have succeeded in greatly reducing the market value of aluminum and its alloys, and thereby vastly extending its uses, and they are now by far the largest producers in the world of these important products. As described in their patents, the Cowles process consists essentially in the use for metallurgical purposes of a body of granular material of high resistance or low conductivity interposed within the circuit in such a manner as to form a continuous and unbroken part of the same, which granular body, by reason of its resistance, is made incandescent, and generates all the heat required. The ore or light material to be reduced—as, for example, the hydrated oxide of aluminum, alum, chloride of sodium, oxide of calcium, or sulphate of strontium—is usually mixed with the body of granular resistance material, and is thus brought directly in contact with the heat at the points of generation, at the same time the heat is distributed through the mass of granular material, being generated by the resistance of all the granules, and is not localized at one point or along a single line. The material best adapted for this purpose is electric light carbon, as it possesses the necessary amount of electrical resistance, and is capable of enduring any known degree of heat when protected from oxygen without disintegrating or fusing; but crystalline silicon or other equivalent of carbon can be employed for the same purpose. This is pulverized or granulated, the degree of granulation depending upon the size of the furnace. Coarse granulated carbon works better than finely pulverized carbon, and gives more even results. The electrical energy is more evenly distributed, and the current can not so readily form a path of highest temperature, and consequently of least resistance through the mass along which the entire current or the bulk of the current can pass. The operation must necessarily be conducted within an air-tight chamber or in a non-oxidizing atmosphere, as otherwise the carbon will be consumed and act as fuel. The carbon acts as a deoxidizing agent for the ore or metalliferous material treated, and to this extent it is consumed, but otherwise than from this cause, it remains unimpaired.
Fig. I. of the accompanying drawings is a vertical longitudinal section through a retort designed for the reduction of zinc ore, according to this process, and Fig. II. is a front elevation of the same. Fig. III. is a perspective view of a furnace adapted to withstand a very high temperature, and Figs. IV. and V. are respectively longitudinal and transverse sections of the same.
This retort consists of a cylinder, A, made of silica or other non-conducting material, suitably embedded in a body, B, of powdered charcoal, mineral wool, or of some other material which is not a good conductor of heat. The rear end of the retort-cylinder is closed by means of a carbon plate, C, which plate forms the positive electrode, and with this plate the positive wire of the electric circuit is connected. The outer end of the retort is closed by means of an inverted graphite crucible, D, to which the negative wire of the electric circuit is attached. The graphite crucible serves as a plug for closing the end of the retort. It also forms a condensing chamber for the zinc fumes, and it also constitutes the negative electrode. The term "electrode" is used in this case as designating the terminals of the circuit proper, or that portion of it which acts simply as an electrical conductor, and not with the intention of indicating the ends of a line between which there is no circuit connection. The circuit between the "electrodes," so called, is continuous, being established by means of and through the body of broken carbon contained in the retort, A. There is no deposit made on either plate of the decomposed constituents of the material reduced. The mouth of the crucible is closed with a luting of clay, or otherwise, and the opening, d, made in the upper side of the crucible, near its extremity, comes entirely within the retort, and forms a passage for the zinc fumes from the retort chamber into the condensing chamber. The pipe, E, serves as a vent for the condensing chamber. The zinc ore is mixed with pulverized or granular carbon, and the retort charged nearly full through the front end with the mixture, the plug, D, being removed for this purpose.
A small space is left at the top, as shown. After the plug has been inserted and the joint properly luted, the electric circuit is closed and the current allowed to pass through the retort, traversing its entire length through the body of mixed ore and carbon. The carbon constituents of the mass become incandescent, generating a very high degree of heat, and being in direct contact with the ore, the latter is rapidly and effectually reduced and distilled. The heat evolved reduces the ore and distills the zinc, and the zinc fumes are condensed in the condensing chamber, precisely as in the present method of zinc making, with this important exception that, aside from the reaction produced by heating carbon in the presence of zinc oxide, the electric current, in passing through the zinc oxide, has a decomposing and disintegrating action upon it, not unlike the effect produced by an electric current in a solution. This action accelerates the reduction, and promotes economy in the process.
Another form of furnace is illustrated by Fig. III., which is a perspective view of a furnace adapted for the reduction of ores and salts of non-volatile metals and similar chemical compounds. Figs. IV. and V. are longitudinal and transverse sections, respectively, through the same, illustrating the manner of packing and charging the furnace.
The walls and floorsL L', of the furnace are made of fire bricks, and do not necessarily have to be very thick or strong, the heat to which they are subjected not being excessive. The carbon plates are smaller than the cross section of the box, as shown, and the spaces between them and the end walls are packed with fine charcoal.
The furnace is covered with a removable slab of fireclay, N, which is provided with one or more vents, n, for the escaping gases.
The space between the carbon plates constitutes the working part of the furnace. This is lined on the bottom and sides with a packing of fine charcoal, O, or such other material as is both a poor conductor of heat and electricity—as, for example, in some cases, silica or pulverized corundum or well-burned lime—and the charge, P, of ore and broken, granular, or pulverized carbon occupies the center of the box, extending between the carbon plates. A layer of granular charcoal, O', also covers the charge on top. The protection afforded by the charcoal jacket, as regards the heat, is so complete, that with the covering-slab removed, the hand can be held within a few inches of the exposed charcoal jacket; but with the top covering of charcoal also removed and the core exposed, the hand cannot be held within several feet. The charcoal packing behind the carbon plates is required to confine the heat and to protect them from combustion.
With this furnace, aluminum can be reduced directly from its ores; and chemical compounds from corundum, cryolite, clay, etc., and silicon, boron, calcium, manganese, magnesium, and other metals are in like manner obtained from their ores and compounds. The reduction of ores according to this process can be practiced, if circumstances require it, without any built furnace.
At present, the Cowles company is engaged mostly in the producing of aluminum bronze and aluminum silver and silicon bronze. The plant, were it run to its full capacity, is capable of turning out eighty pounds of aluminum bronze, containing 10 per cent. of aluminum daily; or, were it to run upon silicon bronze, could turn out one hundred and twenty pounds of that per day, or, we believe, more aluminum bronze daily than can be produced by all other plants in the world combined. This production, however, is but that of the experimental laboratory, and arrangements are making to turn out a ton of bronze daily, and the works have an ultimate capacity of from eight to ten thousand horse power. The energy consumed by the reduction of the ore is almost entirely electrical, only enough carbon being used to unite with the oxygen of the ore to carry it out of the furnace in the form of the carbon monoxide, the aluminum remaining behind. Consequently, the plant necessary to produce aluminum on a large scale involves a large number of the most powerful dynamos. These are to be driven by water-power or natural gas and marine engines of great capacity.
