The Home Medical
Library
By
Kenelm Winslow, B.A.S., M.D.
Formerly Assistant Professor Comparative Therapeutics, Harvard
University; Late Surgeon to the Newton Hospital;
Fellow of the Massachusetts Medical Society, etc.
With the Coöperation of Many Medical
Advising Editors and Special Contributors
IN SIX VOLUMES
First Aid :: Family Medicines :: Nose, Throat, Lungs,
Eye, and Ear :: Stomach and Bowels :: Tumors and
Skin Diseases :: Rheumatism :: Germ Diseases
Nervous Diseases :: Insanity :: Sexual Hygiene
Woman and Child :: Heart, Blood, and Digestion
Personal Hygiene :: Indoor Exercise
Diet and Conduct for Long Life :: Practical
Kitchen Science :: Nervousness
and Outdoor Life :: Nurse and Patient
Camping Comfort :: Sanitation
of the Household :: Pure
Water Supply :: Pure Food
Stable and Kennel
New York
The Review of Reviews Company
1907
Medical Advising Editors
Managing Editor
Albert Warren Ferris, A.M., M.D.
Former Assistant in Neurology, Columbia University; Former Chairman,
Section on Neurology and Psychiatry, New York Academy of Medicine;
Assistant in Medicine, University and Bellevue Hospital Medical
College; Medical Editor, New International Encyclopedia.
Nervous Diseases
Charles E. Atwood, M.D.
Assistant in Neurology, Columbia University; Former Physician, Utica
State Hospital and Bloomingdale Hospital for Insane Patients; Former
Clinical Assistant to Sir William Gowers, National Hospital, London.
Pregnancy
Russell Bellamy, M.D.
Assistant in Obstetrics and Gynecology, Cornell University Medical
College Dispensary; Captain and Assistant Surgeon (in charge),
Squadron A, New York Cavalry; Assistant in Surgery, New York
Polyclinic.
Germ Diseases
Hermann Michael Biggs, M.D.
General Medical Officer and Director of Bacteriological Laboratories,
New York City Department of Health; Professor of Clinical Medicine in
University and Bellevue Hospital Medical College; Visiting Physician
to Bellevue, St. Vincent's, Willard Parker, and Riverside Hospitals.
The Eye and Ear
J. Herbert Claiborne, M.D.
Clinical Instructor in Ophthalmology, Cornell University Medical
College; Former Adjunct Professor of Ophthalmology, New York
Polyclinic; Former Instructor in Ophthalmology in Columbia University;
Surgeon, New Amsterdam Eye and Ear Hospital.
Sanitation
Thomas Darlington, M.D.
Health Commissioner of New York City; Former President Medical Board,
New York Foundling Hospital; Consulting Physician, French Hospital;
Attending Physician, St. John's Riverside Hospital, Yonkers; Surgeon
to New Croton Aqueduct and other Public Works, to Copper Queen
Consolidated Mining Company of Arizona, and Arizona and Southeastern
Railroad Hospital; Author of Medical and Climatological Works.
Menstruation
Austin Flint, Jr., M.D.
Professor of Obstetrics and Clinical Gynecology, New York University
and Bellevue Hospital Medical College; Visiting Physician, Bellevue
Hospital; Consulting Obstetrician, New York Maternity Hospital;
Attending Physician, Hospital for Ruptured and Crippled, Manhattan
Maternity and Emergency Hospitals.
Heart and Blood
John Bessner Huber, A.M., M.D.
Assistant in Medicine, University and Bellevue Hospital Medical
College; Visiting Physician to St. Joseph's Home for Consumptives;
Author of "Consumption: Its Relation to Man and His Civilization; Its
Prevention and Cure."
Skin Diseases
James C. Johnston, A.B., M.D.
Instructor in Pathology and Chief of Clinic, Department of
Dermatology, Cornell University Medical College.
Diseases of Children
Charles Gilmore Kerley, M.D.
Professor of Pediatrics, New York Polyclinic Medical School and
Hospital; Attending Physician, New York Infant Asylum, Children's
Department of Sydenham Hospital, and Babies' Hospital, N. Y.;
Consulting Physician, Home for Crippled Children.
Bites and Stings
George Gibier Rambaud, M.D.
President, New York Pasteur Institute.
Headache
Alonzo D. Rockwell, A.M., M.D.
Former Professor Electro-Therapeutics and Neurology at New York
Post-Graduate Medical School; Neurologist and Electro-Therapeutist to
the Flushing Hospital; Former Electro-Therapeutist to the Woman's
Hospital in the State of New York; Author of Works on Medical and
Surgical Uses of Electricity, Nervous Exhaustion (Neurasthenia), etc.
Poisons
E. Ellsworth Smith, M.D.
Pathologist, St. John's Hospital, Yonkers; Somerset Hospital,
Somerville, N. J.; Trinity Hospital, St. Bartholomew's Clinic, and the
New York West Side German Dispensary.
Catarrh
Samuel Wood Thurber, M.D.
Chief of Clinic and Instructor in Laryngology, Columbia University;
Laryngologist to the Orphan's Home and Hospital.
Care of Infants
Herbert B. Wilcox, M.D.
Assistant in Diseases of Children, Columbia University.
Special Contributors
Food Adulteration
S. Josephine Baker, M.D.
Medical Inspector, New York City Department of Health.
Pure Water Supply
William Paul Gerhard, C.E.
Consulting Engineer for Sanitary Works; Member of American Public
Health Association; Member, American Society Mechanical Engineers;
Corresponding Member of American Institute of Architects, etc.; Author
of "House Drainage," etc.
Care of Food
Janet McKenzie Hill
Editor, Boston Cooking School Magazine.
Nerves and Outdoor Life
S. Weir Mitchell, M.D., LL.D.
LL.D. (Harvard, Edinburgh, Princeton); Former President, Philadelphia
College of Physicians; Member, National Academy of Sciences,
Association of American Physicians, etc.; Author of essays: "Injuries
to Nerves," "Doctor and Patient," "Fat and Blood," etc.; of scientific
works: "Researches Upon the Venom of the Rattlesnake," etc.; of
novels: "Hugh Wynne," "Characteristics," "Constance Trescott," "The
Adventures of François," etc.
Sanitation
George M. Price, M.D.
Former Medical Sanitary Inspector, Department of Health, New York
City; Inspector, New York Sanitary Aid Society of the 10th Ward, 1885;
Manager, Model Tenement-houses of the New York Tenement-house Building
Co., 1888; Inspector, New York State Tenement-house Commission, 1895;
Author of "Tenement-house Inspection," "Handbook on Sanitation," etc.
Indoor Exercise
Dudley Allen Sargent, M.D.
Director of Hemenway Gymnasium, Harvard University; Former President,
American Physical Culture Society; Director, Normal School of Physical
Training, Cambridge, Mass.; President, American Association for
Promotion of Physical Education; Author of "Universal Test for
Strength," "Health, Strength and Power," etc.
Long Life
Sir Henry Thompson, Bart., F.R.C.S., M.B. (Lond.)
Surgeon Extraordinary to His Majesty the King of the Belgians;
Consulting Surgeon to University College Hospital, London; Emeritus
Professor of Clinical Surgery to University College, London, etc.
Camp Comfort
Stewart Edward White
Author of "The Forest," "The Mountains," "The Silent Places," "The
Blazed Trail," etc.
 WALTER REED.
WALTER REED.
In the year 1900, Major Walter Reed, a surgeon in the United States
Army, demonstrated, by experiments conducted in Cuba, that a mosquito
of a single species, Stegomyia fasciata, which has sucked the blood of
a yellow-fever patient may transmit the disease by biting another
person, but not until about twelve days have elapsed. He also proved,
as described in Volume I, Part II, that the malady is not contagious.
"With the exception of the discovery of anæsthesia," said Professor
Welch, of Johns Hopkins University, "Dr. Reed's researches are the
most valuable contributions to science ever made in this country."
General Leonard Wood declared the discovery to be the "greatest
medical work of modern times," which, in the words of President
Roosevelt, "renders mankind his debtor." Major Reed died November 23,
1902.
The Home Medical
Library
VOLUME V :: SANITATION
Edited by
Thomas Darlington, M.D.
Health Commissioner of New York City; Former President Medical
Board, New York Foundling Hospital, etc.; Author of
Medical and Climatological Works
WATER SUPPLY AND PURIFICATION
By WILLIAM PAUL GERHARD, C.E.
Consulting Engineer for Sanitary Works; Author of "House Drainage,"
"Sanitary Engineering," "Household Wastes," etc.
PURE FOOD FOR THE
HOUSEKEEPER
By S. JOSEPHINE BAKER, M.D.
Medical Inspector, New York City Department of Health
THE HOUSE AND GROUNDS
By GEORGE M. PRICE, M.D.
Former Medical Sanitary Inspector, Department of Health, New
York City; Author of "Tenement-House Inspection,"
"Handbook on Sanitation," etc.
New York
The Review of Reviews Company
1907
Copyright, 1907, by
The Review of Reviews Company
THE TROW PRESS, NEW YORK
[Pg 9]
Contents
PART I
| CHAPTER |
|
PAGE |
| I. |
Country Sources of Water Supply |
19 |
| |
Relation of Water to Health—Collection of
Rain Water—Cisterns—Springs—Various
Kinds of Wells—Laws Regulating Supply. |
|
| II. |
Appliances for Distributing Water |
39 |
| |
Pumping Machines—The Hydraulic Ram—Use
of Windmills—Engines—Steam
and Electric Pumps—Reservoirs and
Tanks—Appliances for Country Houses. |
|
| III. |
Purifying Water by Copper Sulphate |
52 |
| |
Clear Water Often Dangerous—Pollution
Due to Plants—Copper Sulphate Method—Directions
for the Copper Cure. |
|
| IV. |
Ridding Stagnant Water of Mosquitoes |
70 |
| |
Malaria Due to Mosquitoes—Cause of Yellow
Fever—Effect of a Mosquito Bite—Destruction
of Larvæ—Best Preventive
Measures—Use of Kerosene. |
|
PART II
| CHAPTER |
|
PAGE |
| I. |
How To Detect Food Adulteration |
87 |
| |
Definition of Adulteration—Food Laws—Permissible[Pg 10]
Adulterants—How to Select
Pure Food—Chemical Tests. |
|
| II. |
Mushroom Poisoning |
112 |
| |
Symptoms and Treatment—Coffee and
Atropine the Best Antidotes—How to Tell
the Edible Kind—"Horse," "Fairy-ring,"
and Other Varieties—Poisonous Species. |
|
PART III
| CHAPTER |
|
PAGE |
| I. |
Soil and Sites |
131 |
| |
Constituents of the Soil—Influence on
Health—Improving Defective Soil—Street
Paving and Tree Planting—Proper Construction
of Houses—Subsoil Drainage. |
|
| II. |
Ventilation |
146 |
| |
What is Meant by Ventilation—Quantity
of Air Required—Natural Agents of Ventilation—Special
Appliances. |
|
| III. |
Warming |
160 |
| |
Various Methods—Materials of Combustion—Chimneys—Fireplaces
and Grates—Stoves—Hot-air
Warming—Hot-water
Systems—Principles of Steam Heating. |
|
| IV. |
Disposal of Sewage |
170 |
| |
Refuse and Garbage—Discharge into Waters—Cremation—Precipitation—Intermittent
Filtration—Immediate Disposal, etc. |
|
| [Pg 11]V. |
Sewers |
182 |
| |
Definition—Materials Used in Construction—Levels
of Trenches—Joints of Pipes—The
Fall and Flow of the Contents—Connections—Tide
Valves—Sewer Gas. |
|
| VI. |
Plumbing |
189 |
| |
Purposes and Requisites—Materials Used—Joints
and Connections—Construction
of Traps—Siphonage and Back Pressure—The
Vent-pipe System. |
|
| VII. |
Plumbing Pipes |
206 |
| |
Construction of House Drains—Fall, Position,
and Connection—Main Traps—Extension
of Vertical Pipes—Fresh-air Inlets—Soil
and Waste Pipes—Branch Pipes, etc. |
|
| VIII. |
Plumbing Fixtures |
216 |
| |
Sinks—Washbasins—Washtubs—Bathtubs—Refrigerators,
etc.—Safes and
Wastes—Pan, Valve, and Hopper Closets—Flush
Tanks—Yard Closets—Drains. |
|
| IX. |
Defects in Plumbing |
231 |
| |
Poor Work—Improper Conditions—How
to Test Traps, Joints, and Connections—Detect
Sewer Gas—Water-pressure,
Smoke, and Scent Tests—Special Appliances. |
|
| [Pg 12]X. |
Infection and Disinfection |
238 |
| |
Physical and Chemical Disinfectants—Use
of Sulphur Dioxide—Formaldehyde—Hydrocyanic
Acid—Chlorine—Carbolic
Acid—Bichloride of Mercury—Formalin—Potassium
Permanganate, etc. |
|
| XI. |
Cost of Conveyed Heating Systems |
254 |
| |
Cost of Hot-air Systems—Cast-iron Hot-water
Heater—Advantages and Disadvantages—Cost
for a Ten-room House—Steam
Heating—Cost of Equipment. |
|
[Pg 13]
The Editor's Preface
The character and scope of this volume render it a most useful book
for the home maker. The question of sanitation is one that closely
affects the life of each individual, and many of its aspects are
treated here in a lucid and comprehensive manner. Designed for wide
distribution, these articles have been written to meet the needs of
the dweller in the more densely populated communities, as well as
those living in the less thickly settled portion of the country.
In large cities the water supply is a problem that is cared for by
regularly constituted sanitary authorities. Pure water is a vital
necessity, but the inhabitant of a city has no need to personally
concern himself with the source of supply. In the country, however,
the home builder must often decide the matter for himself, and it is
the aim of this book to give him the needed directions for avoiding
many errors and pitfalls that abound in this direction.
House construction, with its intricate problems, is also a more
serious matter for the country dweller than for his city brother.
In the matter of food supply, the inhabitant of a country district is
more fortunate. Fresh vegetables[Pg 14] and dairy products are much more
easily obtained, and their freshness and purity more dependable.
The article on water supply by Mr. Gerhard is authoritative, written,
as it is, by a most eminent sanitarian. The publishers are to be
congratulated upon the following valuable contribution to the same
subject as regards the use of copper sulphate and the concise
presentation of plans for mosquito extermination, while the extended
work of Dr. Price and Dr. Baker's "Food Adulteration" are much to be
commended. The two latter have been connected with the Department of
Health of New York City, and have the advantage of experience in an
organization which gives to the citizens of New York the protection to
health that the wise use of science, knowledge, and money afford.
I trust that the notes I have added in the light of recent practice of
the New York City Department of Health may make this material of the
utmost practical value to the householder of to-day.
Through this Department of Health, New York City spent, during 1905,
over $1,500,000, and for 1906 it has appropriated over $1,800,000.
This vast sum of money is used for the sole purpose of safeguarding
its citizens from disease. Sanitation in its varied branches is
pursued as an almost exact science, and the efforts of trained minds
are constantly employed in combating disease and promoting sanitation.
The cities care for their own, but the greater num[Pg 15]ber of the
inhabitants of this country must rely upon their individual efforts.
Therefore, any dissemination of knowledge regarding sanitation is most
worthy. This book has a useful mission. It is pregnant with helpful
suggestions, and I most heartily commend its purpose and its contents.
Thomas Darlington,
President of the Board of Health.
New York City.
[Pg 16]
[Pg 17]
Part I
WATER SUPPLY AND
PURIFICATION
BY
WILLIAM PAUL GERHARD
[Pg 18]
[Pg 19]
CHAPTER I
Country Sources of Water Supply
The writer was recently engaged to plan and install a water-supply
system for a country house which had been erected and completed
without any provision whatever having been made for supplying the
buildings and grounds with water. The house had all the usual
appointments for comfort and ample modern conveniences, but these
could be used only with water borrowed from a neighbor. In all parts
of the country there are numerous farm buildings which are without a
proper water-supply installation. These facts are mentioned to
emphasize the importance of a good water supply for the country home,
and to point out that water is unquestionably the most indispensable
requirement for such structures.
Adequate Water Supply Important
But the advantages of a water supply are not limited to the dwelling
house, for it is equally useful on the farm, for irrigation, and in
the garden, on the golf grounds and tennis courts, in the barns and
stables;[Pg 20] it affords, besides, the best means for the much-desired
fire protection. And, most important of all, an unstinted and adequate
use of water promotes cleanliness and thereby furthers the cause of
sanitation, in the country not less than in the city home.
The water supply for country houses has been so often discussed
recently that the writer cannot hope to bring up any new points. This
article should, therefore, be understood to offer simple suggestions
as to how and where water can be obtained, what water is pure and fit
for use, what water must be considered with suspicion, what water is
dangerous to health, and how a source of supply, meeting the
requirements of health, can be made available for convenient use.
Right here I wish to utter a warning against the frequent tendency of
owners of country houses to play the rôle of amateur engineers. As a
rule this leads to failure and disappointment. Much money uselessly
spent can be saved if owners will, from the beginning, place the
matter in experienced hands, or at least seek the advice of competent
engineers, and adopt their suggestions and recommendations as a guide.
Points to be Borne in Mind
Many are the points to be borne in mind in the search for water.
Science teaches us that all water comes from the clouds, the
atmospheric precipitation being in the form of either rain, or dew, or
snow.[Pg 21] After reaching the earth's surface, the water takes three
different courses, and these are mentioned here because they serve to
explain the different sources of supply and their varied character.
A part of the water runs off on the surface, forming brooks, streams,
and lakes, and if it falls on roofs of houses or on prepared catchment
areas, it can be collected in cisterns or tanks as rain water. Another
part of the water soaks away into pervious strata of the subsoil, and
constitutes underground water, which becomes available for supply
either in springs or in wells. A third part is either absorbed by
plants or else evaporated.
In our search for a source of supply, we should always bear in mind
the essential requirements of the problem. Briefly stated, these are:
the wholesomeness of the water, the adequateness and steadiness of the
supply, its availability under a sufficient pressure, insuring a good
flow, and the legal restrictions with which many water-supply problems
are surrounded.
The first essential requirement is that of wholesomeness. The
quality of a water supply is dependent upon physical properties and
upon chemical and bacteriological characteristics. Water, to be
suitable for drinking, must be neither too hard nor too soft; it
should not contain too many suspended impurities, nor too much foreign
matter in solution. Pure water is colorless and without odor. But it
must be understood that the quality cannot be decided merely by[Pg 22] the
color, appearance, taste, and odor. The chemical and bacteriological
examinations, if taken together, form a much safer guide, and with
these analyses should go hand in hand a detailed survey of the water
source and its surroundings.
Relation of Water to Health
Any pronounced taste in the water renders it suspicious; an offensive
smell points to organic contamination; turbidity indicates presence of
suspended impurities, which may be either mineral or organic. But even
bright and sparkling waters having a very good taste are sometimes
found to be highly polluted. Hence, it should be remembered that
neither bright appearance nor lack of bad taste warrants the belief
that water is free from dangerous contamination.
It is a well-established fact now that there is a relation between the
character of the water supply and the health of a community; and what
is true of cities, villages, and towns, is, of course, equally true of
the individual country house.
How Water Becomes Contaminated
There are numerous ways in which water may become polluted, either at
the source or during storage or finally during distribution. Rain
water, falling pure from the clouds, encounters dust, soot, decaying
leaves[Pg 23] and other vegetable matters, and ordure of birds on the roofs;
its quality is also affected by the roofing material, or else it is
contaminated in the cisterns by leakage from drains or cesspools.
Upland waters contain generally vegetable matter, while surface water
from cultivated lands becomes polluted by animal manure. River water
becomes befouled by the discharge into it of the sewers from
settlements and towns located on its banks. Subsoil water is liable to
infiltration of solid and liquid wastes emanating from the human
system, from leaky drains, sewers, or cesspools, stables, or
farmyards; and even deep well water may become contaminated by reason
of defects in the construction of the well.
During storage, water becomes contaminated in open reservoirs by
atmospheric impurities; a growth of vegetable organisms or algæ often
causes trouble, bad taste, or odor; water in open house tanks and in
cisterns is also liable to pollution. During distribution, water may
become changed in quality, owing to the action of the water on the
material of the pipes.
From what source shall good water be obtained? This is the problem
which confronts many of those who decide to build in the country.
The usual sources, in their relative order of purity, are: deep
springs and land or surface springs, located either above or below the
house, but not too near to settlements; deep subterranean water, made
available by boring or drilling a well; upland or mountain[Pg 24] brooks
from uninhabited regions; underground water in places not populated,
reached by a dug or driven well; lake water; rain water; surface water
from cultivated fields; pond and river water; and finally, least
desirable of all, shallow well water in villages or towns. These
various sources of supply will be considered farther on.
An Ample Volume Necessary
The second essential requirement is ample quantity. The supply must
be one which furnishes an ample volume at all seasons and for all
purposes.
What is a reasonable daily domestic consumption? The answer to this
question necessarily depends upon the character of the building and
the habits and occupation of its inmates. It is a universal experience
that as soon as water is introduced it is used more lavishly, but also
more recklessly and regardless of waste. For personal use, from twenty
to twenty-five gallons per person should prove to be ample per day:
this comprises water for drinking and cooking, for washing clothes,
house and kitchen utensils, personal ablutions, and bathing; but,
taking into account other requirements on the farm or of country
houses, we require at least sixty gallons per capita per diem. To
provide water for the horses, cows, sheep, for carriage washing, for
the garden, for irrigation of the lawn, for fountains, etc., and keep
a suitable reserve in case[Pg 25] of fire, the supply should be not less
than 150 gallons per person per day.
A Good Pressure Required
The third essential requirement is a good water pressure. Where a
suitable source of water is found, it pays to make it conveniently
available, so as to avoid carrying water by hand, which is troublesome
and not conducive to cleanliness. A sufficient pressure is attained by
either storing water at, or lifting it to, a suitable elevation above
the point of consumption. In this respect many farm and country houses
are found to be but very imperfectly supplied. Often the tank is
placed only slightly higher than the second story of the house. As a
result, the water flows sluggishly at the bathroom faucets, and, in
case of fire, no effective fire stream can be thrown. Where a
reservoir is suitably located above the house, the pressure is
sometimes lost by laying pipes too small in diameter to furnish an
ample stream. Elevated tanks should always be placed so high as to
afford a good working pressure in the entire system of pipes. Where a
tower of the required height is objectionable, either on account of
the cost or on account of appearance, pressure tanks may be installed
which have many advantages.
In selecting a source of water supply, the following points should be
borne in mind for guidance: first, the wholesomeness of the water;
next, the cost required[Pg 26] to collect, store, and distribute the water;
finally, where a gravity supply is unavailable, the probable operating
expenses of the water system, cost of pumping, etc.
Collection of Rain Water
The collection of rain water near extensive manufacturing
establishments is not advisable, except where arrangements are
provided for either filtering or distilling the water. In the country,
rain water is pure and good, if the precaution is observed to allow
the first wash from roofs to run to waste. The rain may be either
caught on the roofs, which must always have a clean surface and clean
gutters, or else on artificially prepared catchment areas. As an
example, I quote: "All about the Bermuda Islands one sees great white
scars on the hill slopes. These are dished spaces, where the soil has
been scraped off and the coral rock exposed and glazed with hard
whitewash. Some of these are a quarter acre in size. They catch and
carry the rainfall to reservoirs, for the wells are few and poor, and
there are no natural springs and no brooks." (Mark Twain, "Some
Rambling Notes of an Idle Excursion.")
After the close of the Boer War the English sent about 7,000 Boer
prisoners of war to Bermuda, where they were encamped on some of the
smaller islands of the group, and the entire water supply for the
encampment was obtained by building artificial catchment areas as
described in the above quotation.
[Pg 27]Sometimes, instead of building underground cisterns, rain water is
caught and stored in barrels above ground; if so, these should always
be well covered, not only to avoid pollution, but to prevent the
barrels from becoming mosquito breeders. Cisterns should always be
built with care and made water-tight and impervious. The walls should
be lined with cemented brickwork. In soil consisting of hard pan,
cisterns in some parts of the country are built without brick walls,
the walls of the excavation being simply cemented. I do not approve of
such cheap construction, particularly where the cistern is located
near a privy or cesspool. Pollution of cistern water is often due to
the cracking of the cement lining. Overflows of cisterns should never
be connected with a drain, sewer, or cesspool. Run the overflow into
some surface ditch and provide the mouth with a fine wire screen, to
exclude small animals. It is not recommended to build cisterns in
cellars of houses.
Quality of Water Obtained from Lakes
Lakes yield, as a rule, a supply of clear, bright, and soft water.
This is particularly the case with mountain lakes, because they are at
a distance from sources of contamination. The character of the water
depends upon whether the lake is fed by brooks, that is, by the rain
falling upon the watershed, or also by springs. In one case the water
is surface water exclusively; in the other, it is surface and
underground water[Pg 28] mixed. The purity also depends upon the depth of
the lake and upon the character of its bottom.
Deep lakes furnish a better supply and clearer water than shallow
ones. The solid matter brought into the lake by the brooks or rivers
which feed it does not remain long in suspension, but soon settles at
the bottom, and in this way some lakes acquire the wonderfully clear
water and the beautiful bluish-green color for which they are far
famed.
Strong Winds Dangerous on Lakes
Strong winds or currents at times stir up the mud from the bottom;
hence, in locating the intake, the direction of the prevailing winds
should be considered, if practicable. The suction pipe should always
be placed in deep water, at a depth of at least fifteen to twenty
feet, for here the water is purer and always cooler.
Settlements on the shores of a lake imply danger of sewage
contamination, but the larger the lake, the less is the danger of a
marked or serious pollution, if the houses are scattered and few.
Pools and stagnant ponds are not to be recommended as a source of
supply. In artificially made lakes there is sometimes danger of
vegetable pollution, and trouble with growth of algæ. The bottom of
such lakes should always be cleared from all dead vegetation.
[Pg 29]Surface water may be obtained from brooks flowing through uninhabited
upland or from mountain streams. Such water is very pure and limpid,
particularly where the stream in its downward course tumbles over
rocks or forms waterfalls. But, even then, the watershed of the stream
should be guarded to prevent subsequent contamination. Larger creeks
or rivers are not desirable as a source of supply, for settlements of
human habitations, hamlets, villages, and even towns are apt to be
located on the banks of the river, which is quite generally
used—wrong as it is—as an outlet for the liquid wastes of the
community, thus becoming in time grossly polluted. Down-stream
neighbors are sure to suffer from a pollution of the stream, which the
law should prevent.
The Water of Springs
The water of springs is subterranean, or ground water, which for
geological reasons has found a natural outlet on the surface. We
distinguish two kinds of springs, namely, land or surface springs, and
deep springs. The former furnish water which originally fell as rain
upon a permeable stratum of sand or gravel, underlaid by an impervious
one of either clay or rock. Such water soaks away underground until it
meets some obstacle causing it to crop out on the surface. Such spring
water is not under pressure and therefore cannot again rise. Water
from[Pg 30] deep springs is rain water fallen on the surface of a porous
stratum on a high level, and which passes under an impermeable
stratum, and thus, being under pressure, rises again where an opening
is encountered in the impervious stratum; these latter springs are
really artesian in character.
Deep-spring water is less apt to be polluted than water from surface
or land springs, for it has a chance in its flow through the veins of
the earth to become filtered. Land springs always require careful
watching, particularly in inhabited regions, to prevent surface
contamination.
Not all Spring Water Pure
It is a popular fallacy that all spring water is absolutely pure and
healthful. The above explanation will be helpful in pointing out how,
in some cases, spring water may be nothing but contaminated ground
water. Land springs in uncultivated and uninhabited regions,
particularly in the mountains, yield a good and pure supply. But it is
always advisable, when tapping a spring for water supply, to study its
probable source, and carefully to inspect its immediate surroundings.
The spring should be protected by constructing a small basin, or
reservoir, and by building a house over this. The basin will also
serve to store the night flow of the spring. Before deciding upon a
supply from a spring, its yield should be ascer[Pg 31]tained by one of the
well-known gauging methods. Springs are usually lowest in the months
of October and November, though there is some difference in this
respect between land springs and deep springs. The minimum yield of
the spring determines whether it forms a supply to be relied upon at
all times of the year.
If the spring is located higher than the grounds and buildings to be
supplied, a simple gravity supply line may be carried from it, with
pipes of good size, thus avoiding undue friction in the line, and
stoppages. If lower than the house, the water from the spring must be
raised by some pumping method.
All water found underground owes its origin to the rainfall. If
concealed water is returned to the surface by natural processes it
is called spring water, but if recovered by artificial means it is
called well water.
Different Kinds of Wells
There are numerous kinds of wells, distinguished from one another by
their mode of construction, by their depth from the surface, by the
fact of their piercing an impervious stratum or merely tapping the
first underground sheet of water, and by the height to which the water
in them rises or flows. Thus we have shallow and deep wells,
horizontal wells or infiltration galleries, open or dug wells, tube
wells, non-flowing and flowing wells, bored, drilled, and driven
wells, tile-lined and brick-lined wells, and combination
dug-and-tubular wells.
[Pg 32]When it is desired to provide a water supply by means of wells some
knowledge of the geology of the region, of the character of the strata
and of their direction and dip, will be very useful. In the case of
deep wells, it is really essential. By making inquiries as to similar
well operations in the neighborhood, one may gain some useful
information, and thus, to some extent, avoid guesswork. When one must
drill or bore through rock for a very deep well, which necessarily is
expensive, much money, often uselessly spent, may be saved by
consulting the reports of the State geologist, or the publications of
the United States Geological Survey, or by engaging the services of an
expert hydrogeologist.
"Water Finders"
It used to be a common practice to send for so-called "water finders,"
who being usually shrewd observers would locate by the aid of a hazel
twig the exact spot where water could be found. In searching for water
one sometimes runs across these men even to-day. The superstitious
faith in the power of the forked twig or branch from the hazelnut bush
to indicate by its twisting or turning the presence of underground
water was at one time widespread, but only the very slightest
foundation of fact exists for the belief in such supernatural powers.
In Europe, attention has again, during the past years, been called to
this "method" of finding water,[Pg 33] and it has even received the
indorsement of a very high German authority in hydraulic engineering,
a man well up in years, with a very wide practical experience, and the
author of the most up-to-date hand-book on "Water Supply," but men of
science have not failed to contradict his statements.
Definition of "Ground-water Level"
Water percolating through the soil passes downward by gravity until it
reaches an impervious stratum. The surface of this underground sheet
of water is technically called "water table" or ground-water level.
The water is not at rest, but has a slow and well-defined motion, the
rate of which depends upon the porosity of the soil and also upon the
inclination or gradient of the water table. A shallow well may be
either excavated or driven into this subsoil sheet of water. In
populous districts, in villages, towns, but also near habitations, the
soil from which water is obtained must, of necessity, be impregnated
with organic waste matter. If, in such a surface well, the level of
the water is lowered by pumping, the zone of pollution is extended
laterally in all directions. Ordinary shallow well water should always
be considered "suspicious water." There are two distinct ways in which
surface wells are contaminated: one is by leakage from cesspools,
sewers, privies, etc.; the other, just as important and no less
dangerous, by direct contamination[Pg 34] from the surface. The latter
danger is particularly great in wells which are open at the surface,
and from which water is drawn in buckets or pails. A pump well is
always the safer of the two. Frogs, mice, and other small animals are
apt to fall into the water; dust and dirt settle into it; the wooden
curb and the rotten cover also contribute to the pollution; even the
draw-buckets add to it by reason of being often handled with unclean
hands.
