
Title: Modern Copper Smelting
Author: Donald M. Levy
Release date: October 23, 2019 [eBook #59328]
Language: English
Other information and formats: www.gutenberg.org/ebooks/59328
Credits: Produced by deaurider, Paul Marshall and the Online
Distributed Proofreading Team at http://www.pgdp.net (This
file was produced from images generously made available
by The Internet Archive)

The Colour of the Converter Flame during the
Bessemerising of Copper Matte.
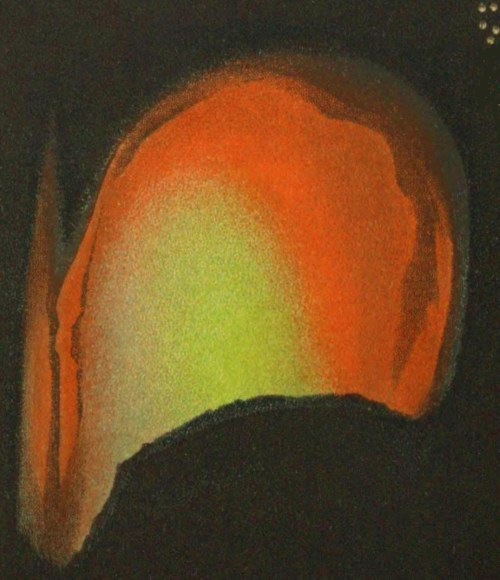
Fig. 2. Flame during the first or “slagging” stage.
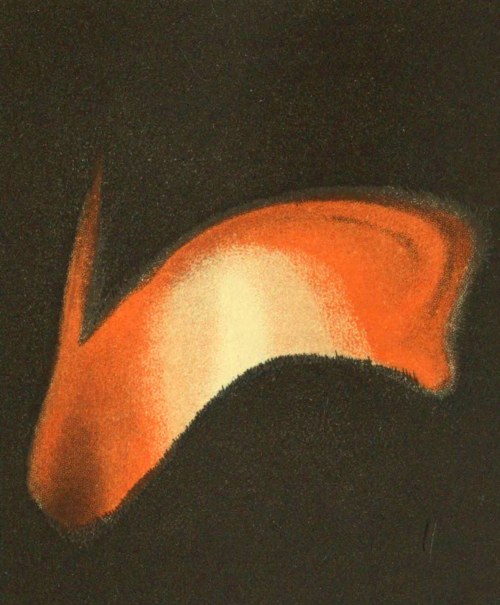
Fig. 3. “White Metal stage.” Slagging of Copper.
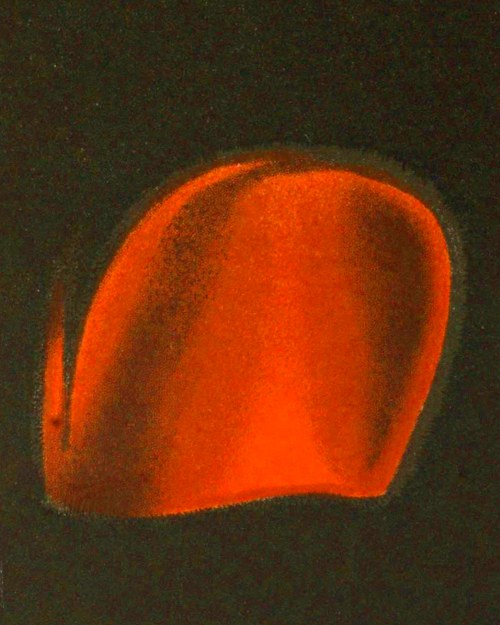
Fig. 4. Flame during second or “blowing to blister” stage.
MODERN
COPPER SMELTING.
BEING
LECTURES DELIVERED AT BIRMINGHAM UNIVERSITY
GREATLY EXTENDED AND ADAPTED, AND WITH
AN INTRODUCTION ON THE HISTORY, USES
AND PROPERTIES OF COPPER.
BY
DONALD M. LEVY, M.Sc., Assoc. R.S.M.,
ASSISTANT LECTURER IN METALLURGY,
UNIVERSITY OF BIRMINGHAM.
With frontispiece, and 76 illustrations.

LONDON:
CHARLES GRIFFIN & COMPANY, LIMITED;
EXETER STREET, STRAND.
1912.
[All Rights Reserved.]
The lectures on “Modern Copper Smelting” embodied in this volume were delivered at the University of Birmingham to the Senior Students in the School of Metallurgy and to others interested in the subject.
They are based largely upon the results of a study of the practice as conducted at a number of the best organised smelters and refineries in the United States of America, at which the author has had the opportunity of spending some considerable time, and it has been felt that there exists a scope, particularly on this side of the Atlantic, for a compact volume dealing broadly with the principles underlying Modern Copper Smelting, illustrated with such examples of working practice from personal observation. The subject-matter of the Lectures has been extended by the addition of an Introduction on the History, Uses, and General Metallurgy of Copper as applied to Modern Practice.
The Copper Industry is already fortunate in the literature at its disposal. It possesses standard works of reference through the publication of Dr. Peters’ classical volumes on the Principles of Copper Smelting, and more recently (during the preparation of the present work) of the volume on the Practice of Copper Smelting—works which have done much to raise copper smelting to a science. The industry is being rendered invaluable service by the Technical Societies and Technical Press, whose publications furnish an admirable record of the constant advance in the theory and practice of the art. Use has been made of these sources of information in the present work, and lists of such references are appended to each of the Lectures.
Grateful acknowledgment is made to several authors and editors who have given permission for the reproduction of illustrations or for the [Pg vi] inclusion of references:—Dr. Peters, Professor Gowland, Mr. Hughes, the Editors of the Engineering and Mining Journal, Mineral Industry, Mines and Minerals, and others. The Institution of Mining and Metallurgy, Messrs. Chambers Bros., The Traylor Engineering Co., and the Power and Mining Machinery Co. have very kindly provided blocks for several of the illustrations; the Anaconda Copper Mining Co. furnished a set of photographs, whilst Figs. 8, 37, and 76 have been reproduced by permission of the American Institution of Mining Engineers.
To the Superintendents and Staffs of the several smelters where opportunities were so freely given for studying modern practice, and particularly to Mr. E. P. Mathewson at Anaconda, Montana, to Mr. J. Parke Channing at the Tennessee Copper Company’s Smelter, and to Mr. W. H. Freeland at Ducktown, Tennessee, the author desires to express his appreciation for much valued information and many other kind services. The frequent references made in this book to the organisation and the methods employed at these works is not only a tribute to the useful information freely imparted, but is also due to the fact that such features are so thoroughly representative of the most advanced practice in copper smelting upon a large scale and of the direction in which all modern work is undoubtedly tending.
The author further thanks Professor Turner of Birmingham University for his interest in this volume, Mr. Frank Levy for reading the proofs, and the publishers, Messrs. Charles Griffin & Co., Ltd., for the care taken in the preparation and production of the work.
University of Birmingham,
May, 1912.
CONTENTS.
| pages | |
| LECTURE I. | |
History of Copper—Development of the Copper Industry—Progress of Smelting Practice—Price and Cost of Production of Copper—Copper Statistics, |
1–17 |
LECTURE II. |
|
The Uses of Copper: as Metal and as Alloy—The Physical Properties of Copper—Effects of Impurities—Mechanical Properties—Chemical Properties, |
18–34 |
LECTURE III. |
|
Compounds of Copper—Copper Mattes—The Varieties of Commercial Copper—Ores of Copper—Preliminary Treatment of Ores—Sampling, |
35–50 |
LECTURE IV. |
|
Modern Copper Smelting Practice—Preliminary Treatment of Ores: Concentration, Briquetting, Sintering—The Principles of Copper Smelting—Roasting, |
51–80 |
LECTURE V. |
|
Reverberatory Smelting Practice:—Functions of the Reverberatory Furnace—Requirements for Successful Working—Principles of Modern Reverberatory Practice—Operation of Modern Large Furnaces—Fuels for Reverberatory Work; Oil Fuel; Analysis of Costs—Condition of the Charge, |
81–112[Pg viii] |
LECTURE VI. |
|
Blast-Furnace Practice:—Functions of the Furnace—Reduction Smelting—Oxidation in the Furnace—The Pyritic Principle—Features of Modern Working: Water-Jacketing, Increase in Furnace Size, External Settling—Constructional Details of the Furnace, |
113–145 |
LECTURE VII. |
|
Modern Blast-Furnace Practice(continued):—Charge Calculations—Working—Disposal of Products—Pyritic Smelting—Sulphuric Acid Manufacture from Smelter Gases, |
146–191 |
LECTURE VIII. |
|
The Bessemerising of Copper Mattes:—Development of the Process—The Converter—Converter Linings—Grade of Matte—Operation of the Process—Systems of Working, |
192–216 |
LECTURE IX. |
|
The Purification and Refining of Crude Copper:—Preliminary Refining and Casting into Anodes—Electrolytic Refining—Bringing to Pitch, and Casting of Merchant Copper, |
217–243 |
Index, |
245–259 |
LIST OF ILLUSTRATIONS.
| Frontispiece
—The Colour of the Converter Flame during the Bessemerising of Copper Matte. |
||
| page | ||
| Fig. 1. | —Fluctuations in the Price of Best Select Copper, | 12 |
| " 2. | —Annual Production of Copper, | 16 |
| " 3. | —Equilibrium Diagram, Cu-Zn Series, | 22 |
| " 4. | —Influence of Arsenic and Antimony on the Electrical Conductivity of Copper, | 25 |
| " 5. | —Relations of Copper and Oxygen, | 27 |
| " 6. | —Microstructure of Copper containing Oxygen (Heyn), | |
| Plate to face | 28 | |
| " 7. | —Relations of Copper and Arsenic, | 29 |
| " 8. | —Freezing-Point Curve of Iron-Copper Sulphides (Mattes), | 38 |
| " 9. | —Outline of Sampling Scheme, Anaconda, | 48 |
| " 10. | —Section through Sampling Mill, | 48 |
| " 11. | —Brunton Sampler, | 49 |
| " 12. | —Outline of Smelting Scheme at the Anaconda Smelter, Montana, U. S. A., | 54 |
| " 13. | —Sketch Plan of Briquetting Plant, | 56 |
| " 14. | —Section through Auger-Former, showing Briquetting Mechanism of Chambers’ Machine, | 56 |
| " 15. | —Chambers’ Briquette-making Machine, | |
| Plate to face | 57 | |
| " 16. | —Dwight-Lloyd Sintering Machine, | 60 |
| " 17. | —O’Harra Furnace (Fraser-Chalmers), illustrating Principle of Mechanical Rabbling by Travelling Ploughs, | 71 |
| " 18. | —Section through Mechanically Rabbled Roaster Furnace (illustrating Improvements for Protecting Driving Mechanism), | 71 |
| " 19. | —MacDougal Roaster—Vertical Section, | 74 |
| " 20. | —Herreshof Furnace—Section indicating Connections for Cooling Rabbles and Spindles, | 74 |
| " 21. | —Spindle Connections and Guide Shields of Evans-Klepetko Roasters, | 76 |
| " 22. | —Rabble-blades and Bases, | 77 |
| " 23. | —Development of the Reverberatory Furnace (Gowland), | 90 |
| " 24. | —Draft Pressure Record of Anaconda Reverberatory Furnace, | 94 |
| " 25. | —Skimming Reverberatory Furnace, Anaconda, | |
| Plate to face | 96 | |
| " 26. | —Transverse Section of Modern Reverberatory Furnace, Anaconda, indicating Foundations, Hearth, and Bracing, | 96 |
| " 27. | —Reverberatory Furnace under Construction, | |
| Plate to face | 96[Pg x] | |
| " 28. | —Sectional Plan and Elevation of Reverberatory Furnace at Anaconda, | 98 |
| " 29. | —Fire-box End of Reverberatory Furnace, showing massive Bracing, Charge Bins, and Charging Levers, Anaconda, | |
| Plate to face | 100 | |
| " 30. | —Interior of Reverberatory Furnace (looking towards Skimming Door), showing Expansion Spaces in Roof, and Charging Holes, Anaconda, | |
| Plate to face | 100 | |
| " 31. | —Shelby Oil-Burner for Reverberatory Furnace Use, | 106 |
| " 32. | —Modern Blast-Furnace Shell of Sectioned Jackets (P. & M. M. Co.), | |
| Plate to face | 122 | |
| " 33. | —Blast-Furnaces under Construction, showing Fixing of Jackets, Bottom Plate, Method of Support, Sectioning, etc. (T. E. Co.), | |
| Plate to face | 124 | |
| " 34. | —Development of the Blast Furnace (Gowland), | 126 |
| " 35. | —Plan of 51-foot Blast Furnace, Anaconda, indicating Position of Crucibles, Spouts, and Connecting Bridge between Old Furnaces, | 128 |
| " 36. | —Longitudinal Section and Part Elevation of 87-foot Blast-Furnace, Anaconda, indicating Crucibles of Old Furnaces, Bridge, and Jacketing, | 128 |
| " 37. | —Copper Contents in the Slags accompanying Mattes of Various Grades, | 132 |
| " 38. | —Water-Jacketed Blast Furnace, lower portion indicating Air and Water Connections, Bottom Supports, End Slag Spouts, etc. (P. & M. M. Co.), | |
| Plate to face | 134 | |
| " 39. | —Tapping Breast of Blast Furnace, Cananea, | 136 |
| " 40. | —Rivetted Steel Water-Jacket, showing Tuyere Holes and Water Inlets, etc. (P. & M. M. Co.), | 137 |
| " 41. | —Transverse Section through Modern Blast Furnace, showing Arrangements of Boshed Lower Jackets, Upper Jackets, and Plates, Stays and Supports, etc., | 138 |
| " 42. | —Interior of Anaconda Blast Furnace, showing Jacketing, Tuyere Holes, and Bridge, | |
| Plate to face | 138 | |
| " 43. | —Showing Upper Jackets, Apron and Mantle Plates and Superstructure of Blast Furnace, Anaconda, | |
| Plate to face | 140 | |
| " 44. | —Charging Blast Furnaces, Anaconda, | |
| Plate to face | 140 | |
| " 45. | —Blast-Furnace Shell, with Air Connections (P. & M. M. Co.), | 142 |
| " 46. | —Details of Tuyere, Cananea Blast Furnace, | 142 |
| " 47. | —V-Shaped Charging Car, indicating Mechanism for Release and Tilting, | 153 |
| " 48. | —End View of Blast Furnace, showing Tilting of Charge Car, Anaconda, | 155 |
| " 49. | —Hodge’s Charging Car, | 155 |
| " 50. | —Freeland Charging Machine (D. S. C. & I. Co.), | 157 |
| " 51. | —Freeland Charger-Details, | 157 |
| " 52. | —Slag Spout, showing Method of Trapping Blast, also Replaceable Nose-Piece of Spout (A), | 159 |
| " 53. | —Details of Slag Spout, Cananea, | 161 |
| " 54. | —Slag Spout, showing Method of Support, | 161 |
| " 55. | —General View of Settler (T. E. Co.), | 163[Pg xi] |
| " 56. | —Method of Lining Settler, Cananea, | 163 |
| " 57. | —Arrangement for Matte and Slag Discharge from Settlers (T. C. C.), | 164 |
| " 58. | —Tap-hole Casting and Detail for Settlers, | 165 |
| " 59. | —Anaconda Blast Furnace (51 feet long), showing Settlers, | |
| Plate to face | 166 | |
| " 60. | —Hoppers of Flue-Dust Chambers and Tracks for Cars underneath, | 167 |
| " 61. | —Slotted Tuyeres, 12 inches by 4 inches (T. C. C.), | 185 |
| " 62. | —Sectional Elevation and Plan of Barrel-Shaped Silica-Lined Converter (Peters), | 196 |
| " 63. | —Latest Form of Silica-Lined Barrel Converter, | 197 |
| " 64. | —Longitudinal Section of Basic-Lined Converter, | 198 |
| " 65. | —Basic-Lined Converter, indicating Tuyeres, Lining, &c., | 199 |
| " 66. | —Composition of a Charge during Bessemerising Operation, | 208 |
| " 67. | —Pouring Slag, Anaconda, | 209 |
| " 68. | —General View of Converter Shop, Anaconda, | |
| Plate to face | 214 | |
| " 69. | —Sectional Plan, Elevation, and Transverse Sections of Refining and Anode-Casting Furnace, Anaconda (Peters), | 220 |
| " 70. | —Indicating Tilting and Pouring Mechanism of Ladle of Casting and Refining Furnaces, | 225 |
| " 71. | —Walker’s Anode-Casting Machine, | |
| Plate to face | 226 | |
| " 72. | —General View of Tank-room of Electrolytic Refinery (Perth Amboy, N.J.), | |
| Plate to face | 226 | |
| " 73. | —Indicating Methods of Suspending and Connecting Electrodes (Perth Amboy, N.J.), | 234 |
| " 74. | —Indicating Connections for Circulation of Electrolyte (Barnett), | 235 |
| " 75. | —Tank-house, showing Anode Crane (Ulke), | 237 |
| " 76. | —Microstructure of Commercial Copper containing Oxygen (Hofman), | |
| Plate to face | 242 | |
TABLES.
| table | page | |
| I. | The Production of Copper, | 15 |
| II. | North American Production of Copper, | 17 |
| III. | Influence of Impurities on the Electrical Conductivity of Copper, | 23 |
| IV. | Analysis of Various Commercial Coppers, | 44 |
| V. | Development in Size of the Reverberatory Furnace, | 89 |
| VI. | Daily Reports. Reverberatory Furnaces, | 102 |
| VII. | Daily Assay Report. Reverberatory Furnaces, | 103 |
| VIII. | Monthly Report. Reverberatory Furnaces, | 104 |
| IX. | Effect on Coke Consumption of Increased Sulphur in the Furnace Charge, | 120 |
| X. | Blast-Furnace Charge Calculations, | 151 |
| XI. | Typical Charging Tables at Pyritic Smelter, | 187 |
| XII. | Changes in Composition during Bessemerising, | 206 |
COPPER SMELTING.
History of Copper—Development of the Copper Industry—Progress of Smelting Practice—Price and Cost of Production of Copper—Copper Statistics.
The History of Copper.—Copper was probably the earliest metal commonly employed by mankind. It occurs in the native condition in various parts of the world, and the natural product thus required no metallurgical treatment prior to use. Its malleability and the property of being readily toughened by simple mechanical treatment were also factors which account for the discovery of its general usefulness in such primitive times.
Although silver and gold were possibly known even earlier, these metals appear to have been employed chiefly for ornamental purposes, and as tokens, rather than for general service.
The alloy of copper and tin, known as bronze, was the first metallic combination in common use by man; its employment was so characteristic in prehistoric times, that archæologists assign to one of the epochs the name of the Bronze Age. As is well known, archæological time is marked by a series of ages, in which the use, first of stone, then of bronze, and ultimately of iron for the manufacture of tools and implements, indicate the development of industrial culture. The dates which can be assigned to those periods vary with the locality; the races in the more Northerly latitudes being later in their development. In our own country, the Stone Ages may be said to date from 3000 b.c. down to 1000 b.c., and the Early and Late Bronze Ages from 1000 b.c. to 500 b.c., and from 500 b.c. to the commencement of the present era, respectively.
It is not unlikely that in many places copper was largely used during the Stone Ages and before the Bronze epoch, since it was only after the art of making fire had been discovered that it became possible to manufacture bronze, whilst native copper could be fashioned without the [Pg 2] aid of heat. Metallic relics of the Bronze Age, in the form of arms, ornaments, and domestic implements have been found in widely distributed localities.
The mention of copper occurs in the Hebrew Scriptures, the metal being termed Nehosheth, from the root Nahásh, to glisten. This was translated as χαλκὸς (chalcos) in the Septuagint, and Aes in the Vulgate; the Greeks and Romans using the terms, however, both for copper and for the alloys brass and bronze.
According to Pliny, the Roman supply was derived chiefly from Cyprus, and the metal thus came to be known as Aes Cyprium, which was gradually shortened to Cyprium, a name afterwards corrupted to Cuprum, from which are derived our modern terms Copper, the German Kupfer, and the French Cuivre.
Copper was well known to the alchemists, and inasmuch as it was largely obtained from Cyprus, an island dedicated to Venus, it was considered to be the metal specially sacred to the Goddess, and was generally known by that name in their writings, and symbolised by the sign ♀. The production of metallic copper on iron by the action of certain liquors from the Hungarian mines and other localities, was likewise known to the alchemists, and was a constant source of inspiration to them; the changes were regarded for some hundreds of years as examples of the transmutation of the elements, until Boyle showed that it was necessary to introduce copper into such solutions before that metal could be precipitated from them.
The Development of the Copper Industry.—The mining and smelting of copper ores on a primitive scale have been carried on from time immemorial; these operations were certainly practised in Greek and Roman days, and the deposits of Britain are said to have been known to the Phœnicians so far back as 1000 b.c. Percy refers to the finding of lumps of copper weighing 42 lbs., carrying a Roman inscription; this metal was found in close proximity to mines in North Wales, which yielded an easily reducible ore, and he concluded that this was smelted in situ by the Romans.