The retail price of standard 10 per cent. aluminum bronze is $1 per pound avoirdupois, which means less than $9 per pound for aluminum, the lowest price at which it has ever been sold, yet the Cowles company has laid a proposition before the Government to furnish this same bronze in large quantities at very much lower prices than this. The Hercules alloy, castings of which will stand over 100,000 pounds to the square inch tensile strain, sells at 75 c. a pound, and is also offered the Government or other large consumers at a heavy discount. The alloys are guaranteed to contain exactly what is advertised; they are standardized into 10 per cent., 7.5 per cent., 5 per cent. and 2.5 per cent. aluminum bronze before shipment.
The current available at the Cowles company's works was, until recently, 330 amperes, driven by an electromotive force of 110 volts and supplied by two Edison dynamos; but the company has now added a large Brush machine that has a current of 560 amperes and 52 volts electromotive force. We shall, on another occasion, give some particulars of experiments in the reduction of refractory ores by the process.—Eng. and Mining Jour.
Optical telegraphy, by reason of its very principle, presents both the advantage and inconvenience of leaving no automatic trace of the correspondence that it transmits. The advantage is very evident in cases in which an optical station falls into the hands of the enemy; on the other hand, the inconvenience is shown in cases where a badly transmitted or badly collated telegram allows an ambiguity to stand subject to dispute. Moreover, in case of warfare between civilized nations that have all the resources of science at their disposal, there may be reason to fear lest one of the enemy's optical stations substitute itself for the corresponding station, and take advantage of the situation to throw confusion into the orders transmitted. The remedy for this appears to reside in the use of cryptography and in the exchange, at various intervals, of certain words that have been agreed upon beforehand, and that the enemy is ignorant of.
As for the automatic preservation of telegrams, the problem has not been satisfactorily solved. It has been proposed to connect the key of the manipulator of the optical apparatus with the manipulator of an ordinary Morse apparatus, thus permitting the telegram to be preserved upon a band of paper. It is unnecessary to say that the space occupied by a dispatch thus transmitted would be considerable; but this is not what has stopped innovators. The principal objection resides in the increase in muscular work imposed by this arrangement upon the telegrapher. Obliged to keep his eye fixed intently at the receiving telescope, while at the same time maneuvering the manipulator and spelling aloud the words that he is receiving, the operator should have a very sensitive manipulator at his disposal, and not be submitted to mental or physical overtaxation. So the apparatus that have been devised have not met with much success.
Two French officers, working independently, have hit upon the same idea of receiving the indications transmitted by the vibration of the luminous fascicle directly upon their travel. The method consists in the use of that peculiar property of selenium of becoming a good conductor under the action of a luminous ray, while in darkness it totally prevents the passage of the electric current. Such modification of the physical properties of selenium, moreover, occurs without the perceptible development of any mechanical work. If, then, in the line of travel of the luminous fascicle emitted by the optical apparatus, or in a portion of such fascicle, we interpose a fragment of selenium connected with the two poles of a local pile, it is easy to see that the current from the latter will be opened or closed according as the luminous ray from the apparatus will or will not strike the selenium, and that the length of time during which the current passes will depend upon the length of the luminous attacks. A Morse apparatus interposed in this annexed circuit will therefore give an automatic inscription of the correspondence exchanged. Such is the principle. But, practically, very great difficulties present themselves, these being connected with the rapid weakening of the electric properties of the selenium, and with the necessity of having recourse to infinitely small mechanical actions only. The problem is nevertheless before us, and it is to be hoped that the perseverance of the scientists who are at work upon it will some day succeed in solving it.
Finally, we may call attention to the attempts made to receive the luminous impression upon a band prepared with gelatino-bromide of silver. In practice this band would unwind uniformly at the focus of the receiving telescope, which would be placed in a box, forming a camera obscura. The velocity of this band prepared for photographing the signals would be regulated by clockwork. The experiments that have been made have not given results that are absolutely satisfactory, by reason of the length of the signals received and the mechanical complication of the device.

The projectors employed for lighting to a distance the surroundings of a stronghold or of a ship have likewise been applied in optical telegraphy. For this purpose Messrs. Sautter, Lemonnier & Co. have added to their usual projecting apparatus some peculiar arrangements that permit of occultations of the luminous focus at proper intervals. Figs. 21 and 22 show the arrangement of the apparatus, the principle of which is as follows: When the axis of the projector points toward the clouds, and in the direction occupied by a corresponding station, the occultations of the luminous source placed in the focus of the apparatus produce upon the clouds, which act as a screen, an alternate series of flashes and extinctions. It is therefore possible with this arrangement, and by the use of the Morse alphabet, to establish an optical communication at a distance. The use of this projector (the principal inconvenience of which is that it requires a clouded sky) even permits two observers who are hidden from each other by the nature of the ground to easily communicate at a distance of 36 or 48 miles.
Figs. 21 and 22.—FRONT VIEW AND LONGITUDINAL SECTION OF THE MANGIN PROJECTOR. (Scale 1/15). A. Elliptical mirror. B. Arm of the same. C. Nut for fixing the mirror. D. Support of the mirror. E. Occultator. F. Support for same. G. Lever for maneuvering the occultator. I. Support of the occultator rod. J. Screw for fixing the mirror support. K. Screw for fixing the support of the occultator rod. L. Screw for fixing the occultator support.
The apparatus shown in Figs. 21 and 22 permits of signaling in three ways:
1. Upon the Clouds.—In this case the mirror, A, is removed, and the projector inclined above the horizon in such a way as to illuminate the clouds to as great a distance as possible. A maneuver of the occultator, E, between the lamp and the mirror arrests the luminous rays of the source, or allows them to pass, and thus produces upon the clouds the dots and dashes of the conventional alphabet.
2. Isolated Communication by Luminous Fascicles.—When it is desired to correspond to a short distance of 2 or 3 miles, and establish a communication between two isolated posts, the mirror, A, is put in place upon its support, B. The luminous fascicle emanating from the source reflected by the mirror is thrown vertically. By revolving the mirror 90° around its horizontal axis the fascicle becomes horizontal, and may thus be thrown in a given direction at unequal intervals and during irregular times, and furnish conventional signs.