Always avoid, in the country, drinking water from farmers' wells
located near cesspools or privies. Such shallow wells are particularly
dangerous after a long-protracted drought. It is impossible to define
by measurement the distance from a cesspool or manure pit at which a
well can be located with safety, for this depends entirely upon local
circumstances. Contamination of shallow wells may, in exceptional
cases, be avoided by a proper location of the well with reference to
the existing sources of impurity. A well should always be placed
above the source of pollution, using the word "above" with reference
to the direction in which the ground water flows.
Precautions Regarding Wells
Other precautions to be observed with reference to surface wells are
the following:
Never dig a well near places where soil contamination has taken or is
taking place. Line the sides of the[Pg 35] well with either brick, stone, or
tile pipe, cemented in a water-tight manner to a depth of at least
twenty feet from the surface, so that no water can enter except from
the bottom, or at the sides near the bottom.
Raise the surface at the top of the well above the grade; arrange it
so as to slope away on all sides from the well; cover it with a
flagstone, and cement the same to prevent foreign matters from
dropping into the well; make sure that no surface water can pass
directly into the well; make some provision to carry away waste water
and drippings from the well.
Shallow wells made by driving iron tubes with well points into the
subsoil water are preferable to dug wells. Use a draw-pump in
preference to draw buckets.
When a well is sunk through an impervious stratum to tap the larger
supply of water in the deeper strata, we obtain a "deep well." Water
so secured is usually of great purity, for the impurities have been
filtered and strained out by the passage of the water through the
soil. Moreover, the nature of the construction of deep wells is such
that they are more efficiently protected against contamination, the
sides being made impervious by an iron-pipe casing. In some rare
cases, even deep wells show pollution due to careless jointing of the
lining, or water follows the outside of the well casing until it
reaches the deeper water sheet. Deep wells usually yield more water
than shallow driven wells, and the supply increases perceptibly when
the water level in the well is lowered[Pg 36] by pumping. While surface
wells draw upon the rainfall percolating in their immediate vicinity,
deep wells are supplied by the rainfall from more remote districts.
Deep wells are either non-flowing or flowing wells. When the
hydrostatic pressure under which the water stands is sufficient to
make it flow freely out on the surface or at the mouth of the well, we
have a flowing, or true artesian well.
Character of Water From Deep Wells
Water from deep wells is of a cool and even temperature. It is usually
very pure, but in some cases made hard by mineral salts in the water.
Sulphur is also at times present, and some wells on the southern
Atlantic coast yield water impregnated with sulphur gases, which,
however, readily pass off, leaving the water in good condition for all
uses. In many cases the water has a taste of iron. No general rule can
be quoted as to the exact amount of water which any given well will
yield, for this depends upon a number of factors. Increasing the
diameter of very deep wells does not seem to have any marked effect in
increasing the supply. Thus, a two-foot well gives only from fifteen
to thirty per cent more water than a three-inch-pipe well. This rule
does not seem to apply to shallow wells of large diameter, for here we
find that the yield is about in proportion to the diameter of the
well.
It is interesting to note the fact that wells located[Pg 37] near the
seashore, within the influence of the tide, vary in the hourly flow.
According to Dr. Honda, of the University of Tokio, there is "a
remarkable concordance between the daily variations in the level of
the tides and the water level in wells." The water in wells one mile
from the seashore was found to stand highest at high tide. The daily
variation amounted to sixteen centimeters, or a little over six
inches. A similar variation was observed by the writer in some flowing
wells located on the north shore of Long Island. Dr. Honda found also
that the water level in wells varied with the state of the barometer,
the water level being lowered with a rise in the barometer.
Where a large supply is wanted a series of wells may be driven, and,
as the expense involved is considerable, it is always advisable to
begin by sinking a smaller test well to find out whether water may be
had.
Ground water may also be recovered from water-bearing strata by
arranging horizontal collecting galleries with loose-jointed sides
through which the water percolates. Such infiltration galleries have
been used in some instances for the supply of towns and of
manufacturing establishments, but they are not common for the supply
of country houses.
Laws Regulating Appropriation of Water
Persons contemplating the establishment of a system of water supply in
the country should bear in mind that the taking of water for supply
purposes is,[Pg 38] in nearly all States, hemmed in by legal restrictions.
The law makes a distinction between subterranean waters, surface
waters flowing in a well-defined channel and within definite banks,
and surface waters merely spread over the ground or accumulated in
natural depressions, pools, or in swamps. There are separate and
distinct laws governing each kind of water. It is advisable, where a
water-supply problem presents itself, to look up these laws, or to
consult a lawyer well versed in the law of water courses.
If it is the intention to take water from a lake, the property owner
should make sure that he owns the right to take such water, and that
the deed of his property does not read "to high-water mark only." The
owner of a property not abutting on a lake has no legal right to
abstract some of the water from the lake by building an infiltration
gallery, or a vertical well of large diameter intended for the same
purpose. On the other hand, an owner may take subterranean water by
driving or digging a well on his own property, and it does not matter,
from the law's point of view, whether by so doing he intercepts partly
or wholly the flow of water in a neighboring well. But, if it can be
shown that the subterranean water flows in a well-defined channel, he
is not permitted to do this. The water from a stream cannot be
appropriated or diverted for supply or irrigation purposes by a single
property holder without the consent of the other riparian owners, and
without compensation to them.
[Pg 39]
CHAPTER II
Appliances for Distributing Water
We have so far discussed only the various sources of potable water. We
must now turn our attention to the mechanical means for making it
available for use, which comprise appliances for lifting, storing,
conveying, distributing, and purifying the water.
The location of the source of supply with reference to the buildings
and grounds decides generally the question whether a gravity supply is
feasible or whether water must be pumped. The former is desirable
because its operating expenses are almost nothing, but it is not
always cheapest in first cost. Rather than have a very long line of
conduit, it may be cheaper to pump water, particularly if wind or
water power, costing nothing, can be used.
Machines for Pumping
When it becomes necessary to pump water, there are numerous machines
from which to choose; only the more important ones will be considered.
We may use pumps operated by manual labor, those run by ani[Pg 40]mal power,
pumping machinery using the power of the wind or that of falling or
running water; then there are hot-air, steam, and electric pumps,
besides several forms of internal-combustion engines, such as gas,
gasoline, and oil engines. Each has advantages in certain locations
and under certain conditions.
Of appliances utilizing the forces of Nature, perhaps the simplest
efficient machine is the hydraulic ram. While other machines for
lifting water are composed of two parts, namely, a motor and a pump,
the ram combines both in one apparatus. It is a self-acting pump of
the impulse type, in which force is suddenly applied and discontinued,
these periodical applications resulting in the lifting of water.
Single-acting rams pump the water which operates them; double-acting
rams utilize an impure supply to lift a pure supply from a different
source.
The advantages of the ram are: it works continuously, day and night,
summer and winter, with but very little attendance; no lubrication is
required, repairs are few, the first cost of installation is small.
Frost protection, however, is essential. The disadvantages are that a
ram can be used only where a large volume of water is available. The
correct setting up is important, also the proper proportioning in size
and length of drive and discharge pipes. The continual jarring tends
to strain the pipes, joints, and valves; hence, heavy piping and
fittings are necessary. A ram of the improved type raises water from
twenty-[Pg 41]five to thirty feet for every foot of fall in the drive pipe,
and its efficiency is from seventy to eighty per cent.
Running water is a most convenient and cheap power, which is often
utilized in water wheels and turbines. These supply power to run a
pump; the water to be raised may come from any source, and the pump
may be placed at some distance from the water wheel. Where sufficient
fall is available—at least three feet—the overshot wheel is used. In
California and some other Western States an impulse water wheel is
much used, which is especially adapted to high heads.
Windmills Used for Driving Pumps
The power of the wind applied to a windmill is much used for driving
pumps. It is a long step forward from the ancient and picturesque
Dutch form of windmill, consisting of only four arms with cloth sails,
to the modern improved forms of wheels constructed in wood and in
iron, with a large number of impulse blades, and provided with devices
regulating the speed, turning the wheel out of the wind during a gale,
and stopping it automatically when the storage tank is filled. The
useful power developed by windmills when pumping water in a moderate
wind, say of sixteen miles an hour velocity, is not very high, ranging
from one twenty-fifth horse-power for an eight and one-half foot wheel
to one and one-half horse-[Pg 42]power for a twenty-five foot wheel. The
claims of some makers of windmills as to the power developed should be
accepted with caution.
The chief advantage is that, like a ram, the windmill may work night
and day, with but slight attention to lubrication, so long as the wind
blows. But there are also drawbacks; it requires very large storage
tanks to provide for periods of calm; the wheel must be placed
sufficiently exposed to receive the full wind force, either on a tower
or on a high hill, and usually this is not the best place to find
water. Besides, a windmill tower, at least the modern one, is not an
ornamental feature in the landscape. It is expensive when built
sufficiently strong to withstand severe winter gales. During the hot
months of the year, when the farmer, the gardener, and the coachman
require most water, the wind is apt to fail entirely for days in
succession.
The Use of Engines
If water is not available, and wind is considered too unreliable,
pumping must be accomplished by using an engine which, no matter of
what form or type, derives its energy from the combustion of fuel, be
the same coal, wood, charcoal, petroleum or kerosene, gas, gasoline,
or naphtha. The use of such pumping engines implies a constant expense
for fuel, operation, maintenance, and repairs. In some modern forms of
engines this expense is small, notably so in the oil engine,[Pg 43] and also
in the gasoline engine; hence these types have become favorites.
Advantages of Pumping Engines
An advantage common to all pumping engines is that they can be run at
any time, not like the windmill, which does not operate in a light
breeze, nor like the ram, which fails when the brook runs low.
Domestic pumping engines are built as simple as possible, so that the
gardener, a farm hand, or the domestic help may run them. Skill is not
required to operate them, and they are constructed so as to be safe,
provided ordinary intelligence is applied.
In using a fuel engine it is desirable, because of the attendance
required, to take a machine of such capacity and size that the water
supply required for two or three days may be pumped to the storage
tank in a few hours.
Expansive Force of Heated Air Utilized
A favorite and extensively used type of domestic pump is the hot-air
engine, in which the expansive force of heated air is used to do
useful work. Among the types are simple and safe machines which do not
easily get out of order. They are started by hand by giving the fly
wheel one or more revolutions. If properly taken care of they are
durable and do not require expensive repairs.
[Pg 44]
Gas and Gasoline Engines
In gas engines power is derived from the explosion of a mixture of gas
and air. Where a gas supply is available, such engines are very
convenient, for, once started, they will run for hours without
attention. They are economical in the consumption of gas, and give
trouble only where the quality of gas varies.
Owing to the unavailability of gas on the farm and in country houses,
two other forms of pumping engines have been devised which are
becoming exceedingly popular. One is the gasoline, the other is the
oil engine. Both resemble the gas engine, but differ from it in using
a liquid fuel which is volatilized by a sprayer. Gasoline engines are
now brought to a high state of perfection.
Kerosene or Crude Oil as Fuel
In recent years, internal-combustion engines which use heavy kerosene
or crude oil as fuel have been introduced. These have two palpable
advantages: first, they are safer than gasoline engines; second, they
cost less to run, for crude oil and even refined kerosene are much
cheaper than gasoline. Oil engines resemble the gas and gasoline
engines, but they have larger cylinders, because the mean effective
pressure evolved from the explosion is much less than that of the
gasoline engines.
[Pg 45]Oil engines for pumping water are particularly suitable in regions
where coal and wood cannot be obtained except at exorbitant cost.
Usually, the engine is so built as to be adapted for other farm work.
It shares this advantage with the gasoline engine. Oil engines are
simple, reliable, almost automatic, compact, and reasonable in first
cost and in cost of repairs. There are many forms of such engines in
the market. To be successful from a commercial point of view, an oil
engine should be so designed and built that any unskilled attendant
can run, adjust, and clean it. The cost of operating them, at eight
cents per gallon for kerosene, is only one cent per hour per
horse-power; or one-half of this when ordinary crude oil is used. The
only attention required when running is periodical lubrication and
occasional replenishing of the oil reservoir. The noise of the
exhaust, common to all engines using an explosive force, can be
largely done away with by using a muffler or a silencer. The smell of
oil from the exhaust likewise forms an objection, but can be overcome
by the use of an exhaust washer.
Steam and Electric Pumps
The well-known forms of steam-pumping engines need not be considered
in detail, because high-pressure steam is not often available in
country houses. Where electric current is brought to the building, or
generated for lighting purposes, water may be pumped[Pg 46] by an electric
pump. Electric motors are easy and convenient to run, very clean, but
so far not very economical. Electric pumps may be arranged so as to
start and stop entirely automatically. Water may be pumped, where
electricity forms the power, either by triplex plunger pumps or by
rotary, screw, or centrifugal pumps.
Pumps Worked by Hand
Space forbids giving a description of the many simpler devices used
for lifting water. In small farmhouses lift and force pumps worked by
hand are now introduced, and the old-fashioned, moss-covered
draw-bucket, which is neither convenient nor sanitary, is becoming a
relic of past times.
Reservoirs and Storage Tanks
The water pumped is stored either in small masonry or earth
reservoirs, or else in storage tanks of either wood, iron, or steel,
placed on a wood or steel tower. Wooden tanks are cheap but unsightly,
require frequent renewal of the paint, and give trouble by leaking,
freezing, and corrosion of hoops. In recent years elevated tanks are
supplanted by pressure tanks. Several such systems, differing but
little from one another, are becoming quite well known. In these water
is stored under suitable pressure in air-tight tanks, filled partly
with water and partly with air.
[Pg 47]
A Simple Pressure System
One system consists of a circular, wrought-steel, closed tank, made
air- and water-tight, a force pump for pumping water into the tank,
and pipe connections. The tank is placed either horizontally or
vertically in the basement or cellar, or else placed outdoors in the
ground at a depth below freezing. Water is pumped into the bottom of
the tank, whereby its air acquires sufficient pressure to force water
to the upper floors.
This simple system has some marked advantages over the outside or the
attic tank. In these, water gets warm in summer and freezes in winter.
Vermin and dust get into the tank, and the water stagnates. In the
pressure tank, water is kept aërated, cool, and clean.
Another pressure tank has an automatic valve, controlled by a float
and connected with suction of pump. It prevents the tank from becoming
water-logged by maintaining the correct amount of air inside.
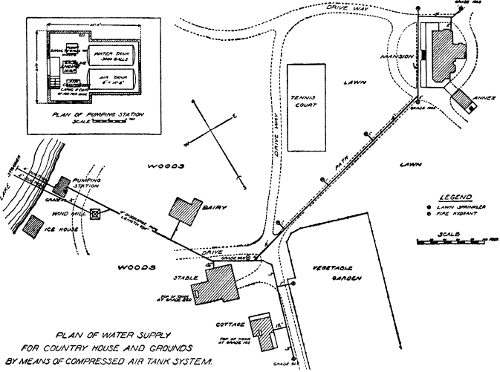
An Ideal System for a Country House
Still another system using pressure tanks is more complete than either
of the others, comprising engine, pump, air compressor, a water tank,
and also an air tank. It is best described by a recent example
constructed from plans and under the direction of the writer. The
buildings supplied with water comprise the mansion, the stable, the
cottage, and a dairy, and[Pg 48]
[Pg 49] the pumping station is placed near the
shore of the lake from which the supply is taken. See Figs. 1 and 2.
 Fig. 2.
Fig. 2.
PRESSURE-TANK PUMPING STATION.
Interior view of pumping station of compressed air-tank system (see
plan on opposite page) showing 3,000 gallon water tank, air tank of
150 pounds pressure and 10 horse-power gasoline engine.
The pump house is about 20 feet by 27 feet, and contains a
water-storage tank 6 feet in diameter and 131/2 feet long, of a
capacity of 3,000 gallons; an air tank of same dimensions as the water
tank, holding air under 150 pounds pressure; a 10 horse-power gasoline
engine, direct-connected, by means of friction clutch,[Pg 50] with an air
compressor and also with a triplex pump of 75 gallons capacity per
minute.
The water in the tank is kept under 75 pounds pressure, and at the
hydrant near the house, located about 100 feet above the pumping
station, there is an available pressure of 33 pounds. The last drop of
water flows from the water tank under the full pressure of 75 pounds
at the pumping station. The suction pipe into the lake is 4 inches and
is provided with well strainers to prevent clogging.
The cost of pumping water by this system is quite reasonable. The
gasoline engine requires per horse-power per hour about 11/4 gallons
of gasoline, and at sixteen cents per gallon this makes the cost for
1,000 gallons pumped about five cents. To this expense should,
however, be added the cost of lubricating oil, repairs, amount for
depreciation, and the small cost for labor in running the engine.
Water pipes forming a distribution system should always be chosen
generous in diameter, in order to avoid undue loss of pressure by
friction. Where fire hydrants are provided, the size of the water main
should not be below four inches. All branches should be controlled by
shut-offs, for which the full-way gate valves are used in preference
to globe valves. Pipe-line material is usually galvanized,
screw-jointed wrought iron for sizes up to four inches.
In conclusion, a word about water purification. Where the quality of
the water supply is not above[Pg 51] suspicion it may be improved by
filtration. A filter should never be installed without the advice of a
qualified expert, for there are numerous worthless devices and few
really efficient ones. Where a filter is not available, the water used
for drinking should be boiled or sterilized if there is the slightest
doubt as to its wholesomeness.
[Pg 52]
CHAPTER III
Purifying Water by Copper Sulphate
From the standpoint of the health of the community, the most vital
problem is to get pure water. Almost equally important, when comfort
and peace of mind is considered, is the procuring of sweet water. The
wise owner of a country home looks to the water supply upon which his
family is dependent. The careful farmer is particular about the water
his stock, as well as his family, must drink. But careless persons
constitute the large majority. Most people in the city and in the
country pay no attention to their drinking water so long as it "tastes
all right."
Clear Water Often Dangerous
Some years ago the inhabitants of Ithaca, N. Y., furnished a pitiful
example of this foolhardy spirit. For a year previous to the breaking
out of the typhoid epidemic, the public was warned, through the local
and the metropolitan press, of the dangerous condition of Ithaca's
water supply. Professors of Cornell College joined in these warnings.
But the people gave no heed, probably because the water was clear
and its taste sweet and agreeable. As was the case in this instance,[Pg 53]
bacteria are tolerated indefinitely, and it is only an alarming
increase in the death rate that makes people careful. Then they begin
to boil the water—when it is too late for some of them.
Bad-Tasting Water not Always Poisonous
But let the taste become bad and the odor repulsive, and a scare is
easily started. "There must be dead things in the water, or it
wouldn't taste so horrible," is the common verdict. Some newspaper
seizes upon the trouble and makes of it a sensation. The ubiquitous
reporter writes of one of "the animals" that it "looks like a wagon
wheel and tastes like a fish." With such a remarkable organism
contaminating one's drink no wonder there is fear of some dread
disease. The water is believed to be full of "germs"; whereas the
pollution is entirely due to the presence of algæ—never poisonous to
mankind, in some cases acting as purifying agents, but at certain
seasons of the year imparting a taste and odor to the water that
cannot be tolerated.
Algæ—what are they? They are aquatic plants. Algæ are not to be
confounded with the water vegetation common to the eye and passing by
the term weeds. Such plants include eelgrass, pickerel weed, water
plantain, and "duckmeat"—all of which have roots and produce flowers.
This vegetation does not lend a bad odor or taste to the water. In
itself it is harm[Pg 54]less, although it sometimes affords a refuge for
organisms of a virulent type.
But when the aquatic vegetation of the flowering variety is eliminated
from consideration, there still remains a group of water plants called
algæ. They comprise one-fifth of the known flowerless plants. They are
the ancestors of the entire vegetable kingdom. Those whose habitat is
the sea number the largest plants known in nature. Certain forms found
in the Pacific are supposed to be 800 feet in length; others are
reported to be 1,500 feet long. The marine variety are familiar as the
brown kelps and the wracks, which are very common along our Northern
coast.
Plants Which Pollute Drinking Water
The fresh-water algæ are usually grass green in color. This green
variety is often seen as a spongy coating to the surface of stagnant
pools, which goes by the name of "frog spawn" or "pond scum." One of
this description, Spirogyra, has done thousands of dollars' worth of
damage by smothering the life out of young water-cress plants in
artificial beds constructed for winter propagation. When the cress is
cut the plants are necessarily left in a weakened condition, and the
algæ form a thick mat over the surface of the water, thus preventing
the growth of the cress plants and oftentimes killing them. The
absolute necessity of exterminating these algæ led to the perfection
of the copper-purification process.
[Pg 55]It is, however, a variety of algæ not easily detected that
contaminates the water. So long as they are in a live, healthy
condition they benefit drinking water by purifying it. Indeed, some
scientists have attributed the so-called self-purification of a stream
entirely to the activities of these plants. Of such, one form,
Chlamydomonas, is bright grass green in appearance. But the largest
group—the plants which have the worst reputation as polluters of
drinking water—are popularly known as the "blue-green algæ"
(Schizophyceæ). The common name tells the color of these plants,
although there are exceptions in this respect, some of them showing
shades of yellow, brown, olive, chocolate, and purplish red. This
variety of algæ flourishes in the summer months, since a relatively
high temperature and shallow stagnant water favor its germination. If
the pond begins to dry up, the death of the organisms takes place, and
the result is a most disagreeable, persistent odor which renders the
water unfit for drinking purposes. This result is chemically due to
the breaking down of highly organized compounds of sulphur and
phosphorus in the presence of the large amount of nitrogen contained
in these plants. Decomposition is not necessary for some of the blue
greens to give off a bad odor, however. A number of them, on account
of their oil-content, produce an odor when in a healthy condition that
is sometimes likened to raw green corn or to nasturtiums, but usually
it cannot be so pleasantly described.
[Pg 56]The Department of Agriculture has been able to solve the problem of
exterminating algæ from water supplies.[1] The department has done
more; for it has succeeded in perfecting a method by which a reservoir
contaminated with typhoid or other pathogenic bacteria can be
purified. The work was begun with an inquiry into the extent of the
trouble from algal pollution. Letters were addressed to some five
hundred engineers and superintendents of water companies scattered all
over the United States. The replies, which came from almost every
State in the Union, were burdened with one complaint—"Algæ are our
worst pest"; and with one prayer—"Come over into Macedonia, and help
us."
A Cheap and Available Remedy for Algæ
Convinced of the need of earnest work, extensive laboratory
experiments were inaugurated. The problem presented was this: the
remedy must not only be readily available, but it must be cheap, that
advantage may be taken of it by the poorest communities, as well as by
those owning large reservoirs. Above all, the remedy must be
absolutely harmless to man; the poison used to exterminate algæ must
not in any way affect the water drinkers. A large number of
sub[Pg 57]stances were used in the experiments before the final decision
rested with copper sulphate. This salt is very poisonous to algæ. On
the other hand, copper in solution just strong enough to destroy algal
growth could not possibly injure man; in fact, the temporary presence
of such a small amount of copper in drinking water could not be
detected.
A Practical Demonstration
The results in the laboratory being successful, the next step was to
make a practical demonstration of the value of the method. This was
first done in the fall of 1901. At Ben, Va., water cress is grown in
large quantities during the winter, when it is a valuable market crop.
Dams are constructed across a stream in such a manner as to enable the
maintenance of a water level not too high for the growth of plants;
when a freeze is threatened the plants can be flooded. In the cress
beds selected for the experiments the water is obtained from a thermal
spring whose temperature throughout the year is about 70° F. This
temperature is particularly favorable to the growth of "frog spawn."
After the cress was cut for market, the algæ frequently developed so
rapidly as to smother the life out of the weakened plants. When this
occurred, the practice was to rake out both water cress and algæ and
reset the entire bed. This was not only expensive; half the time it
failed to exterminate the pest. It was,[Pg 58] therefore, most desirable to
devise a method of ridding the bed of algal growth without injuring
the cress.
The Copper-sulphate Method Tested
Here the copper-sulphate method was put to a practical test. At the
outset a strong solution was sprayed on the algæ which coated the
surface of the pond. This only killed the algal growth with which the
particles of copper came in contact and left the main body of algæ
unaffected. Then trial was made of dissolving the copper directly in
the water, and the result was most satisfactory. The solution used was
that of 1 part of copper to 50,000,000 parts of water.
Growers need have no trouble in the future. They need have no fear of
employing the method, as the copper solution required for killing the
algæ could not possibly injure water cress, provided ordinary care is
used in the work. As to the frequency of treatment required, one or
two applications a year will generally be found sufficient, as this
letter, received from the manager of the Virginia company, goes to
show:
"The 'moss' has given me no trouble at all this winter; in fact, I
have for six months had to resort to the copper sulphate only once....
All the conditions were favorable last fall and early winter for a
riot of 'moss,' but it did not appear at all until just a few days
ago, and then yielded to treatment much more readily than it did when
I first began to use the[Pg 59] copper." This letter was written over three
years after Dr. Moore made his experiment in these cress beds.
Satisfied with the results attained in exterminating algal growth in
water-cress beds, attention was next given to reservoirs. Some fifty
water supplies were treated during the summer of 1904, and in every
case success attended the copper cure. In one respect the results were
surprising. It was found that in practice the copper-sulphate method
worked better than in theoretic experimentation; results in large
reservoirs were more pronounced than in the laboratory. In fact, it
developed that the solution necessary to kill algæ in the laboratory
must contain from five to twenty times as much copper as that
contained in a solution which will exterminate algal growth in its
natural habitat. This is not easily explained, if it can be explained
at all. The test reason advanced is that only the most resistant
organisms stand transplanting to an artificial environment. But, after
all, the important point is that the new method works better in
practice than was expected.
A Prescription for the Copper Cure
Thus the department is able to announce that the process is no longer
in the experimental stage, and also to say what conditions must be
known in determining the proper quantity of copper sulphate for
destroying algæ, together with a prescription for the copper cure.[Pg 60]
Here it is, for the benefit of careful persons who will use the method
with proper intelligence: "The importance of knowing the temperature
of the contaminated water is second only to the necessity of knowing
the organism present. With increase of temperature the toxicity of a
given dilution increases, and vice versa. Assuming that 59° F. is
the average temperature of reservoirs during the seasons when
treatment is demanded, the quantity of copper should be increased or
decreased approximately 2.5 per cent for each degree below or above
59° F.
"Similar scales should be arranged for the organic content and the
temporary hardness of the water. With the limited data at hand it is
impracticable to determine these figures, but an increase of 2 per
cent in the quantity of copper for each part per 100,000 of organic
matter and an increase of 0.5 to 5 per cent in the proportion of
copper for each part per 100,000 of temporary hardness will possibly
be found correct. The proper variation in the increase due to hardness
will depend upon the amount of dissolved carbon dioxide; if very
small, 5 per cent increase is desirable; if large, 0.5 per cent is
sufficient."
The information in this prescription is to be used in connection with
a table[2] published by the Department of Agriculture. This table
gives the number of parts of water to one part of copper sulphate
necessary to kill the various forms of algæ which are listed. The[Pg 61]
formulæ vary from 1 part of copper to 100,000 parts of water,
necessary to destroy the most resistant and very rare forms (three of
these are listed), to 1 part of copper in 25,000,000 parts of water,
which is a sufficiently strong solution to exterminate Spirogyra,
the cress-bed pest. By far the majority of forms do not require a
solution stronger than that of 1 part of copper to 1,000,000 parts of
water.
What the Agricultural Department is Doing
It is true that the department is not now holding out, directly, a
helping hand to the owner of a country place, or to the farmer, in
this campaign of purifying drinking water. In the first place, the
greatest good of the greatest number demands that large reservoirs,
which supply a great number of people with drinking water, ought to be
considered first. Such supplies, moreover, are most frequently
contaminated. Where fifty reservoirs were treated last summer, ten
times that number will be "cured" this summer. It will be readily
seen, therefore, that in conducting such a large number of
experiments—considering preliminary reports, prescribing for
treatment, and keeping proper account of results—the department, with
a limited force and limited facilities, has its hands more than full.
More important still, there is an absolute need of the services of
some expert on the ground. While an[Pg 62] algologist is a functionary not
generally employed by water companies—in fact, a man trained in the
physiology of algæ is difficult to find—nevertheless, it is highly
important, as the department views it, to have the coöperation of an
expert versed to some extent in the biological examination of drinking
water. In other words, the copper cure is not a "patent medicine,"
with printed directions which any person could follow. Intelligence
and care are absolutely essential in the use of this treatment.
Furthermore, each case must be treated as a distinct and separate
case, as a physician would treat a patient.
Actual Purification Simple
Suppose, however, an owner of a country place, which is dependent upon
a fresh-water pond for its water supply, finds that his drinking water
is contaminated, that the taste and odor are such as to render the
water unfit for use. There is no reason why he should not treat the
supply, provided he is properly careful. When the nature of the
polluting organism is definitely determined and the average
temperature of the water observed, then the necessary formula can be
decided upon. First, of course, the pond must be plotted, the depth
found, and the capacity computed. The department will willingly
furnish data for this purpose, together with blanks upon which to
submit details as to contaminating organisms and water tem[Pg 63]perature,
to any applicant. Once the proper solution is determined upon, the
actual work of purification is most simple. In the following
directions the department outlines the most practicable method of
introducing the copper sulphate into a water supply:
Directions for the Copper Cure
"Place the required number of pounds of copper sulphate in a coarse
bag—gunny sack or some equally loose mesh—and, attaching this to the
stern of a row-boat near the surface of the water, row slowly back and
forth over the reservoir, on each trip keeping the boat within ten to
twenty feet of the previous path. In this manner about a hundred
pounds of copper sulphate can be distributed in one hour. By
increasing the number of boats, and, in the case of deep reservoirs,
hanging two or three bags to each boat, the treatment of even a large
reservoir may be accomplished in from four to six hours. It is
necessary, of course, to reduce as much as possible the time required
for applying the copper, so that for immense supplies, with a capacity
of several billion gallons, it would probably be desirable to use a
launch, carrying long projecting spars to which could be attached bags
containing several hundred pounds of copper sulphate.
"The substitution of wire netting for the gunny-sack bag allows a more
rapid solution of the sulphate, and the time required for the
introduction of the salt[Pg 64] may thus be considerably reduced. It is best
to select as warm a day for treatment as circumstances will permit."
Cost of the Treatment
Not difficult, one would say. No—when the proper solution is
determined; to reach that determination is the difficulty. That the
method can be tried "at home" is proved by the results obtained by the
owner of a country home in the vicinity of New York. Tired of
consulting engineers, who looked at his water supply, informed him
that they could do nothing, and then charged him a big fee (to one he
paid $250), this owner resorted to the copper-sulphate treatment. The
cure cost the man just $2—but let his letter to the department tell
the story:
"My place in the country is located at Water Mill, in the township of
Southampton, in Long Island. I purchased it in April, 1902, and was
largely influenced in selecting this piece of land by the beauty of a
pond which bounds it on the east. This little body of water covers
about two acres, is fed by numerous springs, and discharges into Mecox
Bay, the southern boundary of the land. When I bought the place the
pond was filled with clear water. About the middle of the following
June algæ began to show, and in August the surface was almost entirely
covered by the growth. The odor was offensive, and myriads of small
insects hovered over the masses of algæ much of the time.[Pg 65] I consulted
two engineers interested in the storage of water, and they told me
that nothing could be done. The condition was so objectionable that I
planned to plant a thick hedge of willows along the bank to shut off
the view of the pond from the house.... I examined the pond on June
15th and found large masses of algæ covering an area several hundred
feet in length and from twenty to forty feet in width. No
microscopical examination was made of the growth, but I was informed
that it seemed to be largely composed of filaments of Spirogyra and
other Confervæ. On June 18th the treatment was begun.... In one week
the growth had sunk and the pond was clear water. I examined the pond
September 15th and found it still clear.
"The use of the sulphate of copper converted an offensive
insect-breeding pond into a body of beautifully clear water. The pond
was full of fish, but the copper did not seem to harm them."