There are undoubted records of copper mining in this country in the time of Edward III., and in that of Elizabeth; whilst the first authentic accounts of copper smelting date also from the latter period, relating to South Wales. It appears that one of the earliest establishments was situated at Neath—a fact recorded in a publication of 1602. The works probably existed for a century before that date, and the copper smelters at Swansea were established about 120 years afterwards.
The processes employed for the primitive smelting of copper ores were, to a large extent, of the same nature as the crude operations practised [Pg 3] generally for the extraction of metals in remote ages and by primitive races, as recorded from time to time by travellers and explorers. The furnace-hearth was a hole in the ground, working usually on oxide ores with charcoal or wood as fuel. This primitive furnace was later developed, by the addition of walls for enclosing the charge, until the “shaft furnace” provided with an air blast of some kind was attained. The sulphide ores presented rather more difficulty in their treatment, but the production of metallic copper from sulphide materials by super-oxidation, in a process akin to the bessemerising of to-day, was developed in Japan centuries ago, and has been described by Professor Gowland.
It would appear that during the middle ages, the art of reducing copper ores to metal on a comparatively large scale was practised simultaneously in Britain and in Central Europe; first by primitive methods similar to those indicated above, developing later by successive improvements into the employment of small blast furnaces. By about 1700, however, the methods diverged, and it is interesting to note that the different styles of working then introduced have persisted, until recent years, as the methods typical of these two parts of the world. In Wales, where the well-known furnace coal was one of the characteristics of the locality, as it still remains to-day, the smelting processes developed along the lines of reverberatory practice, for which such fuel is eminently suited, and this resulted in the establishing of the representative Welsh process. On the other hand, the enormous forests of Central Europe furnished wood suitable for the making of charcoal, a type of fuel which necessitates close proximity with the furnace charge, so that in these localities smelting was carried out in the shaft furnace, which gradually developed into the small blast furnace. At the present time, the solid fuel suitable for reverberatory practice is only obtainable in very small quantities in Central Europe, and the characteristic method employed there for copper smelting is that in which small blast furnaces are used, except that charcoal has been largely replaced by coke as the fuel.
It is probable that the early ore furnaces of the primitive blast-furnace type in Britain were worked by Germans experienced in that class of work, just as at a later period in the history of the industry, Swansea coppermen were to be found in all parts of the world teaching other nations their art. Gowland reproduces a letter, dated January, 1583, protesting against the introduction of this foreign labour, whilst a second letter, dated July, 1585, which is also quoted, is of particular interest, as it gives evidence of a remarkable knowledge of the art of smelting, and, whilst illustrating an important [Pg 4] feature of modern practice, indicates also the manner in which an astute smelterman was able to work profitably with difficult material so long ago as three and a quarter centuries.
The letter is to the following effect:—
“Ulricke Frosse to Robert Denham. 4th July, 1585.
“To his loving friend, Robert Denham.
“Friend Denham,—I have me heartily commended unto you, you shall understand it we did lack ore more than 14 days ago, for we have found out a way to smelt 24 cwts. of ore every day with one furnace, the Lord be thanked, and if we may have ore enough from your side we may, with God’s help, melt with two furnaces in 40 weeks 560 tons of ore, having reasonable provision made for it, desiring you from hence-forward to send such ores as you have with as much speed as maybe, not caring what ore it is. Your ore of St. Dines is very hard to melt it, hoping we will overcome it what St. Ust ores will do, we long to see it.
“This I rest, the Lord send you good success with your mines. And so I commit you to God. From Neath, the 4th of July, 1585.
“Your friend,
“Ulricke Frosse.
“When you do send any more ore, if you can, send of all sorts, the better it will melt and with more profit.”
The sound principle of obtaining, when possible, one class of copper ore for the purpose of fluxing off the gangue from ore of another class, was thus recognised as a profitable feature of practice from comparatively early times.
Copper mining and smelting in Staffordshire dates back a considerable time, certainly prior to 1686; the mines were situated at Ecton, and the smelter was at Elleston, near Ashbourne, where small blast furnaces were employed. Copper smelting in Lancashire, which is nowadays conducted on a comparatively extensive scale, appears to have commenced in 1720 with Cornish ores and smaller importations from the West Indian and American Colonies. During the 18th century, the chief supply of the world’s copper ore came from the Cornish mines, which even at that time, were deep and extensive. It seems, however, that for some peculiar reason, the Cornishmen were unable to smelt these ores with profit, nor indeed, to do more with them than to send the material to South Wales to be treated. There are numerous explanations for their failure, which have been discussed exhaustively by Percy.
The centre of the copper smelting industry thus came to be located in the South Wales (Swansea) district, where circumstances were very favourable. The study of local conditions is one of great importance for metallurgists, and since this case affords a good example, it will [Pg 5] be of value to refer briefly to those circumstances which rendered the Swansea district such an excellent centre for the industry.
The extensive collieries in the locality rendered available an abundant supply of suitable fuel at a low price, and many of the smelters held a financial interest in them. The large coal was profitably used for home consumption or export, and the small, which, though dirty, still gave the long flame required, was very suitable for smelting work, and was reserved for that purpose.
Further, Swansea was an excellent seaport, situated at a short distance only from Cornwall, the chief source of ore, and was also readily accessible to vessels carrying cupriferous ores and products from South America, Australia, and other parts of the world. This was a great advantage, in that the Swansea copper smelters, having a large variety of ores at their disposal, some with basic gangue, others with siliceous gangue, were in a position to make up furnace charges which were more or less self-fluxing, and thus avoided the necessity for purchasing and using barren fluxes. The finished products were also in a most convenient centre for distribution, at the seaport of Swansea.
At the end of the 18th century, Great Britain was producing 75 per cent. of the world’s copper, the Cornish mines supplying most of the copper ore, and the Swansea smelters extracting most of the world’s supply of metal. Stevens has summarised the position for 1799, showing that “from the Cornish ores 4,923 tons of refined copper were produced, and from the Welsh ores of Anglesea 2,000 tons. The great Mansfeld mine in Germany produced only 372 tons in that year, Spain’s output was insignificant, and in the United States only a few tons were made. Russia and Japan probably ranked next to Great Britain as producers, small amounts of ore from Austria, Scandinavia, and Italy made up the remainder. Thus at the commencement of the 19th century, the copper resources of the United States, Spain, Chili, Mexico, Australia, Tasmania, Canada, and South Africa, which now supply over 90 per cent. of the world’s metal, were either undeveloped, or only yielded a few tons each; Great Britain, which produced nearly 7,000 tons of copper at that time, extracted from its own ore supplies, a hundred years later, only 550 tons.”
It will be remembered that it was in connection with the development of Cornish copper mining that the use of steam power in engineering was introduced and successfully worked out. On account of the increasing depth and extension of the Cornwall mines, the problem of disposing of the underground water became urgent, and led to the introduction of [Pg 6] steam engines for driving the pumps, the Newcomen engine being installed on the Wheal Fortune Mine in 1720. The success of this engine led to increase both in depth and in extent of the workings, until it became impossible to cope with the pumping requirements by this means. At the right moment Watt brought out the modern steam engine, and the first Watt engine was erected in 1777 at Chasewater, in Cornwall. It was the introduction of these improved methods of pumping which have made possible the successful development of present-day mining. Not only has the steam engine thus led to an increase in the supply of copper, by enabling the opening up of vaster deposits to be undertaken, but the development of engineering science which it has brought about, has caused a further consumption of the increasing quantity of copper which it has helped to render available for use.
During the first half of the 19th century Great Britain retained its position as the chief copper producer of the world, and the Swansea smelters possessed advantages such as have been rarely enjoyed by any other body of manufacturers. They were able to impose what conditions they pleased on the producers and sellers of copper ore, as well as on the consumers of the metal, and as business men, were not slow to avail themselves of their opportunities to the greatest possible extent, strengthening their position by the formation of a combination known as the Associated Copper Smelters of Swansea, which controlled the price of the metal from 1850 to 1860. Percy gives an interesting account of the terms imposed by them under the name of returning charges, etc., as well as of the conditions of sampling, analysis, and sale, which were strongly in their favour.
During these years of monopoly, the smelters were, on the whole, conservative in tendency from the metallurgical point of view, and few great developments in either processes or methods were devised: nevertheless, they enjoyed great prosperity, and their business attained such dimensions that Swansea remains one of the greatest centres of smelting industry in the world. The Welsh smeltermen had, moreover, acquired such proficiency in furnace management, and such knowledge of the working and control of copper charges, that their reputation had spread to all quarters of the world.
Though from 1840 onward, the British copper mining industry commenced to decline, still for 20 years longer the Swansea smelting works prospered more and more as new mines were being opened abroad and thus furnished a constantly increasing supply of rich copper ore, cheap to purchase and easy to smelt.
It was this development of foreign copper resources, and the unsatisfactory conditions which the producers received at the hands of [Pg 7] the smelters, which was the cause of the eventual displacement of Swansea from its position as the leading seat of copper manufacture.
In 1830, the production of copper ore in Chili had commenced and developed rapidly, Chili soon becoming one of the chief suppliers of ore to the Welsh smelters, whose independent attitude led to the first introduction of the copper-smelting industry on any large scale in America. Owing to the sailing conditions of the time, the simultaneous coming into port of several ships laden with ore, instead of their arrival at regular intervals, enabled purchases to be made by the smelters at a remarkably low figure, the standard price of the metal being subsequently raised. Mine-owners commenced to seek for a remedy, their ultimate endeavour being to substitute, for the exportation of their ores, smelting operations at or near the mines themselves. In 1842 Lambert introduced reverberatory furnaces into Chili, and so great was his success, that in a short time they were in use throughout that country. In 1857 he erected the first blast furnace in Chili, and the smelting industry thereupon grew so rapidly that, whilst from 1856 to 1865 the copper exports from Chili were in the proportions of ore 21 per cent., regulus 38 per cent., and bars 40 per cent., they subsequently became ore 1½ per cent., regulus 3½ per cent., and bars 95 per cent. The ultimate effect was a widening of the market for the finished Chilian product, so that Continental purchasers were enabled to obtain their supplies of metal direct, instead of being obliged to purchase from the Welsh smelters on the unsatisfactory terms then prevalent.
In 1842 the first large copper mines of Australia (Kapunda and later Burra Burra) were discovered, but developed slowly; and in 1844 the first copper mines of the Lake Superior district began work—on oxide ore, not on native metal.
In 1850 an enormous development in the Chilian mines commenced, half the world’s copper being produced from this source; in 1859–60 the Spanish mines at St. Domingo (Mason and Barry) were re-opened, as well as the Portuguese mine, the Tharsis. These mines were in reality operated in order to supply the wants of the sulphuric acid industry, the ore residues being subsequently smelted for copper at Swansea. In 1862, however, the Henderson wet process for copper was introduced, for which these materials were very suitable, and the Spanish and Portuguese supplies became of considerable importance, soon afterwards coming under the control of a Scottish company.
The competition from these new and abundant supplies of rich ores from Chili, Spain, and Portugal severely injured the production from the British mines; increasing supplies led to a fall in the price, and one [Pg 8] native mine after another shut down, the British supply diminishing with considerable rapidity.
In 1866 the great Calumet and Hecla mine at Lake Superior commenced operations, and speedily became one of the most important sources of copper in the world; the Moonta and Wallaroo mines in Australia opened about the same time, and in 1873 the Arizona mines started producing. In 1876 the enormous Spanish mines at Rio Tinto were re-opened, and soon rendered available large quantities of ore. Later, the Tasmanian supplies entered the markets.
In 1880 a remarkable development in copper mining occurred with the discovery of the Butte camp in Montana; this is now the greatest producer in the world.
The later extensions of the copper mining industry occurred in Utah, Tennessee, and Queensland, whilst within recent years the most important work on a large scale has been commenced in Tanganyika, in Nevada, and in Siberia.
The developments in the smelting industry in most of these localities have proceeded, until the last few years, on very similar lines. During the first periods following the opening up of mines and works, ore was shipped to the custom smelters, most often to Swansea; where, in the early days, many of those connected with the smelting works had some sort of financial interest in the foreign mines. Later, the ore underwent its first smelting to matte in the mining district itself, the matte product being then shipped East for treatment, thus saving much of the freight-charge on useless gangue, as well as smelters’ heavy returning charges, etc. At a later period the smelting operation was carried to a still further stage in the mining district, crude blister copper only being sent to Swansea or elsewhere to be refined.
Gradually, electrolytic refineries were established somewhat nearer to the mining districts, and in the natural course of events, and where local conditions are not prohibitive, the probability is that the whole cycle of operations from mining to the production of refined market metal will be carried out at the great camps themselves.
At present, however, this is not generally the case, since the conditions under which the enormous refineries in the Eastern States of New York, New Jersey, and in Baltimore, etc., operate, allow of the cheaper production of electrolytic copper at points nearer to the distributing markets. At Anaconda, indeed, the fully-equipped electrolytic plant was shut down, owing to the commercial conditions such as have just been indicated, having rendered the refining of the [Pg 9] anode copper at the Eastern refineries more profitable than electrolytic treatment on the spot.
The Chief Features in the Development of Modern Copper Smelting Practice.—In the early days of copper smelting, the reduction of the oxidised ores, which were then chiefly available, was not a problem of very great difficulty, although losses in slag were likely to be very high, and the operation generally wasteful. When, however, mines became deeper and sulphide ores had to be smelted, the problem became rather more complicated. In the first stages of development, such ores were probably roasted until as much sulphur as possible had been driven off, leaving practically an oxide charge to be treated by the older reduction methods involving the attendant extravagance in fuel consumption and large losses of copper in the slag.
From these crude and wasteful methods the Welsh process was gradually worked out, and it will ever rank as one of the finest examples of highly developed smelting practice in the history of metallurgy, particularly when the times and working conditions are borne in mind. The process having received such full treatment from most of the common text-books, it is not proposed to review it in detail here, since, moreover, it has been largely superseded by more modern processes.
As will be explained later, copper smelting of sulphide ores is essentially a fractional oxidation—chiefly of iron and sulphur—followed by the slagging or elimination of extraneous constituents of the ore. The Welsh process embodied a series of roastings and slaggings which, though most admirably adjusted for a substantial concentration of the copper in each succeeding product, allowed of the formation of slags in the first stages which carried but comparatively little copper, on account of the low tenor of the matte; whilst the slags in the later stages of the process, containing more copper on account of their association with higher grade matte, were made in such relatively small quantity that their re-treatment for the recovery of these values did not involve very much loss of efficiency in the furnace operations.
Later modifications of the process were chiefly devised with the view to reducing the number of operations, by eliminating the successive roasting stages, for which purpose oxidised materials, such as roasted or oxidised ores, were added to the charge.
The Best-Selecting process, and the Nicholl and James process are likewise valuable and ingenious modifications of the Swansea method for special work. [Pg 10]
In general, however, up to 1880, there had taken place but little change in principle from the older methods of smelting. The chief improvements involved a slow change in furnace size, and progress in several details in practice. The more important of these advances were—
(a) In Roasting Practice.—1865. Introduction of the mechanically driven furnace (the Brückner cylinder); not, however, adopted for copper smelting till many years afterwards. Later—Arrangements for using roaster gases for sulphuric acid manufacture.
(b) In Reverberatory Furnace Smelting.—1861. Gas firing introduced, but with very little success for copper smelting, even at the present day.
(c) In Blast-Furnace Smelting.—Several very important changes were introduced in the construction of furnaces.
1863. Elongation of the furnace.
Rachette in Germany introduced the elliptical blast furnace. (Intended first for lead smelting; rapidly adopted for copper matte smelting.)
1875. The water-jacketing of blast furnaces.
The Piltz water-jacketed furnace was likewise first employed in lead smelting, and subsequently introduced into copper smelting practice. The principle had, indeed, been utilised in certain branches of iron smelting before this date, but for non-ferrous work the idea was new.
Although the method of water-jacketing was recognised as leading to great improvement in the working of the furnace, its use was at first somewhat restricted, owing to various practical difficulties, and the ultimate great success was effected when in American practice, the plan of working the two principles of elongated furnaces and water-jacketing in conjunction, was adopted.
Commencing from 1880, and onwards, however, when production in the Far West began, enormous advances have been made, both in connection with the principles of working as well as in practical operation. These include—
[Pg 11] With an increased output of ore from the mines, and with increased consumption, stimulated by the growth of the electrical industry, the demand for metal increased so quickly that developments naturally followed with a view to an augmented and rapid production by more efficient and scientific processes; especially since increased competition and poorer ore supplies necessitated a very decided lowering of the costs of production. To meet the enormous present-day demand for metal with the older methods and furnaces would have been impossible. The greatest stimulus to the adoption of these new or modified processes was the shifting of the chief producing centres from the older and more conservative influences to districts like the then newly awakening West, where, with ever-increasing—almost limitless—supplies of ore available, and free from the necessity of considering the capital invested in old plants, the men in charge of the work, untrammelled by old smelting customs which might stand in the way of rapid progress, were in a position to develop their ideas with originality and vigour.
There may, nevertheless, be recalled the important share which British, and especially Swansea, workmen had in this great development of the industry. At many of the greater smelters in these new districts, Welsh furnacemen are still to be found, and large numbers went abroad in former days to take charge of such work, especially during the critical early stages. The principles underlying these modern improvements were, in many cases, first worked out by scientists in Europe.
The Price and Cost of Production of Copper.—The price of copper has been influenced to an enormous extent by financial speculation, so that until recent times it has fluctuated very considerably from year to year, the curve in fig. 1 relating to Best Select copper, indicating this variation over a considerable period. The price of the other qualities of commercial copper follows this line fairly closely, electrolytic copper being from £2 to £4 per ton lower, and standard copper £3 to £6 per ton. The average value of the standard refined metal at the present time (December, 1911) is about £56 per ton in London, and about 12 cents per pound in New York.
On three occasions during the past century, and once at least during the past decade, the market price of copper has been directly affected by more or less artificial conditions consequent on financial manipulation. The first of these instances was the 1850–1860 period, when the Welsh smelters held the monopoly of the copper trade, and were in a position to fix their own price; the second was during the French combination of Secretan during 1887–9, which, as a result of mere market speculation, caused fluctuations of price which amounted on one occasion to no less than £35 per ton within twenty-four hours. The third instance was created by the American combine. [Pg 12]
[Pg 13] In 1899 the Amalgamated Copper Company was formed in the United States. This corporation was established in view of the enormously increasing production of the West, and of the extensive development of electrical industry which involved a greatly increased consumption of copper; and it was probably designed to control the world’s copper industry. Prices were raised gradually for some time, but in 1901 the Trust, as then constituted, failed, owing largely to trade depression in Europe. Heavy losses resulted, as well as expensive law suits, and the price of the metal dropped again with great rapidity. Trade subsequently revived and expanded, the consumption of copper increased and appeared to overtake the rate of production, whilst stocks diminished and the price advanced, until, in 1907, copper was sold at well over £100 per ton. The American financial panic in the autumn of that year again reduced prices to a comparatively low figure, and they have, on the whole, remained fairly steady since, though showing a tendency to decrease. Production has, meanwhile, increased very largely, and a steady price of 12 to 13½ cents per pound yields handsome profits to most of the larger concerns. The present situation in the copper market is such that the enhanced production has again resulted in an accumulation of stocks, which has occasioned restricted output on the part of many of the principal smelters until briskness of trade development shall call forth increased consumption and more satisfactory prices.
The question of price is one involving certain considerations to which attention may be drawn. The present conditions and the comparative steadiness in the copper market have been shown in a recent review to result in part from:—
(1) The concentration of the copper industry in a few strong hands, which, whilst maintaining healthy competition, keeps the market free from such outside pressure as would reduce the price too much, and by restricting unprofitable output, brings production and consumption into equilibrium, making for stability.
(2) The comparative cheapness of money, which has allowed of the financing for large production, with the prospect of absorption not being long delayed.
At the same time, some of the richer and more cheaply worked mines of former times are gradually approaching exhaustion—recent instances of this will be readily recalled, whilst the disadvantages of having to work lower-grade deposits at greater depth have also tended to increase [Pg 14] the price of metal. These conditions, on the other hand, have been counterbalanced by improvements in the mining and metallurgical processes concerned, by the opening up of new districts, and by the economies resulting from amalgamation of interests, involving closer organisation and enormous outputs of material.
Apart from finance, two of the factors most likely to affect the price of the metal considerably are the possible replacement of copper for electrical transmission purposes by conductors of other metals; and further, the enormous prospective production in the newer districts, such as Utah, Nevada, and Tanganyika, in the course of a few years.
The cost of production of the metal is so dependent on local and general circumstances as not to admit of analysis in this place. Questions of locality, transport facilities, proximity to supplies of every kind, problems of labour, capitalisation, bye-products, and numerous similar considerations have such an important bearing on each individual case as to convey a definite meaning only to the man on the spot. In the same way, detail costs of each stage of the copper smelting processes are influenced by similar circumstances.
Broadly speaking, the average total cost of production and marketing at present may be taken as being somewhere about 10 cents per pound of copper; in certain specially favoured cases, 9, 8, or even 7 cents per pound. The newly opened low-grade “porphyry” camps at Utah and elsewhere, which have been commenced under an enormous capitalisation, anticipate a production at a cost of about 6 cents per pound when steady and normal running is in progress.