3. Night Communication upon the Entire Horizon.—When we wish to correspond at a short distance, say two miles, and make signals visible from the entire horizon, the mirror, A, is put in place, so that it shall reflect the luminous fascicle vertically. The fascicle, at a distance of about fifty feet, meets a white balloon which it renders visible from every point in the horizon. The maneuver of the occultator brings the balloon out of darkness or plunges it thereinto again, and thus produces the signs necessary for aerial communication.

These ingenious arrangements, which depend upon the state of the atmosphere, do not appear to have been imitated outside of the navy.
The system of optical communication proposed by Capt. Gaumet, and which he names the Telelogue, is based upon the visibility of colored or luminous objects, and upon the possibility of piercing the opaque curtain formed by the atmosphere between the observer's eye and a signal, by utilizing the difference in brightness that exists between such objects and the atmosphere. It is a question, then, of giving such difference in intensity its maximum of brightness. To do this, Capt. Gaumet proposes to employ silvered signals upon a black background. He uses the simple letters of the alphabet, but changes their value. His apparatus has the form of a large album glued at the back to a sloping desk. Each silvered letter, glued to a piece of black cloth, is seen in relief upon the open register. A sort of index along the side, as in commercial blank-books, permits of quickly finding any letter at will. Such is the manipulator of the apparatus.
The receiver consists of a spy-glass affixed to the board that carries the register. For a range of two and a half miles, the complete apparatus, with a 12×16 inch manipulator and telescope, weighs but four and a half pounds. For double this range, with a 20×28 inch manipulator and telescope, the total weight is thirteen pounds. The larger apparatus, according to the inventor, have a range of seven miles.
For night work the manipulator is lighted either by one lamp, or by two lamps with reflector, placed laterally against it.
This apparatus, although well known, and having been publicly experimented with, has not, to our knowledge, been applied practically. From a military standpoint, its short range will evidently not permit it to compete with optical telegraphic apparatus, properly so called. Perhaps it might rather be of service for private communications between localities not very far apart, since it costs but little and is easily operated.
Optical communications by signals, during day and night, with experienced men, may, in the absence of telephones, telegraphs, and messengers, render important service when the distance involved is greater than two thousand feet.
This mode of correspondence is based upon the use of the Morse alphabet. The signals are divided into night and day ones. The day signals are made with small flags. When these are wanting, sheets of white cardboard may be used. The night signals are made with a lantern provided with a support, which may be fixed to a wall or upon a bayonet.
In day signaling, the dashes of the Morse alphabet are formed by means of two flags (Fig. 23) held simultaneously at arm's length by the signaler. The dots are formed with a single flag held in the right hand (Fig. 24). In this way it is possible, by extremely simple combinations, to establish a correspondence, and produce any conventional signal. By means of relay stations, the signals may be transmitted from one to another to a great distance.
In signaling with the lantern, long and short interruptions of the luminous source are produced by means of a screen.
Various interesting experiments have been made with a view to utilizing luminous captive balloons for optical communications. As we have already seen, this maybe effected by using opaque balloons, and throwing upon them at unequal intervals a luminous fascicle by means of a projector. As for using a luminous source placed in the car of a balloon, that cannot be thought of in the present state of aeronautic science; the continual rotation of the balloon around its axis would render the projection and reception of the signals in a given direction impossible.
For communicating optically from ship to ship during the day, the marine uses flags of different forms and colors, and flames. Between ships and the land there are used what are called semaphore signals, which are made by means of a mast provided with three arms and a disk placed at the upper part. The combinations of signs thus obtained, which are analogous in principle to those of the Chappe telegraph, permit of satisfactorily communicating to a distance.
On board ship, hand signals are used like those employed by the army for communicating between bodies of troops. For night communications the marine employs lights corresponding to the day flags, as well as rockets, and luminous rays projected by means of reflectors and intercepted by screens.
In conclusion, it may be said that optical telegraphy, which has only within a few years emerged from the domain of theory to enter that of practice, has taken a remarkable stride in the military art and in science. It is due to its processes that Col. Perrier has in recent years been enabled to carry out certain geodesic work that would have formerly been regarded as impracticable, notably the prolongation of the arc of the meridian between France and Spain. Very recently, an optical communication established between Mauritius and Reunion islands, to a distance of 129 miles, with 24 inch apparatus, proved that, in certain cases, the costly laying of a submarine cable may be replaced by the direct emission of a luminous ray.
Continued from page 8094.
Mr. F. Von Faund-Szyll has devised an original system of submarine telegraph, which is based upon the well known property that selenium exhibits of modifying its resistance under the influence of luminous rays, and which he styles the Selen-Differenzialrecorder.
Contrary to what is found in the other systems hitherto employed, the Faund-Szyll system utilizes the cable current merely for starting the receiving apparatus, which are operated by means of strong local batteries. The result is that the mechanical work that devolves upon the line current, which is, as well known, very weak, is exceedingly reduced.
The system consists of two essential parts: (1) The receiver, properly so called. (2) The relay as well as the registering apparatus or differenzialrecorder. The receiver consists of a closed box, K, in the interior of which there is a very intense source of light whose rays escape by passing through apertures, a a', in the front part (Fig. 1).
As a source of light, there may be conveniently employed an incandescent lamp, g, capable of giving an intense light, and arranged (as shown in Fig. 2) behind the side that contains the slits, a a'.
The starting apparatus consists of a small galvanometric helix, r, analogous to Thomson's siphon recorder, which is suspended from a cocoon fiber and capable of moving in an extremely powerful magnetic field, N_S. This helix carries, as may be seen in Figs. 1, 3 and 4, a prolongation, v, at its lower end whose form is that of a prism, and which is arranged in front of the partition of the box, K, in such a way that it exactly covers the two slits, a and a when the bobbin is at rest, and in this case prevents the luminous rays of the lamp, g, from escaping from the box. But, as soon as the current sent through the cable reaches the spirals of the bobbin, through the conductors, y y', the sum of the elementary electrodynamic actions that arise causes the helix to revolve to the right or left, according to the polarity of the current, while at the same time the helix slightly approaches one or the other of the poles of the magnet. The prolongation, v, of the helix, being firmly united with the latter, follows it in its motion, and has the effect of permitting the luminous rays to escape through one or the other of the slits, a_a', so that the freeing of the luminous fascicle, if such an expression is allowable, is effected.
In order to prevent oscillations, which could not fail to occur after each emission of a current (so that the helix, instead of returning to a position of equilibrium and stopping there, would go beyond it and alternately uncover the slits, a a'), the apparatus is provided with a liquid deadener. To this end, the prolongation, v, carries a piece, o, which dips into a cup containing a mixture of glycerine and water.