Effect of Copper Sulphate on Fish
Native trout were not injured when the large reservoir at Cambridge,
N. Y., was purified by the copper treatment. A slightly different
result, in this respect, was reported from Elmira, N. Y., however.
Part of the report is as follows:
"The effect of the copper-sulphate treatment on the different animal
life was as follows: numerous 'polly[Pg 66]wogs' killed, but no frogs;
numerous small (less than two inches long) black bass and two large
ones (eight inches long) killed; about ten large 'bullheads' were
killed, but no small ones; numerous small (less than two inches long)
'sunfish' were killed, but no large ones.
"The wind brought the dead fish to the corners of the reservoir, and
it was very little trouble to remove them. No dead fish were seen
twenty-four hours after completion of the treatment."
The injury done by copper sulphate to fish is a more serious matter
than was at first supposed. Brook trout are, apparently, the least
resistant to the salt. A Massachusetts trout pond stocked with
eight-inch trout lost forty per cent as a result of the introduction
of a strong solution of copper sulphate. The Bureau of Fisheries is
working in conjunction with the Division of Plant Physiology in this
matter, and it is hoped to secure reliable information. In the
meantime, owners of ponds stocked with game fish would do well to take
great care before resorting to the copper cure for algæ—that is, if
they hesitate to lose a part of the fish.
Water May be Drunk During Treatment
When a pond or reservoir is treated with the proper amount of copper
sulphate to remove algæ—except in the case of the few very resistant
forms requiring a stronger solution than 1 part of copper to
1,000,000[Pg 67] parts of water—there is no need of discontinuing the use
of the water supply during treatment; the water may be drunk with
impunity. But when water known to be polluted with pathogenic bacteria
is sterilized by means of copper sulphate in strong solution, it is
just as well to discontinue the use of the water for drinking purposes
for not more than twenty-four hours. Even then, this is an overcareful
precaution rather than a necessity.
Experiments conducted with great care and thoroughness demonstrate
that at room temperature, which is near the temperature of a reservoir
in summer, a solution of 1 part of copper to 100,000 parts of water
will destroy typhoid bacteria in from three to five hours. Similar
experiments have proved that a copper solution of like strength is
fatal to cholera germs in three hours, provided the temperature is
above 20° F. As was the case with algæ, bacteria were found to be much
more sensitive to copper when polluting water than when grown in
artificial media.
The Use of Copper Tanks
The toxic effect of metallic copper upon typhoid bacteria in water
gives some hints as to prevention of the disease by the use of copper
tanks. This should not altogether take the place of the boiling of the
water; it is useful in keeping it free from contamination, although
water allowed to stand in copper re[Pg 68]ceptacles for a period of from
twenty-four to forty-eight hours at room temperature would be
effectively sterilized, no matter what its contamination and no matter
how much matter it held in suspension. But in order to insure such
results the copper must be kept thoroughly clean. This polishing is
not, as was popularly supposed, to protect the consumer from "copper
poisoning," but to prevent the metal from becoming so coated with
foreign substances that there is no contact of the copper with the
water, hence no antiseptic quality.
Dr. Henry Kreamer, of Philadelphia, proved that within four hours
typhoid germs were completely destroyed by the introduction into the
polluted water of copper foil.
"Granting the efficiency of the boiling of water for domestic
purposes, I believe that the copper-treated water is more natural and
more healthful.... The intestinal bacteria, like colon and typhoid,
are completely destroyed by placing clean copper foil in the water
containing them.
"Pending the introduction of the copper treatment of water on a large
scale, the householder may avail himself of a method for the
purification of drinking water by the use of strips of copper foil
about three and one-half inches square to each quart of water, this
being allowed to stand overnight, or from six to eight hours at the
ordinary temperature, and then the water drawn off or the copper foil
removed."
[Pg 69]Although a splendid antiseptic, copper in weak solution is not
harmful, no more so than the old copper utensils used by our
forefathers were harmful. Undoubtedly they were of benefit, and the
use of them prevented the growth of typhoid and other bacteria. People
of to-day might well go back to copper receptacles for drinking
water.
[Pg 70]
CHAPTER IV
Ridding Stagnant Water of Mosquitoes
Because of the serious and often fatal injury it inflicts on man, the
most dangerous animal known is the mosquito. Compared with the evil
done by the insect pest, the cobra's death toll is small. This
venomous serpent is found only in hot countries, particularly in
India, while mosquitoes know no favorite land or clime—unless it be
Jersey. Arctic explorers complain of them. In Alaska, it is recorded
by a scientist that "mosquitoes existed in countless millions, driving
us to the verge of suicide or insanity." A traveler on the north shore
of Lake Superior, when the snow was several feet deep, and the ice on
the lake five feet in thickness, relates that "mosquitoes appeared in
swarms, literally blackening the banks of snow in sheltered places."
Mosquitoes Responsible for Yellow Fever
In the temperate zone this evil-breeding insect was, until recent
years, considered more in the light of an exasperating pest. It is now
known, however, that[Pg 71] malaria is due entirely to the bites of
mosquitoes. But it is in the tropical countries that their deadliest
work is done. There, it has been proved beyond question, the
mosquitoes are responsible for the carriage of yellow fever. If, in a
yellow-fever ridden region, one were to live entirely in an inclosure,
carefully protected with proper screens—as certain entomologists
did—there practically would be no danger from the dread disease, even
if all other precautions were neglected.
Effect of a Mosquito Bite
The crime committed by the mosquito against its innocent victim, man,
is more in the nature of manslaughter than of murder, according to the
authorities. There is no premeditated malice. "A mosquito bites
primarily to obtain food," says a leading entomologist; "there is
neither malice nor venom in the intent, whatever there may be in the
act." There isn't great comfort in the intelligence conveyed by the
scientist, nor in his further observation:
"Theoretically, there would seem to be no reason why there should be
any pain from the introduction of the minute lancets of the insects,
and the small amount of bloodletting is usually a benefit rather than
otherwise. Unfortunately, however, in its normal condition the human
blood is too much inclined to clot to be taken unchanged into the
mosquito stomach;[Pg 72] hence, when the insect bites, a minute droplet of
poison is introduced, whose function it is to thin out the fluid and
make it more suitable for mosquito digestion. It is this poison that
sets up the inflammation and produces the irritation or swelling....
The pain is caused entirely by the action of the poison in breaking up
the blood, and, as the first act of a biting mosquito is to introduce
the poison into the wound, the pain and inflammation will be the same,
whether the insect gets its meal or not. In fact, it has been said
that if a mosquito be allowed to suck its fill and then fly, the bite
will not itch, and there is just a basis of justification for this."
To make a scientific inquiry into the habits of the mosquito, and to
do it patiently, one should be far from the maddening swarms, or at
least effectively screened in. Then it would be possible to believe
the statement of the Government's entomologist that not "one mosquito
in a million" ever gets the opportunity to taste the blood of a
warm-blooded animal. As proof of this there are, in this country,
great tracts of marshy land never frequented by warm-blooded animals,
and in which mosquitoes are breeding in countless numbers. The point
is emphasized by the prevalence of mosquitoes in the arctic circle and
other uninhabited regions.
If this gory insect does not live by blood alone, how is it nourished?
Female mosquitoes are by nature vegetarians; they are plant feeders.
Why they should[Pg 73] draw blood at all is a question which remains
unsolved by entomologists—as well as by the suffering victims. The
females have been observed sucking the nectar from flowers; obtaining
nutriment from boiled potatoes, even from watermelon rinds, from which
they extract the juice. As regards the blood habit, the male mosquito
is a "teetotaler." Just how this male insect lives, scientists have
not determined. He may not take nourishment at all. At any rate, the
mouth parts of the male are so different from those of the female that
it is probable his food is obtained differently. The male is often
seen sipping at drops of water, and a taste for molasses is ascribed
to the male mosquito by one authority.
Presence of Mosquitoes Depends Upon Winds
A common remark heard along the Jersey shore, also on Long Island, is
this: "When we have a sea breeze we are not troubled with mosquitoes,
but when there comes a land breeze they are a pest." While this
observation is true, the reasons therefore entertained by the
unscientific mind are erroneous. The matter of the absence or
abundance of mosquitoes in varying winds is closely related to the
inquiry which entomologists have made: how far will mosquitoes fly?
Says one investigator:
"The migration of mosquitoes has been the source of much
misapprehension on the part of the public.[Pg 74] The idea prevalent at our
seaside resorts that a land breeze brings swarms of mosquitoes from
far inland is based on the supposition that these insects are capable
of long-sustained flight, and a certain amount of battling against the
wind. This is an error. Mosquitoes are frail of wing; a light puff of
breath will illustrate this by hurling the helpless creature away, and
it will not venture on the wing again for some time after finding a
safe harbor. The prevalence of mosquitoes during a land breeze is
easily explained. It is usually only during the lulls in the wind that
Culex can fly. Generally on our coast a sea breeze means a stiff
breeze, and during these mosquitoes will be found hovering on the
leeward side of houses, sand dunes, and thick foliage.... While the
strong breezes last, they will stick closely to these friendly
shelters, though a cluster of houses may be but a few rods off, filled
with unsuspecting mortals who imagine their tormentors are far inland
over the salt meadows. But if the wind dies down, as it usually does
when veering, out come swarms upon swarms of females intent upon
satisfying their depraved taste for blood. This explains why they
appear on the field of action almost immediately after the cessation
of the strong breeze; on the supposition that they were blown inland,
this sudden reappearance would be unaccountable."
A sultry, rainy period of midsummer is commonly referred to as "good
mosquito weather." The ac[Pg 75]cepted idea is that mosquitoes are much more
abundant at such times. This is true, and the explanation is simple.
Mosquito larvæ, or wrigglers, as they are termed, require water for
their development. A heavy shower leaves standing water, which, when
the air is full of moisture, evaporates slowly. Then, too, the heat
favors the growth of the microörganisms on which the larvæ feed;
wrigglers found in the water forty-eight hours after their formation
will have plenty of food, and adult mosquitoes will appear six to
eight days after the eggs are laid. Clear weather, with quick
evaporation, interferes with the development of the wrigglers, so that
a season with plenty of rain, but with sunshiny, drying weather
intervening, is not "good mosquito weather."
Destroy the Larvæ
Inasmuch as a generation of mosquitoes appear to torment man within
ten days, at the longest, after the eggs are laid; as a batch laid by
a female mosquito contains from two hundred to four hundred eggs; as
from each egg may issue a larva or wriggler which in six days will be
an adult mosquito on the wing—it is to the destruction of the larvæ
that attention should be directed. The larva is a slender organism,
white or gray in color, comprising eight segments. The last of these
parts is in the form of a tube, through which the wriggler breathes.
Although its habitat is the[Pg 76] water, it must come to the surface to
breathe, therefore its natural position is head down and tail, or
respiratory tube, up. Now, if oil is spread on the surface of a pool
inhabited by mosquito larvæ, the wrigglers are denied access to the
air which they must have. Therefore, they drown, just as any other
air-breathing animal would drown under similar circumstances.
Best Preventive Measures
As to the best methods to employ in ridding a country place, or any
other region, of mosquitoes, the directions furnished by Dr. L. O.
Howard, the Government entomologist, who has been a careful student of
the problem since 1867, are of great value:
"Altogether,[3] the most satisfactory ways of fighting mosquitoes are
those which result in the destruction of the larvæ or the abolition of
their breeding places. In not every locality are these measures
feasible, but in many places there is absolutely no necessity for the
mosquito annoyance. The three main preventive measures are the
draining of breeding places, the introduction of small fish into
fishless breeding places, and the treatment of such pools with
kerosene. These are three alternatives, any one of which will be
efficacious and any one of which may be used where there are reasons
against the trial of the others."
[Pg 77]
Quantity of Kerosene to be Used
"The quantity of kerosene to be practically used, as shown by the
writer's experiments, is approximately one ounce to fifteen square
feet of water surface, and ordinarily the application need not be
renewed for one month.... The writer is now advising the use of the
grade known as lubricating oil, as the result of the extensive
experiments made on Staten Island. It is much more persistent than the
ordinary illuminating oils.... On ponds of any size the quickest and
most perfect method of forming a film of kerosene will be to spray the
oil over the surface of the water.... It is not, however, the great
sea marshes along the coast, where mosquitoes breed in countless
numbers, which we can expect to treat by this method, but the inland
places, where the mosquito supply is derived from comparatively small
swamps and circumscribed pools. In most localities people endure the
torment or direct their remedies against the adult insect only,
without the slightest attempt to investigate the source of the supply,
when the very first step should be the undertaking of such an
investigation.
"The remedy which depends upon draining breeding places needs no
extended discussion. Naturally the draining off of the water of pools
will prevent mosquitoes from breeding there, and the possibility of
such draining and the means by which it may be done will vary with
each individual case. The writer is in[Pg 78]formed that an elaborate bit of
work which has been done at Virginia Beach bears on this method.
Behind the hotels at this place, the hotels themselves fronting upon
the beach, was a large fresh-water lake, which, with its adjoining
swamps, was a source of mosquito supply, and it was further feared
that it made the neighborhood malarious. Two canals were cut from the
lake to the ocean, and by means of machinery the water of the lake was
changed from a body of fresh to a body of salt water. Water that is
somewhat brackish will support mosquitoes, but water that is purely
salt will destroy them."
Employing Fish to Destroy Larvæ
"The introduction of fish into fishless breeding places is another
matter. It may be undesirable to treat certain breeding places with
kerosene, as, for instance, water which is intended for drinking,
although this has been done without harm in tanks where, as is
customary, the drinking supply is drawn from the bottom of the tank.
The value of most small fishes for the purpose of destroying mosquito
larvæ was well indicated by an experience described to us by Mr. C. H.
Russell, of Bridgeport, Conn. In this case a very high tide broke away
a dike and flooded the salt meadows of Stratford, a small town a few
miles from Bridgeport. The receding tide left two small lakes, nearly
side by side and of the same size.[Pg 79] In one lake the tide left a dozen
or more small fishes, while the other was fishless. An examination by
Mr. Russell in the summer of 1891 showed that while the fishless lake
contained tens of thousands of mosquito larvæ, that containing the
fish had no larvæ. The use of carp for this purpose has been
demonstrated, but most small fish will answer as well. The writer
knows of none that will be better than either of the common little
sticklebacks (Gasterosteus aculeatus or Pygosteus pungitius)."
Is mosquito fighting a success? This question is an all-important one,
not only to the summer resident, but also to cities and towns
contiguous to salt-water marshes, or to swampy lands, well suited for
mosquito breeding. The answer is this: Mosquito control is possible;
actual extermination impossible with an insect that develops so
rapidly. The "Jersey mosquito," the unscientific name popularly given
to an insect of huge size and ravenous appetite, has become famous. As
a matter of fact, the species of mosquitoes found in New Jersey are no
more rare or varied than those found on Staten Island or on Long
Island. But until very recently the region lying between Jersey City
and Newark has been particularly favorable to the development of
mosquito larvæ. It has been announced in the press that mosquitoes
have been driven out of the Newark meadows. This is an exaggeration,
of course, but the work accomplished there is remarkable, and other
infected regions may take heart from the[Pg 80] marked success which has
attended the efforts of Dr. John B. Smith, Entomologist of the New
Jersey State Agricultural Experiment Station.
Remarkable Work Accomplished
The salt marsh lying within the limits of the city of Newark covers an
area of about 3,500 acres. It extends from a point on the Passaic
River to the mouth of Bound Creek, where it empties into Newark Bay.
Its length is about eight miles and it has an extreme width of three
miles. The Newark marsh problem was a very complex one. The meadows
are cut into many sections by the several traversing railroads and by
creeks; this materially influences the drainage. The Peddie Street
sewer crosses the marsh in a straight line of about three miles from
the city to the bay. This sewer is twenty feet wide, and its banks are
from three to four feet above the marsh land.
An experiment with machine ditching was made in 1903. The worst parts
of the marsh were selected, and about 40,000 feet of ditches were cut.
These ditches were six inches wide, two feet deep, and the drainage
was perfect from the outset. The section of meadow thus drained became
so dry in consequence that the grass growing there can now be cut by a
machine in summer, whereas formerly the hay could be mown only in
winter. The work was so successful that the Newark Common Council
appropriated $5,000[Pg 81] to complete the mosquito drainage of the marsh.
Of the results obtained up to this spring, Dr. Smith says:
"This Newark marsh problem was an unusual one, and one that would not
be likely to recur in the same way at any other point along the coast.
Nevertheless, of the entire 3,500 acres of marsh, not 100 acres remain
on which there is any breeding whatever, and that is dangerous only in
a few places and under certain abnormal conditions. Including old
ditches cleaned out, about 360,000 running feet of ditches have been
dug on the Newark marshes, partly by machine and partly by hand, and
if the work is not entirely successful, that is due to the defects
which were not included in the drainage scheme. It is a safe
prediction, I think, that Newark will have no early brood of
mosquitoes in 1905, comparable with the invasions of 1903 and 1904."
This prophecy has proved true.
The Campaign on Long Island
The wealthy summer residents along the north shore of Long Island,
keenly alive to the necessity of driving mosquitoes from the region
where they spend so much of their time, have attacked the problem in a
scientific, as well as an energetic way. The North Shore Improvement
Association intrusted the work to Henry Clay Weeks, a sanitary
engineer, with whom was associated, as entomologist, Prof. Charles B.[Pg 82]
Davenport, Professor of Entomology at the University of Chicago and
head of the Cold Spring Biological Laboratory; also F. E. Lutz, an
instructor in biology at the University of Chicago. Prof. N. S.
Shaler, of Harvard University, the most eminent authority in the
country on marine marshes, was retained to make a special examination
of the salt marshes with a view to recommending the best means of
eliminating what were the most prolific breeding grounds of
mosquitoes. A detailed examination of the entire territory was made.
Practically every breeding place of mosquitoes, including the smaller
pools and streams, and even the various artificial receptacles of
water, were located and reported on. Mr. Weeks, with his assistant,
then examined each body of water in which mosquito larvæ had been
found, with a view to devising the best means of preventing the
further breeding of mosquitoes in these plague spots. Finally, a
report was prepared, together with a map on which was located every
natural breeding place.
Investigations in Connecticut
Important investigations have been made in Connecticut by the
Agricultural Experiment Station, under the direction of W. E. Britton
and Henry L. Viereck, and the results have been most encouraging. Dr.
Howard, in his directions for fighting mosquitoes, acknowledges his
indebtedness to the very successful[Pg 83] experiments carried on at Staten
Island. Maryland is aroused to the point of action. Dr. Howard A.
Kelley, of Johns Hopkins University, is to coöperate with Thomas B.
Symons, the State entomologist, in carrying the war to the shores of
Chesapeake Bay. "Home talent," moreover, can accomplish much. To fight
intelligently, let it not be forgotten that the battle should be
directed against the larvæ. These wrigglers are bred for aquatic life;
therefore, it is to all standing water that attention should be
directed. Mosquito larvæ will not breed in large ponds, or in open,
permanent pools, except at the edges, because the water is ruffled by
the wind. Any pool can be rendered free from wrigglers by cleaning up
the edges and stocking with fish. Every fountain or artificial water
basin ought to be so stocked, if it is only with goldfish. The house
owner should not overlook any pond, however small, or a puddle of
water, a ditch, or any depression which retains water. A half-filled
pail, a watering trough, even a tin receptacle will likely be
populated with mosquito larvæ. Water barrels are favorite haunts for
wrigglers.
A Simple Household Remedy
There are those, however, who will obstinately conduct their campaign
against the adult mosquito. If energetic, such persons will search the
house with a kerosene cup attached to a stick; when this is held[Pg 84]
under resting mosquitoes the insects fall into the cup and are
destroyed. Those possessed of less energy daub their faces and hands
with camphor, or with the oil of pennyroyal, and bid defiance to the
pests. With others it is, Slap! slap!—with irritation mental as well
as physical; for the latter, entomologists recommend household
ammonia.
[Pg 85]
Part II
PURE FOOD FOR THE
HOUSEKEEPER
BY
S. JOSEPHINE BAKER
[Pg 86]
[Pg 87]
CHAPTER I
How to Detect Food Adulteration
Adulteration when applied to foodstuffs is a broad, general term, and
covers all classes of misrepresentation, substitution, deterioration,
or addition of foreign substances; adulteration may be either
intentional or accidental, but the housekeeper should be prepared to
recognize it and so protect herself and her household.
Food is considered adulterated when it can be classified under any of
the following headings:
DEFINITIONS OF ADULTERATION.—(1) If any substance has been mixed or
packed with it so as to reduce or lower or injuriously affect its
quality or strength.
(2) If any inferior substance has been substituted for it, wholly or
in part.
(3) If any valuable constituent has been wholly or in part abstracted
from it.
(4) If it consists wholly or in part of diseased or decomposed or
putrid or rotten animal or vegetable substance, or any portion of an
animal unfit for food, whether manufactured or not, or if it is the
product[Pg 88] of a diseased animal or one who has died otherwise than by
slaughter.
(5) If it be colored or coated or polished or powdered, whereby damage
is concealed or it is made to appear better than it really is.
(6) If it contains any added poisonous ingredient or any ingredient
which may render such article injurious to health; or if it contains
any antiseptic or preservative not evident or not known to the
purchaser or consumer.
FOOD LAWS.—There is now in effect in the United States a rigid law
against the offering for sale of any article intended for human
consumption which is adulterated in any way, without the fact and
nature of such adulteration being plainly stated on a label attached
to the package containing the article. This law, however, applies only
to articles of this nature which originate, or are produced, in one
State and offered for sale in another. The purchaser is, therefore, in
a great degree protected, but many foodstuffs or manufactured articles
may have their origin within the State wherein they are sold, and in
this case the only safeguards are those afforded by the laws of the
State, city, or town immediately concerned. If these restraining laws
do not exist or if they are not enforced the housekeeper must rely
upon her own efforts to protect her family from adulterated food.
PERMISSIBLE ADULTERANTS.—In this class are included articles having a
food value such as salt,[Pg 89] sugar, vinegar, spices, or smoke used as
preservatives of meats; or starch when added to the salts composing
baking powder, where a certain amount is permissible for the purpose
of absorbing moisture.
GENERAL DIRECTIONS.—The ability to select fresh, wholesome meats,
poultry, fish, fruits, and vegetables, to determine readily the purity
of dairy products, and to detect adulteration or misrepresentation in
all classes of foodstuffs must, in most instances, be acquired. Common
sense and good reasoning powers are needed here as in every problem of
life. While some adulterants can be detected only by trained chemists
and by means of tests too difficult and involved for general use, the
average housekeeper may amply protect herself from gross imposition by
simply cultivating her powers of observation and by making use of a
few simple tests well within her grasp and easily applied.
First—Sight, Taste, and Smell.—All are of prime importance in
determining the freshness and wholesomeness of foods, especially
meats, poultry, fish, vegetables, and fruits. Avoid all highly colored
bottled or canned fruits or vegetables; pure preserved fruits, jams,
jellies, or relishes may have a good bright color, but never have the
brilliant reds and greens so often shown in the artificially colored
products.[4] The same is[Pg 90] true of canned peas, beans, or Brussels
sprouts; here the natural product is a dull, rather dingy green, and
all bright green samples must be suspected. Foreign articles of this
class are the worst offenders.
All food products should have a clean wholesome odor, characteristic
of their particular class. The odor of decomposition can be readily
detected; stale and musty odors are soon recognized.
It should be rarely necessary to use the sense of taste, but any food
with a taste foreign to the known taste of a similar product of known
purity should be discarded or at least suspected.
Second—Price.—Remember that the best and purest food, however high
priced, is cheapest in the end. Its value in purity, cleanliness, food
value, and strength gives a greater proportionate return than foods
priced lower than one might legitimately expect from their supposed
character. To cite a few instances: pure Java and Mocha coffee cannot
be retailed at twenty cents per pound; therefore, when the housekeeper
pays that price she must expect to get chicory mixed with the coffee;
if it contains no other adulterant, she may consider herself
fortunate. Cheap vanilla is not made from the vanilla bean. These
beans sell at wholesale for from ten to fifteen dollars a pound, and
the cheap[Pg 91] extracts are made from the Tonka bean or from a chemical
product known as vanillin. These substances are not harmful, but they
are not vanilla. Pure virgin olive oil is made from the flesh of
olives after the stones and skin have been removed; cheaper grades are
made from the stones themselves and have little food value, while the
virgin oil is one of the most nutritious and wholesome of foods.
Such instances might be cited almost without end. Good, pure food
demands a good price, and economy defeats its own purpose when it is
practiced at the expense of one of the most vital necessities of
health and life.
Third—Reliable Dealers.—Select your tradesmen with the same care you
bestow in the choice of a physician. A grocer or butcher who has once
sold stale, adulterated, or impure wares has forfeited his right to be
trusted. A man who is honestly trying to build up a good trade must
have the confidence of his customers and it is to his interest to sell
only worthy goods; this confidence he can gain only by proving his
trustworthiness. When you are convinced of your dealer's honesty give
him your trade and do not be lured away by flashy advertisements and
the promise of "something for nothing."
PREPARATION FOR CHEMICAL TESTS.—Although the housekeeper will rarely
need the use of any chemical tests for the purpose of determining the
purity of food, the following directions must be kept[Pg 92] in mind if such
an expedient is deemed necessary. It will be wise, however, in the
majority of cases when the presence of chemical preservatives and
adulterants is suspected, to send the article to a chemist for
analysis.
1. All refuse matter, such as shells, bones, bran, and skin, must be
removed from the edible portion of the food to be tested.
2. If the sample is solid or semi-solid, divide it as finely as
possible. All vegetables and meats may be minced in the common
household chopping machine. Tea, coffee, whole spices, and the like
may be ground or crushed in a mortar or in a spice mill.
3. Milk must be thoroughly stirred or shaken so that the cream is well
mixed with the body of the milk.
FLESH FOODS—Meat.—Fresh, wholesome meat is neither pink nor purple;
these colors indicate either that the animal was not slaughtered or
that it was diseased. Good meat is firm and elastic and when dented
with the finger does not retain the impression; it has the same
consistency and color throughout; the flesh is marbled, due to the
presence of fat distributed among the muscular fibers; it will hardly
moisten the finger when touched; it has no disagreeable odor and has a
slightly acid reaction so that red litmus paper applied to it should
not turn blue.
Wet, sodden, or flabby meat with jellylike fat, a strong putrid odor,
and alkaline reaction should be[Pg 93] avoided. These signs indicate
advanced decomposition, and such meat is unfit for food.
Beef.—This meat should have a fine grain, be firm in texture, with
rosy-red flesh and yellowish-white fat.
Lamb and Mutton should have a clear, hard, white fat with the lean
part juicy, firm, and of rather light-red color. The flesh should be
firm and close of grain.
Veal.—The meat should not be eaten unless the animal was at least six
weeks old before slaughtering. The sale of this immature veal, or "bob
veal" as it is sometimes called, is prohibited by law in many States.
It is unwholesome and may be recognized by its soft, rather mushy
consistency and bluish tinge. Good veal has a firm white fat with the
lean of a pale-red color.
Pork.—This meat when fresh has a fat that is solid and pure white; if
yellow and soft it should be rejected; the lean is pink and the skin
like white translucent parchment.
Poultry.—Good poultry is firm to the touch, pink or yellowish in
color, is fairly plump, and has a strong skin showing an unbroken
surface. It has a fresh odor.
Stale poultry is flabby and shows a bluish color; it becomes green
over the crop and abdomen, and the skin is already broken or easily
pulled apart in handling. The odor of such a bird is disagreeable and
may even be putrid.
Fish.—With the exception of the salted or preserved varieties fish
should always be perfectly fresh[Pg 94] when eaten. Probably no other
article of food is more dangerous to health than fish when it shows
even the slightest traces of decomposition. The ability to recognize
the earliest signs of staleness is of the utmost importance. Fish
deteriorate rapidly and should always be carefully inspected before
purchasing.
Fresh fish are firm to the touch, the scales moist and bright, the
gills red, and the eyes clear and slightly prominent. When held flat
in the hand the fish should remain rigid and the head and tail droop
slightly, if at all.
Stale fish are soft and flabby, the skin is dull and the eyes sunken
and often covered with a film. The tendency of the head and tail to
droop is marked and the fish has a characteristic disagreeable odor.
This odor of decomposition is best detected in the gills.
Lobsters and Crabs.—These shellfish should always be alive when
purchased. This condition is easily demonstrated by their movements,
and the rule should never be disregarded.
Oysters and Clams.—Oysters should not be eaten during the months of
May, June, July, and August; these are their breeding months and they
are unwholesome during that period. That oysters sometimes contain the
germs of typhoid fever is an assured fact; these germs are acquired
not from the natural habitat of the oyster in salt water but from the
fresh-water, so-called "fattening beds," where the oysters are placed
for a season to remove the brackish and salty taste of[Pg 95] the sea and to
render them more plump. These beds are frequently subject to
pollution, and the housekeeper should only purchase oysters from
reliable dealers where the purity of the source of the supply is
unquestioned.
Clams are in season and may be eaten throughout the year.
All shellfish when fresh have an agreeable fresh odor. The shells
should be firmly closed or should close when immersed in water and
touched with the finger. If they have been removed from their shells
when purchased, the flesh of the fish itself should be firm, clean in
appearance and not covered with slime or scum; the odor should be
fresh. The odor of dead or decomposed oysters and clams is pungent and
disagreeable.
MEAT PRODUCTS—Canned or Potted Meats.—The label on cans containing
meat products should state clearly the exact nature of the contents.
Deception as to the character of the meat is easy to practice and
difficult to detect by any but a trained analyst. The presence of
preservatives can also only be detected by chemical analysis. As these
products are practically all put on the market by the large packing
houses and designed for interstate commerce, they are subject to
government inspection, and, therefore, if they bear the government
stamp may be considered pure. The point that the housekeeper may
consider is the length of time the meat has remained in the can. Put
up under[Pg 96] proper precautions these canned goods retain their
wholesomeness for an almost indefinite period. The heads of the cans
should always present a concave surface; if they are convex, it is a
sign of decomposition of the contents. When the can is opened the meat
should have a clean appearance, free from mold or greenish hue, and
the odor should be fresh and not tainted.
Sausages.—If possible, sausages should be homemade, then one may be
assured of their purity and freedom from adulteration.
Owing to the rapid color changes and early decomposition of fresh
meat, artificial colors are often used to conceal the former, and
preservatives like boric acid or saltpeter to retard the latter.
The artificial colors, such as carmine and aniline red, may be
detected by observation or by warming the finely divided material on a
water bath with a five per cent solution of sodium salicylate. This
fluid will extract the color, if present.
Lard.—Good lard is white and granular and has a firm consistency. It
has an agreeable characteristic odor and taste. The choicest leaf lard
is made from the fat about the kidneys of the hog; the cheaper grades
are made from the fat of the whole animal.
FRESH VEGETABLES AND FRUITS—Vegetables.—All green vegetables to be
eaten uncooked should be carefully washed and examined for insects,
dirt, and foreign matter generally. The ova or eggs[Pg 97] of the tapeworm
may be ingested with improperly cleaned vegetables. Running water and
a clean brush (kept for this purpose) should be used.
Green vegetables should have a fresh, unwilted appearance; any sign of
staleness or decay should cause their rejection. Overripe or underripe
vegetables are harmful.
Lettuce, celery, and all leaved or stemmed vegetables should be
examined to see if the outer leaves have been removed; this may be
determined by the distance of the leaves from the stem head. The
general signs of disease in vegetables are softening, change of color,
and mold.
The following characteristics indicate fresh and wholesome vegetables:
Asparagus.—Firm and white in the stalk with a green, compact tip.
Beans and Peas should have green, not yellow, pods, brittle, and
easily snapped open. The vegetable itself should be tender, full and
fleshy, not wrinkled or shrunken.
Cabbage, crisp and firm, with a well-rounded and compact head.