A recent analysis gives interesting information with regard to the cost of production estimated at different plants. Of the American output of about 480,000 tons in 1909—
| Almost 3·5 | per cent. was produced at a cost of | 7·14 | cents per lb. | (Nevada). | |
| 1·8 | " | 7·98 | " | (Baltic, Superior). | |
| 10·5 | " | 8–9 | " | (Utah, etc.). | |
| 48·3 | " | 9–10 | " | (Boston and Montana, Calumet and Hecla, etc.). | |
| 9·0 | " | 10–11 | " | (Utah Consolidated, Tennessee, etc.). | |
| 20·0 | " | 11–12 | " | (Anaconda, Arizona, Cananea). | |
| 1·8 | " | 12–13 | " | ||
| 1·1 | " | 13–14 | " | ||
| 1·4 | " | 14–15 | " | (Tamarack). | |
| 1·1 | " | 15–16 | " | ||
| 1·1 | " | 16–17 | " | ||
| 0·1 | " | 17·09 | " | [Pg 15] |
Copper Statistics.—The outstanding features which attract attention in the statistics of copper production will be most readily seen from the curves of fig. 2. The enormous increase within recent years in the total output of metal, and the overwhelming proportion produced by the United States of America, is clearly indicated. The curves also show the practical extinction of the native supply of Great Britain and the steady output of Spain and Germany.
An analysis of the total production for the year 1910 is given in the following Table I.:—
TABLE I.—THE PRODUCTION OF COPPER
(Short Tons of 2,000 lbs.).
| 1909. | 1910. | |||||
| ┐ | ||||||
| U. S. A., | 549,114 | 543,125 | │ | |||
| Canada, | 26,998 | 28,801 | │ | 1909. | 1910. | |
| Newfoundland, | 1,546 | 1,210 | ├ | North America, | 644,058 | 645,927 |
| Mexico, | 63,085 | 68,899 | │ | |||
| Cuba, | 3,315 | 3,892 | │ | |||
| ┘ | ||||||
| ┐ | ||||||
| Argentina, | 672 | 336 | │ | |||
| Bolivia, | 2,240 | 2,800 | ├ | South America, | 60,911 | 63,101 |
| Chili, | 40,079 | 39,463 | │ | |||
| Peru, | 17,920 | 20,502 | │ | |||
| ┘ | ||||||
| ┐ | ||||||
| Spain and Portugal, | 58,447 | 56,386 | │ | |||
| Germany, | 25,150 | 27,675 | │ | |||
| Russia, | 19,880 | 24,987 | │ | |||
| Norway, | 10,170 | 11,676 | │ | |||
| Hungary, | 5,152 | 5,550 | ├ | Europe, | 127,283 | 135,738 |
| Sweden, | 2,240 | 2,240 | │ | |||
| Italy, | 3,052 | 3,606 | │ | |||
| Austria, | 1,809 | 2,386 | │ | |||
| Turkey, | 896 | 672 | │ | |||
| Great Britain, | 487 | 560 | │ | |||
| ┘ | ||||||
| Japan, | 52,640 | 51,520 | ||||
| Africa, | 16,738 | 17,030 | ||||
| Australasia, | 38,528 | 45,153 | ||||
| Total, | 940,158 | 958,469 | ||||
| ═════ | ═════ | [Pg 16] | ||||
[Pg 17] In Table II. is indicated the distribution of the American production among the various States.
TABLE II.—North American Production of Copper
(in Short Tons of 2,000 lbs.).
| 1909. | 1910. | ||
| Alaska, | 2,028 | 2,504 | |
| Arizona, | 146,021 | 149,803 | |
| California, | 26,679 | 22,897 | |
| Colorado, | 5,244 | 5,063 | |
| Idaho, | 3,885 | 3,108 | |
| Michigan, | 113,624 | 110,700 | |
| Montana, | 156,918 | 143,121 | |
| Nevada, | 25,917 | 31,944 | (about 6,000 tons in 1908) |
| New Mexico, | 2,567 | 1,816 | |
| Utah, | 50,219 | 62,521 | (about 35,000 tons in 1908) |
| Wyoming, | 44 | 90 | |
| South and East, | 11,409 | 9,098 | |
| Other States, | 1,973 | 463 | |
| Totals, | 546,538 | 543,125 | |
| ═════ | ═════ |
There will be noticed a decline in the production of the United States during the year 1910, resulting from the present movement to restrict output whilst the large accumulated stocks of metal are being absorbed. The movement is probably more or less temporary, and is being largely directed by American financiers who are endeavouring to bring about an international agreement on the subject.
Regarding the American output, the marked movement for curtailment in Montana has reduced the output of that State to such an extent, that the position it gained in 1909, of being the greatest producing State once more reverts to Arizona. The increases from Nevada and Utah, in which developments on a large scale are commencing, may be noted.
Percy, John, “Metallurgy (Copper).”
Gowland, William, Presidential Address, Trans. Inst. Mining and Metallurgy, vol. xvi., 1906–7, pp. 265–291.
Stevens, H. J.,“The Copper Handbook.”
Brown, N., and Turnbull, C. C., “A Century of Copper.”
Engineering and Mining Journal, “Copper Production.” May 6th, 1910, p. 891.
Mineral Statistics of the United Kingdom.
Mineral Industry.
The Uses of Copper: as Metal and as Alloy—The Physical Properties of Copper—Effects of Impurities—Mechanical Properties—Chemical Properties.
The Uses of Copper.—Generally speaking, the industrial applications of copper involve its employment in two forms:—
(1) As metal. (2) As a constituent of alloys.
The more limited use in the form of copper salts is of chemical rather than of metallurgical interest.
Copper in the metallic form is employed for three classes of work:—
(a) Electrical Uses.—Of late years the marked growth in the consumption of copper has arisen very largely from its usefulness as a conductor of electricity; the increased demand for the metal with the development of electrical enterprise being a well-marked feature in industrial progress. It is estimated that from 60 to 70 per cent. of all the copper produced is utilised for this purpose, and metal is specially prepared and sold under the designation of “high-conductivity copper.” The demand has, to a large extent, increased irrespective of price up to recent years, owing to the necessity of employing copper for such purposes, though the natural economic factor that an enhanced price of the metal tends to some discouragement of expansion and of fresh electrical enterprise, has exerted considerable effect in checking consumption.
It is merely necessary to enumerate some few of the present aspects of electrical industry in order to realise the enormous absorption of copper in this connection, as, for instance, electrical traction, lighting, and power, the telegraph, and the telephone. With reference to the use of the metal for this work, it is important that certain mechanical as well as electrical requirements should be fulfilled, for in many branches, considerable strength of the material is also [Pg 19] requisite. The demand of the electrical engineer is that as a conductor, the copper shall offer a minimum of resistance to the passage of the current, and for this requirement the metal must be in a condition of very great purity. With but few exceptions, this necessitates the purification of the copper by electro-deposition. Electro-deposited metal as produced at the refineries is, however, not immediately suitable for drawing into wire, owing to the weakness and porosity inherent in the material prepared by this method. It must, therefore, be melted, brought to pitch, cast into bars, and these bars transformed into wire, which operations require to be conducted with much care in order to keep the metal in as pure a condition as possible for its work. It may be noted that within recent years, several processes, notably those of Cowper-Coles and Elmore, have been put into operation for the direct manufacture for electrical purposes, of electrolytic-copper wire of the requisite strength.
The mechanical qualities demanded of the metal for such purposes as telegraph work may be indicated by the two specifications of wire for the British Post Office, which are appended:—
The following figures afford some indication of the increasing demand for copper in two branches only of electrical industry:—
| 1902. | 1907. | ||
| Mileage of wire for | telegraph purposes, | 3·9 | 5·3 |
| "" | telephone purposes, | 10·9 | 28·2 |
| (in million of miles) | |||
(b) Engineering Uses.—Metallic copper finds application in marine shipbuilding and engine work, as well as in railway and locomotive work, where the metal is particularly employed for steam pipes, and for fire-box plates and stays, sometimes also for boiler tubes, on account of its high conductivity for heat, combined with toughness. The [Pg 20] questions of suitable composition, and the other requirements of the metal intended for these purposes, has been a subject for discussion by some of the leading marine and locomotive engineers. Useful information on the subject will be found in the reports of some of these discussions at the Institution of Mechanical Engineers.
The following tests are required for copper plate (best quality) intended for locomotive fire-boxes on the Lancashire and Yorkshire Railway, taken from standard specifications given by their Chief Mechanical Engineer at the Institute of Metals:—
Bending Test.—Pieces of the plate shall be tested both cold and at a red heat by being doubled over on themselves— that is, bent through an angle of 180°—without showing either crack or flaw on the outside of the bend.
Flanging.—Plates must not show any defects in flanging.
Tensile Test.—Ultimate breaking load, 14 tons per square inch; Elongation, 35 per cent. in 8 inches.
Analytical Test.—To be made at contractor’s expense.
The copper upon analysis to give the following results:—Arsenic, not less than 0·35 per cent. nor more than 0·55 per cent.; other foreign elements, exclusive of combined oxygen, not to exceed 0·25 per cent.
Clauses are also inserted as to stamping, inspection, and the giving of testing facilities.
Typical analysis of such plates show—
Impurities, chiefly antimony, lead, iron, nickel, tin, and sulphur, not exceeding 0·25 per cent.
The average test on a number of plates gave—
The effect of temperature and the influence of impurities on the mechanical properties of the metal intended for engineering purposes are of very great importance, and much attention has been devoted to researches in this subject, particularly by Milton and Le Chatelier, whose published experience gives important information of much practical value. The main conclusions arrived at from practice have had [Pg 21] reference to the general effects of impurities in hardening the metal, and the general tendency of heat to soften it and to increase the ductility. The diverse effects of different impurities on strength and ductility will be reviewed in detail at a later stage.
(c) General Industrial Uses.—Copper as metal is also employed to a considerable extent in certain important industries, as in textile manufacture, where it is used for the rollers in calico-printing; and it is in general industrial use in the form of copper heaters, vats, coils, pans, and the like, and occasionally also for roofing and sheathing.
Uses of Copper Alloys.—Between 20 and 30 per cent. of the copper produced is employed in the form of alloys. The more important of these are:—
It is further not unlikely that several classes of ternary alloys, at present still under investigation, may have important industrial application in the future. Among such alloys may be mentioned the copper-aluminium series alloyed with other metals, Monel metal and the Monel steel series, etc.
Of the above alloys, the brasses are by far the most widely used. It may be recalled that the advantages possessed by alloys of copper and zinc are in large measure due to their increased strength and hardness; to the fact that they are more fusible, and more fluid when melted, and so give good castings; that they are characterised by a good colour and high lustre, as well as by the factor of cheapness resulting from the addition of a less costly metal—zinc—in their manufacture.
The uses of the copper alloys may also be arranged in two classes—(a) engineering uses, and (b) general uses. Of the brasses, those containing upwards of 70 per cent. of copper may be rolled cold, whilst the alloys with less than 70 per cent. are hot-rolled.
In the engineering industry large quantities of 70⁄30 brass are utilised in the form of condenser tubes, whilst for the multifarious requirements of general engineering work, very considerable amounts of brass of lower tenor are employed in the forms of taps, pipes, fittings, etc.
Muntz metal, the 60⁄40 brass, finds extended application for the sheathing of ships, whilst [Pg 22] the employment of brass and of the other alloys for all manner of articles of general utility is a matter of common knowledge.
The close connection between properties, constitution, and the equilibrium diagram of these various classes of alloys has become manifest to a marked degree within recent years, and the effects of thermal treatment partly in modifying their constitution, and thereby the properties, and also in controlling the condition and distribution of the constituents, are at the present time having an important bearing on the manipulation of these alloys in the industries manufacturing them and adapting them for their various uses. The study and application of these equilibrium diagrams are highly important to those who have to deal with these alloys on an industrial scale.
The Properties of Copper.—The properties of the metal which render it of such service in the arts and industries are mainly its high electrical conductivity, its great ductility, malleability, and toughness, which enable it to be readily worked up into the different forms in which it is employed, its high thermal conductivity, and its resistance to the various agencies which lead to corrosion. These are consequently the properties to which close study is directed. Of perhaps still greater importance is a knowledge of the influence exerted upon these properties by the circumstances which usually attend working practice; such as, for example, the various common impurities, and the variations of temperature, as well as the previous mechanical and thermal treatment. These can only be indicated in general terms here, references to authorities on the different branches being given later. [Pg 23]
TABLE III. —Influence of Impurities on the Electrical Conductivity.
| Addicks. | Johnson. | |||
|---|---|---|---|---|
| Impurity. Per cent. |
Conductivity. | Impurity. Per cent. |
Conductivity. | |
| Pure copper, | .. | 101 | .. | 101 |
| Copper with— | ||||
| Aluminium, | 0·006 | 98·6 | 0·01 | 99·7 |
| 0·109 | 66·8 | 0·02 | 98·8 | |
| 0·739 | 43·5 | .. | .. | |
| Antimony, | 0·007 | 99·6 | .. | .. |
| 0·022 | 97·2 | .. | .. | |
| 0·047 | 95·4 | 0·05 | 96·9 | |
| Arsenic, | 0·004 | 99·6 | .. | .. |
| 0·007 | 96·8 | .. | .. | |
| 0·013 | 93·2 | 0·04 | 92·4 | |
| 0·140 | 62·3 | 0·06 | 82·0 | |
| Bismuth, | 0·028 | 99·6 | 0·01 | 95·7 |
| 0·045 | 99·3 | .. | .. | |
| Cadmium, | 0·062 | 99·5 | .. | .. |
| 0·113 | 99·1 | .. | .. | |
| 0·427 | 96·1 | .. | .. | |
| Cobalt, | .. | .. | 0·05 | 92·0 |
| Gold, | 0·089 | 98·9 | 0·05 | 99·7 |
| 0·149 | 98·4 | .. | .. | |
| 0·317 | 96·4 | .. | .. | |
| Iron, | 0·042 | 96·8 | .. | .. |
| 0·046 | 92·9 | .. | .. | |
| 0·068 | 89·6 | 0·09 | 98·8 | |
| Lead, | 0·083 | 99·1 | .. | .. |
| 0·052 | 98·7 | 0·06 | 100·6 | |
| 0·347 | 98·3 | .. | .. | |
| Manganese, | .. | .. | 0·02 | 98·8 |
| Nickel, | .. | .. | 0·05 | 91·4 |
| Oxygen, | 0·020 | 100·7 | .. | .. |
| 0·050 | 101·4 | .. | .. | |
| 0·100 | 100·5 | 0·10 | 99·8 | |
| Phosphorus, | 0·08 | 52·3 | 0·004 | 98·5 |
| Platinum, | .. | .. | 0·02 | 93·6 |
| Silicon, | 0·007 | 99·4 | 0·004 | 99·7 |
| 0·042 | 99·0 | 0·01 | 98·4 | |
| Silver, | 0·003 | 100·5 | .. | .. |
| 0·137 | 100·0 | 0·05 | 99·8 | |
| 0·340 | 98·3 | .. | .. | |
| Sulphur, | 0·053 | 100·0 | 0·01 | 98·5 |
| 0·135 | 99·0 | .. | .. | |
| 0·236 | 98·9 | .. | .. | |
| Tellurium, | 0·065 | 100·4 | .. | .. |
| 0·181 | 100·2 | .. | .. | |
| 0·405 | 98·7 | .. | .. | |
| Tin, | 0·052 | 97·6 | 0·05 | 100·5 |
| 0·097 | 92·7 | .. | .. | |
| 0·295 | 79·8 | .. | .. | |
| Zinc, | 0·048 | 98·3 | 0·02 | 98·5 |
| 0·095 | 96·3 | .. | .. | |
Physical Properties.—The colour of copper is familiar, being a fine salmon pink. The appearance of the fractured surface is a useful guide in several respects as to the condition of the metal, [Pg 24] and in the process of manufacture the refiner relies upon this appearance as an important criterion of the progress of the refining operation. Copper containing an excess of oxygen, for example, has a purplish-red colour and a coarse brick-like fracture; this is known as “dry copper,” and the metal is brittle and commercially useless when in that form. The ingot of dry copper is also characterised by a depression running along the surface. Tough copper (“tough-pitch”) the mechanically useful variety resulting from the furnace-refining operation, possesses a bright salmon-coloured fracture, finely granular to silky in appearance, whilst “overpoled copper,” also brittle and industrially valueless whilst in that condition, has a very light salmon-coloured fracture, and is more coarsely fibrous.
The melting point of copper is 1,083° C., and is slightly lowered by the small quantities of impurity usually present in commercial metal. Molten copper is of a pale apple-green colour. The boiling point under ordinary conditions is about 2,300° C. (1,700° C. in vacuo). The electrical conductivity is of much importance. Copper ranks second only to silver as a conductor, the relative conductivity of the best copper being about 98 compared with silver as 100. The resistance of 12 inches of pure copper wire, 0·001 inch in diameter, is 9·612 ohms. The conductivity of the metal is decreased by mechanical working, and it follows the general straight-line law connecting conductivity and temperature.
The effect of even small quantities of impurity on this property is very marked, so much so that only the purest varieties are suitable for electrical work, and for this reason electrolytic refining is often a necessary operation in the manufacture of copper intended for this purpose.
Table III. on preceding page, summarises the results of the work of Addicks and Johnson, and indicates the effects of small amounts of different impurities on the conductivity of the metal.
The notoriously destructive effect of arsenic on the conductivity is very apparent.
The influence of most of the common impurities is of a similar nature, and detailed investigations indicate that the effect is more or less progressive as the quantity increases—within the limits usually present in commercial metal. The results of Hiorns and Lamb’s experiments with reference to arsenic and antimony are indicated in Fig. 4.
The specific gravity of copper naturally varies according to its condition and composition. When pure and in the worked state, its density is 8·95; cast metal, more open and inclined to porosity, has a [Pg 25] density of about 8·2 to 8·6, depending on the purity, rate of cooling, etc. Impurities lower the specific gravity.
The conductivity for heat of the metal is high, being 898 compared with gold as 1,000, and as a conductor it is two and a-half times more efficient than iron. It is this property, combined with its toughness and resistance to corrosion, etc., which largely determines its employment for heaters, steam-coils, and the like.
Power of Dissolving Gases.—When molten, especially under reducing conditions, the metal possesses the property, common to many others, of absorbing gases such as carbon monoxide, hydrogen, hydrocarbons, sulphur dioxide, etc., which are moreover, to a large extent insoluble in the solid material, and are, therefore, often liberated at or about the moment of solidification; though some may remain dissolved. This action is one of the causes of the difficulty which is experienced in making sound castings of the metal, particularly since the gases mentioned are present in quantity during the poling and refining operations. The presence of certain materials in the copper, as in the [Pg 26] case of steel, appears to reduce the dissolving power of the liquid metal for these gases, or possibly to increase their solubility when the copper is solidifying, and in this way tends to minimise their injurious effects. It would seem that one of the functions of the cuprous oxide, which is purposely introduced into the metal when “bringing it up to pitch,” is to exert this action. The ridge in the ingot of overpoled copper is, to some extent, accounted for as being due to the effects of the evolved gases, and this appearance indicates the absence of the requisite quantity of cuprous oxide necessary to counteract the effect.
Copper is also supposed to be capable of holding certain quantities of gas in solution after it has become solid, and the resulting metal is more brittle and often commercially useless. Several of the characteristics of overpoled copper probably arise from this cause also.
Impurities[2] in Copper.—In view of the marked influence of impurities on the properties of metallic copper, it may be advisable in this place briefly to review the results of recent scientific work as to the condition in which they exist in the metal, thus offering some clearer indication of the manner in which they affect the mechanical and other properties. The common impurities in ordinary commercial metal may be oxygen, arsenic, antimony, bismuth, lead, and to smaller extents, iron, sulphur, tellurium, and selenium.
A factor of much importance is that the effect of two or more of the common constituents when present together, may be of even greater moment than that of each one separately, and in this connection Hampe’s classical work should be consulted. The investigation of the joint effects of impurities becomes so complex that systematic study progresses but slowly. Metallographic work is, however, revealing much evidence, and the researches in progress at present at several laboratories will, when published, afford greatly increased knowledge on the subject. Recent papers by F. Johnson give valuable detailed information (see References, p. 34). The importance of oxygen in this connection is particularly marked: its effects are profound, since in addition to its own specific influence as oxide, it also brings about chemical changes in some of the other constituents, thus leading to the formation of entirely new compounds possessing quite different properties. The beneficial influence of certain definite proportions of oxygen in addition to the other constituents of commercial copper is [Pg 27] well known in practice, and has been systematically studied by Hampe, and later by several other workers with more delicate means of investigation at their disposal.
Oxygen in Copper.—Molten copper has the power of dissolving its oxide, Cu2O. When the melted metal is exposed to oxygen, this oxide is produced and passes into solution in the liquid, yielding a series of binary alloys, of which the oxide acts as the second constituent. The equilibrium diagram of the series, as worked out by Heyn[3] (see Fig. 5), affords a good indication of these relationships, and throws light on several features connected with the presence of oxygen in copper.
It will be observed that when molten oxygenated metal containing less than about 0·38 per cent. of oxygen solidifies, copper crystallises out first, whilst later, in between the copper crystals, there solidifies a eutectic of copper and cuprous oxide. This eutectic contains about 3·45 [Pg 28] per cent. of cuprous oxide, equivalent to 0·38 per cent. of oxygen; it melts at a temperature about 18° C. below that of the pure metal. The presence of this material, which is of a blue colour when viewed under the microscope, constituting slightly more fusible, tough, non-conducting areas between the copper crystals, accounts for many of the well-known effects of oxygen in metallic copper.