We shall now describe the differenzialrecorder. Opposite the two slits, a and a', there are two powerful converging lenses, l and l', whose foci coincide with two sorts of selenium plate rheostat, z and z'. The result of this arrangement is that as soon as one of the slits, as a consequence of the displacement of the helix, r, allows a luminous fascicle to escape, this latter falls upon the corresponding lens, which concentrates it and sends it to the selenium plates just mentioned. Under the influence of the luminous rays, the resistance that the selenium offers to the passage of an electric current instantly changes. At M and M' are placed two horseshoe magnets whose poles are provided with pieces of soft iron that serve as cores to exceedingly fine wire bobbins, d. These polarized pieces are arranged in the shape of a St. Andrew's cross, and in such a way that the poles of the same name occupy the two extremities of the same arm of the cross, an arrangement very clearly shown in Fig. 2.
Between the poles of the magnets, M and M', there is a permanent magnet, A, movable around a vertical axis, i. Four spiral springs, f, whose tension may be regulated, permit of centering this latter piece in such a way that when the current is traversing the spirals of the polar bobbins it is equally distant from the four poles, n, s, s', and n'. Under such circumstances it is evident that a difference in the power of attraction of these four poles, however feeble it be, will result in moving the magnet, A, in one direction or the other around its axis. The energy and extent of such motion may, moreover, be magnified by properly acting upon the four regulating springs.
The bobbins of the magnet, M, are mounted in series with the selenium plates, z, the local battery, B, and a resistance box, W. Those of the magnet, M', are in series with z', B', and W'. The local batteries, B and B', are composed of quite a large number of elements. The current from the battery, B, traverses the selenium plates and the bobbins of the magnet, M, and returns to B through the rheostat, W; and the same occurs with the current from B'. The two currents, then, are absolutely independent of one another.
From this description it is very easy to see how the system works. Let us suppose, in fact, that the current which is traversing the spirals of the helix, r, has a direction such that the helix in its movement approaches the pole, S; then the prolongation, v, will uncover the slit, a, which, along with a', had up to this moment been closed, and a luminous fascicle escaping through a will strike the lens, l', and from thence converge upon the selenium plates, z'. This is all the duty that the line current has to perform.
The luminous rays, in falling upon the selenium plates, z', modify the resistance that these offered to the passage of the current produced by the battery, B'. As this resistance diminishes, the intensity of the current in the circuit supplied by the battery, B', increases, the attractive action of the polar pieces of the magnet, M', diminishes, the equilibrium is destroyed, and the piece, A, revolves around the axis, i. If the polarity of the line current were different, the same succession of phenomena would occur, save that the direction of A's rotation would be contrary. As for the rheostats, W W', their object is to correct variations in the selenium's resistance and to balance the resistances of the two corresponding circuits. The magnet, A, will be combined with a registering apparatus so as to directly or indirectly actuate the printing lever. The entire first part of this apparatus, which is very sensitive, may be easily protected from all external influence by placing it in a box, and, if need be, in a room distant from the one in which the employes work.
The differenzialrecorder alone has to be in the work room.
As may be seen, the system is not wanting in originality. Experience alone will permit of pronouncing upon the question as to whether it is as practical as ingenious.—La Lumiere Electrique.
Messrs. Thomson & Bottomley have recently invented a peculiar circuit cutter based upon the use of a metal whose melting point is exceedingly low. Recourse is had to this process for breaking the current within as short a time as possible. In this new device the ends of the conductors are soldered together with the metal in question at one or several points of the circuit. The metal employed is silver or copper of very great conductivity, seeing that the increase of temperature in a conductor, due to a sudden increase of the current, is inversely proportional to the product of the electric resistance by the specific heat of the conductor; that these metals are best adapted for giving constant and definite results; and that the contacts are better than with lead or the other metals of low melting point which are frequently employed in circuit cutters.

Fig. 1 represents one form of the new device. Here, a is the copper or silver wire, and b is a soldering made with a very fusible metal and securing a continuity of the circuit. Each extremity of the wire, a, is connected with a heavy ring, c, of copper or other good conducting metal. The hook, d, with which the upper ring, c, is in contact, communicates metallically with one of the extremities of the conductor at the place where the latter is interrupted for the insertion of the circuit cutter. The hook, e, with which the lower ring, c, is in contact, tends constantly to descend under the action of a spiral spring, f, which is connected metallically with the other extremity of the principal conductor. The hooks, d and e, are arranged approximately in the same vertical plane, and have a slightly rounded upper and lower surface, designed to prevent the rings, c, of the fusible wire, a, from escaping from the hooks. In Fig. 1 the position of the arm, e, when there is no fusible wire in circuit, is shown by dotted lines. When this arm occupies the position shown by entire lines, it exerts a certain traction upon the soldering, b, and separates the two halves of the wire, a, as soon as the intensity of circulation exceeds its normal value. The mode of putting the wire with fusible soldering into circuit is clearly shown in the engraving.
Fig. 2 shows a different mode of mounting the wire. The wire, q, is soldered in the center, and is bent into the shape of a U, and kept in place by the pieces, r and s. In this way the two ends of it tend constantly to separate from each other. Messrs. Thomson & Bottomley likewise employ weights, simply, for submitting the wire to a constant stress. The apparatus is inclosed in a box provided with a glass cover.—La Lumiere Electrique.
Despite the simplicity of their parts, and the slight value of the materials employed, the existing micro-telephonic apparatus keep at relatively high prices, and the use of them is often rejected, to the benefit of speaking tubes, when the distance between stations is not too great. We propose to describe a new style of apparatus that are in no wise inferior to those in general use, and the price of which is relatively low.
The microphone transmitter may have several forms. The most elementary of these consists of two pieces of carbon, from one to one and a quarter inches in length by one-half inch in width, between which are fixed two nails, about two inches in length, whose extremities, filed to a point, enter small conical apertures in the carbons. Fig. 1 gives an idea of the arrangement.
Fig. 2 represents a model which is a little more complicated, but which gives remarkable results. The largest nail is here two inches in length, and the shortest three-quarter inch.