Carrots, light red or yellow, with a regular, conical shape, sweet and
crisp.
Cauliflower, white, compact head; any tinge of yellow or green
generally indicates an inferior quality.
Celery, nearly white in color; large, crisp, and solid stalks, nutty
in flavor.
[Pg 98]Cucumbers, firm, crisp, with a smooth skin and white flesh.
Lettuce, the head close and compact; the leaves clean, crisp, and
sweet. When it is too young or running to seed the taste is bitter.
Pale patches on the leaves are caused by mildew and are a sign of
decay.
Parsnips, buff in color, with unforked roots, sweet and crisp.
Potatoes, underripe, green potatoes are unfit for food; they contain a
poisonous substance which renders them actually harmful. Good potatoes
should have a smooth skin and few eyes; the flesh pale and of a
uniform color and of a firm consistency. A rough skin, with little
depressions, indicates a disease called "scab"; dark-brown patches on
the skin are due to a disease called "smut." Potatoes with such
diseases are of inferior quality. If green on one side, due to
exposure to the sun when growing, the potatoes are unwholesome.
Fruits.—Underripe or green fruit should never be eaten. This
condition may be easily detected by the color and consistency of the
fruit. Diseased or decayed fruit is known by its change of color,
softening, and external mold. Spots on fruit are often caused by a
fungus which lowers its quality and renders it less wholesome.
CEREALS AND THEIR PRODUCTS—Cereals.—Particularly when bought in
original packages cereals are generally pure and unadulterated. When[Pg 99]
bought in bulk there may be found dust, dirt, worms, insects, and
excessive moisture. These may all be determined by careful inspection.
The presence of an undue amount of moisture adds greatly to the weight
of cereals and is therefore a fraud. Cereals should be dry to the
touch and the individual kernels or particles separate and distinct.
Flour.—By this general term is meant the ordinary wheat flour. It
should not be too moist, should have a fine white appearance, remain
lumpy, or hold its form, on pressure, not show any particles which
cannot be crushed, and when a handful is thrown against the wall, part
of it should adhere. The odor and taste should be fresh and clean and
not musty or moldy.
The common adulterants are corn and rice meal. If a sample of the
flour be thrown on the surface of a glassful of water, the corn and
rice, being heavier, will sink; grit and sand may be detected in the
same way. If the flour has been adulterated with mineral substances it
may be shown by burning a portion down to an ash; the ash of pure
flour should not exceed two per cent of the total amount; if mineral
substances are present the amount of ash will be greatly increased.
Alum is sometimes added to flour in order to give it a whiter
appearance and to produce whiter and lighter bread; it is most
unwholesome. It can be detected by the so-called "logwood" test, which
is prepared and used as follows:
[Pg 100]Make two solutions. The first: a five per cent solution of logwood
chips in alcohol. The second: a fifteen per cent solution of ammonium
carbonate in water. Make a paste of one teaspoonful of the flour and
an equal amount of water; mix with it one-quarter of a teaspoonful of
the logwood solution; follow this immediately with one-quarter of a
teaspoonful of the ammonium carbonate solution. If alum is present,
the paste will show a lavender or blue color; if absent, the mass will
become pink, fading to a dirty brown. If the result is doubtful, set
the paste aside for several hours, when the colors will show more
plainly.
Bread.—Bread should be well baked and not too light or too heavy; the
crust should be light brown and adherent to the substance of the
bread. The center should be of even consistency, spongy, and firm; it
should not pit or be soggy or doughy. The pores or holes should be of
practically the same size throughout.
Exceedingly white, light, or porous bread shows the presence of alum.
It may be detected by means of the solutions already mentioned in the
"logwood" test. Mix one teaspoonful of each solution and add three
ounces (six tablespoonfuls) of water; pour this over a lump of bread,
free from crust and about an inch square. After the bread has become
thoroughly soaked, pour off the excess of liquid and dry the bread in
the dish; if alum is present, the mass will show[Pg 101] a violet or blue
tint, more marked on drying; if absent, a brownish color will appear.
Baking Powders.—Baking powders are of three classes, all having
sodium bicarbonate (baking soda) as their alkaline salt. The first
style is the commonly used and wholesome mixture of cream of tartar
and baking soda; the second has calcium phosphate for the acid salt,
and the third contains alum. All have a certain proportion of starch
to absorb moisture. Of these the alum powders are the most harmful and
should be avoided. Practically all of the well-known brands of baking
powder are of the first-mentioned class and wholesome, and are rarely
adulterated.
DAIRY PRODUCTS—Milk.—Pure milk should have a specific gravity of
from 1.027 to 1.033. Its normal reaction is neutral or slightly acid;
it should never be strongly acid. If it is strongly alkaline, i. e.,
turning red litmus paper blue, it is pretty certain that something in
the way of a preservative has been added to it. When left standing for
a few hours the cream should show as a slightly yellowish top layer,
one-tenth or more of the whole amount; the milk below the cream should
be lighter in color and with the slightest bluish tinge. If the color
is of a yellowish tinge throughout, the addition of coloring matter
must be suspected. "Annatto," a vegetable pigment, is used to give a
"rich" tint to milk. To detect it, add one teaspoonful of baking soda
to one quart of milk and immerse in it a strip of unglazed paper; in a
few[Pg 102] hours examine the paper; if annatto is present, it will have
become an orange color.[5]
If the whole milk has a blue and thin appearance, or if the cream is
scant in quantity, it has probably been diluted with water. The
popular idea that chalk is sometimes added to poor milk to make it
appear of better quality is erroneous; chalk would always show as a
precipitate, as it does not dissolve, and the presence of such a
sediment would be a too obvious adulteration to be practiced.
Milk should always be kept at a temperature below 50° F.; above that
temperature the bacteria in it multiply with great rapidity and render
it unfit for use.
Milk may be preserved for several days if "pasteurized" or
"sterilized." Pasteurization consists of heating milk to a temperature
of about 167° F., and maintaining it at that degree for twenty
minutes. Sterilization means keeping the milk at a temperature of 212°
F. for two hours and a half. Immediately after either process the milk
should be cooled, then placed in absolutely clean, covered bottles and
kept on ice. These methods are not only harmless but actually
beneficial in that they destroy any disease germs that might be
present.
Chemical preservatives are occasionally found in[Pg 103] milk. They may be
suspected if the milk is alkaline in reaction and has a disguised
taste. The ones most commonly used are boric and salicylic acids and
formaldehyde; the two former can only be detected by chemical tests
too delicate and intricate to be used by the housewife. Formaldehyde
may be tested for by using a solution of one drop of a ten per cent
solution of ferric chloride to one ounce of hydrochloric acid.[6] Fill
a small porcelain dish one-third full of this solution; add an equal
volume of milk and heat slowly over a flame nearly to the boiling
point, giving the dish a rotary motion to break up the curd. If
formaldehyde is present, the mass will show a violet color, varying in
depth with the amount present; if it is absent, the mass turns brown.
Butter.—Good butter has a fresh, sweet odor and an agreeable taste.
It should be of the same color and consistency throughout, easily cut
and adherent and not crumbly when molded into shapes. Pure butter is
very light in color; nearly all that is sold is colored, in order to
meet the popular demand for "yellow" butter; annatto and other
vegetable and mineral substances are sometimes employed for this
purpose. These coloring matters are generally harmless but may be
detected by dissolving a portion of the butter in alcohol; the natural
color will dissolve, while foreign coloring will not. Butter should
consist of eighty-five[Pg 104] per cent fat, with the remainder water,
casein, and salt. The most common methods of adulteration consist in
an excess of water and the addition of oleomargarine. If an excess of
water has been added it may be shown by melting the butter; the water
and fat will separate in two distinct layers. Oleomargarine has a
distinctive meaty smell, like that of cooked meat, and lacks the
characteristic odor of pure butter. If pure butter is melted in a
spoon, it will not sputter; if oleomargarine is present, it will.
The preservatives sometimes used, namely, boric and salicylic acids
and formaldehyde, can only be detected by chemical tests.
Eggs.—Two methods may be used to detect stale eggs. First: make a
solution of one part of table salt to ten parts of water and immerse
the suspected egg; if it sinks, it is perfectly fresh; if it remains
in the water below the surface, it is at least three days old, and if
it floats, it is five or more days old.
Second: hold the egg between a bright light and the eye. If it is
fresh, it will show a rosy tint throughout, without dark spots, as the
air chamber is small; if not fresh, it will look cloudy, with many
dark spots present.
TEA AND COFFEE.—These substances are extensively adulterated, but the
adulterants are almost without exception harmless.
Tea.—The commonest forms of adulteration of tea are as follows: (a)
Exhausted tea leaves which have[Pg 105] already been used are dried and
added. Their presence may be detected by the weakness of the infusion,
made from a given quantity of the suspected tea, compared with a
similar infusion made from tea known to be pure. (b) Leaves from
other plants are sometimes dried and added; these are easily shown if
an infusion is made and when the leaves are thoroughly wet unrolling
and comparing them. (c) Green teas may be "faced" or colored with
Prussian blue, indigo, French chalk, or sulphate of lime; black teas
may be similarly treated with plumbago or "Dutch pink." If teas so
treated are shaken up in cold water the coloring matter will wash off.
(d) Sand and iron filings are occasionally added for weight;
observation, and the fact that they sink when tea is thrown in water,
will show their presence. Iron filings may be readily found by using a
magnet. (e) The presence of starch may be shown by washing the tea
in cold water, straining it, and testing the solution in the following
manner: dissolve one-half teaspoonful of potassium iodide in three
ounces of water and add as much iodine as the solution will dissolve;
a few drops of this solution added to the suspected sample will give a
blue color if starch is present.
Coffee.—Coffee should always be purchased in the bean, as ground
coffee is much more frequently adulterated and the foreign substances
are more difficult to detect.
The adulterants commonly used are: chicory, peas,[Pg 106] beans, peanuts, and
pellets of roasted wheat flour, rye, corn, or barley.
Fat globules are always present in pure coffee; their presence may be
shown by the fact that imitation coffee sinks in water, while pure
coffee floats.
Chicory is the most frequently used adulterant; it is added for flavor
and to produce a darker infusion, thus giving the impression of
greater strength. It is perfectly harmless and as a drink is actually
preferred by some people. Its detection is comparatively easy. Chicory
grains are dark, gummy, soft, and bitter; coffee grains are hard and
brittle; a small amount put in the mouth will demonstrate the
difference. Chicory will often adhere to the wheels of a coffee
grinder, clogging them on account of its gummy consistency.
When a sample of adulterated coffee is thrown in water the pure coffee
floats and leaves the water unstained; chicory sinks almost instantly,
coloring the water, while peas and beans sink more slowly but also
color the water.
Peas and beans are also detected by the polished appearance of the
broken or crushed grains in marked contrast to the dull surface of
crushed coffee.
The presence of peas, beans, rye, wheat, bread crumbs, and allied
substances may be shown by the fact that they all contain starch.
Make a ten per cent infusion of the suspected coffee; filter it, and
decolorize the solution by boiling it with a piece of animal charcoal.
Test the decolorized solu[Pg 107]tion by slowly adding a few drops of the
"potassium-iodide-iodine solution," directions for preparing which
were given under heading of "Tea." A resulting blue color will
indicate the presence of starch.
COCOA AND CHOCOLATE.—The adulterants of these substances are
generally harmless, as they usually consist of flavoring extracts,
sugar, starch, flour, and animal fats. No tests other than flavor,
consistency, and smoothness need be considered. Good cocoa and
chocolate should be slightly bitter, with a pleasant characteristic
odor and taste; they should have a smooth, even consistency and be
free from grit or harsh particles.
CANNED AND BOTTLED VEGETABLES AND FRUITS.—In general, acid
substances, such as tomatoes and fruits, should not be canned in tin,
as the action of the acid tends to dissolve the tin. It is better,
therefore, to purchase these articles in glass.
After opening the can the odor and appearance of the contents should
be noted. The odor should be clean and fresh, and the slightest trace
of any sour, musty, or disagreeable smell should cause the rejection
of the food. The appearance should be clean, with no mold; the
consistency and color of the fruit or vegetables should be uniform
throughout. If the color is brighter than that of a similar article
when canned at home, the presence of artificial coloring matter must
be suspected. The brilliant green of some[Pg 108] brands of peas, beans, or
Brussels sprouts is produced by the addition of the salts of copper.
This may be proved by leaving the blade of a penknife in the contents
of the can for a short time; if copper is present it will be deposited
on, and discolor, the blade.
Brightly colored fruits should excite suspicion; this same dictum
applies to all brightly colored jams and jellies, as the colors are
usually produced by the addition of carmine or aniline red.
The presence of preservatives, salicylic and boric acids, the
benzoates, etc., can only be proved by delicate chemical tests.
SUGAR.—Pure granulated or powdered sugar is white and clean. The
presence of glucose should be suspected in sugar sold below the market
price; it is perfectly harmless, but has a sweetening power of only
about two-thirds that of sugar and is added on account of its
cheapness and to increase the bulk.
If sand, dirt, or flour are present they may be detected by
observation, or by washing the suspected sample in water; flour will
not dissolve, sand will sink, and dirt will discolor the water.
SPICES.—Spices should be bought whole and ground in a spice mill as
needed; if this is done, there need be little fear of their impurity,
for whole spices are difficult to simulate or adulterate. Ground
spices may be adulterated with bark, flour, starches, or arrowroot;
these adulterants are harmless, but are fraudulent,[Pg 109] as they increase
the bulk and decrease the strength. Their actual presences can only be
demonstrated by a microscopical or chemical examination.
Peppers.—Black pepper is made from the whole berry; white pepper is
made from the same berry with the outer husk removed. The adulterants
are usually inert and harmless substances, such as flour, mustard, or
linseed oil; their presence is obviated by the use of the whole
peppercorns, ground as needed.
Red Pepper.—This may be adulterated with red lead; when pure it will
be entirely suspended in water; if a sediment falls it is probably red
lead.
Mustard.—Practically all of the adulterants of mustard can only be
detected by intricate chemical tests. The presence of turmeric may be
detected by the appearance of an orange-red color when ammonia is
added to a solution of the sample.
Tomato Catsup.—Artificial dyestuffs are common, giving a brilliant
crimson or magenta color. Such catsup does not resemble the natural
dull red or brown color of the homemade article.
Preservatives, such as boric, salicylic, or benzoic acids and their
salts, are sometimes added. While their presence cannot be condoned,
yet they are usually present in small amounts and therefore
practically harmless.
Pickles.—These should be of a dull-green color. The bright emerald
green sometimes observed is due to the presence of the salts of
copper; this may be[Pg 110] proved by dipping the blade of a penknife in the
liquor, as described under the heading of "Canned Goods."
Alum is sometimes used as a preservative and in order to make the
pickles crisp. Its presence may be demonstrated by means of the
"logwood" test mentioned under the heading of "Flour."
VINEGAR.—Cider vinegar is of a brownish-yellow color and possesses a
strong odor of apples.
Wine vinegar is light yellow if made from white wine, and red if made
from red wine.
Malt vinegar is brown and has an odor suggestive of sour beer.
Glucose vinegar has the taste and odor of fermented sugar.
Molasses vinegar has the distinctive odor and taste of molasses.
OLIVE OIL.—Pure olive oil has a pleasant, bland taste and a
distinctive and agreeable odor, unmistakable in character for that of
any other oil. The finest virgin oil is pale green in color, the
cheaper grades are light yellow.
The adulterants consist of cotton-seed, corn, mustard, and peanut
oils.
When pure olive oil is shaken in a glass or porcelain dish with an
equal quantity of concentrated nitric or sulphuric acid[7] it turns
from a pale to a dark green color in a few minutes; if under this
treatment a red[Pg 111]dish to an orange or brown color is produced the
presence of a foreign vegetable oil is to be suspected.
FLAVORING EXTRACTS—Vanilla.—This may be wholly or in part the
extract of the Tonka bean or may be made from a chemical substance
known as vanillin. The best practical working tests as to its purity
are the price, taste, and odor. The distinctive odor and taste of
vanilla are characteristic and cannot be mistaken.[8]
Lemon.—This extract is often made from tartaric or citric acid. They
may be tested for as follows: to a portion of the extract in a test
tube add an equal volume of water to precipitate the oil; filter, and
add one or two drops of the filtrate to a test tube full of cold,
clear limewater; if tartaric acid is present a precipitate will fall
to the bottom of the tube. Filter off this precipitate (if present)
and heat the contents of the tube; if citric acid is present it will
precipitate in the hot limewater.
Footnote.—Dr. Baker wishes to acknowledge her indebtedness to the
following authorities and the volumes mentioned for many helpful
suggestions. Pearman and Moore, "Aids to the Analysis of Foods and
Drugs"; Albert E. Leach, "Food Inspection and Analysis"; Francis
Vacher, "Food Inspector's Hand Book."
[Pg 112]
CHAPTER II
Mushroom Poisoning
Symptoms—Treatment—How to Tell Mushrooms—The Common Kind—Other
Varieties—The Edible Puffball—Poisonous Mushrooms Frequently
Mistaken.
MUSHROOM POISONING.—Vomiting, cramps, diarrhea, pains in legs;
possibly confusion, as if drunk, stupidity, followed by excitement,
and perhaps convulsions. Lips and face may be blue. Pulse may be weak.
First Aid Rule 1.—Rid the stomach and bowels of remaining poison.
Give emetic of mustard, tablespoonful in three glasses of warm water,
unless vomiting is already excessive. When vomiting ceases, give
tablespoonful of castor oil, or compound cathartic pill. Give no
salts. Also empty bowels with injection of tablespoonful of glycerin
in pint of warm soapsuds and water.
Rule 2.—Antidote the poison. Give a cup of strong coffee and fifteen
drops of tincture of belladonna to adult. Repeat both once, after two
hours have passed.
[Pg 113]Rule 3.—Rest and stimulate. Put patient to bed. Give whisky, a
tablespoonful in twice as much water. Give tincture of digitalis, ten
drops every two hours, till two or three doses are taken by adult.
Symptoms.—Vomiting and diarrhea come on in a few hours to half a day,
with cramps in the stomach and legs. The face and lips may grow blue.
There is great prostration. In the case of poisoning by the fly
amanita, stupor may appear early, the patient acting as if drunk, and
difficult breathing may be a noticeable symptom. Afterwards the
patient becomes excited and convulsions develop. The pulse becomes
weak and slow. The patient may die in a few hours, or may linger for
three or four days. If treatment be thorough, recovery may result.
Treatment.—Unless vomiting has already been excessive, the patient
should receive a tablespoonful of mustard mixed with a glassful of
tepid water. After the vomiting ceases he should receive a
tablespoonful of castor oil, or any cathartic except salts. If the
cathartic is vomited, he should receive an injection into the rectum
of a tablespoonful of glycerin mixed with a pint of soapsuds and
water. Coffee and atropine (or belladonna) are the best antidotes.
If a physician be secured, he will probably give a hypodermic
injection of atropine. If a physician is not procurable, the patient
should receive a cup of strong coffee, and a dose of ten or fifteen
drops of tinc[Pg 114]ture of belladonna in a tablespoonful of water, if an
adult. This dose should be repeated once after the lapse of two hours.
The patient should be kept in bed, a bedpan being used when the bowels
move.
When the pulse begins to grow weak, two tablespoonfuls of whisky and
ten drops of the tincture of digitalis should be given to an adult in
quarter of a glass of hot water. The digitalis should be repeated
every two hours till three or four doses have been taken. The patient
must be kept warm with hot-water bottles and blankets.
HOW TO KNOW MUSHROOMS.—One-sixth of one of the poisonous mushrooms
has caused death. It is, therefore, impossible to exert too much care
in selecting them for food. A novice would much better learn all the
characteristics of edible and poisonous mushrooms in the field from an
expert before attempting to gather them himself, and should not trust
to book descriptions, except in the case of the few edible species
described hereafter. It is not safe for a novice to gather the
immature or button mushrooms, because it is much more difficult to
determine their characteristics than those of the full grown. As
reference books, the reader is advised to procure Bulletin No. 15 of
the United States Department of Agriculture, entitled "Some Edible and
Poisonous Fungi," by Dr. W. G. Farlow, which will be sent without
charge on request by the Agricultural Department at Washington;
"Studies of American Fungi," by Atkinson, and Miss Mar[Pg 115]shall's
"Mushroom Book," all of which are fully illustrated, and will prove
helpful to those interested in edible fungi.
There are no single tests by which one can distinguish edible from
poisonous fungi, such as taste, odor, the blackening of a silver
spoon, etc., although contrary statements have been made. Even when
the proper mushrooms have been eaten, ill effects, death itself, may
follow if the mushrooms have been kept too long, have been
insufficiently cooked, have been eaten in too large a quantity
(especially by children), or if the consumer is the possessor of an
unhappy idiosyncrasy toward mushrooms.
No botanic distinction exists between toadstools and mushrooms;
mushrooms may be regarded as edible toadstools. They are all,
botanically speaking, edible or poisonous fungi. A description follows
of the five kinds of fungi most commonly eaten, and the poisonous
species which may be mistaken for them.
EDIBLE MUSHROOMS.—1. The Common Mushroom (Agaricus
Campestris).—The fungi called agarici are those which have gills,
that is, little plates which look like knife blades on the under
surface of the top of the mushroom, radiating outward from the stem
like the spokes of a wheel. This is the species most frequently grown
artificially, and sold in the markets. The top or cap of this mushroom
is white, or of varying shades of brown, and measures from one and a
half to three or even four inches in diameter. It[Pg 116]
[Pg 117] is found in the
latter part of August, in September, and in October, growing in
clusters on pastures, fields, and lawns.
The gills are pink or salmon colored in the newly expanded specimen;
but as it grows older, or after it is picked, the gills turn dark
purple, chestnut brown, or black. This is the important point to
remember, since the poisonous species mistaken for it all have white
gills. The gills end with abrupt upward curves at the center of the
cap without being attached to the stem. In the young mushroom, when
the cap is folded down about the stem, the gills are not noticeable,
as they are covered by a veil or filmy membrane, a part of which
remains attached to the stem (when the top expands), as a ring or
collar about the stem a little more than halfway up from the ground.
The stem is solid and not hollow, and there is no bulbous enlargement
at the base of the stem, surrounded by scales or a collar, as occurs
in the fly amanita and other poisonous species. Neither the
campestris nor any other mushroom should be eaten when over a day
old, since decomposition quickly sets in.
 Fig. 35.
Fig. 35.
THE FIELD MUSHROOM.
(Agaricus Campestris.)
An edible variety; very common.
2. Horse Mushroom (Agaricus Arvensis).—This species may be
considered with the foregoing, but it differs in being considerably
larger (measuring four to ten inches across) and in having a more
shiny cap, of a white or brown hue. The ring about the stem is
noticeably wider and thicker, and is composed of two distinct layers.
The gills are white at first, turning dark[Pg 118]
[Pg 119] brown comparatively late,
and the stem is a little hollow as it matures. In some localities it
is more common than the campestris in fields and pastures, while in
other places it is found only in rich gardens, about hot beds, or in
cold frames. It is not distinguished from the campestris by market
people, but is often sold with the latter.
 Fig. 36.
Fig. 36.
THE HORSE MUSHROOM.
(Agaricus Arvensis.)
This variety is edible.
3. Shaggy Mane, Ink Cap, or Horsetail Fungus (Coprinus
Comatus).—This mushroom possesses the most marked characteristics of
any of the edible species; it would seem impossible to mistake its
identity from written descriptions and illustrations. It is considered
by many superior in flavor to the campestris.
The top or cap does not expand in this mushroom, until it begins to
turn black, but remains folded down about the stem like a closed
umbrella. Mature specimens are usually three to five, occasionally
from eight to ten, inches high. The stem is hollow. The inside of the
cap or gills and the stem are snow white. The outer surface of the
cap, which is white in young plants, becomes of a faint, yellow-brown
or tawny color in mature specimens, and also darker at the top.
Delicate scales often rolled up at their lower ends are seen on the
exterior of the cap, more readily in mature mushrooms, hence the name
"shaggy mane." There is a ring around the stem at the lower margin of
the cap, and it is so loosely attached to either the cap or stem that
it sometimes drops down to the base of the latter.
[Pg 120]
[Pg 121]The most salient feature of shaggy mane is the change which occurs
when it is about a day old; it turns black and dissolves away into an
inky fluid, whence the other common name "ink cap." The mushroom
should not be eaten when in this condition. The ink cap is usually
found growing in autumn, rarely in summer, in richer earth than the
common mushroom. One finds it in heaps of street scrapings, by
roadsides, in rich lawns, in soils filled with decomposing wood and in
low, shaded, moist grounds.
 Fig. 37.
Fig. 37.
THE HORSE-TAIL FUNGUS.
(Coprinus Comatus.)
Edible; cut shows entire plant and section.
4. Fairy-ring Mushroom (Marasmius Oreades).—This species usually
grows on lawns, in clusters which form an imperfect circle or
crescent. The ring increases in size each year as new fungi grow on
the outside, while old ones toward the center of the circle perish.
This mushroom is small and slender, and rarely exceeds two inches in
breadth. The cap and the tough and tubular stem are buff, and the
gills, few in number and bulging out in the middle, are of a lighter
shade of the same color. There is no ring about the stem. Several
crops of the fairy-ring mushroom are produced all through the season,
but the most prolific growth appears after the late fall rains. There
are other fungi forming rings, some of which are poisonous, and they
may not be easily distinguished from the edible species; hence great
care is essential in gathering them. The under surface of the cap is
brown or blackish in the mature plants of poisonous species.
 Fig. 38.
Fig. 38.
THE FAIRY-RING MUSHROOM.
(Marasmius Oreades.)
An edible variety.
5. Edible Puffball (Lycoperdon Cyathiforme).—Edible[Pg 122] puffballs grow
in open pastures, and on lawns and grassplots, often forming rings.
They are spherical in form, generally from one and a half to two
inches, occasionally six inches, in diameter, broad and somewhat
flattened at the top, and tapering at the base, white or brown
outside. They often present an irregularly checkered appearance, owing
to the fact that the white interior shows between the dark raised
parts. The[Pg 123]
[Pg 124] interior is at first pure white and of solid consistency,
but later becomes softer and yellowish, and then contains an
amber-colored juice. After the puffball has matured, the contents
change into a brown, dustlike mass, and the top falls off; and it is
then inedible. All varieties of puffball with a pure white interior
are harmless, if eaten before becoming crumbly and powdery. There is
only one species thought to be poisonous, and that has a yellow-brown
exterior, while the interior is purple-black, marbled with white.
 Fig. 39.
Fig. 39.
THE EDIBLE PUFFBALL.
(Lycoperdon Cyathiforme.)
Upper illustration shows entire plant; lower, a section.
POISONOUS MUSHROOMS FREQUENTLY MISTAKEN.
To escape eating poisonous mushrooms do not gather the buttons, and
be suspicious of those growing in woods and shady spots that show any
bright hue, or have a scaly or dotted cap, or white gills.[9] By so
doing the following species will be avoided.
Fly Amanita (Amanita Muscaria).—Infusions of this mushroom made by
boiling in water are used to kill flies. This species grows in woods
and shady places, by roadsides, and along the borders of fields, and
is much commoner than the campestris in some localities. It prefers
a poor, gravelly soil, and is found in summer.
The stem is hollow and its gills are white. The[Pg 125]
[Pg 126]
[Pg 127] cap is variously
colored, white, orange, yellow, or even brilliant red, and dotted over
with corklike particles or warty scales which are easily rubbed off.
There is a large, drooping collar about the upper part of the hollow,
white stem, and the latter is scaly below with a bulbous enlargement
at its base.
The young mushrooms, or buttons, do not exhibit the dotted cap, and
the bulbous scaly base may be left in the ground when the mushroom is
picked. The fly amanita is usually larger than the common mushroom.
 Fig. 40.
Fig. 40.
A POISONOUS FUNGUS.
(Amanita Muscaria.)
The Fly Agaric.
Death Cup or Deadly Agaric (Amanita Phalloides).—This species is
more fatal in its effects than the preceding. Its salient feature is a
bulbous base surmounted and surrounded by a collar or cup out of which
the stem grows. This is often buried beneath the ground, however, so
that it may escape notice. The gills and stem are white like the
preceding, but the cap is usually not dotted but glossy, white,
greenish, or yellow. There is also a broad, noticeable ring about the
stem, as in the fly amanita. This mushroom frequents moist, shady
spots, also along the borders of fields. It occurs singly, and rarely
in fields or pastures.
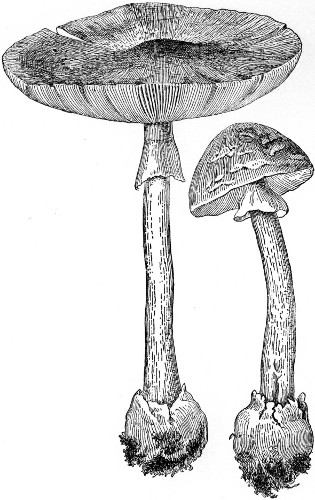 Fig. 41.
Fig. 41.
THE DEADLY AGARIC.
(Amanita Phalloides.)
This variety is very poisonous.
[Pg 128]
[Pg 129]
Part III
THE HOUSE AND GROUNDS
BY
GEORGE M. PRICE
[Pg 130]
Acknowledgment
We beg to tender grateful acknowledgment to author and publisher for
the use of Dr. George M. Price's valuable articles on sanitation. The
following extracts are taken from Dr. Price's "Handbook on
Sanitation," published by John Wiley & Son, and are covered by
copyright.
[Pg 131]
CHAPTER I
Soil and Sites
Definition.—By the term "soil" we mean the superficial layer of the
earth, a result of the geological disintegration of the primitive rock
by the action of the elements upon it and of the decay of vegetable
and animal life.
Composition.—Soil consists of solids, water, and air.
Solids.—The solid constituents of the soil are inorganic and organic
in character.
The inorganic constituents are the various minerals and elements found
alone, or in combination, in the earth, such as silica, aluminum,
calcium, iron, carbon, sodium, chlorine, potassium, etc.
The characteristics of the soil depend upon its constituents, and upon
the predominance of one or the other of its composing elements. The
nature of the soil also depends upon its physical properties. When the
disintegrated rock consists of quite large particles, the soil is
called a gravel soil. A sandy soil is one in which the particles
are very small. Sandstone is consolidated sand. Clay is soil
consisting principally of[Pg 132] aluminum silicate; in chalk, soft calcium
carbonate predominates.
The organic constituents of the soil are the result of vegetable and
animal growth and decomposition in the soil.
Ground Water.—Ground water is that continuous body or sheet of water
formed by the complete filling and saturation of the soil to a certain
level by rain water; it is that stratum of subterranean lakes and
rivers, filled up with alluvium, which we reach at a higher or lower
level when we dig wells.
The level of the ground water depends upon the underlying strata, and
also upon the movements of the subterranean water bed. The relative
position of the impermeable underlying strata varies in its distance
from the surface soil. In marshy land the ground water is at the
surface; in other places it can be reached only by deep borings. The
source of the ground water is the rainfall, part of which drains into
the porous soil until it reaches an impermeable stratum, where it
collects.
The movements of the ground water are in two directions—horizontal
and vertical. The horizontal or lateral movement is toward the seas
and adjacent water courses, and is determined by hydrostatic laws and
topographical relations. The vertical motion of the ground water is to
and from the surface, and is due to the amount of rainfall, the
pressure of tides, and water courses into which the ground water
drains. The[Pg 133] vertical variations of the ground water determine the
distance of its surface level from the soil surface, and are divided
into a persistently low-water level, about fifteen feet from the
surface; a persistently high-water level, about five feet from the
surface, and a fluctuating level, sometimes high, sometimes low.