When oxygen is present in quantities above the eutectic proportion, the first constituent to solidify from the molten over-oxygenated copper is brittle copper oxide, and the presence of such brittle material disseminated through the metal explains why “dry copper” cannot be worked.
The effects of comparatively small quantities of oxygen are greatly increased on account of the fact that one part of oxygen, when present as cuprous oxide, yields a constituent in almost nine times as great a proportion by weight alone, since Cu2O : O :: 142 : 16 or 9 : 1; whilst oxygen existing as oxide-eutectic is represented in the ratio of nearly 30 : 1. The presence of excess of copper oxide in the metal is particularly dangerous when copper is to undergo annealing in a reducing atmosphere, since the reducing gases acting upon the oxides at the crystal boundaries destroy them, thus tending to produce that rottenness in the material which is so often encountered under such circumstances.
The great value and importance of oxygen in copper lies in its property of bringing the metal up to pitch as indicated above.
The effect of carbon on oxygenated copper was the subject of much enquiry in early years. It was thought at one time that the influence of carbon per se in the copper was responsible for the beneficial effects resulting from the melting of brittle “dry” copper with carbon, but the work of Percy, since confirmed, showed that its sole action is in the reduction of the injurious excess of oxide.
In addition to the specific influences of oxygen as just recorded, and to its important physical effects with regard to the solubility of gases, etc., oxygen in copper performs other valuable functions, by forming with reduced impurities which are exceedingly dangerous, oxygenated compounds more infusible and more insoluble; and this has the effect of segregating or distributing such injurious impurities into forms and positions much less harmful.
ab
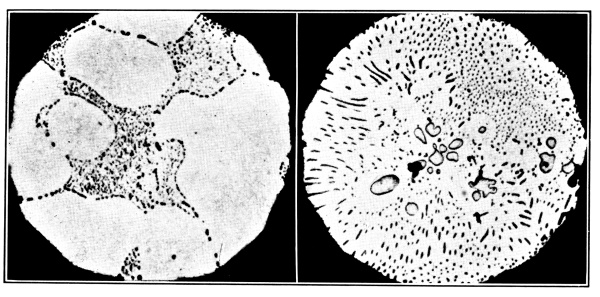
Fig. 6.—Microstructure of Copper containing Oxygen (Heyn).
a. Hypo-eutectic.b. Hyper-eutectic.
Oxygen 0·13 per cent. = 1·16 per cent. Cu2O.
Oxygen 0·53 per cent. = 4·7 per cent. Cu2O.
Arsenic in Copper.—When arsenic and copper are melted together chemical combination occurs, and a series of arsenides is produced; the system, which has been investigated by Friedrich (from whose work the following diagram has been constructed), Hiorns, Bengough & Hill, and others, being one of considerable complexity. With proportions of arsenic such as are usually present in commercial coppers, the compound produced is probably Cu3As (28·3 per cent. of arsenic), which passes into solution in the excess of metal, and on solidification the copper retains this arsenide in solid solution. As in the case of all such solid solutions, the solidification takes place over a range of temperature represented between the liquidus and solidus curves; the purer metal crystallising out first, followed gradually by crystals of copper which become progressively richer and richer in arsenic (still in solid solution). In the case in question, diffusion of the arsenic throughout the crystalline mass proceeds but slowly, and as a result, the metal, as usually obtained in the cast state, shows fringes of such arsenic-rich copper. By annealing, diffusion is greatly assisted, and [Pg 30] the material gradually becomes homogeneous, as is seen on microscopic examination. There appears further to be some decrease of this solubility with fall of temperature when the arsenic is high, leading sometimes to a separation of the arsenide itself at the crystal boundaries.
Antimony appears to form an analogous compound, Cu3Sb, also capable of passing into solid solution in the copper, but to a rather smaller extent than the corresponding arsenide. The fringes are therefore more pronounced, and the decrease of the solubility on further cooling is also more marked.
Bismuth.—The influence of even minute quantities of bismuth on copper is notorious. Bismuth appears to be soluble in liquid copper, but not in the solid metal. In consequence, when copper containing bismuth solidifies, the copper crystals separate first, whilst the liquid bismuth still remains between them, until the metal reaches a temperature of about 268° C.—the melting point of bismuth—when it too solidifies in situ. The presence of such envelopes of very brittle, fusible, and limpid bismuth material explains much of the harmful effect of this impurity. These envelopes are found to consist almost entirely of practically pure bismuth. Oxygen converts the bismuth into a more compactly crystalline oxide, much less fusible and harmful. Arsenical copper tends to the scattering of the bismuth globules among the fringes which are formed during the gradual process of solidification over the range of temperature already indicated, and thus renders this impurity to some extent less dangerous.
Lead behaves in apparently much the same way as bismuth, and the effects produced upon it by the presence of oxygen and arsenic are probably similar.
Selenium and Tellurium probably exist in the form of selenides and tellurides, which are characterised by marked brittleness and fusibility.
Mechanical Properties of Copper.—The mechanical properties of commercial copper are influenced to a vital degree by the conditions associated with working practice, such as composition, previous mechanical and thermal treatment, temperature of working, etc. As has been already indicated, it is the possession by the copper of certain mechanical qualifications which leads to its employment by engineers, and it is, therefore, necessary to consider the influence of the above conditions, when reviewing the mechanical properties of the metal.
Much of the copper employed for general engineering work (apart from electrical and alloying purposes) is of the quality designated as [Pg 31] “tough-pitch” copper. This tough copper generally contains certain impurities which render the metal exceedingly useful for mechanical service, and their presence is, indeed, almost essential in copper intended for such purposes. At the same time, such elements would render it absolutely unfit for the other uses just specified, where purity is practically the first necessity.
The standard works and the papers indicated in the appended list of references should be consulted for details concerning the effect of each circumstance on the several mechanical properties; certain general considerations must, however, be noted here.
Not only should the composition of the metal be carefully considered, but attention must be directed to the actual condition and distribution of each constituent. Owing largely to the difficulties of determining the oxygen contents in copper, and to a want of definite knowledge as to the condition, amount, and effects of the dissolved gases in the metal, the information at present available is not sufficiently concise to allow of a systematised statement being made as to the direct influence of the constituents on the mechanical properties. This is more especially the case since the other attendant circumstances of working practice may react through these to a considerable extent.
Many of the more general results have, however, long been known to engineers from practical working, and these have been placed on record from time to time.
The malleability and ductility of copper are considerable. Cold rolling and hammering causes a reduction in this respect, and the metal is hardened, but the properties are restored by annealing. The annealing effect commences at about 300° C., but proceeds more effectively at higher temperatures, the factors of annealing temperature and duration necessary for annealing being inversely connected. The impurities which influence these properties most adversely are bismuth and tellurium. The effect of other constituents, oxygen per se, sulphur, and iron, in the quantities usually present in commercial copper, is very small. Arsenic and antimony up to 0·4 or 0·5 per cent. have no deleterious effect on the malleability and ductility of copper of the correct pitch, and may even improve the metal when tested in the cold; the hot malleability is, however, somewhat decreased.
The presence of impurities raises the temperature required to bring about the full effects of annealing after the metal has been hardened by mechanical work. This action is probably explained by the interference of the impurities upon the molecular freedom of the metal, [Pg 32] which controls the mechanism of annealing. The conditions, whether reducing or oxidising, during annealing, may exert an important influence on the results.
Hardness.—Pure copper is a comparatively soft metal. It is hardened by mechanical work—the hardness of rolled copper, determined by the Brinell Test, being 74 compared with mild steel as 100—and by the presence of even small quantities of impurities, tin possessing a particularly marked effect in this connection. The worked metal is softened on annealing.
Tensile Strength and Elongation.—The strength of copper, being a property of such practical importance, has been the subject of much extended investigation. The work has, however, been conducted under such a great variety of conditions, many of which have been left unrecorded, that co-ordination of the results is barely possible, and does not allow of establishing on a definite basis the effect of different influences on this property of the metal. Later work, some already published, some still in progress, should eventually allow of more general standardisation than is at present possible. The tensile strength of pure cast copper is 8 to 9 tons per square inch. Mechanical work causes an increase in the value up to 14, or even 16 tons, cold work exerting a still more marked influence; whilst 33 tons and more per square inch has been recorded with cold-drawn fine wire. The elongation varies according to the mechanical work which the metal has undergone; the amount ranges from 35 to 40 per cent. and upwards, measured on a 3-inch length.
Tensile strength is reduced on annealing, but never to so low a degree as that of the cast material, the usual figure being 12 to 14 tons per square inch. The effect of temperature in reducing tensile strength, especially when impurities are present, is important from the industrial point of view. The reduction in strength caused by annealing appears to be considerably smaller in the presence of arsenic and antimony.
Arsenic increases the tensile strength of copper when the metal is of the correct pitch, generally to well over 15 or 16 tons, in the presence of the proportions usually found. Antimony has a similar effect. Some workers state that, within certain limits, the strengthening effect of this element is even more pronounced. Excess of antimony exerts, however, a much more adverse influence than does excess of arsenic. The elongation is increased by the presence of moderate quantities of arsenic.
Oxygen per se, when present in moderate quantity in copper, has but little effect on the tenacity. Bismuth, tellurium, sulphur, and lead [Pg 33] are the impurities which lower the strength, even when present in minute quantities, and especially on heating. Bismuth in the proportion of 0·005 per cent. lowers the malleability and ductility considerably, and recent reports state that 0·02 per cent. bismuth renders copper cold short, that 0·05 per cent. makes it red short, and that 0·005 per cent. is the limit for electrolytic copper which is to be rolled. The deleterious effects of bismuth are, as already explained, to some extent masked by the presence of arsenic and by oxygen.
The strength is increased by the presence of nickel, tin, and zinc in the proportions usually present in the commercial metal; these are, however, generally small.
From the foregoing review, indications will be afforded of the reasons for the choice by engineers of “tough-pitch” copper for much of their work, and the explanation for the 0·3 to 0·5 per cent. arsenic often particularly specified for. The frequent use of arsenical coppers for such purposes as fire-box plates will also be understood, since the arsenic not only improves the mechanical properties of the metal, but ensures the retention of rigidity and strength at the high working temperatures required, to a greater degree than would have been the case had pure copper been employed.
The effect of the above factors on the elastic limit of copper, is also very marked and of much importance, the influence being closely analogous to that produced on the other mechanical properties.
Chemical Properties.—The atomic weight of copper is 63·57. The metal is unchanged in dry air at ordinary temperatures; in the presence of moisture and of carbon dioxide a green coating of basic carbonate is produced. When heated in air, a black scale, consisting of cuprous oxide, Cu2O, is obtained, which is readily detached by quenching and hammering. Water at ordinary temperatures is without effect upon copper; concentrated sulphuric and nitric acids have little action upon it in the cold, but attack it on heating. The best solvent for the metal is dilute nitric acid, which dissolves it very readily. Copper is liable to corrosion when subjected, whilst hot, to the action of chlorine or hydrochloric acid gas; this action has provided an explanation of the corrosion of copper boiler tubes where the coal employed had been exposed to sea water.
Copper is deposited from solution as a dull red, spongy mass, by iron, zinc, or aluminium, but it is more electro-positive than gold or silver, and readily precipitates these metals from solutions of their salts, these effects being extensively made use of in practice. The metal possesses a powerful affinity for sulphur, and this property has very important applications in the smelting processes. [Pg 34]
Copper readily alloys with gold, silver, tin, zinc, and nickel, but not with lead or iron.
Composition and Properties of Metal for Railway and Locomotive Work (p. 20).
Proc. Inst. Mech. Eng., 1893; Dean, p. 139; Blount, p. 164; Watson, p. 168; Gowland, p. 176; Aspinall, p. 193; Tomlinson, p. 182.
Webb, F. W., “Locomotive Fire-box Stays.” Proc. Inst. C.E., 1902.
Milton, J. T., “The Treatment of Copper for Steam Pipes.” Inst. Marine Eng., 1908–9.
Hughes, G., “Non-ferrous Metals in Railway Work.” J. Inst. Metals, Sept. 1911.
Law, E. F, “Alloys.”
Influence of Impurities on the Electrical Conductivity of Copper (p. 24).
Lawrence Addicks, Trans. Amer. Inst. Elect. Eng., 1903, vol. xxii., pp. 695–702; Electro-Chemical Industry, 1902–3, pp. 580–583; Trans. Amer. Inst. Min. Eng., 1906, vol. xxxvi., p. 18.
Walker, A. L., Mineral Industry, 1898, vol. vii., p. 248.
T. Johnson, “Some Features in the Metallurgy of Copper.” Proc. B’ham. Met. Soc., 1906.
Hiorns and Lamb, “Influence of Arsenic and Antimony on Copper.” Journ. Soc. Chem. Ind., May, 1909.
Condition and Influence of Impurities on the Mechanical Properties of Copper (p. 32).
Zeitschrift für Berg. Hutten and Sal. Wesen,
Hampe 1873, xxi., 218; 1876, xxiv., 26
Chemiker Zeitung, 1892, No. 42, p. 16.
Reports, Royal Tech. Testing Institute.
Heyn, E., “Copper and Oxygen.” Charlottenburg, 1900, p. 315.
Metallographist, vol. vi., 1902, p. 48.
Arnold, Engineering, vol. lxi., p. 176. Feb. 7, 1896.
Roberts-Austen, Second Report, Alloys, Research Committee. Proc. Inst. Mech. Eng., April, 1893, p. 114.
Rudeloff, Mittheil. König. Tech. Versuchs. Anstalt., 1894, ii. (b), pp. 292–330; 1898. 16a, pp. 171–219.
Lawrie, Bull. Amer. Inst. Min. Eng., 1909, pp. 857–66. “Bismuth in Wire Bar Copper.”
Johnson, F., “Impurities in Tough Pitch Copper containing Arsenic.” Proc. Inst. of Metals, 1910, vol. iv.; No. 2, p. 163, et seq.
Johnson, F., “The Influence of Impurities on the Properties of Copper.” Metallurgical and Chemical Engineering, Oct. 1910, p. 570. “Annealing of Copper and Diseases of Copper.” Ibid., Feb. 1911, p. 87. “Notes on the Metallurgy of Wrought Copper.” Ibid., August, 1911, p. 396.
See also Standard Specifications for Copper Wire-Bars (recommendations by the Committee of the American Society for Testing Materials). Eng. and Min. Journ., Jan. 20, 1912, p. 181.
Compounds of Copper—Copper Mattes—The Varieties of Commercial Copper—Ores of Copper—Preliminary Treatment of Ores, Sampling.
Compounds of Copper.—From the smelting point of view, the three most important classes of copper compounds are the oxides, the sulphides, and the silicates.
Copper Oxides.—Of the oxides, two are of importance—cuprous oxide, Cu2O, and cupric oxide, CuO—the first-named particularly, having extensive connection with smelting practice.
Cuprous oxide is black when in the massive form, and has a red hematite colour when powdered. It is readily formed by the oxidation of copper, and melts at a red heat without decomposition; further heating in the presence of air produces the cupric oxide which is less fusible. As has been already indicated, cuprous oxide dissolves in the molten metal. It is easily reduced to metallic copper by heating with carbon, the metal being also obtained if the oxide be heated in the presence of reducing gases; it combines readily with silica when heated, yielding fusible silicates.
When cuprous oxide is heated with sulphide of iron, the copper, having a greater affinity for the sulphur than iron possesses, enters into combination with it, forming copper sulphide and iron oxide, and if sufficient silica be present, a silicate of iron slag is produced. When melted with copper sulphide, cuprous oxide yields metallic copper with liberation of sulphur dioxide according to the equation—
Cu2S + 2Cu2O ➡ 6Cu + SO2.
This reaction is a quantitative one, and takes place in the Direct Process of Nicholl and James as operated at Swansea. The excess of either constituent remains unchanged. The reaction is of great importance in the processes of copper extraction, since upon it depends the liberation of metallic copper from the sulphide, both in the old roaster process and in the modern converter operation.
Sulphides of Copper.—Of the sulphides Cu2S and CuS, the former only is of metallurgical importance. It is grey black, brittle, and [Pg 36] crystalline, its melting point is about 1,135° C., and its specific gravity, when cold, about 5·5. Owing to the great affinity of sulphur for copper, this element acts as practically the universal carrier of the metal in smelting work, detaching the copper from all other forms of combination, and collecting it as sulphide, mixed with the sulphides of other metals, particularly that of iron—copper sulphide and iron sulphide alloying in all proportions.
When copper sulphide is melted with an excess of sulphur, it remains unchanged; when melted with copper and subsequently cooled, the sulphide and metal separate as such, although it is believed that small amounts of copper are present in solid solution in the sulphide on solidification, but that they separate from it during a dimorphic change in the material, which occurs at about 103° C. The sulphide reacts with iron with liberation of some metallic copper, and the formation of some iron sulphide which associates itself with the rest of the copper sulphide, forming a matte. This matte is not further affected by iron, so that it is not possible to completely decompose copper sulphide by this means.
When heated in a powdered condition in excess of air, copper sulphide is oxidised, oxides of copper and sulphur being produced. There occur probably several intermediate reactions, and several intermediate products are formed, but the main effect is represented by the equations—
| Cu2S + 2O ➡ 2Cu + SO2 | |
| 2Cu + O ➡ Cu2O | |
which take place simultaneously, the copper represented in the first equation being oxidised spontaneously according to the second, and the resultant is the reaction Cu2S + 3O ➡ Cu2O + SO2.
In the furnace operations, some of the SO2 in the presence of air and oxidisable material, and in contact with the heated brickwork becomes oxidised to SO3, which, interacting with the oxides and sulphides present, combines to form copper sulphate and cupric oxide. At a higher temperature the sulphate is again decomposed to CuO and SO3, some of which passes off and is free to oxidise more sulphide; the rest is decomposed to SO2 and oxygen. These reactions occur during the roasting of charges containing copper sulphides.
Copper Mattes.—On smelting a furnace charge which contains both copper and sulphur, the sulphur appears to have a stronger attraction for the copper than for any of the other metals usually present, and only when this affinity has been [Pg 37] satisfied is the excess sulphur free to combine with other constituents of the charge. The fusible copper sulphide which is thus produced, has the power of mixing completely with any more sulphides which may be present, especially with sulphide of iron.
The fused sulphides resulting from such furnace operations are termed copper-mattes. They may contain from a mere trace to upwards of 80 per cent. of copper, and in ordinary work, sulphide of iron is the other constituent present in the greatest proportion, but sulphides of nickel, silver, zinc, or lead, etc., may also be found, as well as arsenides and antimonides.
These facts relating to the collection of the copper as a constituent of a fused sulphide product, form the basis of modern copper-smelting work. In view of the practical importance of the mixed sulphides, the diagram representing their equilibrium requires notice. A number of workers have studied the question with widely differing results. Röntgen made an exhaustive investigation of the system FeS—Cu2S, and published a very complete diagram of the series, working with FeS and Cu2S in the pure state.
The sulphides as commonly met with, especially in smelting practice, do not however, occur as materials of the composition denoted by the formulæ Cu2S and FeS. The ordinary commercial sulphide of iron corresponds more closely to the impure eutectic of the iron-FeS system, containing about 85 per cent. of FeS and 15 per cent. of iron, and melts at about 970° C., whereas the pure FeS has a melting point of upwards of 1,180° C. At the elevated temperatures of the copper-smelting furnace, pure FeS tends to lose sulphur and to assume the composition of the eutectic. There are, further, good reasons for believing that copper sulphide behaves in a somewhat similar manner, so that the series of sulphides constituting the mattes of practice are not represented by pure materials so well as by a series composed of mixtures of the respective eutectics.
The diagram of this series of industrial sulphides was worked out by Hofman, Caypless, and Harrington, and gives a fair summary of the melting points of the series of mattes. It is reproduced in fig. 8. The temperatures may be supplemented by Gibb’s determinations of 1,121° C. for the 71·7 per cent. copper matte, and 1,098° C. for the 80 per cent. matte.
The problem of the constitution of mattes is, however, a very complex one, and is not yet satisfactory settled. An interesting view was put forward by Gibb and Philp. Mattes corresponding to the formula 5Cu2S . FeS (copper 71·7 per cent.), when examined microscopically, appeared to be homogeneous, and indicated some form of combination between the [Pg 38] sulphides in these proportions. Lower-grade mattes were assumed to consist of this compound substance and excess FeS. Iron sulphide was held to be capable of carrying a certain quantity of copper in solution, and mattes might, therefore, carry this copper, according to the amount of excess FeS which they contained. Within certain limits the lower the grade of the matte—i.e., the more FeS present—the more copper was held in solution, and with a fall of temperature this solubility was lessened, and moss copper was set free in the solid matte.
Deposition of copper may also be accounted for by a variation in the solubility for copper, accompanying the well-marked dimorphic change occurring in FeS at 130° C. whilst another possible cause of the separation of moss copper is the partial decomposition of Cu2S, being effected, as previously indicated, by the free iron of the iron-FeS eutectic which constitutes the iron sulphide component of copper mattes. The whole subject is thus of considerable complexity, and involves questions of thermal and chemical equilibrium.