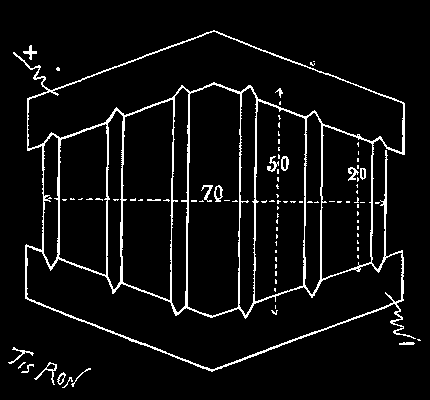
The receivers may be Bell telephones of the simplest form found in the market (Fig. 3); but for these there may be substituted a bar of soft iron, cast iron, or steel, one of the extremities of which is provided with a bobbin upon, which is wound insulated copper wire 0.02 inch in diameter. The apparatus is mounted like an ordinary Bell telephone. A horseshoe electro may also be used, and the poles be made to act (Fig. 4). The current sent by the transmitter suffices to produce a magnetic field in which the variations in intensity produced by the microphone succeed perfectly in reproducing speech and music. With four Leclanche elements, the sounds are perceived very clearly. The elements used may be bichromate of potash ones, those of Lelande and Chaperon, etc.

To this apparatus there may be added a second bobbin of coarser wire into which is passed a current from a local pile. This produces a much intenser magnetic field, and, consequently, louder sounds. This modification, however, is really useful only for long distances.
Any arrangement imaginable may be given the transmitter and receiver; but, aside from the fact that the ones just indicated are the simplest, they give results that are at least equal, if not superior, to all others.
We shall insist here only upon the arrangement of the microphone, which is new (at least in practice), and upon the uselessness of having well magnetized steel bars and wires of extreme fineness in the receiver.
We have stated that the nail microphones are the simplest. The nails may be replaced by copper or any other metal, or they may be well nickelized; but common nails answer very well, and do not oxidize much. An apparatus of this kind (Fig. 5) that has been for more than a year in a laboratory filled with acid vapors is yet working very well. These apparatus possess the further advantage of being very strong, and of undergoing violent shocks without breaking or even getting out of order. They may be used either with or without induction coils. We have not yet measured their range, but can cite the following fact:
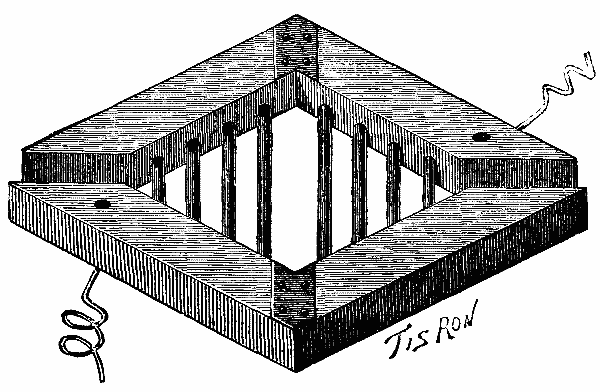
One of these apparatus, quite crudely mounted, was put into a circuit with a resistance of 300 ohms. With a single already exhausted bichromate element, giving scarcely 2 volts, musical sounds and speech reached the receiver without being notably weakened. Such resistance represents a length of eighteen miles of ordinary telegraph wire. After this, 700 ohms were overcome with 3.4 volts. This result was obtained by direct transmission, and without an induction coil, and it is probable that it might be much exceeded without sensibly increasing the electromotive force of the current.—Le Genie Civil.
We give herewith, from the Elektrotechnische Zeitschrift, a few interesting details in regard to the measuring apparatus of Messrs. Kapp and Crompton.
It is evident that when we use permanent magnets or springs as directing forces in measuring instruments, we cannot count upon an absolute constancy in the indications, as the magnetism of the magnetized pieces, or the tension of the springs, modifies in time. The apparatus require to be regulated from time to time, and hence the idea of substituting electro-magnets for permanent ones.
If we suppose (Fig. 1) a magnetized needle, n s, placed between the extremities of a soft iron core, N S, and if we group the circuit in such a way that the current, after traversing the coil, e e, of the electro, traverses a circle, d d, situated in a plane at right angles with the plane of the needle's oscillation, it is evident that we shall have obtained an apparatus that satisfies the aforesaid conditions. It seems at first sight that in such an instrument the directing force should be constant from the moment the electro was saturated, and it would be possible, were sufficiently thin cores used, to obtain a constancy in the directing magnetic field for relatively feeble intensities. In reality, the actions are more complex. The needle, n s, is, in fact, induced to return to its position of equilibrium by two forces, the first of which (the attraction of the poles, N S) rapidly increases with the intensity so as to become quickly and perceptibly constant, while the second (the sum of the elementary electrodynamic actions that are exerted between the spirals, e e, and the needle, n s) increases proportionally to the intensity of the current. If we represent these two sections graphically by referring the magnetic moments as ordinates and the current intensities as abscissas to two co-ordinate axes (Fig. 2), we shall obtain for the first force the curve, O A B, which, starting from A, becomes sensibly parallel with the axis of X, and for the second the right line, O D. The resultant action is represented by the curve, O E E' F. It will be seen that this action, far from being constant, increases quite rapidly with the intensity of the current, so that the deflections would become feebler and feebler for strong intensities, of current; and this, as well known, would render the apparatus very defective from a practical point of view.
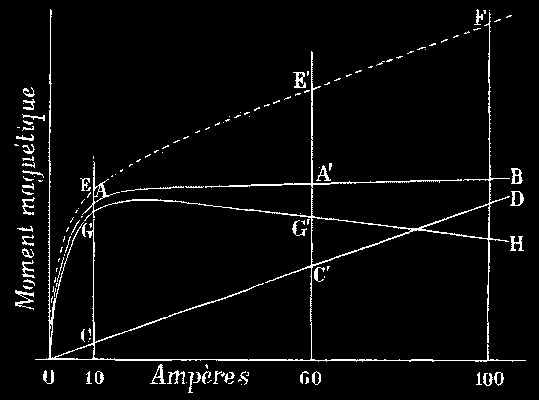
But the action of the spirals can be annulled without sensibly diminishing the magnetism of the core by arranging a second system of spirals identical with the first, but placed in a plane at right angles therewith, or, more simply still, by having a single system of spirals comprising the coil of the electro-magnet, but distributed in a plane that is oblique with respect to the needle's position of rest. It then becomes possible, by properly modifying such angle of inclination, to obtain a total directing action that shall continue to increase with the intensity, and which, graphically represented, shall give the curve, O G G' H, for example (Fig. 2).