Ground Air.—Except in the hardest granite rocks and in soil
completely filled with water the interstices of the soil are filled
with a continuation of atmospheric air, the amount depending on the
degree of porosity of the soil. The nature of the ground air differs
from that of the atmosphere only as it is influenced by its location.
The principal constituents of the air—nitrogen, oxygen, and carbonic
acid—are also found in the ground air, but in the latter the relative
quantities of O and CO2 are different.
AVERAGE COMPOSITION OF ATMOSPHERIC AIR IN 100 VOLUMES
| Nitrogen |
79.00 |
per cent. |
| Oxygen |
20.96 |
" |
| Carbonic acid |
0.04 |
" |
AVERAGE COMPOSITION OF GROUND AIR
| Nitrogen |
79.00 |
per cent. |
| Oxygen |
10.35 |
" |
| Carbonic acid |
9.74 |
" |
Of course, these quantities are not constant, but vary in different
soils, and at different depths, times, etc. The greater quantity of
CO2 in ground air is due to the process of oxidation and
decomposition taking place[Pg 134] in the soil. Ground air also contains a
large quantity of bacterial and other organic matter found in the
soil.
Ground air is in constant motion, its movements depending upon a great
many factors, some among these being the winds and movements of the
atmospheric air, the temperature of the soil, the surface temperature,
the pressure from the ground water from below, and surface and rain
water from above, etc.
Ground Moisture.—The interstices of the soil above the ground-water
level are filled with air only, when the soil is absolutely dry; but
as such a soil is very rare, all soils being more or less damp, soil
usually contains a mixture of air and water, or what is called ground
moisture.
Ground moisture is derived partly from the evaporation of the ground
water and its capillary absorption by the surface soil, and partly by
the retention of water from rains upon the surface. The power of the
soil to absorb and retain moisture varies according to the physical
and chemical, as well as the thermal, properties of the soil.
Loose sand may hold about 2 gallons of water per cubic foot; granite
takes up about 4 per cent of moisture; chalk about 15 per cent; clay
about 20 per cent; sandy loam 33 to 35 per cent; humus[10] about 40
per cent.
Ground Temperature.—The temperature of the soil is due to the direct
rays of the sun, the physicochemi[Pg 135]cal changes in its interior, and to
the internal heat of the earth.
The ground temperature varies according to the annual and diurnal
changes of the external temperature; also according to the character
of the soil, its color, composition, depth, degree of organic
oxidation, ground-water level, and degree of dampness. In hot weather
the surface soil is cooler, and the subsurface soil still more so,
than the surrounding air; in cold weather the opposite is the case.
The contact of the cool soil with the warm surface air on summer
evenings is what produces the condensation of air moisture which we
call dew.
Bacteria.—Quite a large number of bacteria are found in the soil,
especially near the surface, where chemical and organic changes are
most active. From 200,000 to 1,000,000 bacteria have been found in 1
c.c. of earth. The ground bacteria are divided into two
groups—saprophytic and pathogenic. The saprophytic bacteria are the
bacteria of decay, putrefaction, and fermentation. It is to their
benevolent action that vegetable and animal débris is decomposed,
oxidized, and reduced to its elements. To these bacteria the soil owes
its self-purifying capacity and the faculty of disintegrating animal
and vegetable débris.
The pathogenic bacteria are either those formed during the process of
organic decay, and which, introduced into the human system, are
capable of producing various diseases, or those which become lodged in
the[Pg 136] soil through the contamination of the latter by ground water and
air, and which find in the soil a favorable lodging ground, until
forced out of the soil by the movements of the ground water and air.
Contamination of the Soil.—The natural capacity of the soil to
decompose and reduce organic matter is sometimes taxed to its utmost
by the introduction into the soil of extraneous matters in quantities
which the soil is unable to oxidize in a given period. This is called
contamination or pollution of soil, and is due: (1) to surface
pollution by refuse, garbage, animal and human excreta; (2) to
interment of dead bodies of beasts and men; (3) to the introduction of
foreign deleterious gases, etc.[11]
Pollution by Surface Refuse and Sewage.—This occurs where a large
number of people congregate, as in cities, towns, etc., and very
seriously contaminates the ground by the surcharge of the surface soil
with sewage matter, saturating the ground with it, polluting the
ground water from which the drinking water is derived, and increasing
the putrefactive changes taking place in the soil. Here the pathogenic
bacteria abound, and, by multiplying, exert a very marked influence
upon the health by the possible spread of infectious diseases. Sewage
pollution of the soils and of the source of water supply is a matter
of grave im[Pg 137]portance, and is one of the chief factors of high
mortality in cities and towns.
Interment of Bodies.—The second cause of soil contamination is also
of great importance. Owing to the intense physicochemical and organic
changes taking place within the soil, all dead animal matter interred
therein is easily disposed of in a certain time, being reduced to the
primary constituents, viz., ammonia, nitrous acid, carbonic acid,
sulphureted and carbureted hydrogen, etc. But whenever the number of
interred bodies is too great, and the products of decomposition are
allowed to accumulate to a very great degree, until the capacity of
the soil to absorb and oxidize them is overtaxed, the soil, and the
air and water therein, are polluted by the noxious poisons produced by
the processes of decomposition.
Introduction of Various Foreign Materials and Gases.—In cities and
towns various pipes are laid in the ground for conducting certain
substances, as illuminating gas, fuel, coal gas, etc.; the pipes at
times are defective, allowing leakage therefrom, and permitting the
saturation of the soil with poisonous gases which are frequently drawn
up by the various currents of ground air into the open air and
adjacent dwellings.
Influence of the Soil on Health.—The intimate relations existing
between the soil upon which we live and our health, and the marked
influence of the soil on the life and well-being of man, have been
recognized from time immemorial.
[Pg 138]The influence of the soil upon health is due to: (1) the physical and
chemical character of the soil; (2) the ground-water level and degree
of dampness; (3) the organic impurities and contamination of the soil.
The physical and chemical nature of the soil, irrespective of its
water, moisture, and air, has been regarded by some authorities as
having an effect on the health, growth, and constitution of man. The
peculiar disease called cretinism, as well as goitre, has been
attributed to a predominance of certain chemicals in the soil.
The ground-water level is of great importance to the well-being of
man. Professor Pettenkofer claimed that a persistently low water level
(about fifteen feet from the surface) is healthy, the mortality being
the lowest in such places; a persistently high ground-water level
(about five feet from the surface) is unhealthy; and a fluctuating
level, varying from high to low, is the most unhealthy, and is
dangerous to life and health. Many authorities have sought to
demonstrate the intimate relations between a high water level in the
soil and various diseases.
A damp soil, viz., a soil wherein the ground moisture is very great
and persistent, has been found inimical to the health of the
inhabitants, predisposing them to various diseases by the direct
effects of the dampness itself, and by the greater proneness of damp
ground to become contaminated with various patho[Pg 139]genic bacteria and
organisms which may be drawn into the dwellings by the movements of
the ground air. As a rule, there is very little to hinder the ground
air from penetrating the dwellings of man, air being drawn in through
cellars by changes in temperature, and by the artificial heating of
houses.
The organic impurities and bacteria found in the soil are especially
abundant in large cities, and are a cause of the evil influence of
soil upon health. The impurities are allowed to drain into the ground,
to pollute the ground water and the source of water supply, and to
poison the ground air, loading it with bacteria and products of
putrefaction, thus contaminating the air and water so necessary to
life.
Diseases Due to Soil.—A great many diseases have been thought to be
due to the influence of the soil. An ætiological relation had been
sought between soil and the following diseases: malaria, paroxysmal
fevers, tuberculosis, neuralgias, cholera, yellow fever, bubonic
plague, typhoid, dysentery, goitre and cretinism, tetanus, anthrax,
malignant Œdema, septicæmia, etc.
Sites.—From what we have already learned about the soil, it is
evident that it is a matter of great importance as to where the site
for a human habitation is selected, for upon the proper selection of
the site depend the health, well-being, and longevity of the
inhabitants. The requisite characteristics of a healthy site for
dwellings are: a dry, porous, permeable soil; a low and nonfluctuating
ground-water level, and a[Pg 140] soil retaining very little dampness, free
from organic impurities, and the ground water of which is well drained
into distant water courses, while its ground air is uncontaminated by
pathogenic bacteria. Exposure to sunlight, and free circulation of
air, are also requisite.
According to Parkes, the soils in the order of their fitness for
building purposes are as follows: (1) primitive rock; (2) gravel, with
pervious soil; (3) sandstone; (4) limestone; (5) sandstone, with
impervious subsoil; (6) clays and marls; (7) marshy land, and (8) made
soils.
It is very seldom, however, that a soil can be secured having all the
requisites of a healthy site. In smaller places, as well as in cities,
commercial and other reasons frequently compel the acquisition of and
building upon a site not fit for the purpose; it then becomes a
sanitary problem how to remedy the defects and make the soil suitable
for habitation.
Prevention of the Bad Effects of the Soil on Health.—The methods
taught by sanitary science to improve a defective soil and to prepare
a healthy site are the following:
- (1) Street paving and tree planting.
- (2) Proper construction of houses.
- (3) Subsoil drainage.
Street Paving serves a double sanitary purpose. It prevents street
refuse and sewage from penetrating the ground and contaminating the
surface soil, and it[Pg 141] acts as a barrier to the free ascension of
deleterious ground air.[12]
Tree Planting serves as a factor in absorbing the ground moisture
and in oxidizing organic impurities.
The Proper Construction of the House has for its purpose the
prevention of the entrance of ground moisture and air inside the house
by building the foundations and cellar in such a manner as to entirely
cut off communication between the ground and the dwelling. This is
accomplished by putting under the foundation a solid bed of concrete,
and under the foundation walls damp-proof courses.
The following are the methods recommended by the New York City
Tenement House Department for the[Pg 142] water-proofing and damp-proofing of
foundation walls and cellars:
Water-proofing and Damp-proofing of Foundation Walls.—"There shall
be built in with the foundation walls, at a level of six (6) inches
below the finished floor level, a course of damp-proofing consisting
of not less than two (2) ply of tarred felt (not less than fifteen
(15) pounds weight per one hundred (100) square feet), and one (1) ply
of burlap, laid in alternate layers, having the burlap placed between
the felt, and all laid in hot, heavy coal-tar pitch, or liquid
asphalt, and projecting six (6) inches inside and six (6) inches
outside of the walls.
"There shall be constructed on the outside surface of the walls a
water-proofing lapping on to the damp-proof course in the foundation
walls and extending up to the soil level. This water-proofing shall
consist of not less than two (2) ply of tarred felt (of weight
specified above), laid in hot, heavy coal-tar pitch, or liquid
asphalt, finished with a flow of hot pitch of the same character. This
water-proofing to be well stuck to the damp course in the foundation
walls. The layers of felt must break joints."
Water-proofing and Damp-proofing of Cellar Floors.—"There shall be
laid, above a suitable bed of rough concrete, a course of
water-proofing consisting of not less than three (3) ply of tarred
felt (not less than fifteen (15) pounds weight per one hundred (100)
square feet), laid in hot, heavy coal-tar[Pg 143]
[Pg 144] pitch, or liquid asphalt,
finished with a flow of hot pitch of the same character. The felt is
to be laid so that each layer laps two-thirds of its width over the
layer immediately below, the contact surface being thoroughly coated
with the hot pitch over its entire area before placing the upper
layer. The water-proofing course must be properly lapped on and
secured to the damp course in the foundation walls."
Other methods of damp-proofing foundations and cellars consist in the
use of slate or sheet lead instead of tar and tarred paper. An
additional means of preventing water and dampness from coming into
houses has been proposed in the so-called "dry areas," which are open
spaces four to eight feet wide between the house proper and the
surrounding ground, the open spaces running as deep as the foundation,
if possible. The dry areas are certainly a good preventive against
dampness coming from the sides of the house.
 Fig. 4.
Fig. 4.
CONCRETE FOUNDATION AND DAMP-PROOF COURSE.
Subsoil Drainage.—By subsoil drainage is meant the reducing of the
level of the ground water by draining all subsoil water into certain
water courses, either artificial or natural. Subsoil drainage is not a
modern discovery, as it was used in many ancient lands, and was
extensively employed in ancient Rome, the valleys and suburbs of which
would have been uninhabitable but for the draining of the marshes by
the so-called "cloacæ" or drains, which lowered the ground-water
level of the low parts of the city and made them fit to build upon.
The drains for the conduction of sub[Pg 145]soil water are placed at a
certain depth, with a fall toward the exit. The materials for the
drain are either stone and gravel trenches, or, better, porous
earthenware pipes or ordinary drain tile. The drains must not be
impermeable or closed, and sewers are not to be used for drainage
purposes. Sometimes open, V-shaped pipes are laid under the regular
sewers, if these are at the proper depth.
By subsoil drainage it is possible to lower the level of ground water
wherever it is near or at the surface, as in swamps, marsh, and other
lands, and prepare lands previously uninhabitable for healthy sites.
[Pg 146]
CHAPTER II
Ventilation
Definition.—The air within an uninhabited room does not differ from
that without. If the room is occupied by one or more individuals,
however, then the air in the room soon deteriorates, until the
impurities therein reach a certain degree incompatible with health.
This is due to the fact that with each breath a certain quantity of
CO2, organic impurities, and aqueous vapor is exhaled; and these
products of respiration soon surcharge the air until it is rendered
impure and unfit for breathing. In order to render the air pure in
such a room, and make life possible, it is necessary to change the air
by withdrawing the impure, and substituting pure air from the outside.
This is ventilation.
Ventilation, therefore, is the maintenance of the air in a confined
space in a condition conducive to health; in other words, "ventilation
is the replacing of the impure air in a confined space by pure air
from the outside."
Quantity of Air Required.—What do we regard as impure air? What is
the index of impurity? How much air is required to render pure an air
in a given space, in a given time, for a given number of people?[Pg 147] How
often can the change be safely made, and how? These are the problems
of ventilation.
An increase in the quantity of CO2 [carbon dioxide gas], and a
proportionate increase of organic impurities, are the results of
respiratory vitiation of the air; and it has been agreed to regard the
relative quantity of CO2 as the standard of impurity, its increase
serving as an index of the condition of the air. The normal quantity
of CO2 in the air is 0.04 per cent, or 4 volumes in 10,000; and it
has been determined that whenever the CO2 reaches 0.06 per cent, or 6
parts per 10,000, the maximum of air vitiation is reached—a point
beyond which the breathing of the air becomes dangerous to health.
We therefore know that an increase of 2 volumes of CO2 in 10,000 of
air constitutes the maximum of admissible impurity; the difference
between 0.04 per cent and 0.06 per cent. Now, a healthy average adult
at rest exhales in one hour 0.6 cubic foot of CO2. Having determined
these two factors—the amount of CO2 exhaled in one hour and the
maximum of admissible impurity—we can find by dividing 0.6 by 0.0002
(or 0.02 per cent) the number of cubic feet of air needed for one
hour,==3,000.
Therefore, a room with a space of 3,000 cubic feet, occupied by one
average adult at rest, will not reach its maximum of impurity (that
is, the air in such a room will not be in need of a change) before one
hour has elapsed.
[Pg 148]The relative quantity of fresh air needed will differ for adults at
work and at rest, for children, women, etc.; it will also differ
according to the illuminant employed, whether oil, candle, gas,
etc.—an ordinary 3-foot gas-burner requiring 1,800 cubic feet of air
in one hour.
It is not necessary, however, to have 3,000 cubic feet of space for
each individual in a room, for the air in the latter can safely be
changed at least three times within one hour, thus reducing the air
space needed to about 1,000 cubic feet. This change of air or
ventilation of a room can be accomplished by mechanical means oftener
than three times in an hour, but a natural change of more than three
times in an hour will ordinarily create too strong a current of air,
and may cause draughts and chills dangerous to health.
In determining the cubic space needed, the height of the room as well
as the floor space must be taken into consideration. As a rule the
height of a room ought to be in proportion to the floor space, and in
ordinary rooms should not exceed fourteen feet, as a height beyond
that is of very little advantage.[13]
Forces of Ventilation.—We now come to the question of the various
modes by which change in the air of a room is possible. Ventilation is
natural or[Pg 149] artificial according to whether artificial or mechanical
devices are or are not used. Natural ventilation is only possible
because our buildings and houses, their material and construction, are
such that numerous apertures and crevices are left for air to come in;
for it is evident that if a room were hermetically air-tight, no
natural ventilation would be possible.
The properties of air which render both natural and artificial
ventilation possible are diffusion, motion, and gravity. These three
forces are the natural agents of ventilation.
There is a constant diffusion of gases taking place in the air; this
diffusion takes place even through stone and through brick walls. The
more porous the material of which the building is constructed, the
more readily does diffusion take place. Dampness, plastering,
painting, and papering of walls diminish diffusion, however.
The second force in ventilation is the motion of air or winds. This is
the most powerful agent of ventilation, for even a slight,
imperceptible wind, traveling about two miles an hour, is capable,
when the windows and doors of a room are open, of changing the air of
a room 528 times in one hour. Air passes also through brick and stone
walls. The objections to winds as a sole mode of ventilation are their
inconstancy and irregularity. When the wind is very slight its
ventilating influence is very small; on the other hand, when the wind
is strong it cannot be[Pg 150] utilized as a means of ventilation on account
of the air currents being too strong and capable of exerting
deleterious effects on health.
The third, the most constant and reliable, and, in fact, principal
agent of ventilation is the specific gravity of the air, and the
variations in the gravity and consequent pressure which are results of
the variations in temperature, humidity, etc. Whenever air is warmer
in one place than in another, the warmer air being lighter and the
colder air outside being heavier, the latter exerts pressure upon the
air in the room, causing the lighter air in the room to escape and be
displaced by the heavier air from the outside, thus changing the air
in the room. This mode of ventilation is always constant and at work,
as the very presence of living beings in the room warms the air
therein, thus causing a difference from the outside air and effecting
change of air from the outside to the inside of the room.
Methods of Ventilation.—The application of these principles of
ventilation is said to be accomplished in a natural or an artificial
way, according as mechanical means to utilize the forces and
properties of air are used or not. But in reality natural ventilation
can hardly be said to exist, since dwellings are so constructed as to
guard against exposure and changes of temperature, and are usually
equipped with numerous appliances for promoting change of air.
Windows, doors, fireplaces, chimneys, shafts, courts, etc.,[Pg 151] are all
artificial methods of securing ventilation, although we usually regard
them as means of natural ventilation.
Natural Ventilation.—The means employed for applying the properties
of diffusion are the materials of construction. A porous material
being favorable for diffusion, some such material is placed in several
places within the wall, thus favoring change of air. Imperfect
carpenter work is also a help, as the cracks and openings left are
favorable for the escape and entrance of air.
Wind, or the motion of air, is utilized either directly, through
windows, doors, and other openings; or indirectly, by producing a
partial vacuum in passing over chimneys and shafts, causing suction of
the air in them, and the consequent withdrawal of the air from the
rooms.
The opening of windows and doors is possible only in warm weather; and
as ventilation becomes a problem only in temperate and cold weather,
the opening of windows and doors cannot very well be utilized without
causing colds, etc. Various methods have therefore been proposed for
using windows for the purposes of ventilation without producing
forcible currents of air.
The part of the window best fitted for the introduction of air is the
space between the two sashes, where they meet. The ingress of air is
made possible whenever the lower sash is raised or the upper[Pg 152] one is
lowered. In order to prevent cold air from without entering through
the openings thus made, it has been proposed by Hinkes Bird to fit a
block of wood in the lower opening; or else, as in Dr. Keen's
arrangement, a piece of paper or cloth is used to cover the space left
by the lifting or lowering of either or both sashes. Louvers or
inclined panes or parts of these may also be used. Parts or entire
window panes[Pg 153] are sometimes wholly removed and replaced by tubes or
perforated pieces of zinc, so that air may come in through the
apertures. Again, apertures for inlets and outlets may be made
directly in the walls of the rooms. These openings are filled in with
porous bricks or with specially made bricks (like Ellison's conical
bricks), or boxes provided with several openings. A very useful
apparatus of this kind is the so-called Sheringham valve, which
consists of an iron box fitted into the wall, the front of the box
facing the room having an iron valve hinged along its lower edge, and
so constructed that it can be opened or be closed at will to let a
current of air pass upward. Another very good apparatus of this kind
is the Tobin ventilator, consisting of horizontal tubes let through
the walls, the outer ends open to the air, but the inner[Pg 154] ends
projecting into the room, where they are joined by vertical tubes
carried up five feet or more from the floor, thus allowing the outside
air to enter upwardly into the room. This plan is also adapted for
filtering and cleaning the incoming air by placing cloth or other
material across the lumen of the hori[Pg 155]zontal tubes to intercept dust,
etc. McKinnell's ventilator is also a useful method of ventilation,
especially of underground rooms.
 Fig. 5.
Fig. 5.
HINKES BIRD WINDOW. (Taylor.)
 Fig. 6.
Fig. 6.
ELLISON'S AIR INLETS. (Knight.)
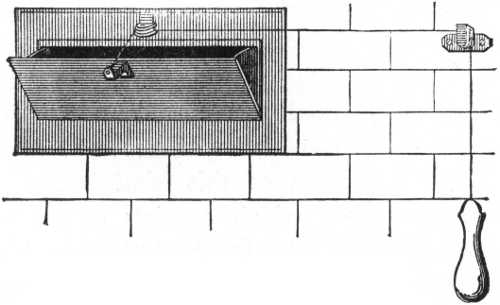 Fig. 7.
Fig. 7.
SHERINGHAM VALVE. (Taylor.)
 Fig. 8.
Fig. 8.
THE TOBIN VENTILATOR. (Knight.)
 Fig. 9.
Fig. 9.
McKINNELL'S VENTILATOR. (Taylor.)
To assist the action of winds over the tops of shafts and chimneys,
various cowls have been devised. These cowls are arranged so as to
help aspirate the air from the tubes and chimneys, and prevent a down
draught.
The same inlets and outlets which are made to utilize winds may also
be used for the ventilation effected by the motion of air due to
difference in the specific gravity of outside and inside air. Any
artificial warming of the air in the room, whether by illuminants or
by the various methods of heating rooms, will aid in ventilating it,
the chimneys acting[Pg 156] as powerful means of removal for the warmer air.
Various methods have also been proposed for utilizing the chimney,
even when no stoves, etc., are connected with it, by placing a
gaslight within the chimney to cause an up draught and consequent
aspiration of the air of the room through it.
 Fig. 10.
Fig. 10.
VENTILATING THROUGH CHIMNEY. (Knight.)
The question of the number, relative size, and position of the inlets
and outlets is a very important one, but we can here give only an
epitome of the requirements. The inlet and outlet openings should be
about twenty-four inches square per head. Inlet openings should be
short, easily cleaned, sufficient in number to insure a proper
distribution of air; should be protected from heat, provided with
valves so as to regulate the inflow of air, and, if possible, should
be placed[Pg 157] so as to allow the air passing through them to be warmed
before entering the room.[14] Outlet openings should be placed near
the ceiling, should be straight and smooth, and, if possible, should
be heated so as to make the air therein warmer, thus preventing a down
draught, as is frequently the case when the outlets become inlets.
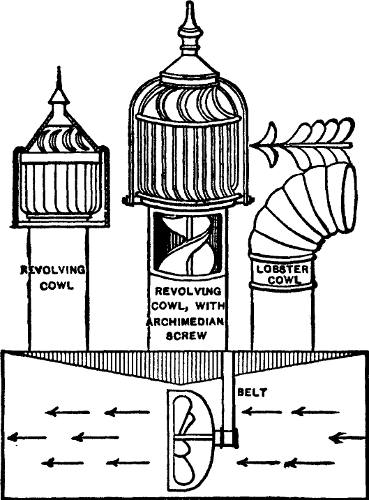 Fig. 11.
Fig. 11.
COWL VENTILATOR. (Knight.)
Artificial Ventilation.—Artificial ventilation is accomplished either
by aspirating the air from the building, known as the vacuum or
extraction method, or by[Pg 158] forcing into the building air from without;
this is known as the plenum or propulsion method.
The extraction of the air in a building is done by means of heat, by
warming the air in chimneys or special tubes, or by mechanical means
with screws or fans run by steam or electricity; these screws or fans
revolve and aspirate the air of the rooms, and thus cause pure air to
enter.
 Fig. 12.
Fig. 12.
AN AIR PROPELLER.
The propelling method of ventilation is carried out[Pg 159] by mechanical
means only, air being forced in from the outside by fans, screws,
bellows, etc.
Artificial ventilation is applicable only where a large volume of air
is needed, and for large spaces, such as theaters, churches, lecture
rooms, etc. For the ordinary building the expense for mechanical
contrivances is too high.
On the whole, ventilation without complex and cumbersome mechanisms is
to be preferred.[15]
[Pg 160]
CHAPTER III
Warming
Ventilation and Heating.—The subject of the heating of our rooms and
houses is very closely allied to that of ventilation, not only because
both are a special necessity at the same time of the year, but also
because we cannot heat a room without at the same time having to
ventilate it by providing an egress for the products of combustion and
introducing fresh air to replace the vitiated.
Need of Heating.—In a large part of the country, and during the
greater period of the year, some mode of artificial heating of rooms
is absolutely necessary for our comfort and health. The temperature of
the body is 98° to 99° F., and there is a constant radiation of heat
due to the cooling of the body surface. If the external temperature is
very much below that of the body, and if the low temperature is
prolonged, the radiation of heat from the body is too rapid, and
colds, pneumonia, etc., result. The temperature essential for the
individual varies according to age, constitution, health, environment,
occupation, etc. A child, a sick person, or one at rest requires a
relatively higher tem[Pg 161]perature than a healthy adult at work. The mean
temperature of a room most conducive to the health of the average
person is from 65° to 75° F.
The Three Methods of Heating.—The heating of a room can be
accomplished either directly by the rays of the sun or processes of
combustion. We thus receive radiant heat, exemplified by that of
open fires and grates.
Or, the heating of places can be accomplished by the heat of
combustion being conducted through certain materials, like brick
walls, tile, stone, and also iron; this is conductive heat, as
afforded by stoves, etc.
Or, the heat is conveyed by means of air, water, or steam from one
place to another, as in the hot-water, hot-air, and steam systems of
heating; this we call convected heat.
There is no strict line of demarcation differentiating the three
methods of heating, as it is possible that a radiant heat may at the
same time be conductive as well as convective—as is the case in the
Galton fireplace, etc.
Materials of Combustion.—The materials of combustion are air, wood,
coal, oil, and gas. Air is indispensable, for, without oxygen, there
can be no combustion. Wood is used in many places, but is too bulky
and expensive. Oil is rarely used as a material of combustion, its
principal use being for illumination. Coal is the best and cheapest
material for combustion.[Pg 162] The chief objection against its use is the
production of smoke, soot, and of various gases, as CO, CO2, etc. Gas
is a very good, in fact, the best material for heating, especially if,
when used, it is connected with chimneys; otherwise, it is
objectionable, as it burns up too much air, vitiates the atmosphere,
and the products of combustion are deleterious; it is also quite
expensive. The ideal means of heating is electricity.
Chimneys.—All materials used for combustion yield products more or
less injurious to health. Every system of artificially heating houses
must therefore have not only means of introducing fresh air to aid in
the burning up of the materials, but also an outlet for the vitiated,
warmed air, partly charged with the products of combustion. These
outlets are provided by chimneys. Chimneys are hollow tubes or shafts
built of brick and lined with earthen pipes or other material inside.
These tubes begin at the lowest fireplace or connection, and are
carried up several feet above the roof. The thickness of a chimney is
from four to nine inches; the shape square, rectangular, or,
preferably, circular. The diameter of the chimney depends upon the
size of the house, the number of fire connections, etc. It should be
neither too small nor too large. Square chimneys should be twelve to
sixteen inches square; circular ones from six to eight inches in
diameter for each fire connection. The chimney consists of a shaft,
or vertical tube, and cowls placed over chimneys on the roof to
prevent down[Pg 163] draughts and the falling in of foreign bodies. That part
of the chimney opening into the fireplace is called the throat.
Smoky Chimneys.—A very frequent cause of complaint in a great many
houses is the so-called "smoky chimney"; this is the case when smoke
and coal gas escape from the chimney and enter the living rooms. The
principal causes of this nuisance are:
(1) A too wide or too narrow diameter of the shafts. A shaft which is
too narrow does not let all the smoke escape; one which is too wide
lets the smoke go up only in a part of its diameter, and when the
smoke meets a countercurrent of cold air it is liable to be forced
back into the rooms.
(2) The throat of the chimney may be too wide, and will hold cold air,
preventing the warming of the air in the chimneys and the consequent
up draught.
(3) The cowls may be too low or too tight, preventing the escape of
the smoke.
(4) The brickwork of the chimney may be loose, badly constructed, or
broken into by nails, etc., thus allowing smoke to escape therefrom.
(5) The supply of air may be deficient, as when all doors and windows
are tightly closed.
(6) The chimney may be obstructed by soot or some foreign material.
(7) The wind above the house may be so strong that its pressure will
cause the smoke from the chimney to be forced back.
[Pg 164](8) If two chimneys rise together from the same house, and one is
shorter than the other, the draught of the longer chimney may cause an
inversion of the current of air in the lower chimney.
(9) Wet fuel when used will cause smoke by its incomplete combustion.
(10) A chimney without a fire may suck down the smoke from a
neighboring chimney; or, if two fireplaces in different rooms are
connected with the same chimney, the smoke from one room may be drawn
into the other.
Methods of Heating. Open Fireplaces and Grates.—Open fireplaces and
fires in grates connected with chimneys, and using coal, wood, or gas,
are very comfortable; nevertheless there are weighty objections to
them. Firstly, but a very small part of the heat of the material
burning is utilized, only about twelve per cent being radiated into
the room, the rest going up the chimney. Secondly, the heat of grates
and fireplaces is only local, being near the fires and warming only
that part of the person exposed to it, leaving the other parts of the
room and person cold. Thirdly, the burning of open fires necessitates
a great supply of air, and causes powerful draughts.
The open fireplace can, however, be greatly improved by surrounding
its back and sides by an air space, in which air can be warmed and
conveyed into the upper part of the room; and if a special air inlet
is provided for supplying the fire with fresh air to be[Pg 165] warmed, we
get a very valuable means of heating. These principles are embodied in
the Franklin and Galton grates. A great many other grates have been
suggested, and put on the market, but the principal objection to them
is their complexity and expense, making their use a luxury not
attainable by the masses.
 Fig. 13.
Fig. 13.
A GALTON GRATE. (Tracy.)
Stoves.—Stoves are closed receptacles in which fuel is burned, and
the heat produced is radiated toward the persons, etc., near them, and
also conducted,[Pg 166] through the iron or other materials of which the
stoves are made, to surrounding objects. In stoves seventy-five per
cent of the fuel burned is utilized. They are made of brick, tile, and
cast or wrought iron.
Brick stoves, and stoves made of tile, are extensively used in some
European countries, as Russia, Germany, Sweden, etc.; they are made of
slow-conducting material, and give a very equable, efficient, and
cheap heat, although their ventilating power is very small.
Iron is used very extensively because it is a very good conductor of
heat, and can be made into very convenient forms. Iron stoves,
however, often become superheated, dry up, and sometimes burn the air
around them, and produce certain deleterious gases during combustion.
When the fire is confined in a clay fire box, and the stove is not
overheated, a good supply of fresh air being provided and a vessel of
water placed on the stove to reduce the dryness of the air, iron
stoves are quite efficient.