The appearance, chemical constitution, and physical properties of mattes vary according to the rate of cooling, and are further influenced by the nature and amount of the impurities they contain, and the following statement must be understood to be more or less general:—Usually low-grade mattes (up to 20 per cent. or so of copper) [Pg 39] are more or less stony in fracture, with a bluish-purple colour; as the copper contents increase, a reddening of the colour occurs, and also an increase in the crystalline character and brittleness. Considerable quantities of moss copper are present in these mattes. Beyond 30 per cent. of copper, increased softness and brittleness result, with a darkening towards blue-black in the colour, whilst with the 60 to 70 per cent. mattes the colour becomes in general of a steel-grey hue.
Increase in the copper contents leads to an increase in the density—a matter which has important applications in connection with the economical separation of matte from slag, and the slag-losses in smelting practice.
| The specific gravity of the | 13 | per cent. | copper matte | is about | 4·80. |
| " | 43 | " | " | " | 5·18. |
| " | 60 | " | " | " | 5·42. |
| " | 80 | " | " | " | 5·55. |
| (Gibb and Philp.) | |||||
The density in the fluid state, which is the important condition in smelting, is less than this, and may indeed be somewhat different, owing to changes in the constitution of the material.
Copper Silicate is formed by the action of copper oxide and silica on heating. The silicate is decomposed when heated in the presence of sulphides, resulting in the formation of sulphide of copper and silicate of the second metal, in consequence of the great affinity of copper and sulphur. Upon this fact depends the extraction of copper from various silicate ores, as well as the cleaning of slags high in copper, which are often added to the sulphide charges in the furnace with this object. When heated with iron, the silicate is reduced to metallic copper with the production of silicate of iron; it is also reduced by carbon in the presence of metallic oxides capable of uniting with the silica which is liberated.
The Varieties of Commercial Copper.—The copper employed industrially comes into the market in widely differing forms. Different varieties are named according to the method of manufacture, the uses for which they are intended, the locality in which they are produced, or by special trade names. The most important variety is:—
Electrolytically-refined High-conductivity Copper, which is largely used for electrical work. The methods by which it is produced ensure that most of the impurities inimical to high conductivity have been removed, and the metal is specially free from arsenic, antimony, and bismuth, as well as from silver and gold. As ordinarily produced at the electrolytic refinery, it is in the form of cathode plates, often about [Pg 40] 3 feet × 2 feet 6 inches by ¾ inch thick, weighing 150 to 170 lbs. It is then remelted in order to bring it “up to pitch,” and to give it the necessary mechanical properties, so that it may be transformed at once into the particular form suitable for the electrical purposes intended. Such metal often comes into the market in the form of wire-bar ingots, cakes, or billets, weighing from 70 to 500 lbs. when in bar form, and from 100 to 400 lbs. when in other shapes. Electrolytic copper is also suitable for the manufacture of alloys.
Lake Copper.—The copper ores of the Lake Superior district are particularly pure, and on smelting and furnace-refining yield a metallic product of great purity which also possesses good mechanical properties. It is, therefore, particularly suitable for electrical work. By reason of its satisfactory properties, Lake copper realises prices which usually rule somewhat higher than those of ordinary electrolytic copper as quoted on the New York market.
Best Select Copper.—For the production of copper alloys, such as best brass, etc., it is essential that the copper should be pure. The impurities which are present in ordinary tough copper, and which may be valuable for imparting strength to the material, have a very harmful effect when present in alloys. In the older Welsh process of manufacturing copper, a special method was employed for obtaining metal free from these impurities, especially arsenic and antimony. This was known as the “best selecting” process.
The principle underlying the method was to conduct the furnace operations to the stage at which a small quantity of copper, known as “copper bottoms,” was obtained. The metal so produced has the property of collecting from the rest of the matte-charge in the furnace, the gold, the silver, and the great bulk of the other impurities, owing to its greater solvent power for them. As a result, the greater part of the matte (“white metal”) was left pure, and from this material the copper was extracted by continuing the furnace operations in the usual manner, the resulting product being known as “best select” (B.S.) copper.
The process was later used principally for the extraction of the gold in the charge, rather than for obtaining specially pure copper. The product is essentially a British one, and was largely used for the manufacture of high quality alloys.
“Tough Pitch Copper.”—The operation of “bringing copper up to pitch” has for its object the imparting to the metal of the toughness and mechanical strength required for industrial service. The process resolves itself into the adjustment of the correct proportion of oxygen, the function of which is largely to eliminate the gases from [Pg 41] the copper, or to overcome their deleterious effects, as well as to convert the otherwise more injurious metalloid impurities into a less harmful form.
In modern practice, practically all copper is brought up to pitch, but it is useful to distinguish between tough-pitch furnace-refined copper and tough-pitch electrolytic copper.
The former is the brand to which the general term “tough pitch copper” is best applied, this name having been given to the product from the refining furnaces of the old Welsh and similar processes. Before the converter method was introduced into copper practice, the furnace processes for extracting copper from the ores resulted in the production of a crude “blister” copper, into which several injurious constituents, if originally present in the ore, found their way. The principal impurity was usually arsenic. Although this was also removable by special refining methods, and with some difficulty, it was known, as has been indicated, that when arsenic is present under suitable conditions and in proper proportions, it is capable of imparting considerable strength and rigidity to the metal. Such copper being particularly suited for various engineering and mechanical uses, the arsenic being sometimes even specified for and purposely added—as in fire-box plates and stay bolts, though it is never employed for conductivity work or for the manufacture of alloys if any considerable proportion be present—the metal found a ready market when brought to pitch.
Tough pitch copper may thus vary largely in composition, especially in arsenical contents, up to about the 0·5 per cent. already indicated as being mechanically very useful. The actual process, as used for bringing all classes of metal to pitch, will be described in detail later, it being practically the same whether conducted on furnace-refined metal, converter metal, or on electrolytic copper, as a necessary preliminary to casting into the various forms of ingot in which it is to be marketed.
In preparing the tough metal from crude copper, the more oxidisable impurities (iron, sulphur, etc.) are first removed by a thorough oxidation during or after melting down, this being known as “airing.” The operation oxidises some of the copper, and it is probable that the copper oxide thus formed plays an important part in getting rid of impurities. By the time they have been thoroughly expelled, the metal is considerably over-oxidised. Samples taken at this stage exhibit the following characteristics:—The ingot has a depression down the centre line, the material is very brittle, the fracture is brick-like in texture and purple-red in colour, whilst much copper oxide and oxidule-eutectic are seen on examination under the microscope. This [Pg 42] material is known as Dry Copper; it is merely an intermediate product, and is commercially useless. The excess of oxygen is removed by “poling”—that is, reduction, effected largely by charcoal, as well as by reducing gases—successive samples showing less and less of the characteristics of dry copper. The surface becomes level, the metal exceedingly tough, the fracture fine-grained to silky in texture, and a fine salmon-pink in colour. With satisfactory mechanical properties, the metal has now become tough pitch copper.
If the poling—that is, the reduction of the oxidised constituents of the tough pitch copper—be carried too far, the metal becomes brittle again, being known as over-poled copper. The fracture then tends to become coarse and fibrous, the colour lighter, and the upper surface of the ingot exhibits a ridge. The reasons for these effects have not yet been quite fully explained, but there is no doubt that they arise from the removal of oxygen from the oxygenated constituents, and the withdrawal from the metal of the protecting influence of the cuprous oxide. Such influences are to some extent physical, since they prevent the retention of the reducing gases; partly mechanical, in their effects on the properties of the metal per se, and partly chemical, as the oxide had probably entered into chemical combination with some of the objectionable impurities, producing compounds, in which form they were much less harmful. The removal of this oxygen from the metal breaks down such combinations, leaving the reduced impurity again to exercise its destructive effects on the properties. Over-poled copper, like dry copper, being brittle, is commercially useless as such, and is really an intermediate product, the metal being brought to pitch again by further aëration to make it “dry,” after which it may be poled back to correct pitch. As already stated, the over-poling effects are not due to any intrinsic action of carbon directly on the copper itself.
Summarising, it may be stated that the most important commercial varieties of copper are:—
And, in addition, Lake Copper and some Converter Bars.
A number of unrefined metallic products met with in practice include:—
Converter Bars.—The product from the Bessemer operation on copper mattes. Most converter metal is subsequently electrolytically refined, [Pg 43] but several varieties of Australian and American copper are put on the market direct in this form. Being produced from fairly pure ores, which carry but little silver and gold, the converter metal may be sufficiently pure to render electrolytic refining unnecessary, and too low in gold and silver values to make such an operation profitable.
Cathode Copper is the product from the electrolytic refinery, and is usually remelted, brought up to pitch, and cast into ingots previous to use.
Black Copper is produced by the smelting of oxide ores, and is subsequently refined.
Cement Copper is produced by wet processes, usually by precipitation from copper-bearing solutions by means of iron, the product being a rather impure reddish-brown spongy mass. Many varieties contain arsenic. It requires melting and subsequent refining to adapt it for service.
Blister Copper was the name given to the crude metal from the older type of furnace operations. Such copper contained large quantities of gas, particularly SO2, which, tending to escape at the moment of solidification in the mould, gave a blistered appearance to the surface. It contained 96 to 98 per cent. of metallic copper, and was subsequently refined. The term is generally applied still to all crude copper exhibiting similar features.
Chili Bar is an impure copper imported from Chili for refining. The composition varies, the metal usually containing 96 to 98 per cent. of copper, with indefinite quantities, sometimes small, of undesirable impurities.
Appended is a series of representative analyses of various copper products, compiled from different sources. The composition of such material as tough pitch copper and the various cruder varieties is, however, subject to very great variation.
The Sources of Copper.—Copper ores usually consist of various minerals of copper mixed with those of many other metals, and accompanied by very varied gangue, according to the locality in which they are found.
They are best classified under three groups:—
The most important points to be noted with regard to the distribution of these different classes are that—
(1) Native ores are localised in their occurrence, being chiefly confined to the Lake Superior district. [Pg 44]
TABLE IV.—Analyses of Various Commercial Coppers.
| Copper | Gold | Silver | Lead | ||
|---|---|---|---|---|---|
| 1. | Electrolytic conductivity copper, | 99·89 | nil | nil | nil |
| 2. | Lake copper, | 99·77 | nil | 0·029 | nil |
| 3. | Best select copper, | 99·75 | .. | .. | 0·024 |
| 4. | Tough pitch copper, | 99·41 | .. | .. | 0·070 |
| 99·25 | .. | 0·36 | 0·0103 | ||
| 5. | Copper fire-box plate | ||||
| (ran 500,000 miles, Met. Ry.), | 98·70 | 0·0001 | 0·0346 | 0·4085 | |
Intermediate Products— |
|||||
| Refined converter copper, | 99·25 | .. | 0·36 | 0·0103 | |
| 99·08 | .. | 0·30 | 0·0085 | ||
| Cathode copper, | .. | .. | 0·001 | 0·00054 | |
| Black copper, | 94·39 | .. | 0·11 | 0·19 | |
| 97·70 | .. | 0·2133 | 0·78 | ||
| Cement copper (Spanish), | 51·90 | .. | 2·35 | 1·45 | |
| 76·93 | 0·10 | .. | trace | ||
| Blister copper, | .. | 0·0009 | 0·04 | 0·042 | |
| Chili bar, | 98·60 | .. | .. | trace | |
| Arsenic | Antimony | Bismuth | Iron | ||
|---|---|---|---|---|---|
| 1. | Electrolytic conductivity copper, | 0·016 | trace | nil | 0·042 |
| 2. | Lake copper, | nil | trace | nil | 0·0077 |
| 3. | Best select copper, | 0·025 | trace | 0·011 | 0·10 |
| 4. | Tough pitch copper, | 0·320 | trace | 0·010 | 0·010 |
| 0·0211 | 0·630 | 0·0044 | .. | ||
| 5. | Copper fire-box plate | ||||
| (ran 500,000 miles, Met. Ry.), | 0·3726 | 0·0346 | 0·0360 | 0·0069 | |
Intermediate Products— |
|||||
| Refined converter copper, | 0·0211 | 0·0630 | 0·0044 | .. | |
| 0·0290 | 0·0254 | 0·0035 | trace | ||
| Cathode copper, | 0·00034 | 0·0008 | 0·0003 | .. | |
| Black copper, | trace | .. | .. | .. | |
| 0·052 | 0·2380 | 0·0035 | 0·17 | ||
| Cement copper (Spanish), | 2·95 | 0·50 | 0·95 | 7·00 | |
| 1·32 | 0·02 | .. | 7·6 | ||
| Blister copper, | 0·108 | 0·157 | 0·055 | 0·4 | |
| Chili bar, | 0·100 | trace | nil | 0·009 | |
| Nickel | Tin | Oxygen | Sulphur | ||
|---|---|---|---|---|---|
| 1. | Electrolytic conductivity copper, | 0·006 | .. | 0·008 | nil |
| 2. | Lake copper, | 0·0146 | nil | 0·070 | .. |
| 3. | Best select copper, | 0·061 | .. | 0·143 | .. |
| 4. | Tough pitch copper, | 0·060 | .. | 0·120 | .. |
| .. | .. | 0·284 | .. | ||
| 5. | Copper fire-box plate | ||||
| (ran 500,000 miles, Met. Ry.), | 0·3039 | .. | 0·0181 | 0·0064 | |
Intermediate Products— |
|||||
| Refined converter copper, | .. | .. | 0·284 | .. | |
| .. | .. | 0·12 | 0·01 | ||
| Cathode copper, | .. | .. | 0·005 | .. | |
| Black copper, | 2·04 | 0·07 | .. | 0·80 | |
| .. | .. | .. | 0·796 | ||
| Cement copper (Spanish), | .. | .. | 16·00 | 5·10 | |
| .. | .. | .. | 0·48 | ||
| Blister copper, | 0·0–0·2 | 0·0–0·5 | .. | 0·112 | |
| Chili bar, | .. | .. | .. | 0·909 | |
[Pg 45] (2) Sulphide ores supply the bulk of the world’s copper, constituting upwards of 80 per cent. of the total.
(3) The oxidised ores are found in most copper districts, though usually to only a limited extent. They are often gossan deposits produced by weathering or by decomposition of sulphides, hence are generally found nearer the surface, changing to sulphide with depth. The supply of copper from oxidised ores, which was at one time very large, is decreasing rapidly, and the greater proportion of the copper now obtained from them comes from the more recently developed deposits, of which those at Tanganyika afford an example.
More than 200 minerals which contain copper are known, but most of them are unimportant from the smelting point of view. The characteristics of the more noteworthy may be fully studied from text-books of economic mineralogy.
Copper Ores—Native Copper.—Occurs extensively in the Lake Superior district of Michigan, in Precambrian rocks, sparingly in New Mexico and China, but seldom anywhere else in workable quantities by itself. Copper barilla or copper sand, an impure native metal from Chili, was formerly of importance. Native copper constitutes about 20 per cent. of the North American supply. It yields metal of exceptional purity, and the brands of Lake copper reach a very high standard, both as regards electrical and mechanical properties. A still purer variety is the native metal from Yunnan, China.
The Lake Superior copper occurs in three formations:—
(a) Vein deposits, from which the enormous masses of copper are taken out.
(b) Copper-bearing ash beds, of amygdaloidal diabase. Chief mine, Quincy.
(c) Beds of conglomerate in which the cementing material consists partly of copper. This last class of deposits yields three-quarters of the Lake copper supply. Their average copper content is 2·9 per cent. The chief mines are the Calumet and Hecla, the Tamarack and the Atlantic, all situated on one ore chute measuring 3 miles in length, and worked to a depth of 4,000 feet.
Sulphide Ores: Chalcopyrite (Copper Pyrites) is by far the most widely distributed ore of copper, and furnishes the greater proportion of the world’s supply.
The formula when pure is Cu2S. Fe2S3 (Cu 34·4, Fe 30·5, and S 35·1 per cent.), but usually the ore is not in this condition, being mechanically mixed with large quantities of iron pyrites, and very often with pyrrhotite. It occurs principally in the older crystalline rocks, often in bedded veins. [Pg 46]
The value of copper veins below the limit of surface decomposition is nearly always due to chalcopyrite. Silver and gold are often carried, as well as other metals. It occurs extensively in Montana, Arizona, Tennessee, Canada, Chili, Japan, Spain, Cornwall, etc.
Chalcopyrite ores vary considerably in copper contents; thus Tennessee ores contain about 2·5 per cent. of copper, Montana ores 5 to 5½ per cent. (with gold and silver valued at about £11 per ton of copper), whilst the Arizona ores vary, being often rich.
Chalcocite (also known as copper glance or redruthite) is much less important. The copper contents are 79·8 per cent. when pure, but such a condition is rare, although the ore seldom contains less than 50 per cent. of copper. Below this proportion it often tends to pass into bornite, and then to chalcopyrite. It is found in Montana, is an important ore in Arizona (Clifton district), and occurs also in Cornwall.
Other important sulphides include:—
Bornite (Erubescite, Peacock copper ore), 3Cu2S. Fe2S3, occurring in Cornwall, which passes with depth into chalcopyrite.
Tetrahedrite (Fahl ore), a very complex sulphide of copper, iron, lead, zinc, with arsenic, etc. It is often rich, and carries silver values.
Oxidised Ores.—The most important of the oxidised ores are—
Malachite, CuCO3. Cu(OH)2, containing, when pure, 57·3 per cent. copper (73·7 CuO); is widely distributed, but usually occurs as such in non-paying quantities except in a few particular localities. It is found in the upper parts of the veins. Whenever found with sulphide ores, it is an extremely useful material to mix in the charge, as it supplies oxygen as well as copper. Malachite is still an important source of the metal in Mexico, Chili, and Bolivia, though not quite so much so as formerly, whilst it is specially important in the Tanganyika (Katanga) deposits, of which it constitutes the greater portion so far developed.
Cuprite, Cu2O, contains 88·8 per cent. copper, when pure. It is widely distributed, but is never found by itself in paying deposits, though in the early days of mining and smelting it was an important source of metal, since it was easily reduced, and consequently was cheaply worked.
Melaconite, CuO, contains 79·8 per cent. copper, when pure; is fairly widely distributed, although hardly ever in sufficient quantity to pay. In one or two localities, however—viz., Tennessee, North Carolina, and Virginia—it was formerly an important source of the metal. The deposits were at first very promising, as they consisted largely of very rich melaconite ore; this was however, quickly worked out, the [Pg 47] ordinary heavy chalcopyrite with 2·5 per cent. copper being struck below.
Other oxidised ores include—
Azurite, 2CuCO3. Cu(OH)2, and Atacamite, CuCl3. 3Cu(OH)2, from Chili.
In modern work, the chief ore smelted is impure chalcopyrite. Carbonate and oxidised ores, when they can be obtained, are mixed with it, increasing the concentration and shortening the process; except under certain special circumstances.
Preliminary Treatment of Ores.—The treatment of ores preparatory to smelting includes the processes of sampling, wet concentration, agglomeration of fines, and roasting.
Sampling.—Since sampling is not a part of the extraction process proper, in copper smelting, it will be convenient to deal with the subject separately here.
It is important that ores and all other products entering or leaving the works, as well as many of the intermediate products of the various operations, should be properly sampled and assayed. Great attention is paid to this point at the best organised smelters, since only by this means can the work of the plant and of its several departments be properly checked and controlled. Each works has its own special method of taking samples from the stocks, the Anaconda practice, for example, being to pass the whole of the first-class ore, amounting in quantity to 25,000 tons per month, through the sampling mill, whilst of the poorer, second-class, ore for concentrating, every fifth car-load is sampled.
There are many different types of sampling plant, and the methods employed vary also, but the principle is much the same in each case—namely, to use some automatic device which cuts out and deflects a certain proportion of the stream of ore on its course through the mill;—the deflected portion being crushed finer, and a part of it again cut out and deflected; repeating the operation in this way three or four times.
The sampling process and plant at Anaconda is so representative of the best practice, that it may be reviewed in brief, as an example. [Pg 48]
[Pg 49] The Anaconda Sampling Plant is entirely automatic in its action. The mill is built in two sections, each of which treats 1,800 tons daily. Each section consists of a set of four sampling machines with intermediate crushers. The ore goes from the bin to a Blake crusher, breaking to 3-inch to 4-inch size; the crushed ore is elevated and fed down a chute to the first sample cutter, which takes out one-fifth (400 lbs. per ton) as a sample, and deflects the rest down another chute. The sample is crushed further in a Blake crusher, and passes a second sample cutter (rather smaller in size), which again takes out one-fifth (80 lbs. for every original ton of ore), and rejects the rest down the “rejects-chute.” The sample is now crushed in rolls, a third cut of one-fifth (16 lbs. of the ton) taken as before, the rest rejected. The sample passes to a final set of crushing rolls, and the last cut of one-fifth is taken. Hence each ton of ore is represented eventually by 3·2 lbs. of sample.
The sample cutter employed is of the Brunton form. It consists essentially of a curved boat of 120° arc, which rotates to and fro on a central spindle. The top is open; one side has one hole cut in, the other has two, the area of the latter being together four times that of the single one, so that the falling stream is cut continually, and one-fifth is deflected to one side, falling down a chute to the next crusher, whilst the other four-fifths fall from the other side to the rejects-chute.