This arrangement, which is adopted in Mr. Kapp's instruments, gives very good results, as may be easily seen by reference to Figs. 3 and 4, in which the current intensities or differences of potential are referred as ordinates and the degrees of deflection of the needle as abscissas. The unbroken lines represent the curves obtained with the apparatus just described, while the dotted ones give the curve of deflection of an ordinary tangent galvanometer. These curves show that for strong intensities of current Mr. Kapp's instrument is more advantageous than the tangent galvanometer. Mr. Crompton has constructed an amperemeter upon the same principle, which is shown in Fig. 5.—La Lumiere Electrique.
Professor A. Vogel, in a communication to the "Sitzungsberichte der Munchener Akademie," brings into prominence the fact that the hemlock plant, which yields coniine in Bavaria, contains none in Scotland. Hence he concludes that solar light plays a part in the generation of the alkaloids in plants. This view is corroborated by the circumstance that the tropical cinchonas, if cultivated in our feebly lighted hothouses, yield scarcely any alkaloids. Prof. Vogel has proved this experimentally. He has examined the barks of cinchona plants obtained from different conservatories, but has not found in any of them the characteristic reaction of quinine. Of course it is still possible that quinine might be discovered in other conservatory-grown cinchonas, especially as the specimens operated upon were not fully developed. But as the reaction employed indicates very small quantities of quinine, it may be safely assumed that the barks examined contained not a trace of this alkaloid, and it can scarcely be doubted that the deficiency of sunlight in our hothouses is one of the causes of the deficiency of quinine.
It will at once strike the reader as desirable that specimens of cinchonas should be cultivated in hothouses under the influence of the electric light, in addition to that of the sun.
If sunlight can be regarded as a factor in the formation of alkaloids in the living plant, it has, on the other hand, a decidedly injurious action upon the quinine in the bark stripped from the tree. On drying such bark in full sunlight the quinine is decomposed, and there are formed dark-colored, amorphous, resin-like masses. In the manufacture of quinine the bark is consequently dried in darkness.
This peculiar behavior of quinine on exposure to sunlight finds its parallel in the behavior of chlorophyl with the direct rays of the sun. It is well known that the origin of chlorophyl in the plant is entirely connected with light, so that etiolated leaves growing in the dark form no chlorophyl. But as soon as chlorophyl is removed from the sphere of vegetable life, a brief exposure to the direct rays of the sun destroys its green color completely.
Prof. A. Vogel conjectures that the formation of tannin in the living plant is to some extent influenced by light. This supposition is supported by the fact that the proportion of tannin in beech or larch bark increases from below upward—that is, from the less illuminated to the more illuminated parts, and this in the proportions of 4:6 and 5:10.
Sunny mountain slopes of a medium height yield, according to wide experience, on an average the pine-barks richest in tannin. In woods in level districts the proportion of tannin is greatest in localities exposed to the light, while darkness seems to have an unfavorable effect. Here, also, we must refer to the observation that leaves exceptionally exposed to the light are relatively rich in tannin.
We may here add that in the very frequent cases where a leaf is shadowed by another in very close proximity, or where a portion of a leaf has been folded over by some insect, the portion thus shaded retains a pale green color, while adjacent leaves, or other portions of the same leaf, assume their yellow, red, or brown autumnal tints. If, as seems highly probable, these tints are due to transformation products of tannin, we may not unnaturally conclude that they will be absent where tannin has not been generated.—Jour. of Science.
There are a number of interesting facts, some of which are known to most persons, and many of them have been long recognized, of which, however, it must be owned that the explanation is somewhat obscure, and the connections existing between them have been but recently pointed out. As an example of this, it is well known that salt water freezes at a lower temperature than fresh water, and hence sea-water may be quite liquid while rivers and ponds are covered with ice. Again, it is noticed that mixtures of salts often have a fusing-point lower than that of either of the constituent salts, and of this fact we often take advantage in fluxing operations. Further, it is well known that certain alloys can be prepared, the melting-points of which are lower than the melting-point of either of the constituent metals alone. Thus, while potassium melts at 62.5° C., and sodium at about 98°, an alloy of these metals is fluid at ordinary temperatures, and fusible metal melts below the temperature of boiling water, or more than 110° lower than the melting-point of tin, the most fusible of the three metals which enter into the composition of this alloy. But though these and many similar facts have been long known, it is but recently, owing largely to the labors of Dr. Guthrie, that fresh truths have been brought to light, and a connection shown to exist throughout the whole which was previously unseen, though we have still to acknowledge that at present there is much at the root of the matter which is but imperfectly understood. Still Dr. Guthrie proves a relationship to exist between the several facts we have previously mentioned, and also between a number of other phenomena which at first sight appear to be equally isolated and unexpected, and we are asked to regard them all as examples of what he has called "eutexia."
We may define a eutectic substance as a body composed of two or more constituents, which constituents are in such proportion to one another as to give to the resultant compound body a minimum temperature of liquefaction—that is, a lower temperature of liquefaction than that given by any other proportion.2 It will be seen at once by this definition that the temperature of liquefaction of a eutectic substance is lower than the temperature of liquefaction of either or any of the constituents of the mixture. And, further, it is plain that those substances only can be eutectic which we can obtain both as liquid and solid, and hence the property of eutexia is closely connected with solution.
Following in the natural divisions adopted by Dr. Guthrie, we may consider eutexia in three aspects:
If a dilute aqueous saline solution be taken at ordinary temperatures, and then slowly cooled to some point below zero on the Centigrade scale, the following series of changes will in general be observed: On reaching a point below zero, the position of which is dependent upon the nature of the salt and the amount of dilution, it will be found that ice is formed; this will float upon the surface of the solution, and may be readily removed. If the ice so removed be afterward pressed, or carefully drained, it will be found to consist of nearly pure water, the liquid draining away being a strong saline solution which had become mechanically entangled among the crystals of ice during solidification. If we further cool the brine which remains, we notice a tolerably uniform fall of temperature with accompanying formation of ice. But at length a point is reached at which the temperature ceases to fall until the whole of the remaining mother-liquor has solidified, with the production of a compound called a cryohydrate,3 which possesses physical properties different from those of either the ice or the salt from which it is formed.
If, on the other hand, we commence with a saturated saline solution, in general it is noticed on cooling the liquid a separation of salt ensues, which salt sinks to the bottom of the mass, and may be removed. The salt so separating may be either anhydrous or a "hydrate" of greater concentration than the mother-liquor. So long as this separation proceeds the temperature falls, but at length a point is reached at which the thermometer remains stationary until the whole is solidified, with the production of a cryohydrate. This temperature of solidification is the same whether we start with a dilute or a saturated solution, and the composition of the cryohydrate is found to be constant. The temperature of production of the cryohydrate is identical with the lowest temperature which can be produced on employing a mixture of ice and the salt as a freezing mixture or cryogen.