Hot-air Warming.—In small houses the warming of the various rooms and
halls can be accomplished by placing the stove or furnace in the
cellar, heating a large quantity of air and conveying it through
proper tubes to the rooms and places to be warmed. The points to be
observed in a proper and efficient hot-air heating system are the
following:
(1) The furnace must be of a proper size in proportion to the area of
space to be warmed. (2) The[Pg 167] joints and parts of the furnace must be
gas-tight. (3) The furnace should be placed on the cold side of the
house, and provision made to prevent cellar air from being drawn up
into the cold-air box of the furnace. (4) The air for the supply of
the furnace must be[Pg 168] gotten from outside, and the source must be pure,
above the ground level, and free from contamination of any kind.[16]
(5) The cold-air box and ducts must be clean, protected against the
entrance of vermin, etc., and easily cleaned. (6) The air should not
be overheated. (7) The hot-air flues or tubes must be short, direct,
circular, and covered with asbestos or some other non-conducting
material.
 Fig. 14.
Fig. 14.
A HOT-AIR FURNACE.
The cold air from outside comes to the COLD-AIR INTAKE through the
cold-air duct, enters the furnace from beneath, and is heated by
passing around the FIRE POT and the annular combustion chamber above.
It then goes through pipes to the various registers throughout the
house. The coal is burnt in the fire pot, the gases are consumed in
the combustion chamber above, while the heat eventually passes into
the SMOKE FLUE. The WATER PAN supplies moisture to the air.
Hot-water System.—The principles of hot-water heating are very
simple. Given a circuit of pipes filled with water, on heating the
lower part of the circuit the water, becoming warmer, will rise,
circulate, and heat the pipes in which it is contained, thus warming
the air in contact with the pipes. The lower part of the circuit of
pipe begins in the furnace or heater, and the other parts of the
circuit are conducted through the various rooms and halls throughout
the house to the uppermost story. The pipes need not be straight all
through; hence, to secure a larger area for heating, they are
convoluted within the furnace, and also in the rooms, where the
convoluted pipes are called radiators. The water may be warmed by
the low- or high-pressure system; in the latter, pipes of small
diameter may be employed; while in the former, pipes of a large
diameter will be required. The character, etc., of the boilers,
furnace, pipes, etc., cannot be gone into here.
[Pg 169]Steam-heating System.—The principle of steam heating does not differ
from that of the hot-water system. Here the pressure is greater and
steam is employed instead of water. The steam gives a greater degree
of heat, but the pipes must be stronger and able to withstand the
pressure. There are also combinations of steam and hot-water heating.
For large houses either steam or hot-water heating is the best means
of warming, and, if properly constructed and cared for, quite
healthy.[17]
[Pg 170]
CHAPTER IV
Disposal of Sewage
Waste Products.—There is a large amount of waste products in human
and social economy. The products of combustion, such as ashes,
cinders, etc.; the products of street sweepings and waste from houses,
as dust, rubbish, paper, etc.; the waste from various trades; the
waste from kitchens, e. g., scraps of food, etc.; the waste water from
the cleansing processes of individuals, domestic animals, clothing,
etc.; and, finally, the excreta—urine and fæces—of man and animals;
all these are waste products that cannot be left undisposed of, more
especially in cities, and wherever a large number of people
congregate. All waste products are classified into three distinct
groups: (1) refuse, (2) garbage, and (3) sewage.
The amount of refuse and garbage in cities is quite considerable;
in Manhattan, alone, the dry refuse amounts to 1,000,000 tons a year,
and that of garbage to 175,000 tons per year. A large percentage of
the dry refuse and garbage is valuable from a commercial standpoint,
and could be utilized, with proper facilities for collection and
separation. The disposal of refuse and garbage has not as yet been
satisfac[Pg 171]torily dealt with. The modes of waste disposal in the United
States are: (1) dumping into the sea; (2) filling in made land, or
plowing into lands; (3) cremation and (4) reduction by various
processes, and the products utilized.
Sewage.—By sewage we mean the waste and effete human matter and
excreta—the urine and fæces of human beings and the urine of domestic
animals (the fæces of horses, etc., has great commercial value, and is
usually collected separately and disposed of for fertilizing
purposes).
The amount of excreta per person has been estimated (Frankland) as 3
ounces of solid and 40 ounces of fluid per day, or about 30 tons of
solid and 100,000 gallons of fluid for each 1,000 persons per year.
In sparsely populated districts the removal and ultimate disposal of
sewage presents no difficulties; it is returned to the soil, which, as
we know, is capable of purifying, disintegrating, and assimilating
quite a large amount of organic matter. But when the number of
inhabitants to the square mile increases, and the population becomes
as dense as it is in some towns and cities, the disposal of the human
waste products becomes a question of vast importance, and the proper,
as well as the immediate and final, disposal of sewage becomes a
serious sanitary problem.
It is evident that sewage must be removed in a[Pg 172] thorough manner,
otherwise it would endanger the lives and health of the people.
The dangers of sewage to health are:
(1) From its offensive odors, which, while not always directly
dangerous to health, often produce headaches, nausea, etc.
(2) The organic matter contained in sewage decomposes and eliminates
gases and other products of decomposition.
(3) Sewage may contain a large number of pathogenic bacteria (typhoid,
dysentery, cholera, etc.).
(4) Contamination of the soil, ground water, and air by percolation of
sewage.
The problem of sewage disposal is twofold: (1) immediate, viz., the
need of not allowing sewage to remain too long on the premises, and
its immediate removal beyond the limits of the city; and (2) the final
disposition of the sewage, after its removal from the cities, etc.
Modes of Ultimate Disposal of Sewage.—The chief constituents of
sewage are organic matter, mineral salts, nitrogenous substances,
potash, and phosphoric acid. Fresh-mixed excrementitious matter has an
acid reaction, but within twelve to twenty hours it becomes alkaline,
because of the free ammonia formed in it. Sewage rapidly decomposes,
evolving organic and fetid matters, ammonium sulphide, sulphureted and
carbureted hydrogen, etc., besides teeming with[Pg 173] animal and bacterial
life. A great many of the substances contained in sewage are valuable
as fertilizers of soil.
The systems of final disposal of sewage are as follows:
- (1) Discharge into seas, lakes, and rivers.
- (2) Cremation.
- (3) Physical and chemical precipitation.
- (4) Intermittent filtration.
- (5) Land irrigation.
- (6) "Bacterial" methods.
Discharge into Waters.—The easiest way to dispose of sewage is to
let it flow into the sea or other running water course. The objections
to sewage discharging into the rivers and lakes near cities, and
especially such lakes and rivers as supply water to the
municipalities, are obvious. But as water can purify a great amount of
sewage, this method is still in vogue in certain places, although it
is to be hoped that it will in the near future be superseded by more
proper methods. The objection against discharging into seas is the
operation of the tides, which cause a backflow and overflow of sewage
from the pipes. This backflow is remedied by the following methods:
(1) providing tidal flap valves, permitting the outflow of sewage, but
preventing the inflow of sea water; (2) discharging the sewage
intermittently, only during low tide; and (3) providing a constant
outflow by means of steam-power pressure.
[Pg 174]Cremation.—Another method of getting rid of the sewage without
attempting to utilize it is by cremation. The liquid portion of the
sewage is allowed to drain and discharge into water courses, and the
more or less solid residues are collected and cremated in suitable
crematories.
Precipitation.—This method consists in separating the solid matters
from the sewage by precipitation by physical or chemical processes,
the liquid being allowed to drain into rivers and other waters, and
the precipitated solids utilized for certain purposes. The
precipitation is done either by straining the sewage, collecting it
into tanks, and letting it subside, when the liquid is drawn off and
the solids remain at the bottom of the tanks, a rather unsatisfactory
method; or, by chemical processes, precipitating the sewage by
chemical means, and utilizing the products of such precipitation. The
chemical agents by which precipitation is accomplished are many and
various; among them are lime, alum, iron perchloride, phosphates, etc.
Intermittent Filtration.—Sewage may be purified mechanically and
chemically by method of intermittent filtration by passing it through
filter beds of gravel, sand, coke, cinders, or any such materials.
Intermittent filtration has passed beyond the experimental stage and
has been adopted already by a number of cities where such a method of
sewage disposal seems to answer all purposes.
[Pg 175]Land Irrigation.—In this method the organic and other useful
portions of sewage are utilized for irrigating land, to improve garden
and other vegetable growths by feeding the plants with the organic
products of animal excretion. Flat land, with a gentle slope, is best
suited for irrigation. The quantity of sewage disposed of will depend
on the character of the soil, its porosity, the time of the year,
temperature, intermittency of irrigation, etc. As a rule, one acre of
land is sufficient to dispose of the sewage of 100 to 150 people.
Bacterial Methods.—The other biological methods, or the so-called
"bacterial" sewage treatment, are but modifications of the filtration
and irrigation methods of sewage disposal. Properly speaking the
bacterial purification of sewage is the scientific application of the
knowledge gained by the study of bacterial life and its action upon
sewage.
In intermittent filtration the sewage is passed through filter beds of
sands, etc., upon which filter beds the whole burden of the
purification of the sewage rests. In the bacterial methods the work of
purification is divided between the septic tanks where the sewage is
first let into and where it undergoes the action of the anaërobic
bacteria, and from these septic tanks the sewage is run to the contact
beds of coke and cinders to further undergo the action of the aërobic
bacteria, after the action of which the nitrified sewage is in a
proper form to be utilized for[Pg 176] fertilization of land, etc. The septic
tanks are but a modification of the common cesspool, and are
constructed of masonry, brick, and concrete.
There are a number of special applications of the bacterial methods of
sewage treatment, into which we cannot go here.
Sewage Disposal in the United States.—According to its location,
position, etc., each city in the United States has its own method of
final disposition of sewage. Either one or the other, or a combination
of two of the above methods, is used.
The following cities discharge their sewage into the sea: Portland,
Salem, Lynn, Gloucester, Boston, Providence, New York, Baltimore,
Charleston, and Savannah.
The following cities discharge their sewage into rivers and lakes:
Philadelphia, Cincinnati, St. Louis, Albany, Minneapolis, St. Paul,
Washington, Buffalo, Detroit, Richmond, Chicago, Milwaukee, and
Cleveland.
"Worcester uses chemical precipitation. In Atlanta a part of the soil
is cremated, but the rest is deposited in pits 8 × 10 feet, and 5 feet
deep. It is then thoroughly mixed with dry ashes from the crematory,
and afterwards covered with either grain or grass. In Salt Lake City
and in Woonsocket it is disposed of in the same way. In Indianapolis
it is composted with marl and sawdust, and after some months used as a
fertilizer. A portion of the sew[Pg 177]age is cremated in Atlanta, Camden,
Dayton, Evansville, Findlay, Ohio; Jacksonville, McKeesport, Pa.;
Muncie, and New Brighton. In Atlanta, in 1898, there were cremated
2,362 loads of sewage. In Dayton, during 30 days, there were cremated
1,900 barrels of 300 pounds each." (Chapin, Mun. San. in U. S.)
The Immediate Disposal of Sewage.—The final disposition of sewage is
only one part of the problem of sewage disposal; the other part is how
to remove it from the house into the street, and from the street into
the places from which it is finally disposed.
The immediate disposal of sewage is accomplished by two methods—the
so-called dry, and the water-carriage methods. By the dry method
we mean the removal of sewage without the aid of water, simply
collecting the dry and liquid portions of excreta, storing it for some
time, and then removing it for final disposal. By the water-carriage
method is understood the system by which sewage, solid and liquid, is
flushed out by means of water, through pipes or conduits called
sewers, from the houses through the streets to the final destination.
The Dry Methods.—The dry or conservacy method of sewage disposal is a
primitive method used by all ancient peoples, in China at the present
time, and in all villages and sparsely populated districts; it has for
its basic principle the return to mother earth of all excreta, to be
used and worked over in its natural[Pg 178] laboratory. The excreta are
simply left in the ground to undergo in the soil the various organic
changes, the difference in methods being only as regards the vessels
of collection and storage.
The methods are:
- (1) Cesspool and privy vault.
- (2) Pail system.
- (3) Pneumatic system.
The Privy Vault is the general mode of sewage disposal in villages,
some towns, and even in some large cities, wherever sewers are not
provided. In its primitive and unfortunately common form, the privy
vault is nothing but a hole dug in the ground near or at some distance
from the house; the hole is but a few feet deep, with a plank or rough
seat over it, and an improvised shed over all. The privy is filled
with the excreta; the liquids drain into the adjacent ground, which
becomes saturated, and contaminates the nearest wells and water
courses. The solid portion is left to accumulate until the hole is
filled or the stench becomes unbearable, when the hole is either
covered up and forgotten, or the excreta are removed and the hole used
over again. This is the common privy as we so often find it near the
cottages and mansions of our rural populace, and even in towns. A
better and improved form of privy is that built in the ground, and
made water-tight by being constructed of bricks set in cement, the
privy being placed at a distance from the house, the shed[Pg 179] over it
ventilated, and the contents of the privy removed regularly and at
stated intervals, before they become a nuisance. At its best, however,
the privy vault is an abomination, as it can scarcely be so well
constructed as not to contaminate the surrounding soil, or so often
cleaned as to prevent decomposition and the escape of poisonous gases.
The Pail System is an economic, simple, and, on the whole, very
efficient method of removing fresh excreta. The excreta are passed
directly into stone or metal water- and gas-tight pails, which, after
filling, are hermetically covered and removed to the places for final
disposal. This system is in use in Rochedale, Manchester, Glasgow, and
other places in England.
The pails may also be filled with dried earth, ashes, etc., which are
mixed with the excreta and convert it into a substance fit for
fertilization.
The Pneumatic System is a rather complicated mechanical method
invented by Captain Lieurneur, and is used extensively in some places.
In this system the excreta are passed to certain pipes and
receptacles, and from there aspirated by means of air exhausts.
The Water-carriage System.—We now come to the modern mode of using
water to carry and flush all sewage material. This method is being
adopted throughout the civilized world. For it is claimed a reduction
of the mortality rate issues wherever it is[Pg 180] introduced. The
water-carriage system presupposes the construction and existence of
pipes from the house to and through the street to the place of final
disposition. The pipes running from the house to the streets are
called house sewers; and when in the streets, are called street
sewers.
The Separate and Combined Systems.—Whenever the water-carriage system
is used, it is either intended to carry only sewage proper, viz.,
solid and liquid excreta flushed by water, or fain water and other
waste water from the household in addition. The water-carriage system
is accordingly divided into two systems: the combined, by which all
sewage and all waste and rain water are carried through the sewers,
and the separate system, in which two groups of pipes are used—the
sewers proper to carry sewage only, and the other pipes to dispose of
rain water and other uncontaminated waste water. Each system has its
advocates, its advantages and disadvantages. The advantages claimed
for the separate system are as follows:
(1) Sewers may be of small diameter, not more than six inches.
(2) Constant, efficient flow and flushing of sewage.
(3) The sewage gained is richer in fertilizing matter.
(4) The sewers never overflow, as is frequently the case in the
combined system.
[Pg 181](5) The sewers being small, no decomposition takes place therein.
(6) Sewers of small diameter need no special means of ventilation, or
main traps on house drains, and can be ventilated through the house
pipes.
On the other hand, the disadvantages of the separate system are:
(1) The need of two systems of sewers, for sewage and for rain water,
and the expense attached thereto.
(2) The sewers used for sewage proper require some system for
periodically flushing them, which, in the combined system, is done by
the occasional rains.
(3) Small sewers cannot be as well cleaned or gotten at as larger
ones.
The separate system has been used in Memphis and in Keene, N. H., for
a number of years with complete satisfaction. Most cities, however,
use the combined system.
[Pg 182]
CHAPTER V
Sewers
Definitions.—A sewer is a conduit or pipe intended for the passage of
sewage, waste, and rain water.
A House Sewer is the branch sewer extending from a point two feet
outside of the outer wall of the building to its connection with the
street sewer, etc.
Materials.—The materials from which sewers are manufactured is
earthenware "vitrified pipes."
Iron is used only for pipes of small diameter; and as most of the
sewers are of greater diameter than six inches, they are made of other
material than iron.
Cement and brick sewers are frequently used, and, when properly
constructed, are efficient, although the inner surface of such pipes
is rough, which causes adherence of sewage matter.
The most common material of which sewers are manufactured is
earthenware, "vitrified pipes."
"Vitrified pipes are manufactured from some kind of clay, and are
salt-glazed inside. Good vitrified pipe must be circular and true in
section, of a uniform thickness, perfectly straight, and free from
cracks or other defects; they must be hard, tough,[Pg 183] not porous, and
have a highly smooth surface. The thicknesses of vitrified pipes are
as follows:
| 4 inches diameter |
1/2 inch thick |
| 6 " " |
1/16 " " |
| 8 " " |
3/4 " " |
| 12 " " |
1 " " |
The pipes are made in two- and three-foot lengths, with spigot, and
socket ends." (Gerhardt.)
Sewer pipes are laid in trenches at least three feet deep, to insure
against the action of frosts.
Construction.—The level of the trenches in which sewers are laid
should be accurate, and a hard bed must be secured, or prepared, for
the pipes to lie on. If the ground is sandy and soft, a solid bed of
concrete should be laid, and the places where the joints are should be
hollowed out, and the latter embedded in cement.
Joints.—The joints of the various lengths must be gas-tight, and are
made as follows: into the hub (the enlargement on one end of the pipe)
the spigot end of the next length is inserted, and in the space left
between the two a small piece, or gasket, of oakum is rammed in; the
remaining space is filled in with a mixture of the best Portland
cement and clean, sharp sand. The office of the oakum is to prevent
the cement from getting on the inside of the pipe. The joint is then
wiped around with additional cement.
[Pg 184]Fall.—In order that there should be a steady and certain flow of the
contents of the sewer, the size and fall of the latter must be
suitable; that is, the pipes must be laid with a steady, gradual
inclination or fall toward the exit. This fall must be even, without
sudden changes, and not too great or too small.
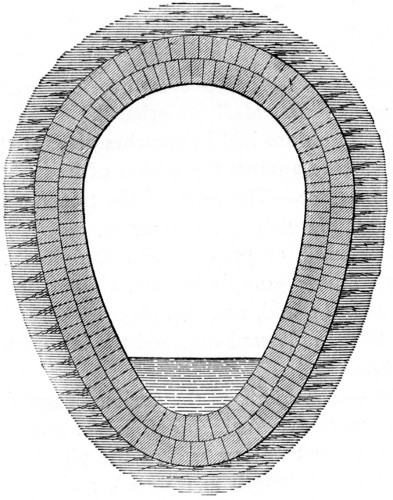 Fig. 15.
Fig. 15.
A BRICK SEWER.
The following has been determined to be about the right fall for the
sizes stated:
| [Pg 185]4-inch pipe |
1 foot in 40 feet |
| 6 " " |
1 " " 60 " |
| 9 " " |
1 " " 90 " |
| 12 " " |
1 " " 120 " |
Flow.—The velocity of the flow in sewers depends on the volume of
their contents, the size of the pipes, and the fall. The velocity
should not be less than 120 feet in a minute, or the sewer will not be
self-cleansing.
Size.—In order for the sewer to be self-cleansing, its size must be
proportional to the work to be accomplished, so that it may be fully
and thoroughly flushed and not permit stagnation and consequent
decomposition of its contents. If the sewer be too small, it will not
be adequate for its purpose, and will overflow, back up, etc.; if too
large, the velocity of the flow will be too low, and stagnation will
result. In the separate system, where there is a separate provision
for rain water, the size of the sewer ought not to exceed six inches
in diameter. In the combined system, however, when arrangements must
be made for the disposal of large volumes of storm water, the size of
the sewer must be larger, thus making it less self-cleansing.
Connections.—The connections of the branch sewers and the house
sewers with the main sewer must be carefully made, so that there shall
be no impediment to the flow of the contents, either of the branches
or of the main pipe. The connections must[Pg 186] be made gas-tight; not at
right angles or by T branches, but by bends, curves, and Y branches,
in the direction of the current of the main pipe, and not opposite
other branch pipes; and the junction of the branch pipes and the main
pipe must not be made at the crown or at the bottom of the sewer, but
just within the water line.
Tide Valves.—Where sewers discharge their contents into the sea, the
tide may exert pressure upon the contents of the sewer and cause
"backing up," blocking up the sewer, bursting open trap covers, and
overflowing into streets and houses. To prevent this, there are
constructed at the mouth of the street sewers, at the outlets to the
sea, proper valves or tide flaps, so constructed as to permit the
contents of the sewers to flow out, yet prevent sea water from backing
up by immediately closing upon the slightest pressure from outside.
House Sewers.—Where the ground is "made," or filled in, the house
sewer must be made of cast iron, with the joints properly calked with
lead. Where the soil consists of a natural bed of loam, sand, or rock,
the house sewer may be of hard, salt-glazed, and cylindrical
earthenware pipe, laid in a smooth bottom, free from projections of
rock, and with the soil well rammed to prevent any settling of the
pipe. Each section must be wetted before applying the cement, and the
space between each hub and the small end of the next section must be
completely and uniformly[Pg 187] filled with the best hydraulic cement. Care
must be taken to prevent any cement being forced into the pipe to form
an obstruction. No tempered-up cement should be used. A straight edge
must be used inside the pipe, and the different sections must be laid
in perfect line on the bottom and sides.
Connections of the house sewer (when of iron) with the house main pipe
must be made by lead-calked joints; the connection of the iron house
pipe with the earthenware house sewer must be made with cement, and
should be gas-tight.
Sewer Air and Gas.—Sewer gas is not a gas at all. What is commonly
understood by the term is the air of sewers, the ordinary atmospheric
air, but charged and contaminated with the various products of organic
decomposition taking place in sewers. Sewer air is a mixture of gases,
the principal gases being carbonic acid; marsh gas; compounds of
hydrogen and carbon; carbonate and sulphides of ammonium; ammonia;
sulphureted hydrogen; carbonic oxide, volatile fetid matter; organic
putrefactive matter, and may also contain some bacteria, saprophytic
or pathogenic.
Any and all the above constituents may be contained in sewer air in
larger or smaller doses, in minute or toxic doses.
It is evident that an habitual breathing of air in which even minute
doses of toxic substances and gases are floating will in time impair
the health of[Pg 188] human beings, and that large doses of those substances
may be directly toxic and dangerous to health. It is certainly an
error to ascribe to sewer air death-dealing properties, but it would
be a more serious mistake to undervalue the evil influence of bad
sewer air upon health.
Ventilation.—To guard against the bad effects of sewer air, it is
necessary to dilute, change, and ventilate the air in sewers. This is
accomplished by the various openings left in the sewers, the so-called
lamp and manholes which ventilate by diluting the sewer air with the
street air. In some places, chemical methods of disinfecting the
contents of sewers have been undertaken with a view to killing the
disease germs and deodorizing the sewage. In the separate system of
sewage disposal, where sewer pipes are small and usually
self-cleansing, the late Colonel Waring proposed to ventilate the
sewers through the house pipes, omitting the usual disconnection of
the house sewer from the house pipes. But in the combined system such
a procedure would be dangerous, as the sewer air would be apt to enter
the house.
Rain storms are the usual means by which a thorough flushing of the
street sewers is effected. There are, however, many devices proposed
for flushing sewers; e. g., by special flushing tanks, which either
automatically or otherwise discharge a large volume of water, thereby
flushing the contents of the street sewers.
[Pg 189]
CHAPTER VI
Plumbing
Purpose and Requisites for House Plumbing.—A system of house plumbing
presupposes the existence of a street sewer, and a water-supply
distribution within the house. While the former is not absolutely
essential, as a house may have a system of plumbing without there
being a sewer in the street, still in the water-carriage system of
disposal of sewage the street sewer is the outlet for the various
waste and excrementitious matter of the house. The house-water
distribution serves for the purpose of flushing and cleaning the
various pipes in the house plumbing.
The purposes of house plumbing are: (1) to get rid of all excreta and
waste water; (2) to prevent any foreign matter and gases in the sewer
from entering the house through the pipes; and (3) to dilute the air
in the pipes so as to make all deleterious gases therein innocuous.
To accomplish these results, house plumbing demands the following
requisites:
(1) Receptacles for collecting the waste and excreta. These
receptacles, or plumbing fixtures, must[Pg 190] be adequate for the purpose,
small, noncorrosive, self-cleansing, well flushed, accessible, and so
constructed as to easily dispose of their contents.
(2) Separate Vertical Pipes for sewage proper, for waste water, and
for rain water; upright, direct, straight, noncorrosive, water- and
gas-tight, well flushed, and ventilated.
(3) Short, direct, clean, well-flushed, gas-tight branch pipes to
connect receptacles with vertical pipes.
(4) Disconnection of the house sewer from the house pipes by the
main trap on house drain, and disconnection of house from the house
pipes by traps on all fixtures.
(5) Ventilation of the whole system by the fresh-air inlet, vent
pipes, and the extension of all vertical pipes.
Definitions.—The House Drain is the horizontal main pipe receiving
all waste water and sewage from the vertical pipes, and conducting
them outside of the foundation walls, where it joins the house sewer.
The Soil Pipe is the vertical pipe or pipes receiving sewage matter
from the water-closets in the house.
The Main Waste Pipe is the pipe receiving waste water from any
fixtures except the water-closets.
Branch Soil and Waste Pipes are the short pipes between the fixtures
in the house and the main soil and waste pipes.
[Pg 191]Traps are bends in pipes, so constructed as to hold a certain volume
of water, called the water seal; this water seal serves as a barrier
to prevent air and gases from the sewer from entering the house.
Vent Pipes are the special pipes to which the traps or fixtures are
connected by short-branch vent pipes, and serve to ventilate the air
in the pipes, and prevent siphonage.
The Rain Leader is the pipe receiving rain and storm water from the
roof of the house.
Materials Used for Plumbing Pipes.—The materials from which the
different pipes used in house plumbing are made differ according to
the use of each pipe, its position, size, etc. The following materials
are used: cement, vitrified pipe, lead; cast, wrought, and galvanized
iron; brass, steel, nickel, sheet metal, etc.
Cement and Vitrified Pipes are used for the manufacture of street
and house sewers. In some places vitrified pipe is used for house
drains, but in most cities this is strongly objected to; and in New
York City no earthenware pipes are permitted within the house. The
objection to earthenware pipes is that they are not strong enough for
the purpose, break easily, and cannot be made gas-tight.
Lead Pipe is used for all branch waste pipes and short lengths of
water pipes. The advantage of lead pipes is that they can be easily
bent and shaped, hence their use for traps and connections. The
dis[Pg 192]advantage of lead for pipes is the softness of the material, which
is easily broken into by nails, gnawed through by rats, etc.
Brass, Nickel, Steel, and other such materials are used in the
manufacture of expensive plumbing, but are not commonly employed.
Sheet Metal and Galvanized Iron are used for rain leaders,
refrigerator pipes, etc.
Wrought Iron is used in the so-called Durham system of plumbing.
Wrought iron is very strong; the sections of pipe are twenty feet
long, the connections are made by screw joints, and a system of house
plumbing made of this material is very durable, unyielding, strong,
and perfectly gas-tight. The objections to wrought iron for plumbing
pipes are that the pipes cannot be readily repaired and that it is too
expensive.
Cast Iron is the material universally used for all vertical and
horizontal pipes in the house. There are two kinds of cast-iron pipes
manufactured for plumbing uses, the "standard and the extra heavy."
The following are the relative weights of each:
| |
Standard. |
Extra Heavy. |
| 2-inch pipe, |
4 lbs. per foot |
51/2 lbs. |
| 3 " " |
6 " " " |
91/2 " |
| 4 " " |
9 " " " |
13 " |
| 5 " " |
12 " " " |
17 " |
| 6 " " |
15 " " " |
20 " |
| 7 " " |
20 " " " |
27 " |
| 8 " " |
25 " " " |
331/2 " |
[Pg 193]
[Pg 194]The light-weight pipe, though extensively used by plumbers, is
generally prohibited by most municipalities, as it is not strong
enough for the purpose, and it is difficult to make a gas-tight joint
with these pipes without breaking them.
Cast-iron pipes are made in lengths of five feet each, with an
enlargement on one end of the pipe, called the "hub" or "socket," into
which the other, or "spigot," end is fitted. All cast-iron pipe must
be straight, sound, cylindrical and smooth, free from sand holes,
cracks, and other defects, and of a uniform thickness.
The thickness of cast-iron pipes should be as follows:
| 2-inch pipe, |
5/16 inches thick |
| 3 " " |
" " " |
| 4 " " |
3/8 " " |
| 5 " " |
7/16 " " |
| 6 " " |
1/2 " " |
Cast-iron pipes are sometimes coated by dipping into hot tar, or by
some other process. Tar coating is, however, not allowed in New York,
because it conceals the sand holes and other flaws in the pipes.
Joints and Connections.—To facilitate connections of cast-iron pipes,
short and convenient forms and fittings are cast. Some of these
connections are named according to their shape, such as L, T, Y, etc.
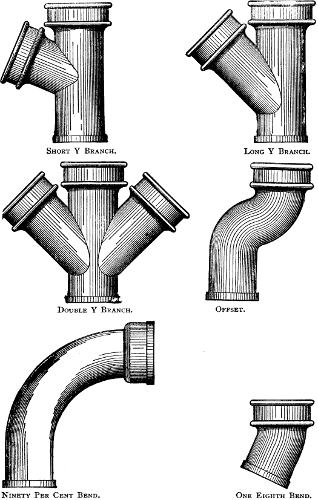 Fig. 16.
Fig. 16.
DIFFERENT FORMS AND FITTINGS.
Iron Pipe is joined to Iron Pipe by lead-calked joints. These
joints are made as follows: the spigot[Pg 195]
[Pg 196] end of one pipe is inserted
into the enlarged end, or the "hub," of the next pipe. The space
between the spigot and hub is half filled with oakum or dry hemp. The
remaining space is filled with hot molten lead, which, on cooling, is
well rammed and calked in by special tools made for the purpose. To
make a good, gas-tight, lead-calked joint, experience and skill are
necessary. The ring of lead joining the two lengths of pipe must be
from 1 to 2 inches deep, and from 1/2 to 3/4 of an inch thick; 12
ounces of lead must be used at each joint for each inch in the
diameter of the pipe. Iron pipes are sometimes connected by means of
so-called rust joints. Instead of lead, the space between the socket
and spigot is filled in with an iron cement consisting of 98 parts of
cast-iron borings, 1 part of flowers of sulphur, and 1 part of sal
ammoniac.
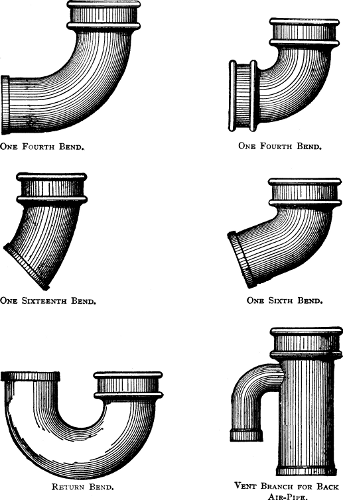 Fig. 17.
Fig. 17.
All connections between Lead Pipes and between Lead and Brass or
Copper pipes must be made by means of "wiped" solder joints. A wiped
joint is made by solder being poured on two ends of the two pipes, the
solder being worked about the joint, shaped into an oval lump, and
wiped around with a cloth, giving the joint a bulbous form.
All connections between Lead Pipes and Iron Pipes are made by
means of brass ferrules. Lead cannot be soldered to iron, so a brass
fitting or ferrule is used; it is jointed to the lead pipe by a wiped
joint, and to the iron pipe by an ordinary lead-calked joint.
[Pg 197]
[Pg 198]Putty, Cement, and Slip joints should not be tolerated on any
pipes.
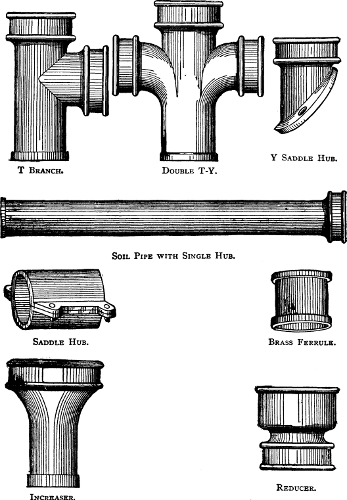 Fig. 18.