The above description is quite general, several details for certain classes of ore having been omitted, but it gives a fair idea of the general principles underlying such work.
The final sample, say 3,200 lbs. per 1,000 tons of ore, is mixed on an iron plate on the floor, quartered several times by a Brunton shovel, and the chosen sample then ground in an Englehardt mill (small Gates’ crusher with two discharges). The material is passed through a 1-foot riddle of 100 mesh wire cloth, the very small quantity of coarser stuff remaining, is bucked down and added, and the whole is then thoroughly mixed in a canister of 1 foot side gripped at opposite corners, and rotated mechanically. [Pg 50]
Constitution of Copper Mattes.
Keller. Mineral Industry, vol. ix., 1900, p. 240. “Elimination of Impurities from Copper Mattes.”
Röntgen. Metallurgie, vol. iii., 1906, p. 479.
Hofman, Caypless, and Harrington. Trans. Amer. Inst. Min. Eng., vol. xxxviii., 1908, pp. 142–153.
Gibb and Philp. Trans. Amer. Inst. Min. Eng., vol. xxxvi., 1906, p. 665.
Heyn and Bauer. Metallurgie, vol. iii., 1906, p. 84.
Fulton and Goodner. Trans. Amer. Inst. Min. Eng., vol. xxxix., 1908, pp. 584–620.
Refining of Copper.
H. O. Hofman, R. Hayden, and H. B. Hallowell, “A Study in the Refining and Overpoling of Electrolytic Copper.” Trans. Inst. Amer. Min. Eng.
Hofman, Green, and Yerxa, “A Laboratory Study of the Stages in Refining Copper.” Trans. Amer. Inst. Min. Eng., 1904, vol. xxxiv., pp. 671–95.
Stahl, “Ueber Raffination, Analyse and Eigenschaften des Kupfers.” Berg. and Hüttenmännische Zeitung, 1889, vol. xlviii., pp. 323–4; 1890, vol. xlix., p. 399; 1893, vol. lii., p. 19; 1901, vol. lx., pp. 77–79.
Keller. Mineral Industry, vol. vii., p. 245, et seq.
Sampling.
D. W. Brunton, “Modern Practice in Ore Sampling.” Mining and Scient. Press, Oct. 30, 1909. “Theory and Practice of Ore Sampling.” Trans. Amer. Inst. Min. Eng., vol. xxv., p. 826.
Modern Copper Smelting Practice—Preliminary Treatment of Ores: Concentration, Briquetting, Sintering—The Principles of Copper Smelting—Roasting.
Modern Copper Smelting Practice.—Until recently, modern smelting practice has been understood to involve the production of a matte containing from 40 to 50 per cent. of copper, which is then bessemerised.
There are however proceeding at present (owing to the successful working of basic-lined converters) developments which indicate that such practice may, within a few years, be modified very considerably in the direction of the converter treatment of lower-grade mattes. Until such operations become successfully established and generally adopted, the production and subsequent bessemerising of 40 to 50 per cent. matte will be here dealt with as constituting modern practice; particularly since, generally speaking, the principles involved are equally applicable to the modified methods now being developed.
Preliminary Treatment of the Ore.—The factors which have to be considered in drawing up a scheme of treatment for the supply of ores shipped to a smelter are exceedingly numerous, and will be discussed in due order. There are no hard and fast principles which determine such schemes, yet a number of considerations must be noted concerning the treatment preliminary to the actual smelting of the ores.
Such preliminary treatment may include—
A. Concentration or Wet Dressing.—In treating the ores of copper, it may be noted that in general—
Native Ores, unless very massive, are usually dressed in a special manner peculiar to themselves—e.g., stamp-milling.
Oxide Ores are rarely wet-dressed. They present much difficulty in treatment on account of their comparatively low density, which makes efficient wet concentration almost impossible, whilst heavy losses in the tailings generally accompany such operations. [Pg 52]
Sulphide Ores.—No definite rules can be laid down as to whether the ore should be wet-dressed or not; the treatment depends altogether on attendant circumstances, such as—(a) the character of the ore, (b) the concentration of the copper desired in the first smelting operation, and (c) the smelting method and furnaces adopted.
Wet concentration is only profitable when the copper ore is of low grade, and then only under suitable conditions. Thus the low tenor may be due to admixture with much gangue or with other sulphides, or both. A massive low-grade pyritic ore carrying but little gangue is not suitable for such treatment, since the mixed sulphides are not separated from one another by wet dressing, and consequently but little enrichment of the copper in the dressed product would be possible; apart altogether from other considerations. Such is the case, for instance, with the Tennessee ores carrying about 2·0 per cent. of copper and only 25 to 35 per cent. of gangue.
An ore with a self-fluxing or almost self-fluxing gangue might allow of its copper being concentrated more cheaply and conveniently by direct smelting than by wet dressing, this depending, of course, on the local conditions.
In other cases a balance has to be struck as to whether the circumstances are more favourable for removing the excess of gangue by means of crushing and treatment in a stream of water, or by slagging it off in a furnace with the addition of suitable fluxes. In many cases, with low-grade ores, the former treatment is the cheaper.
The case of the low-grade ores of the Butte, Montana, district, affords a good example of these considerations. This ore contains 5 to 5½ per cent. copper, with a large quantity of highly siliceous gangue. It was found that the purchase and carriage of sufficient flux, and the cost of carrying out this fluxing operation was so expensive that it was cheaper to build a concentrator and smelter at Anaconda, 30 miles away—in a locality where a suitable water supply was available for the dressing—and to convey the ore this distance in order to concentrate it by a wet method. The dressed ore assays 9 to 10 per cent. of copper.
It is important to note that the process of wet dressing involves crushing the ore, and yields the product in a more or less finely divided form. Most copper sulphide minerals are exceedingly brittle, and break up to a very small size on crushing for concentration, so that the copper concentrates usually include a large quantity of fine material.
There are two general types of furnace available for smelting—reverberatory furnaces and blast furnaces—and the questions [Pg 53] of the desirability and of the degree of crushing and concentration depend to a large extent on the plant and furnaces adopted or proposed.
Blast-furnace treatment has hitherto often been considered the most economical process for smelting copper ores, especially with regard to fuel costs, but for many reasons it is not a convenient or efficient furnace for the direct treatment of fine material. When it is desired to employ the blast furnace, it is necessary to make up charges consisting, to as great an extent as possible, of coarse material. In consequence, when concentrating ores with a view to subsequent blast-furnace treatment, the degree of crushing and dressing has to be modified with these factors in view; otherwise a further preliminary manipulation of the fine concentrates that are produced is rendered necessary. Such modified dressing schemes involve a maximum of coarse breaking and screening, the crushing and separating stages being thus very gradual, and the units in the plant are multiplied, whilst the process is rendered complex in consequence. With the greatest care, moreover, large quantities of fines are bound to be produced, and have to be dealt with by some means other than immediate blast-furnace treatment.
Dressing schemes and plant for sulphide copper ores are thus often complicated, particularly for the recovery of the values from the finer material, and cannot be discussed at any length here. Reference should be made to Richards or other standard works on the subject.
As representative of wet-dressing practice, the Anaconda scheme may be noted, as summarised below.
There are eight mills, each treating 1,000 tons of ore per day, and conducting the—
The muddy water goes to enormous settling ponds, where the slime settles down, gradually drains, and dries, and it is afterwards used for various purposes during the smelting operations; being dug out in the form of a fine clay. A new form of centrifugal apparatus (the Peck) [Pg 54] is now being installed for the separation of this material. The subsequent treatment of the products from the concentrating operation is indicated in the diagram (fig. 12), from which it will be seen that the—
B. Agglomeration of Fines.—It has just been seen that the wet concentration of ores (considered advisable in a large number of cases) results in the production of a considerable quantity of fine concentrate, a form of material not well suited for immediate blast-furnace treatment.
In addition, smelters often receive considerable amounts of fines in the smelting-ore supply, which it is not unusual to screen out and to treat separately from the coarser materials.[4]
The alternatives for the treatment of fines, and more particularly of fine concentrate, include smelting in reverberatory furnaces (usually after roasting); blowing into the converter (a new process still in the experimental stage); and blast-furnace treatment after suitable preparation.
Blast furnaces have many advantages which lead to their extended use in copper smelting practice, but one important feature, which also applies to the smelting of other metals, has always to be borne in mind in this connection—viz., that material in a finely divided state cannot be treated directly in a blast furnace without heavy losses, and the working of the furnace on such charges is not efficient.
No material less than ¼ to ⅜ inch in size, especially when in the form of sulphides, should be fed as such into a modern blast furnace. Fines in the furnace lead to—
and their presence is often the cause of much trouble at many of the modern smelters. The agglomerating of the fines is, therefore, a very important preliminary in any scheme of treatment involving the employment of the blast furnace on such material. Agglomerating is usually performed by one of two methods—(1) briquetting, (2) sintering. Of these, briquetting has hitherto been in very general use, but several advantages connected with the sintering process and the resulting product are leading to its adoption with much success in several localities, and attracting for it considerable attention at present. [Pg 56]
(a) Briquetting.—Among the advantages of briquetting is the fact that it utilises large quantities of the copper-bearing slime produced at the concentrating plant, this material often possessing good binding properties which render it very suitable for briquette-making.
[Pg 57] The type of plant in use at different smelters varies considerably, the method adopted being either the stamping out of the briquettes, or by the application of steady pressure, the production of bars which are then cut up to convenient size.
The constituents used depend naturally on the materials available at the smelter, briquettes, both with lime and without, being made.
The Briquetting Plant at Anaconda.—The operation of this plant affords a good example of the process. Its working is very successful in using up much fine concentrate, as well as the slime from the ponds, which acts as binding material and at the same time supplies copper. Briquette, indeed, constitutes one of the biggest items of the charge for the Anaconda blast furnaces. There are four Chambers’ machines in use, making 840 tons of briquettes daily. The briquettes consist of slime, fine first-class ore screenings (< ⅜-inch size), fine concentrate from the dressing plant, and coke (which is recovered from the reverberatory furnace gratings). The quantities used daily are somewhat as follows, though they are naturally subject to some variation, depending on supplies:—
| Slime, | 500 | tons. |
| First-class ore screenings, | 300 | " |
| Fine concentrate, | 200 | " |
| Coke, | 70 | " |
and the composition of the briquettes is about—
| Copper, | 5·0 | per cent. |
| Ferrous Oxide, | 16 | " |
| Silica, | 45 to 50 | " |
| Sulphur, | 15 | " |
| Lime, | 0·7 | " |
| Moisture, | 15·0 | " |
| Coke, | 5·0 | " |
The different materials are stored in bins, and fed through doors to conveyors, which discharge on to an elevator leading to a divided hopper, each division of which feeds a pug-mill. The pug mills are long troughs in which inter-moving bladed spindles rotate, churning up the materials; the mixing being assisted by a water supply from above. The mixture passes down a chute to one end of an auger machine, from which it issues, through a steel ring, in the form of a continuous slab, 6 inches × 4 inches in section, to a cutter 10 feet distant, which slices off bricks 10 inches long, each of which weighs about 10 lbs. The [Pg 58] bricks pass to a traveller, thence by another to feed bins. The briquettes are not dried, but are used just as made with 15 per cent. of moisture, and are generally the last item of the charge to be added on the car. They crumble slightly, but are sufficiently strong to stand the handling during charging.
Many similar methods, including hand processes, are employed.
(b) Sintering Processes.—This method of treating fines involves roasting reactions, as well as the mechanical process of agglomerating. Whilst it thus furthers the concentration obtained in the subsequent furnace operation, since it eliminates some sulphur, it also utilises the fuel value of the fines, and yields a product which works well in the blast furnace. Several processes have been introduced, and the M‘Murty-Rogers method installed at Wallaroo, S. Australia, illustrates very well the principles upon which this class of treatment depends. It is a sintering and roasting process similar in type to the Huntingdon-Heberlein method for lead smelting, but lime is not used as a rule. It is employed primarily for fine concentrates which are somewhat siliceous.
Charge.—Must contain 15 to 35 per cent. silica, and 15 to 25 per cent. sulphur.
Pots.—8 feet 6 inches in diameter, when used for ore, and 4 feet 6 inches deep; with vertical sides. There is a false grate 10 inches above the bottom, pierced with ⅝-inch holes.
Blast.—1,000 cubic feet per minute at 13 to 20 ozs. pressure per square inch.
Capacity, 8 to 10 tons. Time, 8 to 10 hours.
Method.—Cover the grate with a layer of roasted material, light small fire of wood, blow, and gradually charge in the ore whilst the blast is on. Lime is unnecessary, but water is essential in the process, and the ore must be very wet; 6 to 9 per cent. water being used for ore charges, and 3 to 4 per cent. with rich mattes, otherwise working is not uniform, and the losses by dusting are great. With the requisite quantity of water present, the working is regular and uniform, there is little dust, and the roasting is efficiently performed.
Products.—If ore is charged, a sintered mass of matte and ferrous silicate results; if poor matte is used, the product is a rich matte and ferrous silicate; and if rich matte is used, metallic copper and ferrous silicate are obtained. At the end of the blow the charge is tipped out and fed into the blast furnace.
Costs.—The method as employed at Wallaroo to treat 400 to 500 tons of material per week, operated at a cost of 3s. 6d. per ton, or about 1s. more per ton than for ordinary roasting. [Pg 59]
Though this particular process is only, to the author’s knowledge, employed at a few smelters, sintering or blast-roasting methods on the same principle have been introduced at several other works, and their adoption promises to lead to very successful results, being particularly suited for the class of material indicated above. The advantages claimed for the process are that—
(a) It saves heavy mechanical losses, such as those of the dust resulting from calcining operations and from the charging of hot calcines into reverberatory furnaces.
(b) It gives a product suitable for blast-furnace smelting—often the cheapest and most convenient method of working.
(c) It results in efficient roasting and good reduction of sulphur, yields the product in an advantageous form for subsequent smelting, and promotes a satisfactory removal of impurities in the slag.
In addition, the process offers the possibility in the future of being so modified as to leave in the adequately compacted products so much sulphide that their fuel values can be realised in the blast furnace. In other words, after the preliminary sintering process, to smelt the (fine) sulphide-concentrates pyritically in the blast furnace.
Of the more recent types of machine for conducting the process of sintering, that of Dwight and Lloyd is in operation at several smelters. The moistened ore falls on to an endless chain conveyor, composed of separate grids carried on wheels. The conveyor carries the ore through the flame from a small furnace which starts its ignition, and it is then drawn over a long suction chamber where air is sucked through the hot mass, thus effectually roasting and sintering it. The chamber has special devices which ensure the drawing in of the air through the charge only, and so prevent inward leakage (see Fig. 16).
The sintered cakes are finally discharged automatically into cars. Details regarding the machine vary at different smelters; at one works the length is 30 feet, the rate of travel 8 inches per minute, and the vacuum in the suction chamber 6 ozs.
The size of the particles should not exceed ¼ inch, and not more than 25 per cent. of the charge should be so large. Some 3 to 5 per cent. tends to pass through the grids, and so be drawn into the suction chamber; this is cleared out at intervals through special doors. Water is necessary, and from 6 to 10 per cent. must be employed in uniformly moistening the charge, which, by the addition of suitable fluxes, is often made of such proportions that in subsequent blast-furnace smelting a satisfactory slag is produced without further additions. The sulphur reduction by the process is very considerable. [Pg 60]
[Pg 61] Such blast-roasting methods, with suitable modifications, promise to assume considerable importance in the developments of modern smelting practice.
c. Roasting.—Roasting is often a very important preliminary stage in the scheme of treatment of copper ores. It was formerly considered an essential operation in smelting processes for sulphide ores, the material being crushed and concentrated largely with a view to such subsequent treatment. This is not the practice in modern smelting. Roasting is now only conducted where the necessity for it arises, as in the case where wet dressing, having been considered advisable, has resulted in the production of large amounts of fine concentrate, and where reverberatory furnaces are installed for the smelting of this material. Preliminary roasting of the concentrates then conduces to the production of a matte of converter grade in one smelting operation.
The Principles of Copper Smelting.—Copper extraction from sulphide ores is essentially an oxidation process, the iron and sulphur being oxidised and the oxide of iron slagged away. All such smelting processes, both the older and the more modern ones, are based on this fact, and underlying all of them are certain fundamental principles which it is essential to keep in mind in considering every phase of the subject.
These may be summarised as follows:—
(1) In the melting down of a furnace charge, the copper has first claim on any sulphur which may be present.
(2) Only such sulphur as remains in excess after the copper has been satisfied, is free to combine with other constituents of the charge.
These fundamental principles can best be illustrated by following the reactions during the smelting of a typical charge. Thus—
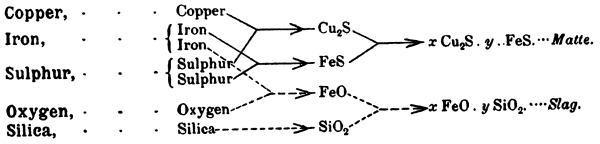
The copper takes up sufficient sulphur to form Cu2S; the remaining sulphur combines with any iron which is available, forming FeS. These two sulphides, dissolving in all proportions, constitute the matte product of smelting. [Pg 62]
The iron in excess of that required by the sulphur becomes oxidised, and the resulting oxide combines with silica in the charge, forming the silicate slag of the smelting operation.[5]
It will thus be apparent that, in general, the larger the amount of sulphur present in a furnace charge, the more FeS will there be in the matte after melting, and the smaller will be the proportion of copper. In consequence, the grade of the matte will be lower.
The proportion of sulphur in the charge thus controls the concentration of the copper by the smelting operation, and, in order to effect the desired concentration, oxygen is required in order to burn off sulphur and to oxidise iron. There are two general methods of supplying this necessary oxygen.
(1) By a preliminary oxidation of the charge outside the smelting furnace—Roasting.
(2) By oxidation inside the smelting furnace itself—The pyritic principle (to be considered later).
Modern Practice as regards Roasting.—In modern copper smelting, the tendency is to do away with roasting as much as possible.
Objections to Roasting.—(1) Expense involved by a separate preliminary process. This includes
(2) Heavy mechanical and other losses during the process.
(3) Loss of the fuel value of the iron and sulphur for smelting.
(4) Necessity, in the majority of cases, of having the ore in a fine state of division in order to conduct efficient roasting, thus militating against its subsequent use in the blast furnace, unless the product receives preliminary agglomeration.
Thus at Tennessee, the cost of roasting was about 40 cents, or 1s. 8d. per ton of ore (equivalent to ½d. on every pound of copper produced). The cost for the year 1903 amounted to £19,000, employing 170 men out of a total staff of 900 at mines and smelters. The conditions for roasting were here exceptionally favourable. The closing of the roast-yards set at liberty £34,000, which had been tied up in this manner.
Advantages of Roasting.—Illustrative of the conditions under [Pg 63] which roasting is advantageously conducted in modern practice, the case of the Butte second-class ores may be quoted.
These ores contain about 5 per cent. of copper in the form of sulphides, finely disseminated through large quantities of siliceous gangue. Direct smelting in a blast furnace would not yield a matte of the desired “converter” grade, except at very heavy expense and difficulty. The ore is, therefore, wet-dressed up to 9 to 10 per cent. copper, and the coarse concentrates now help to yield a good matte, when smelted in the blast furnace. By the wet-dressing treatment, however, a considerable quantity of fine material is unavoidably produced, for which the most convenient treatment in such large quantities, under prevailing conditions, is in the reverberatory furnace. The atmosphere of this type of furnace being to a great extent neutral, the charge would tend simply to melt down without very much reduction of sulphur, resulting in the production of very low-grade matte. Roasting of these fine concentrates is, therefore, desirable for reducing the sulphur to such an extent as will yield a high-grade converter matte.[6] Roasting being thus often advisable as a preliminary, its inclusion in a smelting scheme under suitable conditions entails the following advantages over the direct reverberatory treatment of unroasted ores:—
(1) It ensures satisfactory concentration on smelting.
(2) It leaves reverberatory furnace smelting practically a remelting operation, and so affords exact control of the concentration effected.
(3) The roaster gases may be utilised for making acid.
In modern practice the work of the reverberatory plant is controlled at the roasters. The reverberatory foreman smelts whatever mixture is sent from the roasting plant, and if the grade of the resulting matte is not satisfactory, it is in the roasting operations that the required change is made for the correct adjustment of the sulphur and for controlling the consequent tenor of the matte.
The Reactions of Roasting.—The operation of roasting is the exposing of a substance to the effects of heat and air, in order to oxidise it, and to render it more suitable for subsequent smelting operations.[7]
In the case of the ordinary sulphide copper ores, roasting not only (a) reduces sulphur, and so ensures good concentration on smelting, but (b) by oxidising the iron, provides a ready flux for siliceous [Pg 64] gangues. The more important reactions occurring to the usual constituents of the copper ores which are roasted, may be summarised as follows:—
Iron Pyrites.—First loses free sulphur at a low temperature: it is generally assumed that FeS is left, but the residual sulphide rarely attains this composition—
FeS2 ➡ FeS + S.
Iron Sulphide.—Sulphur has a great affinity for oxygen, to form SO2 and it may be assumed that this reaction first takes place thus—
FeS + O2 ➡ (Fe) + SO2 (i.)