It will be readily seen that in the formation of a cryohydrate we have an example of eutexia, since the constituents are present in such proportion as to give to the resultant compound body a minimum temperature of liquefaction.
Although it has been long known that on mixing certain salts the resulting substance possessed a lower melting-point than either of the constituent salts alone, still but few determinations of the melting-points of mixtures of salts have been made, and even these are often of small value, on account of the very considerable range of temperature observed during solidification. This is due largely to the fact that eutectic mixtures were not known, as equivalent proportions of various salts have been employed, while eutectic mixtures are seldom found to possess any simple arithmetical molecular relationship between their constituents.
Eutectic salt alloys closely resemble cryohydrates in behavior. If for simplicity we confine our attention to a fused mixture of two salts in any proportion other than eutectic, it is found that, on cooling, the thermometer falls steadily, until at length that salt which is in excess of the proportion required for a eutectic mixture begins to separate out. If this is removed, the thermometer falls until a fixed point is reached at which the temperature remains stationary until the whole of the mixture solidifies. On remelting, the temperature of solidification is found to be quite fixed, and the mixture is evidently eutectic.
It is of interest to notice that from our knowledge of the cryohydrates it becomes possible to predict the existence, composition, and temperature of solidification of a eutectic alloy, if we are previously furnished with the melting-points of mixtures of the substances in question. Or, in other cases, we may predict from the curve of melting-points that no eutectic alloy is possible.
As an example, we may take the determinations of the melting-points of mixtures of potassium and sodium nitrate by M. Maumené.5 These are graphically represented in Fig. 1, the curve being derived from the mean of the temperatures given in the memoir. From this diagram we should be led to expect a eutectic mixture, since the curve dips below a horizontal line passing through the melting-point of the more fusible of its constituents. From our curve we should expect a eutectic mixture with about 35 per cent. KNO3, and with a temperature of solidification below 233°. Dr. Guthrie gives 32.9 per cent. at 215°. This agreement is as good as might be expected when one remembers that the melting-points, not being of eutectic mixtures, are difficult to determine, and a considerable range is given; that analyses of mixtures of potassium and sodium salts are apt to vary; and that the two observers differ by ±7° in the temperatures given for the melting-points of the original salts.
Dr. Tilden has drawn my attention to an interesting example of the lowering of melting-point by the mixture of salts. The melting-point of monohydrochloride of turpentine oil is 125°, while that of the dihydrochloride is 50°; but on simply stirring together these compounds in a mortar at common temperatures, they immediately liquefy. Two molecules of the monohydrochloride and one molecule of the dihydrochloride form a mixture which melts at about 20°.
Although many fusible alloys have been long known, I believe no true eutectic metallic alloy had been studied until Dr. Guthrie6 worked at the subject, employing the same methods as with his cryohydrates. It is found if two metals are fused together and the mixture allowed to cool, that the temperature falls until a point is reached at which that metal which is present in a proportion greater than is required to form the eutectic alloy begins to separate. If this solid be removed as it forms, the temperature gradually falls until a fixed point is reached, at which the eutectic alloy solidifies. Here the thermometer remains stationary until the whole has become solid, and, on remelting, this temperature is found to be quite fixed. In addition to the di-eutectic alloys, we have also tri- and tetra-eutectic alloys, and as an example of the latter we may take the bismuth-tin-lead-cadmium eutectic alloy, melting at 71°.
We have already seen with salt eutectics that, given the curve of melting-points of a mixture in various proportions, we may predict the existence, composition, and melting-point of the eutectic alloy. As a matter of course, the same thing holds good for metallic eutectics. An interesting example of this is furnished by the tin-lead alloys, the melting-points of which have been determined by Pillichody.7 From these determinations we obtain the curve given in Fig. 2, and from this curve, since it dips below a horizontal line passing through the melting-point of the more fusible constituent, we are at once able to predict a eutectic alloy. We should further expect this to have a constitution between PbSn3 and PbSn4 and a melting-point somewhat below 181°. On melting together tin and lead, and allowing the alloy to cool, we find our expectation justified; for by pouring off the fluid portion which remains after solidification has commenced, and repeating this several times with the portion so removed, we at length obtain an alloy which solidifies at the constant temperature of 180°, when the melting-point of tin is taken as 228°. On analysis 1.064 grm. of this alloy gave 0.885 grm. SnO2, which corresponds to Sn 65.43 per cent., or PbSn3.3. This, therefore, is the composition of the eutectic alloy, and it finds its place naturally on the curve given in Fig. 2.
It will be seen that the subject of eutexia embraces many points of practical importance and of theoretical interest. Thus it has been shown by Dr. Guthrie that the desilverizing of lead in Pattinson's process is but a case of eutexia, the separation of lead on cooling a bath of argentiferous lead poor in silver being analogous to the separation of ice from a salt solution. Dr. Guthrie has also shown that eutexia may reasonably be supposed to have played an important part in the production and separation of many rock-forming minerals.
It is with considerable diffidence that I suggest the following as an explanation of the multitude of facts to which previous reference has been made.
In a mixture of two substances, A and B, we have the following forces active, tending to produce solidification:
1. The cohesion between the particles of A.
2. The cohesion between the particles of B.
3. The cohesion between the particles of A and the particles of B.
With regard to this last factor, it will be seen that there are three cases possible:
1. The cohesion of the mixture A B may be greater than the cohesion of A + the cohesion of B.
2. The cohesion of A B may be equal to the cohesion of A + the cohesion of B.
3. The cohesion of A B may be less than the cohesion of A + the cohesion of B.
Now, since cohesion tends to produce solidification, we should in the first case expect to find the melting-point of the mixture higher than the mean of the melting-points of its constituents, or the curve of melting-points would be of the form given in a, Fig. 3. Here no eutectic mixture is possible.
In the second case, where cohesionA B = cohesion A + B, we should obtain melting-points for the mixture which would agree with the mean of the melting-points of the constituents, the curve of melting-points would be a straight line, and again no eutectic mixture would be possible.
In the third case, however, where cohesionA B is less than cohesion A + B, we should find the melting-points of the mixture lower than the mean of the melting-points of its constituents, and the curve of melting-points would be of the form given in e, Fig. 3. Here, in those cases where the difference of cohesion on mixture is considerable, the curve of melting-points may dip below the line e f. This is the only case in which a eutectic mixture is possible, and it is, of course, found at the lowest point of the curve.