Fig. 18.
Traps.—We have seen that a trap is a bend in a pipe so constructed as
to hold a quantity of water sufficient to interpose a barrier between
the sewer and the fixture. There are many and various kinds of traps,
some depending on water alone as their "seal," others employing
mechanical means, such as balls, valves, lips, also mercury, etc., to
assist in the disconnection between the house and sewer ends of the
pipe system.
The value of a trap depends: (1) on the depth of its water seal; (2)
on the strengths and permanency of the seal; (3) on the diameter and
uniformity of the trap; (4) on its simplicity; (5) on its
accessibility; and (6) on its self-cleansing character.
The depth of a trap should be about three inches for water-closet
traps, and about two inches for sink and other traps.
Traps must not be larger in diameter than the pipe to which they are
attached.
The simpler the trap, the better it is.
Traps should be provided with cleanout screw openings, caps, etc., to
facilitate cleaning.
The shapes of traps vary, and the number of the various kinds of traps
manufactured is very great.
Traps are named according to their use: gully, grease, sediment,
intercepting, etc.; according to their shape: D, P, S, V, bell,
bottle, pot, globe, etc.;[Pg 199]
[Pg 200] and according to the name of their
inventor: Buchan, Cottam, Dodd, Antill, Renk, Hellyer, Croydon, and
others too numerous to mention.
The S trap is the best for sink waste pipes; the running trap is the
best on house drains.
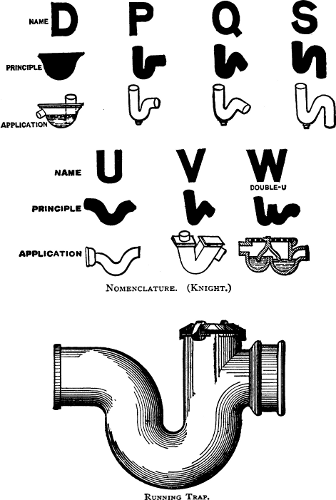 Fig. 19.
Fig. 19.
FORMS OF TRAPS.
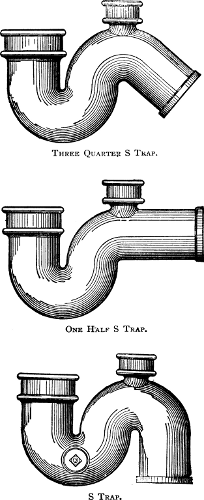 Fig. 20.
Fig. 20.
FORMS OF TRAPS.
Loss of Seal by Traps.—The seals of traps are not always secure, and
the causes of unsealing of traps are as follows:
(1) Evaporation.—If a fixture in a house is not used for a long
time, the water constituting the seal in the trap of the fixture will
evaporate; the seal will thus be lost, and ingress of sewer air will
result. To guard against evaporation, fixtures must be frequently
flushed; and during summer, or at such times as the house is
unoccupied and the fixtures not used, the traps are to be filled with
oil or glycerin, either of which will serve as an efficient seal.
(2) Momentum.—A sudden flow of water from the fixture may, by the
force of its momentum, empty all water in the trap and thus leave it
unsealed. To prevent the unsealing of traps by momentum, they must be
of a proper size, not less than the waste pipe of the fixture, the
seal must be deep, and the trap in a perfectly straight position, as a
slight inclination will favor its emptying. Care should also be taken
while emptying the fixture to do it slowly so as to preserve the seal.
(3) Capillary Attraction.—If a piece of paper, cotton, thread,
hair, etc., remain in the trap, and a part[Pg 201]
[Pg 202] of the paper, etc.,
projects into the lumen of the pipe, a part of the water will be
withdrawn by capillary attraction from the trap and may unseal it. To
guard against unsealing of traps by capillary attraction, traps should
be of a uniform diameter, without nooks and corners, and of not too
large a size, and should also be well flushed, so that nothing but
water remains in the trap.
Siphonage and Back Pressure.—The water in the trap, or the "seal,"
is suspended between two columns of air, that from the fixture to the
seal, and from the seal of the trap to the seal of the main trap on
house drain. The seal in the trap is therefore not very secure, as it
is influenced by any and all currents and agitations of air from both
sides, and especially from its distal side. Any heating of the air in
the pipes with which the trap is connected, any increase of
temperature in the air contents of the vertical pipes with which the
trap is connected, and any evolution of gases within those pipes will
naturally increase the weight and pressure of the air within them,
with the result that the increased pressure will influence the
contents of the trap, or the "seal," and may dislodge the seal
backward, if the pressure is very great, or, at any rate, may force
the foul air from the pipes through the seal of the traps and foul the
water therein, thus allowing foul odors to enter the rooms from the
traps of the fixtures. This condition, which in practice exists
oftener than it is ordinarily thought,[Pg 203] is called "back pressure." By
"back pressure" is therefore understood the forcing back, or, at
least, the fouling, of the water in traps, due to the increased
pressure of the air within the pipes back of the traps; the increase
in air pressure being due to heating of pipes by the hot water
occasionally circulating within them, or by the evolution of gases due
to the decomposition of organic matter within the pipes.
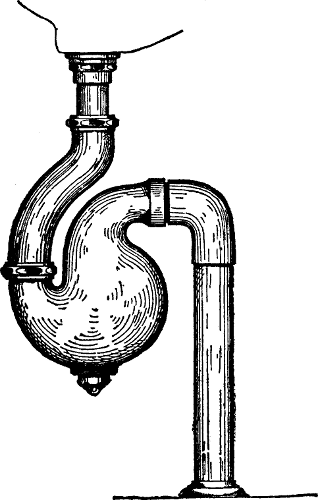 Fig. 21.
Fig. 21.
NON-SYPHONING TRAP.
Copyright by the J. L. Mott Iron Works.
A condition somewhat similar, but acting in a reverse way, is
presented in what is commonly termed "siphonage." Just as well as the
seal in traps may[Pg 204] be forced back by the increased pressure of the air
within the pipes, the same seal may be forced out, pulled out,
aspirated, or siphoned out by a sudden withdrawal of a large quantity
of air from the pipes with which the trap is connected. Such a sudden
withdrawal of large quantities of air is occasioned every time there
is a rush of large column of water through the pipes, e. g., when a
water-closet or similar fixture is suddenly discharged; the water
rushes through the pipes with a great velocity and creates a strong
down current of air, with the result that where the down-rushing
column passes by a trap, the air in the trap and, later, its seal are
aspirated or siphoned out, thus leaving the trap without a seal. By
"siphonage" is therefore meant the emptying of the seal in a trap by
the aspiration of the water in the trap due to the downward rush of
water and air in the pipes with which the trap is connected.
To guard against the loss of seal through siphonage "nonsiphoning"
traps have been invented, that is, the traps are so constructed that
the seal therein is very large, and the shape of the traps made so
that siphonage is difficult. These traps are, however, open to the
objection that in the first place they do not prevent the fouling of
the seals by back pressure, and in the second place they are not
easily cleansable and may retain dirt in their large pockets. The
universal method of preventing both siphonage and back pressure is by
the system of vent pipes, or[Pg 205] what plumbers call "back-air" pipes.
Every trap is connected by branches leading from the crown or near the
crown of the trap to a main vertical pipe which runs through the house
the same as the waste and soil pipes, and which contains nothing but
air, which air serves as a medium to be pressed upon by the
"back-pressure" air, or to be drawn upon by the siphoning, and thus
preventing any agitation and influence upon the seal in the traps; for
it is self evident that as long as there is plenty of air at the
distal part of the seal, the seal itself will remain uninfluenced by
any agitation or condition of the air within the pipes with which the
trap is connected.
The vent-pipe system is also an additional means of ventilating the
plumbing system of the house, already partly ventilated by the
extension of the vertical pipes above the roof and by the fresh-air
inlet. The principal objection urged against the installation of the
vent-pipe system is the added expense, which is considerable; and
plumbers have sought therefore to substitute for the vent pipes
various mechanical traps, also nonsiphoning traps. The vent pipes are,
however, worth the additional expense, as they are certainly the best
means to prevent siphonage and back pressure, and are free from the
objections against the cumbersome mechanical traps and the filthy
nonsiphoning traps.
[Pg 206]
CHAPTER VII
Plumbing Pipes
The House Drain.—All waste and soil matter in the house is carried
from the receptacles into the waste and soil pipes, and from these
into the house drain, the main pipe of the house, which carries all
waste and soil into the street sewer. The house drain extends from the
junction of the soil and waste pipes of the house through the house to
outside of the foundations two to five feet, whence it is called
"house sewer." The house drain is a very important part of the
house-plumbing system, and great care must be taken to make its
construction perfect.
Material.—The material of which house drains are manufactured is
extra heavy cast iron. Lighter pipes should never be used, and the use
of vitrified pipes for this purpose should not be allowed.
Size.—The size of the house drain must be proportional to the work
to be performed. Too large a pipe will not be self-cleansing, and the
bottom of it will fill with sediment and slime. Were it not for the
need of carrying off large volumes of storm water, the house drain
could be a great deal smaller than it[Pg 207]
[Pg 208] usually is. A three-inch pipe
is sufficient for a small house, though a four-inch pipe is made
obligatory in most cities. In New York City no house drains are
allowed of smaller diameter than six inches.
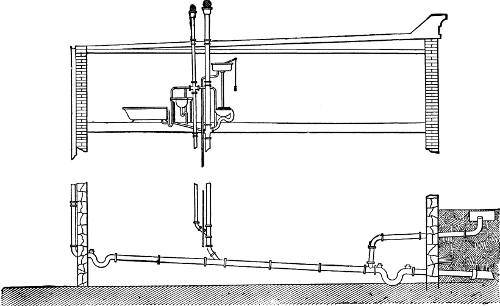 Fig. 22.
Fig. 22.
SYSTEM OF HOUSE DRAINAGE, SHOWING THE PLUMBING OF A HOUSE. (H.
Bramley.)
Fall.—The fall or inclination of the house drain depends on its
size. Every house drain must be laid so that it should have a certain
inclination toward the house sewer, so as to increase the velocity of
flow in it and make it self-flushing and self-cleansing. The rate of
fall should be as follows:
| For 4-inch pipe |
1 in 40 feet |
| " 5 " " |
1 " 50 " |
| " 6 " " |
1 " 60 " |
Position.—The house drain lies in a horizontal position in the
cellar, and should, if possible, be exposed to view. It should be hung
on the cellar wall or ceiling, unless this is impracticable, as when
fixtures in the cellar discharge into it; in this case, it must be
laid in a trench cut in a uniform grade, walled upon the sides with
bricks laid in cement, and provided with movable covers and with a
hydraulic-cement base four inches thick, on which the pipe is to rest.
The house drain must be laid in straight lines, if possible; all
changes in direction must be made with curved pipes, the curves to be
of a large radius.
Connections.—The house drain must properly connect with the house
sewer at a point about two feet outside of the outer front vault or
area wall of the[Pg 209] building. An arched or other proper opening in the
wall must be provided for the drain to prevent damage by settling.
All joints of the pipe must be gas-tight, lead-calked joints, as
stated before. The junction of the vertical soil, waste, and
rain-leader pipes must not be made by right-angle joints, but by a
curved elbow fitting of a large radius, or by "Y" branches and 45°
bends.
When the house drain does not rest on the floor, but is hung on the
wall or ceiling of the cellar, the connection of the vertical soil and
waste pipes must have suitable supports, the best support being a
brick pier laid nine inches in cement and securely fastened to the
wall.
Near all bends, traps, and connections of other pipes with the house
drain suitable hand-holes should be provided, these hand-holes to be
tightly covered by brass screw ferrules, screwed in, and fitted with
red lead.
"No steam exhaust, boiler blow-off, or drip pipe shall be connected
with the house drain or sewer. Such pipes must first discharge into a
proper condensing tank, and from this a proper outlet to the house
sewer outside of the building must be provided."
Main Traps.—The disconnection of the house pipes from the street
sewer is accomplished by a trap on the house drain near the front
wall, inside the house, or just outside the foundation wall but[Pg 210]
usually inside of the house. The best trap for this purpose is the
siphon or running trap. This trap must be constructed with a cleaning
hand-hole on the inside or house side of the trap, or on both sides,
and the hand-holes are to be covered gas-tight by brass screw
ferrules.
Extension of Vertical Pipes.—By the main trap the house-plumbing
system is disconnected from the sewer, and by the traps on each
fixture from the air in the rooms; still, as the soil, waste, and
drain pipes usually contain offensive solids and liquids which
contaminate the air in the pipes, it is a good method to ventilate
these pipes. This ventilation of the soil, waste, and house drain
pipes prevents the bad effects on health from the odors, etc., given
off by the slime and excreta adhering in the pipes, and it is
accomplished by two means: (1) by extension of the vertical pipes to
the fresh air above the roof, and (2) by the fresh-air inlet on the
house drain.
By these means a current of air is established through the vertical
and horizontal pipes.
Every vertical pipe must be extended above the roof at least two feet
above the highest coping of the roof or chimney. The extension must be
far from the air shafts, windows, ventilators, and mouths of chimneys,
so as to prevent air from the pipes being drawn into them. The
extension must be not less than the full size of each pipe, so as to
avoid friction from the circulation of air. The use of covers, cowls,[Pg 211]
return bends, etc., is reprehensible, as they interfere with the free
circulation of air. A wire basket may be inserted to prevent foreign
substances from falling into pipes.
Fresh-air Inlet.—The fresh-air inlet is a pipe of about four inches
in diameter; it enters the house drain on the house side of the main
trap, and extends to the external air at or near the curb, or at any
convenient place, at least fifteen feet from the nearest window. The
fresh-air inlet pipe usually terminates in a receptacle covered by an
iron grating, and should be far from the cold-air box of any hot-air
furnace. When clean, properly cared for, and extended above the
ground, the fresh-air inlet, in conjunction with the open extended
vertical pipe, is an efficient means of ventilating the air in the
house pipes; unfortunately most fresh-air inlets are constantly
obstructed, and do not serve the purpose for which they are made.
The Soil and Waste Pipes.—The soil pipe receives liquid and solid
sewage from the water-closets and urinals; the waste pipe receives all
waste water from sinks, washbasins, bath tubs, etc.
The material of which the vertical soil and waste pipes are made is
cast iron.
The size of main waste pipes is from three to four inches; of main
soil pipes, from four to five inches. In tenement houses with five
water-closets or more, not less than five inches.
[Pg 212]The joints of the waste and soil pipe should be lead calked. The
connections of the lead branch pipes or traps with the vertical lines
must be by Y joints, and by means of brass ferrules, as explained
above.
The location of the vertical pipes must never be within the wall,
built in, nor outside the house, but preferably in a special
three-foot square shaft adjacent to the fixtures, extending from the
cellar to the roof, where the air shaft should be covered by a
louvered skylight; that is, with a skylight with slats outwardly
inclined, so as to favor ventilation.
The vertical pipes must be accessible, exposed to view in all their
lengths, and, when covered with boards, so fitted that the boards may
be readily removed.
Vertical pipes must be extended above the roof in full diameter, as
previously stated. When less than four inches in diameter, they must
be enlarged to four inches at a point not less than one foot below the
roof surface by an "increaser," of not less than nine inches long.
All soil and waste pipes must, whenever necessary, be securely
fastened with wrought-iron hooks or straps.
Vertical soil and waste pipes must not be trapped at their base, as
the trap would not serve any purpose, and would prevent a perfect flow
of the contents.
Branch Soil and Waste Pipes.—The fixtures must be near the vertical
soil and waste pipes in order that the branch waste and soil pipes
should be as short as[Pg 213] possible. The trap of the branch soil and waste
pipes must not be far from the fixture, not more than two feet from
it, otherwise the accumulated foul air and slime in the waste and soil
branch will emit bad odors.
The minimum sizes for branch pipes should be as follows:
| Kitchen sinks |
2 inches |
| Bath tubs |
11/2 to 2 " |
| Laundry tubs |
11/2 to 2 " |
| Water-closets |
not less than 4 " |
Branch soil and waste pipes must have a fall of at least one-quarter
inch to one foot.
The branch waste and soil pipes and traps must be exposed, accessible,
and provided with screw caps, etc., for inspection and cleaning
purposes.
Each fixture should be separately trapped as close to the fixture as
possible, as two traps on the same line of branch waste or soil pipes
will cause the air between the traps to be closed in, forming a
so-called "cushion," that will prevent the ready flow of contents.
"All traps must be well supported and rest true with respect to their
water level."
Vent Pipes and Their Branches.—The purpose of vent pipes, we have
seen, is to prevent siphoning of traps and to ventilate the air in the
traps and pipes. The material of which vent pipes are made is cast
iron.
The size of vent pipes depends on the number of[Pg 214] traps with which they
are connected; it is usually two or three inches. The connection of
the branch vent to the trap must be at the crown of the trap, and the
connection of the branch vent to the main vent pipe must be above the
trap, so as to prevent friction of air. The vent pipes are not
perfectly vertical, but with a continuous slope, so as to prevent
condensation of air or vapor therein.
The vent pipes should be extended above the roof, several feet above
the coping, etc.; and the extension above the roof should not be of
less than four inches diameter, so as to avoid obstruction by frost.
No return bends or cowls should be tolerated on top of the vent pipes.
Sometimes the vent, instead of running above the roof, is connected
with the soil pipe several feet above all fixtures.
 Fig. 23.
Fig. 23.
LEADER PIPE.
Rain Leaders.—The rain leader serves to collect the rain water from
the roof and eaves gutter. It usually discharges its contents into the
house drain, although some leaders are led to the street gutter, while
others are connected with school sinks in the yard. The latter
practice is objectionable, as it may lead the foul air from the school
sink into the rooms, the windows[Pg 215] of which are near the rain leader;
besides, the stirring up of the contents of the school sink produces
bad odors. When the rain leader is placed within the house, it must be
made of cast iron with lead-calked joints; when outside, as is the
rule, it may be of sheet metal or galvanized-iron pipe with soldered
joints. When the rain leader is run near windows, the rules and
practice are that it should be trapped at its base, the trap to be a
deep one to prevent evaporation, and it should be placed several feet
below the ground, so as to prevent freezing.
[Pg 216]
CHAPTER VIII
Plumbing Fixtures
The receptacles or fixtures within the house for receiving the waste
and excrementitious matter and carrying it off through the pipes to
the sewer are very important parts of house plumbing. Great care must
be bestowed upon the construction, material, fitting, etc., of the
plumbing fixtures, that they be a source of comfort in the house
instead of becoming a curse to the occupants.
Sinks.—The waste water from the kitchen is disposed of by means of
sinks. Sinks are usually made of cast iron, painted, enameled, or
galvanized. They are also made of wrought iron, as well as of
earthenware and porcelain. Sinks must be set level, and provided with
a strainer at the outlet to prevent large particles of kitchen refuse
from being swept into the pipe and obstructing it. If possible the
back and sides of a sink should be cast from one piece; the back and
sides, when of wood, should be covered by nonabsorbent material, to
prevent the wood from becoming saturated with waste water.[18] No
woodwork should[Pg 217] inclose sinks; they should be supported on iron legs
and be open beneath and around. The trap of a sink is usually two
inches in diameter, and should be near the sink; it should have a
screw cap for cleaning and inspection, and the branch vent pipe should
be at the crown of the trap.
Washbasins.—Washbasins are placed in bathrooms, and, when properly
constructed and fitted, are a source of comfort. They should not be
located in bedrooms, and should be open, without any woodwork around
them. The washbowls are made of porcelain or marble, with a socket at
the outlet, into which a plug is fitted.
Wash Tubs.—For laundry purposes wooden, iron-enameled, stone, and
porcelain tubs are fitted in the kitchen or laundry room. Porcelain is
the best material, although very expensive. The soapstone tub is the
next best; it is clean, nonabsorbent, and not too expensive. Wood
should never be used, as it soon becomes saturated, is foul, leaks,
and is offensive. In old houses, wherever there are wooden tubs, they
should be covered with zinc or some nonabsorbent material. The wash
tubs are placed in pairs, sometimes three in a row, and they are
generally connected with one lead waste pipe one and a half to two
inches in diameter, with one trap for all the tubs.
Bath Tubs.—Bath tubs are made of enameled iron or porcelain, and
should not be covered or inclosed by any woodwork. The branch waste
pipe should be[Pg 218] trapped and connected with the main waste or soil
pipe. The floor about the tub in the bathroom should be of
nonabsorbent material.[19]
Refrigerators.—The waste pipes of refrigerators should not connect
with any of the house pipes, but should be emptied into a basin or
pail; or, if the refrigerator is large, its waste pipe should be
conducted to the cellar, where it should discharge into a properly
trapped, sewer-connected and water-supplied open sink.
Boilers.—The so-called sediment pipe from the hot-water boiler in the
kitchen should be connected with the sink trap at the inlet side of
the trap.
Urinals.—As a rule, no urinals should be tolerated within a house;
they are permissible only in factories and office buildings. The
material is enameled iron or porcelain. They must be provided with a
proper water supply to flush them.
Overflows.—To guard against overflow of washbasins, bath tubs, etc.,
overflow pipes from the upper portion of the fixtures are commonly
provided. These pipes are connected with the inlet side of the trap of
the same fixture. They are, however, liable to become a nuisance by
being obstructed with dirt and not being constantly flushed; whenever
possible they should be dispensed with.
Safes and Wastes.—A common usage with plumb[Pg 219]ers in the past has been
to provide sinks, washbasins, bath tubs, and water-closets, not only
with overflow pipes, but also with so-called safes, which consist of
sheets of lead turned up several inches at the edge so as to catch all
drippings and overflow from fixtures; from these safes a drip pipe or
waste is conducted to the cellar, where it empties into a sink. Of
course, when such safe wastes are connected with the soil or waste
pipes, they become a source of danger, even if they are trapped, as
they are not properly cared for or flushed; and their traps are
usually not sealed. Even when discharging into a sink in the cellar,
safes and safe waste are very unsightly, dirty, liable to accumulate
filth, and are offensive. With open plumbing, and with the floors
under the fixtures of nonabsorbent material, they are useless.
Water-closets.—The most important plumbing fixtures within the house
are the water-closets. Upon the proper construction and location of
the water-closets greatly depends the health of the inhabitants of the
house. Water-closets should be placed in separate, well-lighted,
perfectly ventilated, damp-proof, and clean compartments, and no
water-closet should be used by more than one family in a tenement
house. The type and construction of the water-closets should be
carefully attended to, as the many existing, old, and obsolete types
of water-closets are still being installed in houses, or are left
there to foul the air of rooms and apartments. There are many
water-closets[Pg 220] on the market, some of which will be described; the
best are those made of one piece, of porcelain or enameled
earthenware, and so constructed as always to be and remain clean.
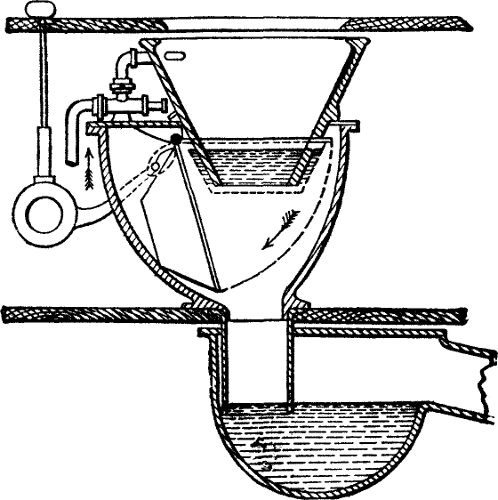 Fig. 24.
Fig. 24.
PAN WATER-CLOSET. (Gerhard.)
The Pan Closet.—The water-closet most commonly used in former times
was a representative of the group of water-closets with mechanical
contrivances. This is the pan closet, now universally condemned and
prohibited from further use. The pan closet consists of four principal
parts: (1) basin of china, small and round; (2) a copper six-inch pan
under the basin; (3) a large iron container, into which the basin with
the pan under it is placed; and (4) a D trap, to which the container
is joined. The pan is attached with a lever to a handle, which, when
pulled, moves the pan; this describes a half circle and drops the
contents into the[Pg 221] container and trap. The objections to pan closets
are the following:
(1) There being a number of parts and mechanical contrivances, they
are liable to get out of order.
(2) The bowl is set into the container and cannot be inspected, and is
usually very dirty beneath.
(3) The pan is often missing, gets out of order, and is liable to be
soiled by adhering excreta.
(4) The container is large, excreta adhere to its upper parts, and the
iron becomes corroded and coated with filth.
(5) With every pull of the handle and pan, foul air enters rooms.
(6) The junctions between the bowl and container, and the container
and trap, are usually not gas-tight.
(7) The pan breaks the force of the water flush, and the trap is
usually not completely emptied.
Valve and Plunger Closets are an improvement upon the pan closets,
but are not free from several objections enumerated above. As a rule,
all water-closets with mechanical parts are objectionable.
Hopper Closets are made of iron or earthenware. Iron hopper closets
easily corrode; they are usually enameled on the inside. Earthenware
hoppers are preferable to iron ones. Hopper closets are either long or
short; when long, they expose a very large surface to be fouled,
require a trap below the floor, and are, as a rule, very difficult to
clean or to keep clean. Short hopper closets are preferable, as they
are easily kept[Pg 222] clean and are well flushed. When provided with
flushing rim, and with a good water-supply cistern and large supply
pipe, the short hopper closet is a good form of water-closet.
The washout and washdown water-closets are an improvement upon the
hopper closets. They are manufactured from earthenware or porcelain,
and are so shaped that they contain a water seal, obviating the
necessity of a separate trap under the closet.
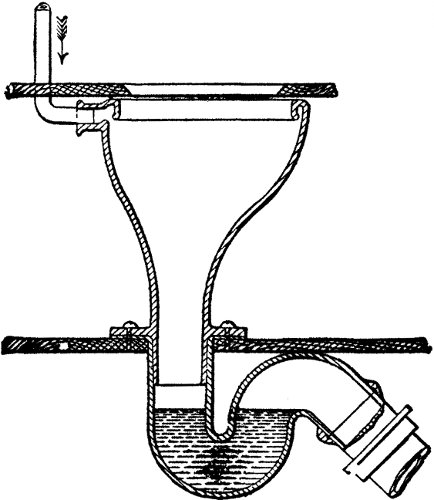 Fig. 25.
Fig. 25.
LONG HOPPER WATER-CLOSET. (Gerhard.)
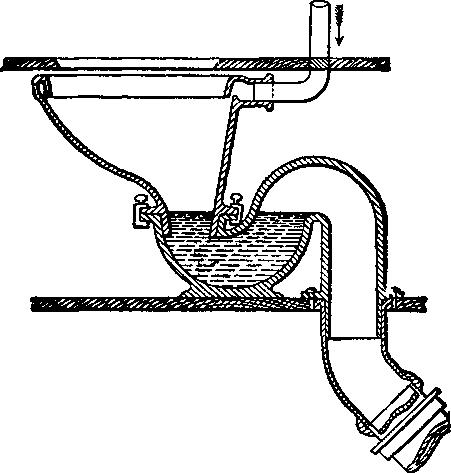 Fig. 26.
Fig. 26.
SHORT HOPPER WATER-CLOSET. (Gerhard.)
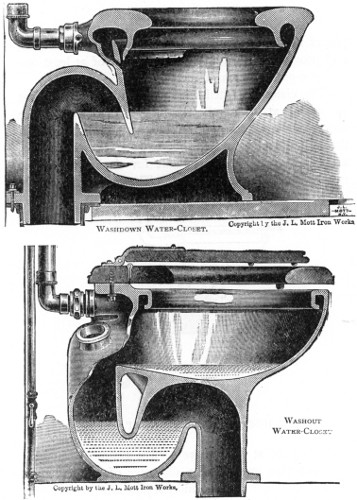 Fig. 27.
Fig. 27.
STYLES OF WATER-CLOSETS.
Flush Tanks.—Water-closets must not be flushed directly from the
water-supply pipes, as there is a possibility of contaminating the
water supply. Water-closets should be flushed from flush tanks, either
of iron or of wood, metal lined; these cisterns should be[Pg 223] placed not
less than four feet above the water-closet, and provided with a
straight flush pipe of at least one and one-quarter inch diameter.
The cistern is fitted with plug and handle, so that by pulling at the
handle the plug is lifted out of the socket of the cistern and the
contents permitted to rush through the pipe and flush the
water-closet. A separate ball arrangement is made for closing the
water supply when the cistern is full. The cistern must have a
capacity of at least three to five gallons of water; the flush pipe
must have a diameter of not less than one and one-quarter inch, and
the pipe must be straight, without bends, and the arrangement within
the closets such as to flush all parts of the bowl at the same time.
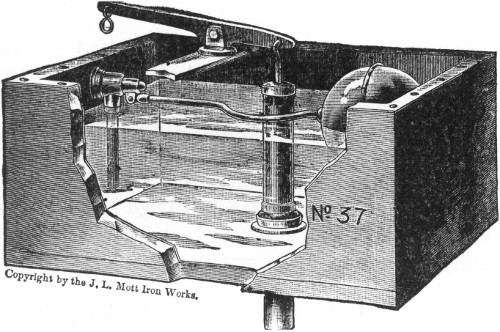 Fig. 28.
Fig. 28.
FLUSHING CISTERN.
[Pg 224]
[Pg 225]Yard Closets.—In many old houses the water-closet accommodations are
placed in the yard. There are two forms of these yard closets commonly
used—the school sink and the yard hopper.
The school sink is an iron trough from five to twelve or more feet
long, and one to two feet wide and one foot deep, set in a trench
several feet below the surface with an inclination toward the exit; on
one end of the trough there is a socket fitted with a plug, and on the
other a flushing apparatus consisting simply of a water service-pipe.
Above the iron trough brick walls are built up, inclosing it; over it
are placed wooden seats, and surrounding the whole is a wooden[Pg 226] shed
with compartments for every seat. The excreta are allowed to fall into
the trough, which is partly filled with water, and once a day, or as
often as the caretaker chooses, the plug is pulled up and the excreta
allowed to flow into the sewer with which the school sink is
connected. These school sinks are, as a rule, a nuisance, and are
dangerous to health. The objections to them are the following:
(1) The excreta lies exposed in the iron trough, and may decompose
even in one day; and it is always offensive.
(2) The iron trough is easily corroded.
(3) The iron trough, being large, presents a large surface for
adherence of excreta.
(4) The brickwork above the trough is not flushed when the school sink
is emptied, and excreta, which usually adheres to it, decomposes,
creating offensive odors.
(5) The junction of the iron trough with the brickwork, and the
brickwork itself, is usually defective, or becomes defective, and
allows foul water and sewage to pass into the yard, or into the wall
adjacent to the school sink. By the Tenement House Law of New York,
the use of school sinks is prohibited even in old buildings.
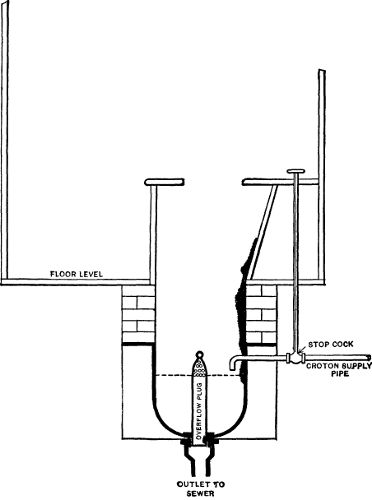 Fig. 29.
Fig. 29.
SCHOOL SINK AFTER SEVERAL MONTHS' USE.
(J. Sullivan.)