The iron is however instantly oxidised by the excess oxygen always present—
(Fe) + O ➡ FeO (?) (ii.)
Or, combining (i.) and (ii.)—
FeS + 3,O ➡ FeO + SO2.
This sulphur oxidation is an important source of heat, and in the early stages of roasting, sulphur is seen burning with the familiar blue flame, and the mass becomes red hot; stirring being required to prevent the material from sintering by the heat generated within itself.
The oxidation of the iron generally proceeds further, yielding higher and more stable oxides—
| 2FeO + O ➡ Fe2O3. | |
| 3FeO + O ➡ Fe3O4. | |
The SO2 in the presence of oxygen and in contact with strongly heated material further tends to form SO3, which is a powerful oxidising agent, and plays a considerable part in the various oxidising reactions which occur.
Pyrrhottite behaves in much the same way; it may be regarded as consisting of xFeS + a little extra sulphur. It does not roast quite so easily as pyrites, partly on account of physical characteristics, and partly because, in the case of pyrites, the greater amount of excess sulphur which is first driven off, tends to leave the mass more porous and so assists oxidation.
Copper Sulphide.—Its characteristics on oxidation have already been indicated in Lecture III., p. 36. It melts easily, often at roasting temperatures, hence careful heating and attention are required when much is present. [Pg 65]
The reactions are probably analogous to those of FeS oxidation, in the primary oxidation of the sulphur and the instantaneous oxidation of the nascent copper—
| Cu2S + O2 ➡ (2Cu) + SO2 | |
| (2Cu) + O ➡ Cu2O, | |
thus
Cu2S + 3.O ➡ Cu2O + SO2;
this being accompanied by simultaneous action of the following nature:—
| Cu2O + SO2 + 2,O ➡ 2CuO + SO3 |
| CuO + SO3 ➡ CuSO4 |
| CuSO4 + Cu2O ➡ 3CuO + SO2. |
In addition to the tendency to melt, copper sulphide roasts less perfectly than the FeS, usually yielding oxides which are accompanied by small quantities of sulphate.
Chalcopyrite is the commonest copper ore, and the material most frequently subjected to roasting in copper smelting practice.
Consisting of Cu2S. Fe2S3, and accompanied usually by a large excess of FeS2, it behaves very much like a mixture of these sulphides when treated in the roaster furnace, hence the reactions on roasting follow on the lines just indicated.
In practice the roasting is never carried to such a degree that all the sulphur is eliminated, since it is essential to retain some sulphur in order to collect the copper in the form of matte, and also because the time, and the cost of the fuel required to roast all of it off, would be prohibitive. Consequently, the products from the roasting of chalcopyrite consist principally of oxides of iron and copper, together with a certain amount of copper sulphate, very little iron sulphate, and some undecomposed sulphides.
The actual form in which the sulphur is present at the end of the roasting operation is not usually of very special importance in practice, especially where the previous experience with the roasted material determines the extent to which the roasting is conducted, since the greater part of the sulphur eventually produces the sulphide and constitutes the matte, on smelting the roasted charge; although some is also eliminated as SO2 by interaction with oxides. In modern roasting practice, therefore, all that is usually required is to roast the ore down to, say, 5 per cent., 6 per cent., 8 per cent., or whatever proportion of sulphur is necessary to yield the required grade of converter-matte in the reverberatories, as judged by previous [Pg 66] experience of the furnace plant and working. Much SO2 is evolved during the roasting, though it is usually largely diluted with nitrogen from the air used up.
Other Foreign Constituents of Copper Ores—Zinc Sulphide.—ZnS is sometimes present. Some remains unchanged on roasting, as the heat in ordinary practice is not great enough to thoroughly decompose it. Some oxide and some sulphate are also produced.
2ZnS + 7,O ➡ ZnO + ZnSO4 + SO2
is suggested by Peters as a probable reaction occurring to this material under roasting conditions.
Lead Sulphide is also occasionally present with copper ores. It melts readily, and is not entirely decomposed at the temperatures employed for the roasting of copper ores. The reactions on oxidation are largely analogous to those for other sulphides.
| PbS + O2 ➡ Pb + SO2 | |
| Pb + O ➡ PbO | |
or,
PbS + 3.O ➡ PbO + SO2.
Also,
2PbO + SO3 ➡ PbSO4.PbO (basic sulphate).
Arsenides are partly left as the corresponding oxides, whilst some As4O6 is evolved, and some basic arsenate generally remains.
Favourable Conditions for Successful Roasting.
(a) The sulphide should be in a finely divided form, so as to ensure good contact with the air.
(b) The air should be supplied in a gentle current, so as to continually provide fresh oxygen, and sweep away the inert gases which are produced.
(c) The ore should be heated to a dull red heat, which is a condition favourable for commencing the ignition and reactions. The temperature should, of course, be well below a melting heat (Peters).
The Apparatus for Roasting depends to some extent on the class of material to be dealt with, which may be in the form of either (a) lump ores, or (b) fine ores.
(a) Roasting of Lump Ores.—In modern copper-smelting work, the practice of roasting lump ores is practically obsolete. The conditions under which its use might still be justified are those associated with newer mining districts, where rapid concentration of heavy sulphide ore into matte is required, before the time is ripe for [Pg 67] smelting the material pyritically, and where further, it is desired to employ the blast furnace for the smelting operations under these circumstances.
The advantages possessed by the method are—
(1) No preliminary crushing is required.
(2) The product is largely in the form of lumps, and hence immediately suitable for blast-furnace work.
(3) The plant and appliances required are simple.
The two methods employed are—(A) open-air roasting, (B) roasting in kilns.
A. Open-air Roasting of Lump Ores.—This method is conducted in heaps or stalls, and the features just considered apply particularly to this branch of roasting practice. The modern tendency is to avoid heap-roasting altogether, and it is only conducted when the conditions are exceptional.
Amongst the many grave objections to open-air roasting are—
(a) It is very slow, since a long period of time is required for the oxidising effect to penetrate through massive lumps of ore.
(b) A large amount of capital is tied up in the material at the roast-yards.
(c) The losses occasioned by wind and rain are very considerable.
(d) It is difficult to use up a large quantity of fines in the roast-heaps.
(e) Difficulties arise owing to damage by the fume, and from interference by litigation.
There is one special instance of a modern smelter making a great success of heap-roasting—namely, at Rio Tinto—but the circumstances are peculiar, as the roasting is followed by leaching operations of the immense ore heaps in situ.
This branch of roasting need not be considered at length, and the older standard text-books give full descriptions of the various methods employed. The following particulars are important, however, when under exceptional circumstances such work has to be undertaken:—
The maximum and best average size under ordinary conditions is 40 feet by 24 feet, by 7 feet high above the bed of fuel. The height is important, and varies with the quantity of sulphur in the ore. The lower the sulphur content, the higher the pile; with about 40 per cent. sulphur, the best height is 6 to 7 feet; with 15 per cent. of sulphur, up to 9 feet; and if still less sulphur be present, the height may even be a little greater. Such a heap holds about 240 tons, and if the quantity of ore to be dealt with exceeds this, a number of such piles [Pg 68] should be constructed. The time occupied in roasting is about 70 days, with 10 days more for removing and rebuilding.
The selection of a proper site is important.
(a) The prevailing direction of the wind must be considered, so as to keep the fumes away from the works and offices.
(b) The yards must be protected from winds, so as to prevent losses of dust, as well as uneven burning.
(c) The ground must be perfectly dry or drained.
Along the upper edges of the roast-yard a deep trench should be cut, so as to catch rain-water, and prevent it from washing soluble copper salts out of the pile; drainage trenches must also be provided to carry any copper-bearing liquors to some point where the copper can conveniently be precipitated on scrap iron. Enormous losses of copper may occur if these precautions are not observed; thus, at one period in the old roasting process in Tennessee, as much as 34 per cent. of the copper in the heaps was lost in 186 days.
Preparing of the Floor.—Remove roots and subsoil, fill space with broken stone or rough tailings, cover with 4 to 6 inches of clayey loam, and beat down well. The floor is then fairly impervious, and does not crack on drying. The ground should be given a gentle slope so as to facilitate draining. A layer (about 6 inches thick) of fine ore is next put down, then 9 inches of fuel; channels are now mapped out by means of logs set in both directions, leading to rough chimneys. The pile is then constructed, with the lower parts of the very coarse materials, smaller stuff being put towards the top and sides. On the very top and at the outside of the pile are placed the fines, but this top cover is only put on when the burning is well started. This process is still worked at Tyee, B.C., and at some other localities, but is most probably only a temporary plan, to be replaced by a more efficient method as development progresses.
B. Kilns for Lump Ores.—Kilns possess the advantage that they permit of arrangements being made for the recovery of SO2 for acid manufacture, and the subject belongs more properly to that branch of technology. Few large smelting works employ kilns for roasting lump ores, though there are important exceptions at works both in Britain and on the Continent of Europe. Kilns are used at the Cape Copper Company’s smelter at Britton Ferry, for this purpose.
(b) The Roasting of Fines.—Fines (and particularly fine concentrates) are the usual materials subjected to roasting. The finer the particles, the more rapid and complete is the oxidation, but the losses by dust are heavier. The size limit is thus liable to some variation, but often the material roasted is that under ⅜-inch in size. [Pg 69]
Roasting Furnaces—Requirements.—For the roasting of fines there is simply required a place where the material can be gently heated in the presence of a constantly renewed air supply. The fuel has itself a reducing action, it must therefore be separated from the charge, and hence the furnace employed is of the reverberatory type. Muffles are never used for the oxidising roasting of copper ores. Since only a moderate temperature is necessary for the operation, the furnace needs but a small fireplace, and it is provided with a large hearth area. The fuel used is one yielding the fairly long oxidising flame required.
Developments of Roasting Practice.—The main objects sought in roasting practice have been—
A. Fixed Hearth.—In Great Britain from 1583 onward, roasting in small reverberatory furnaces seems to have been the usual method, and up to 1850 the furnaces appear to have been only of moderate dimensions, with a single hearth, 16 feet × 13 feet 6 inches, constructed of firebricks set on end, and with a fire-box 7 feet × 2 feet 3 inches × 18 inches. Rabbling was done by a long rake, the material being charged and worked through one door. This method of working wasted time, made the process intermittent, and caused continual cooling down of the furnace, involving large fuel costs and much labour. The first improvements were to lengthen the hearth, to add more working doors, and to put the charge into the furnace by a hopper passing through the roof. It was next found best to elongate the hearth still further, and to drop the level of the bed in stages by about 2 inches at a time, thus ensuring better control of working. By this means the best type of hand-calciner was arrived at, consisting of four beds, each 16 feet × 16 feet, the whole charge being moved forward from one bed to the next at each stage of the process. [Pg 70]
In roasting, the ore is first placed in the coolest part of the furnace, and is worked towards the fire, so that the charge travels in one direction, and the flame and furnace gases in the opposite direction to meet it.
The advantages of this system are that—
The capacity of the four-bedded hand-roaster is 7 to 15 tons per twenty-four hours, depending on the sulphur proportion in the charge and in the roasted product.
It is a very useful form of furnace when labour is cheap. The furnace works very efficiently, but in the New World, where manual labour was dear, labour costs became prohibitive, and in order to economise in this direction, mechanical rabbling was introduced.
The O’Harra Calciner (1885) was essentially the old type of furnace, double hearthed and mechanically rabbled. It consisted of long straight furnace hearths. The rabbles were ploughs dragged through the furnace by means of endless chains which were carried over grooved pulleys, situated outside the furnace, at the ends. This was an important invention, giving a continuous feed and discharge, a much larger output, and efficient and regular stirring without much hand labour. The rabbles became cooled on issuing from the hearth. The capacity was 50 tons per day from furnaces of 90 feet × 9 feet hearths, giving a roasting capacity of 61 lbs. of ore per square foot of hearth area, compared with about 33 lbs. per square foot with the old hand calciner. In working the early forms of this furnace there were many mechanical troubles and breakdowns, and the subsequent modifications of this form consisted largely of devices for the purpose of overcoming such difficulties.
Modifications and Improvements.—Allen, instead of a rope to carry the ploughs, used small wheeled carriages, running on a track which was laid along the floor.
Brown (important) ran the carriages along narrow corridors at either side of the hearth, so as to protect the ropes and carriages from the very corrosive action of the furnace gases. A continuous narrow slit along the inner wall of the corridors allowed the arm carrying the plough to travel forward. [Pg 71]
Wethey; Keller; worked on very similar principles. The chief improvements were in details, and had for their object the prevention of wear and tear, and of the break-down of parts.
Prosser.—Very similar; used at Swansea Works.
Ropp.—The carriage runs underneath the bed, and supports a vertical shaft which passes through a slot along the furnace hearth and carries the arms furnished with ploughs.
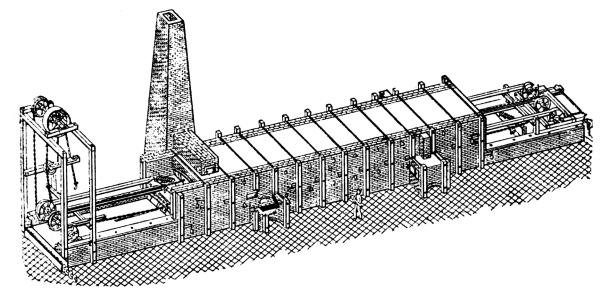
Fig. 17.—O’Harra Furnace (Fraser-Chalmers), illustrating
Principle of Mechanical Rabbling by Travelling Ploughs.
The Ropp and Prosser calciners work very successfully. The hearth is about 105 feet long × 11 feet wide, with a capacity of about 36 tons per day.
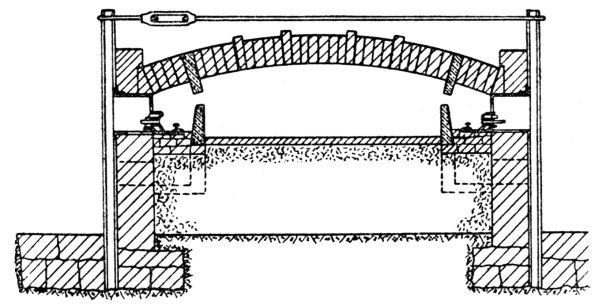
Fig. 18.—Section through Mechanically Rabbled Roaster Furnace
(illustrating Improvements for Protecting Driving Mechanism).
Brown Horse-Shoe Furnace operates on the same principle as the above, except that the hearth is bent round in order to save space.
Pearse-Turrett (1892 at Argo).—In this type of furnace the bed is curved round in the form of a circle. The rabbling ploughs are carried [Pg 72] at the ends of arms which are attached to an upright rotating spindle. The spindle is set in the centre of the space enclosed by the circular hearth.
In all the above classes of furnace, the firing is done, when necessary, from fireplaces built at intervals along the sides of the furnace; either coal or gas being employed as fuel.
B. Rotating Hearths.—This type of furnace is still reverberatory, but instead of making use of mechanical rabbling, the hearth rotates, in order to give agitation to the materials and assist their discharge.
(a) Intermittent Working—The Brückner Roaster.—The details and working of this roaster are familiar. The furnace was invented in 1864 for gold and silver ore-roasting in Colorado, and was later introduced for the roasting of copper ores, being at one time the furnace most commonly used for the purpose. It was employed all over the Western States, and at one works alone, 56 were at one time in use.
The usual length was 18 feet 6 inches and the diameter, 8 feet 6 inches; giving an output of about 12 tons per twenty-four hours. It was furnished with a removable fireplace, used to start the roasting. The operation could then be allowed to proceed by itself, the fireplace being wheeled away to another hearth, and being eventually brought back to the first hearth for about three hours, in order to give the required higher finishing temperature. Several dust chambers were attached to this, as to all forms of roasting furnaces, which by their nature and manner of work are apt to produce considerable quantities of dust.
The advantages of the Brückner cylinder lay largely in the fact that it afforded good control of the sulphur contents in the charge, since the ore could be retained in the furnace until the sulphur was sufficiently low. The furnace is simple to work, and not so liable to get out of order as many other forms. It possesses however, distinct disadvantages in that its working is intermittent, its use involves comparatively high fuel costs, whilst the discharging presents considerable difficulty and trouble to the labour employed, on account of the awkwardness and the high temperature of the discharge, and the sulphurous gases evolved.
Its use has now been very largely discontinued.
Improvements—(b) Continuous Working.—The continuous type of roasting furnace of this class involves the use of sloping cylindrical hearths which rotate, and so agitate and help to discharge the materials.
Oxland (1868) first introduced this type in Cornwall, for the roasting of tin ores. [Pg 73]
The Oxland furnace was an inclined cylinder, the material was fed in at the top, and by the rotation of the cylinder the charge gradually travelled downwards, approaching nearer and nearer to the fire, and being discharged close to the fire-box.
White (1872) improved this furnace, and the White cylinder is largely used in South Wales. The cylinder revolves slowly by friction gearing; inside are four lines of projecting brick-work which form a shelf, thus assisting the agitation of the charge.
The White-Howell Furnace is somewhat similar to the White, but is unlined for the greater part of its length, except at the lower end near the fire-box, where it is much wider and is bricked. It is stated to work more satisfactorily than the older form, having a larger capacity and using but little fuel.
The furnace is employed at the Cape Copper Works, South Wales, for matte-roasting. It is here 60 feet long, 7 feet diameter, inclined 6 inches in 60 feet, makes 8 revolutions per hour, and has a capacity of 10 tons of charge per day.
Argall Furnace.—Consists essentially of four narrow tubes bound together, each 28 feet long, 2 feet diameter, and lined. It works rapidly, having a capacity of 40 to 50 tons per day, but is used more for the roasting of cupriferous gold ores than at the copper smelters.
C. The MacDougal Type.—The most important form of modern roaster furnace, and that most generally employed, is the MacDougal type. The first furnace on this principle was invented by Parkes in 1860. The design embodied two hearths, one above the other. Vertically down the centre of these passed a spindle, supporting arms from which were suspended the ploughs, and the rotation of this spindle carried the arms over the beds.
As devised by Parkes, various mechanical difficulties were found, and the working was intermittent, but the principle was recognised as important. MacDougal in 1873 introduced his modification of the furnace, primarily for the roasting of pyrites, at a Liverpool works, and this form has now supplanted many of the older types for copper ore roasting, and is in operation at most of the new smelting works.
Principles of the MacDougal Type.—The furnace consists of an iron cylinder lined with brick. Six circular hearths are constructed inside, one above the other, and the vertical spindle carrying the arms and ploughs for each hearth passes through the centre of the furnace. The ore is ploughed towards openings on each hearth, which communicate with the hearth below; the charge thus travelling from the outer edge [Pg 74] towards the centre, through the central opening to the middle of the next floor, then outwards to the openings at the edge, and so on. The original MacDougal furnace was 12 feet high and 6 feet in diameter. It was improved by Herreshof in the direction of better rabbling mechanism and greater ease of repair. The central spindle was an air-cooled shaft, the supporting arms were made so as to be easily removable from the shaft to facilitate repairs, and the furnace was enlarged. Herreshof used air-cooling for the spindle and arms, as shown in Fig. 20.
Evans, and subsequently Klepetko, in working the furnaces in Montana, introduced, in about 1892, various marked improvements. The dimensions were increased, enlarging the output. The spindle and arms were water-cooled, which improvement removed much of the great difficulty in working the MacDougal furnace, where the rapid wearing out of working parts, and the difficulty of their removal, repair, and renewal interfered greatly with efficient working. [Pg 75]
Many of these troubles have now been overcome in the Evans-Klepetko type, and in the still further improvements since made at Anaconda. The general arrangement of the floors, spindles, arms and other details shown in the Herreshof furnace (Fig. 20) are preserved in the Evans-Klepetko and similar types of roaster; the chief alterations are in matters of detail, the results of which have however, been important.
Furnaces of this improved kind are now used all over the West; there are 64 at Anaconda, Mont.; 32 at the International Smelter, Tooele, Utah; 24 at Garfield, Utah; 16 at Steptoe, Nevada; and also at Balakala, Cal., Cerro de Pasco, Peru, and other large smelting centres.
Important Advantages.—Of the marked advantages of this type of furnace, the following are perhaps the most striking and important:—
(1) There is a great saving of floor space by having the six hearths one above the other.
(2) The use of a central common spindle carrying the arms and ploughs simplifies the mechanism.
(3) The form is convenient for the compact arrangement of a roasting plant of many units for feeding, discharge, and supervision.
(4) Very little heat is lost by radiation, as the heat passes mostly from one hearth to another.
(5) Very little fuel is required, none with heavy sulphides (except for starting), as the heat of oxidation of the iron and sulphur usually yields a high enough temperature to keep the operation going. The fuel costs are lower than in other types of roaster.
(6) Thorough rabbling, greater uniformity and better mixing of product, continuous and regular feed and discharge are obtained.
(7) The roasting is thorough, and perfect control of the degree of oxidation is ensured by adjusting the rate of passage of the ore through the furnace, which is regulated by varying the ore feed and the speed of rotation of the rabbles.
(8) Great saving in labour costs and difficulties. The labour in roasting plants is extremely arduous, on account of the high temperature of the material, and is dangerous on account of the atmosphere.