If it be true, as above suggested, that the force of cohesion is at its minimum in the eutectic alloy, we should expect to find, in preparing a eutectic substance, either that actual expansion took place, or that the molecular volume would gradually increase in passing along our curve of melting-points, from either end, for each molecule added, and that it would obtain its greatest value at the point corresponding to the eutectic alloy.
Of this I have no direct evidence as yet, but it is a point of considerable interest, and I may possibly return to it at some future time.—Chemical News.
Read before the Birmingham Philosophical Society, January 22, 1885.
Guthrie, Phil. Mag. [5], xvii., p. 462.
Guthrie, Phil. Mag., 4th Series, xlix., pp. 1, 206, 266; 5th Series, i., pp. 49, 354, 446, vi., p. 35.
F. Guthrie, Phil. Mag. [5], xvii., p. 469; F.B. Guthrie, Journ. Chem. Soc,. 1885, p. 94.
Comptes Rendus, 1883, 2, p. 45.
Phil. Mag., 5th Series, xvii., p. 462.
Dingler's Polyt. Jour., 162, p. 217; Jahresberichte, 1861, p. 279.
Dr. Conrad Berens, of the University of Pennsylvania, reaches the following:
1. Chinoline tartrate is a powerful agent, producing death by asphyxia.
2. The drug increases the force and frequency of the respirations by stimulating the vagus roots in the lung.
3. It paralyzes respiration finally by a secondary depressant action upon the respiratory center.
4. It does not cause convulsions.
5. It lessens and finally abolishes reflex action by a direct action upon the cord, and by a slight action upon the muscles and nerves.
6. It diminishes or abolishes muscular contractility respectively when applied through the circulation or directly.
7. It coagulates myosin and albumen.
8. It causes insalivation by paralysis of the secretory fibers of the chorda tympani; increases the flow of bile; has no action upon the spleen.
9. It lowers blood-pressure by paralyzing the vaso-motor centers and by a direct depressant action upon the heart muscle.
10. It diminishes the pulse rate by direct action upon the heart.
11. It lowers the temperature by increasing the loss of heat.
12. It is a powerful antiseptic; and, finally,
13. Its paths of elimination are not known.
Being called upon to make a good many brief and rapid analyses of urine on "clinic days" of our medical department, I devised the following modification of Knop's method of estimating urea; and after using it for a year with perfectly satisfactory results, venture to describe and recommend it as especially adapted for physicians' use, by reason of simplicity, cheapness, and accuracy. In perfecting and testing it I was assisted greatly by J. Torrey, Jr., then working with me.
The apparatus consists of the glass tube, A, which is about 8 cm. long and 2½ cm. in diameter, joined to the tube, B, which is about 25 or 30 cm. in length in its longer arm and 8 or 10 in its shorter, and has a diameter of about 5 mm. Near the bend is an outlet tube, c, provided with "ball valve" or pinch cock. d, e, f, g, are marks upon the tubes. C is a rubber cork with two holes through which the bent tube, D, passes. D is of such size and length as to hold about 1 c.c., and one of its ends may be a trifle longer than the other.
The apparatus is used as follows: Remove the cork and pour in mercury until it stands at e and g, then fill up to the mark, f, with sodium or potassium hypobromite (made by shaking up bromine with a strong solution of sodium or potassium hydroxide). Next carefully fill the tube in the cork with the urine, being careful especially not to run it over or leave air bubbles in it. This can easily be done by using a small pipette, but if accidentally a little runs over, it should be wiped off the end of the cork with blotting paper. The cork is then to be inserted closely into the tube; the urine tube being so small, the urine will not run out in so doing. The mercury is then drawn out through c till it stands in B at d. Its level in A will of course not be changed greatly. Now, incline the apparatus till the surface of the hypobromite touches the urine in the longer part of the urine tube, and then bring it upright again. The urine will thus be discharged into the hypobromite, which will of course decompose the urea, liberating nitrogen, which will cause the mercury to rise in B. Shake until no further change of level is seen, and mark the level of mercury in B with a rubber band, then remove the cork, draw out the liquid with a pipette, dry out the tube above the mercury with scrap of blotting paper, pour back the mercury drawn out, and repeat the process to be sure that no error was made.
If now two or three marks have been made upon the tube, B, indicating the height of the mercury when solutions containing known per cents. of urea are used, an accurate opinion can be at once formed as to the condition of the urine as regards urea.
As is well known, normal urine contains about 2.5-3 per cent. of urea, so that graduations representing 2, 2.5, 3, and 4 per cent. are usually all that are needed, though of course many more can be easily made.
The results obtained with this apparatus have been repeatedly compared with those of more elaborate ones, and no practical difference observed. Evidently the same apparatus, differently graduated, might be employed to determine the carbonate present in such a substance as crude soda ash or other similar mixture. In such a case the weighed material would be put upon the mercury with water and the small tube filled with acid.
Bowdoin College Chemical Laboratory.
—F.C. Robinson, in Amer. Chem. Jour.
A Catalogue containing brief notices of many important scientific papers heretofore published in the Supplement, may be had gratis at this office.
Sent by mail, postage prepaid, to subscribers in any part of the United States or Canada. Six dollars a year, sent, prepaid, to any foreign country.
All the back numbers of The Supplement, from the commencement, January 1, 1876, can be had. Price, 10 cents each.
All the back volumes of The Supplement can likewise be supplied. Two volumes are issued yearly. Price of each volume, $2.50 stitched in paper, or $3.50 bound in stiff covers.
Combined Rates.—One copy of Scientific American and one copy of Scientific American Supplement, one year, postpaid, $7.00.
A liberal discount to booksellers, news agents, and canvassers.
MUNN & CO., Publishers,
361 Broadway. New York, N.Y.
In connection with the Scientific American, Messrs. MUNN & CO. are solicitors of American and Foreign Patents, have had 40 years' experience, and now have the largest establishment in the world. Patents are obtained on the best terms.
A special notice is made in the Scientific American of all inventions patented through this Agency, with the name and residence of the Patentee. By the immense circulation thus given, public attention is directed to the merits of the new patent, and sales or introduction often easily effected.
Any person who has made a new discovery or invention can ascertain, free of charge, whether a patent can probably be obtained, by writing to MUNN & CO.
We also send free our Hand Book about the Patent Laws, Patents, Caveats, Trade Marks, their costs, and how procured. Address
Munn & Co., 361 Broadway, New York.
Branch Office, cor. F and 7th Sts., Washington, D.C.