Yard Hopper Closets.—Where the water-closet accommodations cannot,
for some reason, be put within the house, yard hopper closets are
commonly employed. These closets are simply long, iron-enameled
hoppers,[Pg 227] trapped, and connected with a drain pipe discharging into
the house drain. These closets are flushed from cisterns, but, in such
case, the cisterns must be protected from freezing; this is
accomplished in some[Pg 228] houses by putting the yard hopper near the house
and placing the cistern within the house; however, this can hardly be
done where several hoppers must be employed. In most cases, yard
hoppers are flushed by[Pg 229]
[Pg 230] automatic rod valves, so constructed as to
flush the bowl of the hopper whenever the seat is pressed upon. These
valves, as a rule, frequently get out of order and leak, and care must
be taken to construct the vault under the hopper so that it be
perfectly water-tight. An improved form of yard hopper has been
suggested by Inspector J. Sullivan, of the New York Health Department,
and used in a number of places with complete satisfaction. The
improvement consists in the doors and walls of the privy apartment
being of double thickness, lined with builders' lining on the inside,
and the water service-pipes and cistern being protected by felt or
mineral wool packing.
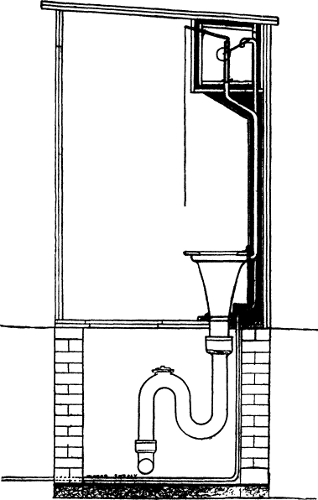 Fig. 30.
Fig. 30.
J. SULLIVAN'S IMPROVED YARD HOPPER CLOSET.
 Fig. 31.
Fig. 31.
A MODERN WATER-CLOSET.
(J. L. Mott Iron Works.)
Yard and Area Drains.—The draining of the surface of the yard or
other areas is done by tile or iron pipes connecting with the sewer or
house drain in the cellar. The "bell" or the "lip" traps are to be
condemned and should not be used for yard drains. The gully and trap
should be made of one piece; the trap should be of the siphon type and
should be deep enough in the ground to prevent the freezing of seal in
winter.
[Pg 231]
CHAPTER IX
Defects in Plumbing
The materials used in house plumbing are many and various, the parts
are very numerous, the joints and connections are frequent, the
position and location of pipes, etc., are often inaccessible and
hidden, and the whole system quite complicated. Moreover, no part of
the house construction is subjected to so many strains and uses, as
well as abuses, as the plumbing of the house. Hence, in no part of
house construction can there be as much bad work and "scamping" done
as in the plumbing; and no part of the house is liable to have so many
defects in construction, maintenance, and condition as the plumbing.
At the same time, the plumbing of a house is of very great importance
and influence on the health of the tenants, for defective materials,
bad workmanship, and improper condition of the plumbing of a house may
endanger the lives of its inhabitants by causing various diseases.
Defects in Plumbing.—The defects usually found in plumbing are so
many that they cannot all be enumerated here. Among the principal and
most common defects, however, are the following:
[Pg 232]Materials.—Light-weight iron pipes; these crack easily and cannot
stand the strain of calking. Sand-holes made during casting; these
cannot always be detected, especially when the pipes are tar-coated.
Thin lead pipe; not heavy enough to withstand the bending and drawing
it is subjected to.
Location and Position.—Pipes may be located within the walls and
built in, in which case they are inaccessible, and may be defective
without anyone being able to discover the defects. Pipes may be laid
with a wrong or an insufficient fall, thus leaving them unflushed, or
retarding the proper velocity of the flow in the pipes. Pipes may be
put underground and have no support underneath, when some parts or
lengths may sink, get out of joint, and the sewage run into the ground
instead of through the pipes. The pipes may be so located as to
require sharp bends and curves, which will retard the flow in them.
Joints.—Joints in pipes may be defective, leaking, and not
gas-tight because of imperfect calking, insufficient lead having been
used; or, no oakum having been used and the lead running into the
lumen of the pipe; or, not sufficient care and time being taken for
the work. Joints may be defective because of iron ferrules being used
instead of brass ferrules; through improperly wiped joints; through
bad workmanship, bad material, or ignorance of the plumber. Plumbers
often use T branches instead of Y branches; sharp bends instead of
bends of forty-five degrees or more;[Pg 233] slip joints instead of
lead-calked ones; also, they often connect a pipe of larger diameter
with a pipe of small diameter, etc.
Traps.—The traps may be bad in principle and in construction; they
may be badly situated or connected, or they may be easily unsealed,
frequently obstructed, inaccessible, foul, etc.
Ventilation.—The house drain may have no fresh-air inlet, or the
fresh-air inlet may be obstructed; the vent pipes may be absent, or
obstructed; the vertical pipes may not be extended.
Condition.—Pipes may have holes, may be badly repaired, bent, out
of shape, or have holes patched up with cement or putty; pipes may be
corroded, gnawed by rats, or they may be obstructed, etc.
The above are only a few of the many defects that may be found in the
plumbing of a house. It is, therefore, of paramount importance to have
the house plumbing regularly, frequently, and thoroughly examined and
inspected, as well as put to the various tests, so as to discover the
defects and remedy them.
Plumbing Tests.—The following are a few minor points for testing
plumbing:
(1) To test a trap with a view to finding out whether its seal is lost
or not, knock on the trap with a piece of metal; if the trap is empty,
a hollow sound will be given out; if full, the sound will be dull.
This is not reliable in case the trap is full or half-full with slime,
etc. Another test for the same purpose is as[Pg 234] follows: hold a light
near the outlet of the fixture; if the light is drawn in, it is a sign
that the trap is empty.
(2) Defects in leaded joints can be detected if white lead has been
used, as it will be discolored in case sewer gas escape from the
joints.
(3) The connection of a waste pipe of a bath tub with the trap of the
water-closet can sometimes be discovered by suddenly emptying the bath
tub and watching the contents of the water-closet trap; the latter
will be agitated if the waste pipe is discharged into the trap or on
the inlet side of trap of the water-closet.
(4) The presence of sewer gas in a room can be detected by the
following chemical method: saturate a piece of unglazed paper with a
solution of acetate of lead in rain or boiled water, in the proportion
of one to eight; allow the paper to dry, and hang up in the room where
the escape of sewer gas is suspected; if sewer gas is present, the
paper will be completely blackened.
The main tests for plumbing are: (1) the Hydraulic or water-pressure
test; (2) the Smoke, or sight test, and (3) the Scent, or
peppermint, etc., test.
The Water-pressure Test is used to test the vertical and horizontal
pipes in new plumbing before the fixtures have been connected. It is
applied as follows: the end of the house drain is plugged up with a
proper air-tight plug, of which there are a number on the market. The
pipes are then filled with water to a[Pg 235] certain level, which is
carefully noted. The water is allowed to stand in the pipes for half
an hour, at the expiration of which time, if the joints show no sign
of leakage, and are not sweating, and if the level of the water in the
pipes has not fallen, the pipes are water-tight. This is a very
reliable test, and is made obligatory for testing all new plumbing
work.
The Smoke Test is also a very good test. It is applied as follows:
by means of bellows, or some exploding, smoke-producing rocket, smoke
is forced into the system of pipes, the ends plugged up, and the
escape of the smoke watched for, as wherever there are defects in the
pipes the smoke will appear. A number of special appliances for this
test are manufactured, all of them more or less ingenious.
The Scent Test is made by putting into the pipes a certain quantity
of some pungent chemical, like peppermint oil, etc., the odor of which
will escape from the defects in the pipes, if there are any. Oil of
peppermint is commonly used in this country for the test. The
following is the way this test is applied: all the openings of the
pipes on the roof, except one, are closed up tightly with paper, rags,
etc. Into the one open pipe is poured from two to four ounces of
peppermint oil, followed by a pail of hot water, and then the pipe
into which the oil has been put is also plugged up. This is done,
preferably, by an assistant. The inspector then proceeds to slowly
follow the course of the various pipes, and will detect the smell of
the oil wherever[Pg 236] it may escape from any defects in the pipes. If the
test is thoroughly and carefully done, if care is taken that no
fixture in the house is used and the traps of same not disturbed
during the test, if the openings of the pipes on the roofs are plugged
up tightly, if the main house trap is not unsealed (otherwise the oil
will escape into the sewer), and if the handling of the oil has been
done by an assistant, so that none adheres to the inspector—if all
these conditions are carried out, the peppermint test is a most
valuable test for the detection of any and all defects in plumbing.
Another precaution to be taken is with regard to the rain leader. If
the rain leader is not trapped, or if its trap is empty, the
peppermint oil may escape from the pipes into the rain leader. Care
must be taken, therefore, that the trap at the base of the rain leader
be sealed; or, if no trap is existing, to close up the connection of
the rain leader with the house drain; or, if this be impossible, to
plug up the opening of the leader near the roof.
Instead of putting the oil into the opening of a pipe on the roof, it
may be put through a fixture on the top floor of the house, although
this is not so satisfactory.
Various appliances have been manufactured to make this test more easy
and accurate. Of the English appliances, the Banner patent drain
grenade, and Kemp's drain tester are worthy of mention. The former
consists "of a thin glass vial charged with pun[Pg 237]gent and volatile
chemicals. One of the grenades, when dropped down any suitable pipe,
such as the soil pipe, breaks, or the grenade may be inserted through
a trap into the drain, where it is exploded." (Taylor.) Kemp's drain
tester consists of a glass tube containing a chemical with a strong
odor; the tube is fitted with a glass cover, held in place by a string
and a paper band. When the tester is thrown into the pipes and hot
water poured after it, the paper band breaks, the spring opens the
cover, and the contents of the tube fall into the drain.
Recently Dr. W. G. Hudson, an inspector in the Department of Health of
New York, has invented a very ingenious "peppermint cartridge" for
testing plumbing. The invention is, however, not yet manufactured, and
is not on the market.
[Pg 238]
CHAPTER X
Infection and Disinfection
Disinfection is the destruction of the infective power of infectious
material; or, in other words, disinfection is the destruction of the
agents of infection.
An infectious material is one contaminated with germs of infection.
The germs of infection are organic microörganisms, vegetable and
animal—protozoa and bacteria.
The germs of infection once being lodged within the body cause certain
reactions producing specific pathological changes and a variety of
groups of symptoms which we know by the specific names of infectious
diseases, e. g., typhoid, typhus, etc.
Among the infectious diseases known to be due to specific germs are
the following: typhoid, typhus, relapsing fevers, cholera, diphtheria,
croup, tuberculosis, pneumonia, malaria, yellow fever, erysipelas,
septicæmia, anthrax, tetanus, gonorrhea, etc.; and among the
infectious diseases the germs of which have not as yet been discovered
are the following: scarlet fever, measles, smallpox, syphilis,
varicella, etc.
The part of the body and the organs in which the[Pg 239] germs first find
their entrance, or which they specifically attack, vary with each
disease; thus, the mucous membranes, skin, internal organs,
secretions, and excretions are, severally, either portals of infection
or the places where the infection shows itself the most.
The agents carrying the germs of infection from one person to the
other may be the infected persons themselves, or anything which has
come in contact with their bodies and its secretions and excretions;
thus, the air, room, furniture, vessels, clothing, food and drink,
also insects and vermin, may all be carriers of infection.
Sterilization is the absolute destruction of all organic life,
whether infectious or not; it is therefore more than disinfection,
which destroys the germs of infection alone.
A Disinfectant is an agent which destroys germs of infection.
A Germicide is the same; an agent destroying germs.
An Insecticide is an agent capable of destroying insects; it is not
necessarily a disinfectant, nor is a disinfectant necessarily an
insecticide.
An Antiseptic is a substance which inhibits and stops the growth of
the bacteria of putrefaction and decomposition. A disinfectant is
therefore an antiseptic, but an antiseptic may not be a disinfectant.
A Deodorant is a substance which neutralizes or destroys the
unpleasant odors arising from matter un[Pg 240]dergoing putrefaction. A
deodorant is not necessarily a disinfectant, nor is every disinfectant
a deodorant.
The ideal disinfectant is one which, while capable of destroying the
germs of disease, does not injure the bodies and material upon which
the germs may be found; it must also be penetrating, harmless in
handling, inexpensive, and reliable. The ideal disinfectant has not as
yet been discovered.
For successful scientific disinfection it is necessary to know: (1)
the nature of the specific germs of the disease; (2) the methods and
agents of its spread and infection; (3) the places where the germs are
most likely to be found; (4) the action of each disinfectant upon the
germs; and (5) the best methods of applying the disinfectant to the
materials infected with germs of disease.
Disinfection is not a routine, uniform, unscientific process; a
disinfector must be conversant with the basic principles of
disinfection, must make a thorough study of the scientific part of the
subject, and moreover must be thoroughly imbued with the importance of
his work, upon which the checking of the further spread of disease
depends.
Physical Disinfectants
The physical disinfectants are sunlight, desiccation, and heat.
Sunlight is a good disinfectant provided the in[Pg 241]fected material or
germs are directly exposed to the rays of the sun. Bacteria are killed
within a short time, but spores need a long time, and some of them
resist the action of the sun for an indefinite period. The
disadvantages of sunlight as a disinfectant are its superficial
action, its variability and uncertainty, and its slow action upon most
germs of infection. Sunlight is a good adjunct to other methods of
disinfection; it is most valuable in tuberculosis, and should be used
wherever possible in conjunction with other physical or chemical
methods of disinfection.[20]
Desiccation is a good means of disinfection, but can be applied only
to very few objects; all bacteria need moisture for their existence
and multiplication, hence absolute dryness acts as a good germicide.
Meat and fish, certain cereals, and also fruit, when dried, become at
the same time disinfected.
Heat is the best, most valuable, all-pervading, most available, and
cheapest disinfectant. The various ways in which heat may be used for
disinfection are burning, dry heat, boiling, and steam.
Burning is of course the best disinfectant, but it not only destroys
the germs in the infected materials, but the materials themselves; its
application is therefore limited to articles of little or no value,
and to rags, rubbish, and refuse.
Dry Heat.—All life is destroyed when exposed to[Pg 242] a dry heat of 150°
C. for one hour, although most of the bacteria of infection are killed
at a lower temperature and in shorter time. Dry heat is a good
disinfectant for objects that can stand the heat without injury, but
most objects, and especially textile fabrics, are injured by it.
Boiling.—Perhaps the best and most valuable disinfectant in existence
is boiling, because it is always at command, is applicable to most
materials and objects, is an absolutely safe sterilizer and
disinfectant, and needs very little if any preparation and apparatus
for its use. One half hour of boiling will destroy all life; and most
bacteria can be killed at even a lower temperature. Subjection to a
temperature of only 70° C. for half an hour suffices to kill the germs
of cholera, tuberculosis, diphtheria, plague, etc. Boiling is
especially applicable to textile fabrics and small objects, and can
readily be done in the house where the infection exists, thus
obviating the necessity of conveying the infected objects elsewhere,
and perhaps for some distance, to be disinfected.
Steam.—Of all the physical disinfectants steam is the most valuable
because it is very penetrating, reliable, and rapid; it kills all
bacteria at once and all spores in a few minutes, and besides is
applicable to a great number and many kinds of materials and objects.
Steam is especially valuable for the disinfection of clothing,
bedding, carpets, textile fabrics, mattresses, etc. Steam can be used
in a small way, as well as in[Pg 243] very large plants. The well-known
Arnold sterilizers, used for the sterilization of milk, etc., afford
an example of the use of steam in a small apparatus; while municipal
authorities usually construct very large steam disinfecting plants. A
steam disinfector is made of steel or of wrought iron, is usually
cylindrical in shape, and is covered with felt, asbestos, etc. The
disinfector has doors on one or both ends, and is fitted inside with
rails upon which a specially constructed car can be slid in through
one door and out through the other. The car is divided into several
compartments, in which the infected articles are placed; when thus
loaded it is run into the disinfector. The steam disinfectors may be
fitted with thermometers, vacuum formers, steam jackets, etc.
Gaseous Chemical Disinfectants
Physical disinfectants, however valuable and efficient, cannot be
employed in many places and for many materials infected with disease
germs, and therefore chemicals have been sought to be used wherever
physical disinfectants could not for one or more reasons be employed.
Chemicals are used as disinfectants either in gaseous form or in
solutions; the gaseous kinds are of especial value on account of their
penetrating qualities, and are employed for the disinfection of rooms,
holds of ships, etc. There are practically but two chemicals which are
used in gaseous[Pg 244] disinfection, and these are sulphur dioxide and
formaldehyde.
Sulphur Dioxide.—Sulphur dioxide (SO2) is a good surface
disinfectant, and is very destructive to all animal life; it is one of
the best insecticides we have, but its germicidal qualities are rather
weak; it does not kill spores, and it penetrates only superficially.
The main disadvantages of sulphur dioxide as a disinfectant are: (1)
that it weakens textile fabrics; (2) blackens and bleaches all
vegetable coloring matter; (3) tarnishes metal; and (4) is very
injurious and dangerous to those handling it.
There are several methods of employing sulphur in the disinfection of
rooms and objects, e. g., the pot, candle, liquid, and furnace
methods.
In the pot methods crude sulphur, preferably ground, is used; it is
placed in an iron pot and ignited by the aid of alcohol, and in the
burning evolves the sulphur dioxide gas. About five pounds of sulphur
are to be used for every 1,000 cubic feet of space. As moisture plays
a very important part in developing the disinfecting properties of
sulphur dioxide, the anhydrous gas being inactive as a disinfectant,
it is advisable to place the pot in a large pan filled with water, so
that the evaporated water may render the gas active. For the purpose
of destroying all insects in a room an exposure of about two hours to
the gas are necessary, while for the destruction of bacteria an
exposure of at least fifteen to sixteen hours is required.
[Pg 245]In the application of disinfection with sulphur dioxide, as with any
other gas, it must not be forgotten that gases very readily escape
through the many apertures, cracks, and openings in the room and
through the slits near doors and windows; and in order to confine the
gas in the room it is absolutely necessary to hermetically close all
such apertures, cracks, etc., before generating the gaseous
disinfectant. The closing of the openings, etc., is done by the
pasting over these strips of gummed paper, an important procedure
which must not be overlooked, and which must be carried out in a
conscientious manner.
When sulphur is used in candle form the expense is considerably
increased without any additional efficiency. When a solution of
sulphurous acid is employed, exposure of the liquid to the air
suffices to disengage the sulphur dioxide necessary for disinfection.
The quantity of the solution needed is double that of the crude drug,
i. e., ten pounds for every 1,000 cubic feet of room space.
Formaldehyde.—At present the tendency is to employ formaldehyde gas
instead of the sulphur so popular some time ago. The advantages of
formaldehyde over sulphur are: (1) its nonpoisonous nature; (2) it is
a very good germicide; (3) it has no injurious effect upon fabrics and
objects; (4) it does not change colors; and (5) it can be used for the
disinfection of rooms with the richest hangings, bric-a-brac, etc.,
without danger to these. Formaldehyde is evolved either[Pg 246] from paraform
or from the liquid formalin; formerly it was also obtained by the
action of wood-alcohol vapor upon red-hot platinum.
Formaldehyde gas has not very great penetrating power; it is not an
insecticide, but kills bacteria in a very short time, and spores in an
hour or two.
Paraform (polymerized formaldehyde; trioxymethylene) is sold in
pastilles or in powder form, and when heated reverts again to
formaldehyde; it must not burn, for no gas is evolved when the heating
reaches the stage of burning. The lamps used for disinfection with
paraform are very simple in construction, but as the evolution of the
gas is very uncertain, this method is used only for small places, and
it demands two ounces of paraform for every 1,000 cubic feet of space,
with an exposure of twelve hours. Formaldehyde is also used in the
form of the liquid formalin either by spraying and sprinkling the
objects to be disinfected with the liquid, and then placing them in a
tightly covered box, so that they are disinfected by the evolution of
the gas, or by wetting sheets with a formalin solution and letting
them hang in the room to be disinfected.
The method most frequently employed is to generate the formaldehyde in
generators, retorts, and in the so-called autoclaves, and then to
force it through apertures into the room.
Of the other gaseous disinfectants used, hydrocyanic acid and chlorine
may be mentioned, although they[Pg 247] are very rarely used because of their
irritating and poisonous character.
Hydrocyanic Acid is frequently used as an insecticide in ships, mills,
and greenhouses, but its germicidal power is weak.
Chlorine is a good germicide, but is very irritating, poisonous, and
dangerous to handle; it is evolved by the decomposition of chlorinated
lime with sulphuric acid. Chlorine gas is very injurious to objects,
materials, and colors, and its use is therefore very limited.
Chemicals Used as Disinfectants
Solution of chemicals, in order to be effective, must be used
generously, in concentrated form, for a prolonged time, and, if
possible, warm or hot. The strength of the solution must depend upon
the work to be performed and the materials used. The method of
applying the solution differs. It may consist in immersing and soaking
the infected object in the solution; or the solution may be applied as
a wash to surfaces, or used in the form of sprays, atomizers, etc. The
most important solutions of chemicals and the ones most frequently
employed are those of carbolic acid and bichloride of mercury.
Carbolic Acid.—In the strength of 1:15,000 carbolic acid prevents
decomposition; a strength of 1:1,000 is needed for the destruction of
bacteria, and a three per cent to five per cent solution for the
destruc[Pg 248]tion of spores. Carbolic acid is used, as a rule, in two per
cent to five per cent solutions, and is a very good disinfectant for
washing floors, walls, ceilings, woodwork, small objects, etc. The
cresols, creolin, lysol, and other solutions of the cresols are more
germicidal than carbolic acid, and are sometimes used for the same
purposes.
Bichloride of Mercury (corrosive sublimate) is a potent poison and a
powerful germicide; in solutions of 1:15,000 it stops decomposition;
in solutions of 1:2,000 it kills bacteria in two hours; and in a
strength of 1:500 it acts very quickly as a germicide for all
bacteria, and even for spores. Corrosive sublimate dissolves in
sixteen parts of cold and three parts of boiling water, but for
disinfecting purposes it should be colored so that it may not be
inadvertently used for other purposes, as the normal solutions are
colorless and may accidentally be used internally. The action of the
bichloride is increased by heat.
Formalin is a forty per cent solution of formaldehyde gas, and its
uses and methods of employment have already been considered.
Potassium Permanganate is a good germicide, and weak solutions of it
are sufficient to kill some bacteria, but the objections against its
use are that solutions of potassium permanganate become inert and
decompose on coming in contact with any organic matter. Furthermore,
the chemical would be too expensive for disinfecting purposes.
[Pg 249]Ferrous Sulphate (copperas) was formerly very extensively used for
disinfecting purposes, but is not so used at present, owing to the
fact that it has been learned that the germicidal power of this
material is very slight, and that its value depends mostly upon its
deodorizing power, for which reason it is used on excreta in privy
vaults, etc.
Lime.—When carbonate of lime is calcined the product is common lime,
which, upon being mixed with water, produces slaked lime; when to the
latter considerable water is added, the product is milk of lime, and
also whitewash. Whitewash is often used to disinfect walls and
ceilings of cellars as well as of rooms; milk of lime is used to
disinfect excreta in privy vaults, school sinks, etc. Whenever lime is
used for disinfecting excreta it should be used generously, and be
thoroughly mixed with the material to be disinfected.
Disinfection of Rooms
Practical disinfection is not a routine, uniform, and thoughtless
process, but demands the detailed, conscientious application of
scientific data gained by research and laboratory experiments.
Disinfection to be thorough and successful cannot be applied to all
objects, material, and diseases in like manner, but must be adjusted
to the needs of every case, and must be performed conscientiously.
Placing a sulphur candle in a room, spilling a quart of carbolic acid
or a couple[Pg 250] of pounds of chlorinated lime upon the floors or objects,
may be regarded as disinfection by laymen, but in municipal
disinfection the disinfector must be thoroughly versed in the science
of disinfection and be prepared to apply its dictates to practice.
Rooms.—In the disinfection of rooms the disinfectant used varies with
the part of the room as well as with the character of the room. When a
gaseous disinfectant is to be used sulphur dioxide or formaldehyde is
employed, with the tendency lately to replace the former by the
latter. Wherever there are delicate furnishings, tapestries, etc.,
sulphur cannot be used on account of its destructive character; when
sulphur is employed it is, as a rule, in the poorer class of tenement
houses where there is very little of value to be injured by the gas,
and where the sulphur is of additional value as an insecticide.
Whenever gaseous disinfectants are used the principal work of the
disinfector is in the closing up of the cracks, apertures, holes, and
all openings from the room to the outer air, as otherwise the gaseous
disinfectant will escape. The closing up of the open spaces is
accomplished usually by means of gummed-paper strips, which are
obtainable in rolls and need only to be moistened and applied to the
cracks, etc. Openings into chimneys, ventilators, transoms, and the
like must not be overlooked by the disinfector. After the openings
have already been closed up the disinfectant is applied and the
disinfector quickly leaves the room, being careful to close the door[Pg 251]
behind him and to paste gummed paper over the door cracks. The room
must be left closed for at least twelve, or better, for twenty-four
hours, when it should be opened and well aired.
Walls and Ceilings of rooms should be disinfected by scrubbing with a
solution of corrosive sublimate or carbolic acid; and in cases of
tuberculosis and wherever there is fear of infection adhering to the
walls and ceilings, all paper, kalsomine, or paint should be scraped
off and new paper, kalsomine, or paint applied.
Metal Furniture should first be scrubbed and washed with hot soapsuds,
and then a solution of formalin, carbolic acid, or bichloride applied
to the surfaces and cracks.
Wooden Bedsteads should be washed with a disinfecting solution and
subjected to a gaseous disinfectant in order that all cracks and
openings be penetrated and all insects be destroyed.
Bedding, Mattresses, Pillows, Quilts, etc., should be packed in clean
sheets moistened with a five per cent solution of formalin, and then
carted away to be thoroughly disinfected by steam in a special
apparatus.
Sheets, Small Linen and Cotton Objects, Tablecloths, etc., should be
soaked in a carbolic-acid solution and then boiled.
Rubbish, Rags, and Objects of Little Value found in an infected room
are best burned.
Glassware and Chinaware should either be boiled or subjected to dry
heat.
[Pg 252]Carpets should first be subjected to a gaseous disinfectant, and then
be wrapped in sheets wetted with formalin solution and sent to be
steamed. Spots and stains in carpets should be thoroughly washed
before being steamed, as the latter fixes the stains.
Woolen Goods and Wool are injured by being steamed, and hence may be
best disinfected by formalin solutions or by formaldehyde gas.
Books are very difficult to disinfect, especially such books as were
handled by the patient, on account of the difficulty of getting the
disinfectant to act on every page of the book. The only way to
disinfect books is to hang them up so that the leaves are all open,
and then to subject them to the action of formaldehyde gas for twelve
hours. Another method sometimes employed is to sprinkle a five per
cent solution of formalin on every other page of the book; but this is
rather a slow process.[21]
Stables need careful and thorough disinfection. All manure, hay, feed,
etc., should be collected, soaked in oil, and burned. The walls,
ceilings, and floors should then be washed with a strong disinfecting
solution applied with a hose; all cracks are to be carefully cleaned
and washed. The solution to be used is preferably lysol, creolin, or
carbolic acid. After this[Pg 253] the whole premises should be fumigated with
sulphur or formaldehyde, and then the stable left open for a week to
be aired and dried, after which all surfaces should be freshly and
thickly kalsomined.
Food cannot be very well disinfected unless it can be subjected to
boiling. When this is impossible it should be burned.
Cadavers of infected persons ought to be cremated, but as this is not
always practicable, the next best way is to properly wash the surface
of the body with a formalin or other disinfecting solution, and then
to have the body embalmed, thus disinfecting it internally and
externally.
Disinfectors, coming often as they do in contact with infected
materials and persons, should know how to disinfect their own persons
and clothing. So far as clothing is concerned the rule should be that
those handling infected materials have a special uniform[22] which is
cleaned and disinfected after the day's work is done. The hands should
receive careful attention, as otherwise the disinfector may carry
infection to his home. The best method of disinfecting the hands is to
thoroughly wash and scrub them for five minutes with green soap,
brush, and water, then immerse first for one minute in alcohol, and
then in a hot 1:1,000 bichloride solution. The nails should be
carefully scrubbed and cleaned.
[Pg 254]
CHAPTER XI
Cost of Conveyed Heating Systems[23]
In our variable climate, with its sudden and extreme changes in
temperature, the matter of heating and ventilation demands the serious
attention of all houseowners and housebuilders.
The most common method of heating the modern dwelling is by a hot-air
furnace in the cellar, with sheet-metal ducts for conveying the heated
air to the various rooms. The advantages of a furnace are cheapness of
installation and, in moderate weather, a plentiful supply of warm but
very dry air. The disadvantages are the cost of fuel consumed, the
liability of the furnace to give off gas under certain conditions, and
the inability to heat certain rooms with some combinations of
temperature and wind. The cost of installing a furnace and its proper
ducts in a ten-room house is from $250 to $350; such a furnace will
consume fifteen to twenty tons of anthracite coal in a season in the
latitude of New York City. The hot-air system works better with
compact square houses than with long, "rangy" structures. For a[Pg 255] house
fully exposed to the northwest blasts, one of the other systems should
be considered.
Perhaps the next most popular arrangement is a sectional cast-iron
hot-water heater, with a system of piping to and from radiators in the
rooms to be heated. Hot-water heating has many advantages, some of
which are the warmth of the radiators almost as soon as the fire is
started and after the fire is out; the moderation of the heat; the
freedom from sudden changes in amount of heat radiated; the absence of
noise in operation, and the low cost in fuel consumed. Some of the
disadvantages are the high cost of installation and the lack of easy
or ready control (as the hot water cools slowly, and shutting the
radiator valves often puts the whole system out of adjustment). A
hot-water heating plant for a ten-room house will cost $400 to $600,
according to the type of boiler; the corresponding fuel consumption
will be twelve to sixteen tons of coal per season.
The third system in common use is by steam through radiators or coils
of pipe connected to a cast-iron sectional boiler, or a steel tubular
boiler set in brickwork. This system is in use in practically all
large buildings; and its advantages are the moderate cost of
installation (as the single-pipe system is very efficient and the
pressure to be provided against in connections and fittings is
slight); the ease of control (since any good equipment will furnish
steam in twenty minutes from the time the fire is[Pg 256] started, and fresh
coal thrown upon the fire with a closing of dampers will stop the
steam supply in five minutes—or any radiator may be turned on or off
in an instant); the ability to heat the entire house in any weather,
or any single room or suite of rooms only; and, lastly, the moderate
fuel consumption.
The disadvantages of steam heat are no heat, or next to none, without
the production of steam, involving some noise in operation, and danger
of explosion. Steam equipment in a ten-room house will cost $300 to
$550, the lower price being for a sectional boiler and the higher for
a steel boiler set in brickwork. The fuel consumed will be from ten to
fifteen tons per season.
Both hot-water and steam systems require supplementary means of
ventilation. Placing the radiators in exposed places, as beneath
windows, in the main hall near the front door, in northwest corners
and near outside walls, will insure some circulation of air; and, if
one or two open fire places be provided on each floor, there will be,
in most cases, sufficient ventilation without the use of special
ducts.
TRANSCRIBER'S NOTE.
1) Figure numbers (which aren't contiguous) have been preserved.
2) Part III, Chapter V. The table showing thickness of vitrified pipes
reads:
| 4 inches diameter |
1/2 inch thick |
| 6 " " |
1/16 " " |
| 8 " " |
3/4 " " |
| 12 " " |
1 " " |
The thickness figure for the 6 inch pipe has been left as originally
printed, but probably is incorrect (logically it should be somewhere
between 1/2 inch and 3/4 inch thick).
3) A larger version of figure 1 can be viewed by clicking on the figure image.
 WALTER REED.
WALTER REED.