The Evans-Klepetko-MacDougal Roasting Furnace Plant at Anaconda.—The roasting plant at Anaconda formerly consisted of 56 Brückner cylinders, which were eventually all scrapped and replaced by new plant of the MacDougal type, subsequently greatly modified and improved as one difficulty after another had to be overcome. [Pg 76]
The saving in working costs resulting from this replacement of the Brückners by MacDougal roasters is reckoned at about 5 cents (2½d.) on every ton of calcines treated.
The roasters are arranged in four rows of 16 each, running east and west. The charge cars travel along tracks at a height of 20 feet above, discharging into rows of bins, one situated over each calciner.
Details of Furnace.—Height, 18 feet 3½ inches; diameter, 16 feet. Six hearths. The spindle is made in three lengths, each to carry the arms for two hearths; it is 18 inches in diameter and is water-cooled. The rabble arms are 6 feet long, half round, and flanged on the lower side; they too, are hollow and water-cooled. The rabble-blades were formerly cast in one piece with base plate, so as to slide on to the arms, but are made now with detachable blades, which slide into grooves on the base plate, so as to facilitate removal for repairs; the blades are 6 inches square and 1½ inches thick (Fig. 32).
The arms on separate floors are set alternately at right angles. Of the two arms for each floor, one carries six blades, the other seven, so that the furrows resulting from one set of blades are turned over by the other. The blades are set so as to direct the ore from the outer to the inner edge or vice versâ, according to the particular hearth. The spindle and connections are protected from falling ore by shields which are bolted on. The rabbles move slowly, making a 2½-inch furrow in a 5-inch layer of material.
Capacity.—40 to 45 tons per day each, reducing the sulphur in the charge from 30 per cent. to about 8·0 per cent. The output of the plant is about 3,000 tons of calcines daily.
System of Working.—Since reverberatory furnaces are used essentially as remelting furnaces only, the roasting plant is operated so as to yield a product of such composition as will directly produce a suitable matte and slag on melting in the reverberatories. The fluxes required for the calculated reverberatory charge are, therefore, sent through the roasters mixed with the fine concentrate; such practice possessing many advantages. The charge thus consists of fine concentrate from the concentrator settling tanks, and screened lime-rock flux (too fine to be used in the blast furnaces). The limestone lightens the charge, decreases the tendency to clotting of the pure sulphides, chemically [Pg 77] assists oxidation, preheats and thoroughly mixes the flux, and ensures a uniformly mixed charge for the reverberatory furnaces; whilst the extra cost involved is but very small.
Three per cent. of lime is used; 40 tons of concentrates, 1¼ tons of lime-rock, and 1¼ tons of flue-dust being charged per twenty-four hours per furnace, through an automatic gravity feed, the opening of which is closed and opened by an eccentric. The speed of the eccentric and the extent of the opening are adjustable.
Working.—Charge contains 25 to 35 per cent. sulphur.
1st Hearth.—Temperature about 230° C. (black heat). This is practically a drying floor, and the wet ore wears the rabbles away rather quickly. Special forms of plough are being introduced. About 4 per cent. of sulphur is driven off from the pyrites.
2nd Hearth.—Hotter; not quite red, except near outer edge. About 5 per cent. of sulphur burnt off.
3rd Hearth.—Bright red heat (about 700° C.). Sulphur can be seen burning off the ridges of calcines, with a blue flame. 5 per cent. of sulphur eliminated. There is some clotting, and the sinter sticks to the rabble-blades, and has to be barred off occasionally.
4th Hearth.—Bright red heat (about 750° C.), uniformly bright, but the flame has ceased. Sulphur loss, 4 per cent.
5th Hearth.—The hottest (800° C.). Bright red.
Bottom Hearth.—Cooler, dark red (about 650° C.). The doors on this floor are left open. The charge is guided towards openings at the outer edge to discharge chutes whilst still red hot, and it is fed from here whilst hot into the reverberatory furnace-bins.
Efficient dust catchers and settlers are essential on the roasting plant. The gases escaping at a temperature of about 315° C. contain 2 per cent. of SO2 by volume, 5 per cent. by weight. The ore takes 2¼ hours to pass through the furnace. Practically no fuel is required except to warm up the roaster on commencing work.
Labour.—The requirements are small. There is one general foreman for the plant, and two helpers for each set of four furnaces. The conditions are rather trying, especially during the discharge of the calcines into the reverberatory charge cars.
Roasting Ores poorer in Sulphur, in MacDougal Roasters.—The Anaconda concentrates carry sufficient sulphur (33 per cent.) to supply all the heat necessary for carrying out the roasting operations. When the sulphur is below this requisite quantity, some extra heating may be required, though, on the other hand, the reduction which is necessary in the sulphur contents is lessened, depending, of course, on the proportions of copper and iron in the charge. At Garfield, Utah, where the concentrate only contains 20 per cent. of sulphur, the fuel required for all roaster purposes is equivalent to 0·2 per cent. of the charge, one of the calcines’ outlets being converted into a fireplace. Here the output per furnace per day approaches 55 tons, roasting the sulphur from 20 per cent. down to 10 to 11 per cent. The flue-dust losses at this plant are 6 per cent., so efficient dust catching appliances are essential.
The Costs of Roasting in the MacDougal Furnace.—Ricketts has recently published a valuable analysis of the costs of the roasting operations at the Cananea Smelter. The figures must, however, be understood to apply strictly to the conditions prevailing at this particular camp.
The roaster plant consists of 32 improved MacDougal furnaces. The charge supplied to the roasters assays—
| Copper, | 5·2 | per cent. |
| Iron, | 28·4 | " |
| Sulphur, | 29·9 | " |
| Silica, | 23·6 | " |
| Alumina, | 3·7 | " |
[Pg 79] whilst the product (“calcines”) has an average composition of
| Copper, | 6·3 | per cent. |
| Iron, | 34·5 | " |
| Sulphur, | 7·7 | " |
| Silica, | 28·6 | " |
| Alumina, | 4·4 | " |
The plant operated on the following quantities of material, from February to July, 1911, inclusive:—
| Concentrates, | 32,929 | short tons | = | 76·08 | per cent. | of charge. |
| Fine sulphide ores, | 9,590 | " | = | 22·16 | " | " |
| Limestone, | 762 | " | = | 1·76 | " | " |
| Total charge, | 43,281 | " | = | 100·00 | " | " |
| Weight of “calcines” produced, | 35,533 | " | = | 82·10 | " | " |
| Shrinkage, | 7,748 | " | = | 17·90 | " | " |
| ════ | ════ |
The total costs of roasting (from roaster charge-bins to reverberatory furnace) worked out at 38·45 cents per ton, the distribution of these costs being as follows:—
| Total Costs. | Cost per Dry Ton. | |||
| Sampling, | $ 222·62 | $ 0·0051 | ||
| Bedding, | 2,016·27 | 0·0466 | ||
| Reclaiming, | 3,072·71 | 0·0710 | ||
| Operating furnaces, | 7,680·58 | 0·1775 | ||
| Hauling calcines, | 911·01 | 0·0210 | ||
| General expenses, | 1,639·99 | 0·0379 | ||
| Total direct costs, | $ 15,543·38 | $ 0·3591 | ||
| Cost of flux, | 1,097·85 | 0·0254 | ||
| Total costs, | $ 16,641·23 | $ 0·3845 | ||
| ════════ | ════════ | |||
| Analysis of Cost— | ||||
| (1) | Operating— | |||
| Labour, | $ 7,398·56 | $ 0·1709 | ||
| Power, | 1,577·64 | 0·0365 | ||
| Fuel, | 773·08 | 0·0179 | ||
| Water, | 78·71 | 0·0018 | ||
| Sundries, | 10·30 | 0·0002 | ||
| Flux, | 1,097·85 | 0·0254 | ||
| $ 10,936·14 | $ 0·2527 | |||
| (2) | Repairs— | $ 2,507·35 | $ 0·0579 | |
| Labour, | ||||
| Shop expense, | 361·00 | 0·0083 | ||
| Supplies, | 2,836·74 | 0·0656 | ||
| Total costs, | $ 16,641·23 | $ 0·3845 | ||
| [Pg 80] | ════════ | ════════ | ||
Peters, E. D., “Principles” and “Practice of Modern Copper Smelting.”
Cloud, T. C., “The M‘Murty-Rogers Process for Desulphurising Copper Ores.” Trans. Inst. Min. and Met., vol. xvi., 1906–7, p. 311.
Hofman, H. O., “Recent Progress in Blast Roasting.” Bulletin Amer. Inst. Min. Eng., No. 42, June, 1910.
Austin, L. S., “The Washoe Plant of the Anaconda Copper Mining Company.” Trans. Amer. Inst. Min. Eng., vol. xxxviii., 1906, p. 560.
Rickets, L. D., “Developments in Cananea Practice.” Engineering and Mining Journal, Oct. 7th, 1911, p. 693.
Redick F. Moore, “Recent Reverberatory Smelting Practice.” Engineering and Mining Journal, May 14th, 1910, p. 1021.
See also—
Pulsifer, H. B., “Important Factors in Blast Roasting.” Met. and Chem. Eng., 1912, vol. x., No. 3, March, pp. 153–159. (With good Bibliography.)
Editorial Correspondence, “Sinter-Roasting with Dwight-Lloyd Machines at Salida, Col.” Ibid., 1912, vol. x., No. 2, Feb., p. 87.
Dwight, A. S., “Efficiency in Ore-Roasting.” School of Mines Quarterly, 1911, vol. xxxiii., No. 1, Nov., pp. 1–17.
Functions of the Reverberatory Furnace—Requirements for Successful Working—Principles of Modern Reverberatory Practice—Operation of Modern Large Furnaces—Fuels for Reverberatory Work; Oil Fuel; Analysis of Costs—Condition of the Charge.
The Functions of the Reverberatory Furnace. —The reverberatory is essentially the furnace for the smelting of fine material, as the comparatively still atmosphere, the absence of blast, and the opportunities for settling prevent the heavy losses by dust which necessarily accrue with the other types of smelting furnace. The atmosphere of the furnace is practically neutral, it therefore exercises little influence on the reactions taking place in the charge, and the reverberatory is, in consequence, mainly a melting furnace.
Its functions are:—
(a) To allow of the formation, from the mixture of sulphides and oxides in the roasted materials from the calciners, of a copper matte and a slag.
(b) To maintain such a high temperature as to render these products perfectly fluid, and thus to allow the matte and slag to settle and separate thoroughly.
In spite of the neutral atmosphere, however, the smelting of the roasted materials usually results in a higher concentration than would be expected from the calculation of the sulphur, copper, and iron in the charge. The reason of this is that the smelting operation results in some further elimination of the sulphur, which causes the production of a higher grade matte. This additional elimination of sulphur in the reverberatory furnace smelting of the roasted charge is due to the reactions which take place on melting, between the oxides, sulphates, and sulphides of copper, all of which exist in the products from the roasters. These reactions are expressed by the equations—
Cu2S + 2Cu2O ➡ 6Cu + SO2
Cu2S + CuSO4 ➡ 3Cu + 2SO2,
which indicate a further addition of copper to the matte, and a [Pg 82] corresponding loss of sulphur. Thus a typical reverberatory charge of the following composition:—
| Silica, | 27·2 | per cent. |
| Iron, | 31·0 | " |
| Lime, | 2·3 | " |
| Sulphur, | 8·4 | " |
| Copper, | 8·3 | " |
should theoretically yield, on melting down, a matte running—
| [8] Cu (8·3) ➡ Cu2S 10·4 | Cu 8·3 | Cu 30 | per cent. | ||||||
| = | S 8·4 | or | S 30 | " | |||||
| S (8·4−2·1) ➡ FeS 17·6 | Fe 11·3 | Fe 40 | " |
In actual practice however, the matte resulting from the reverberatory smelting of the charge had the composition—
| Cu | 45 | per cent. | |
| S | 27 | " | |
| Fe | 28 | " |
the 3 per cent. loss of sulphur causing a 15 per cent. increase in the copper contents of the matte.
Experience in the working of the plant enables the management to determine this important factor with fair accuracy, and thus from a knowledge of the composition of the roaster product, to regulate and control the grade of the matte produced at the reverberatories. In modern reverberatory practice, therefore, the control of the furnace products is carried out at the roasting plant, and the reverberatory furnace has simply to melt the charge and ensure good settling.
Anaconda Practice affords a good illustration. The foreman of the reverberatory furnaces simply charges what is sent him from the roasters, and practically nothing else is put in,[9] his duty being to smelt this mixture and to obtain from it a clean slag and fluid matte. He is not responsible for the grade of the matte, and if this is not satisfactory, some change is made in the working at the roasters. The reverberatory foreman does not learn the composition of the materials passing into his furnace until he is furnished with the daily assay reports on the following day.
Reverberatory smelting is essentially a British process, developed in Wales, as already explained, owing to a plentiful supply of good furnace [Pg 83] coal yielding a long flame, and also of good refractory material. Many Swansea workmen were, in the early days of American development, and are still, employed in charge of such copper furnaces, and it is largely due to British technical skill and to American genius for organisation and development that reverberatory smelting in the large furnaces at modern works has become so very successful.
The Principles of Modern Practice.—Success in modern reverberatory work has been due to the recognition of the fact, that with the maintenance of constant high temperature on large masses of material, thorough fusion and separation of the products can be very efficiently conducted.
The Requirements for Successful Reverberatory Work.—Since the action in the furnace is performed mainly by the effects of heat, it is necessary that—
The temperature required for the formation of slag and for obtaining a thorough fluidity of the materials is from 1,400° to 1,600° C., and the methods of achieving the proper conditions can best be stated as the avoiding of all circumstances likely to cool the furnace or to interfere with the melting down of the charge.
A. To ensure rapidity of melting, it is essential that a very large quantity of coal shall be burned as rapidly as possible. This requires—
In localities where a supply of suitable coal is not available, other methods of heating, such as the use of oil or gaseous fuel, are necessary.
B. To prevent heat losses as much as possible, it is necessary—
A. i.—Enlarged Grate Area.—In the older methods of working, there was a general tendency to employ a furnace of standard size, and improvements in the economy of the process were in the direction of reducing the fuel bill as much as possible for the given size of furnace. This was effected by keeping the grate area fairly small.
In modern practice, economical working still involves having the ratio of size of hearth to size of fire-box as large as possible, but instead of reducing the dimensions of the fire-grate to suit the hearth, a large grate is built to commence with, and the hearth is constructed of such a size as will utilise all the heat available. From this principle of burning a large quantity of fuel and melting with it as much charge as possible, the efficient and economical working of large furnaces has been developed.
A grate area of about 28 square feet is now regarded as the minimum for economical work at modern smelters, and fire-boxes up to 128 square feet in area are usual in practice.
In small fire-boxes, only small quantities of fuel can be burned at once, and in consequence, fresh firing is continually required, which interferes greatly with the work of the furnace and decreases the rapidity of heating. Each addition of cold fuel has a cooling effect on the fire and furnace gases, the temperature in the hearth being found to drop for a period of five or ten minutes by as much as 100° C., the flame becoming smoky, red, and cold. A similar time is required for the original temperature to be attained once more. Cold air is also admitted every time the fire-box doors are opened for charging.
The advantages of large grate area therefore include:—
The most rapid and economical smelting at the present day requires that at least 0·7 lb. of coal be burned per minute per square foot of hearth area.
A. ii.—Draft.—The charge in a reverberatory furnace hearth is melted chiefly by the heat from the hot gases passing over it, and in giving up their heat to the charge, the gases become cooled down. The [Pg 85] heating of the charge is made continuous by the continual addition of fresh fuel in the fire-box, and by the drawing of the flames over the hearth by means of flues situated at the other end of the furnace and leading to the stack. The flues and stack must be large enough to cause sufficient draft through the furnace for the heated gases to be drawn over the charge with sufficient rapidity, and much unsuccessful work has been due to the fact that these requirements have not been fulfilled. There should be a suction equivalent to at least 1 inch to 1·5 inches water pressure up the stack, this being readily measured by water-manometers—a feature of modern working.
Reverberatories may be worked either by forced or natural draft, the latter being usually preferred, though it necessitates a large stack and spacious flues.
Forced draft by fan or blower under the fire-grate has been in use at several smelters, the ashpit then being closed. It was at one time adopted at Anaconda, but was given up later. The use of forced draft has the advantage that leakages of cold air into the furnace are to a large extent prevented, hot gases tending to be forced out rather than cold air drawn in, but the objections to its use include the facts that—
A. iii.—Firing and Grating.—This question is closely connected with the dimensions of the grate, since the use of a small fire-box necessitates methods of firing and grating which are not conducive to the most rapid and efficient combustion of the fuel. In addition to the cooling action of frequent fresh fuel charges in the small fireplace, attendant disadvantages include the closing up of the spaces in the grate by which air enters for burning the fuel, and the consequent necessity for frequent grating with small beds of fuel, which entails numerous objections.
The addition of fresh coal to the fire causes the production of large quantities of volatile hydrocarbons which require an increased air supply for proper combustion, and this air admission is just prevented by the blanketing action of the fresh fuel added. This is indicated by the red smoky flame, and means waste and cooling. The difficulty is [Pg 86] overcome by the arranging of a series of air-holes at the fire-box end of the furnace, near the fire-bridge, and by the opening of these directly after firing, the volatiles are immediately burnt up. This is an important feature in successful working, and with a large fire-grate and this air-admission, the effect of adding even 1½ tons of fuel on to the fire at once causes little difference in the furnace temperature. The flame is observed through a window let into the off-take flue, which allows of the changes in appearance being noted by the fireman on the fire-box platform.
The fire is kept moderately shallow, to allow of rapid burning of the fuel, though deep enough to keep up the enormous body of heat necessary in the furnace.
B. i.—Avoiding Leakage of Cold Air.—The admission of cold air was the cause of much waste in the older processes of working. Each time the doors were opened, either at the fire-box, or during charging on to the hearth, large quantities of cold air were admitted; air entered through the working door whilst slag was skimmed off, whilst matte was being tapped, and whilst the furnace hearth was being clayed; all of which operations occupied considerable time. The doors were opened during the levelling down of the fresh charges, and at later periods when the charge was stirred and the half-fused masses sticking to the bottom were worked up.
In modern practice, an essential feature of working is to keep all the doors closed as much as possible, and, as will be indicated shortly, every means is taken to eliminate the heat losses from the causes just referred to. Air leakage is also occasioned by bad grating, which causes the formation of channels in a few parts of the bed of fuel, admitting excess of air at these places, instead of causing it to come regularly through the bed in all parts. Channelling is now checked by the drop of suction-pressure in the flues, as registered by the manometer.
B. ii.—Prevention of Radiation through Walls and Roof.—Such heat losses are now minimised by thickening these parts, and blanketing the outside of the roof with sand, keeping the construction together by very heavy bracing.
B. iii.—Prevention of Cooling of the Hearth on Withdrawal and on Charging.—By far the most important cause of heat losses in working was occasioned by the withdrawal of the whole of the melted products, the charging of fresh cold ores, and the efficiency of the furnace was very greatly reduced in consequence. In the older methods, fully three-quarters of the time and fuel, and almost all the labour, were [Pg 87] spent in manipulating the charges and bringing them up to the point of fusion, the actual smelting operation being responsible for but a small proportion. The withdrawal of the hot slag and matte abstracts much of the heat of the furnace, and the cold charge which is fed in, not only cools the furnace hearth on which it rests, but being a poor conductor, prevents the heat from again penetrating through it to the hearth and to the undermost portion of the charge. It has been estimated through the use of pyrometers, that the temperature in the furnace after such withdrawal and recharging may drop to less than 700° C.—a dull red heat—and there is no way under such circumstances of heating up the hearth again, except by conduction through the charge. Some hours’ hard firing were thus required to bring the furnace to the desired temperature again, after which it was necessary to re-open the working doors, in order to stir the materials so as to prevent the half-fused masses, still lying on the hearth, from sticking to it. This also occasioned delay in the operations, and caused much waste of fuel, heat, and labour.
B. iv.—Utilising the Heat of Melted Charges for the Heating of Fresh Additions.—All the above difficulties, and many others, have been overcome by maintaining a deep pool of hot molten matte in the furnace, and by feeding hot charges upon this matte layer. These are two of the most vital and successful changes introduced into modern reverberatory practice, and will be reviewed in detail subsequently.
B. v.—Utilising the Heat of the Escaping Gases as much as possible.—Improvements in this direction have been brought about—
Modern Reverberatory Practice.—The requirements for the successful operation of the reverberatory furnace, and the methods for ensuring its efficient working which have just been reviewed, involve the application of the following principles, which are the essential factors in modern reverberatory smelting practice:—
[Pg 88] 1. Control of Furnace Products at the Roasters.—This feature has already been indicated in dealing with roasting practice. The importance of this system in the economy and efficiency of the furnace working is very marked.
(a) The roasting plant affords the most ready means of control over the desired sulphur elimination, this being its sole function. The modern roaster is so designed as to allow of almost perfect regulation in this respect, since amount of feed and rate of passage of the sulphides through the furnace are under perfect control.
(b) The work of the reverberatory is thus confined to one object only, that of rapid melting down, to which the foreman can give his sole attention free from the necessity of manipulating the grade of the matte at the same time.
In modern work it is usual to pass the whole of the charge (concentrates as well as flux) intended for the reverberatories, through the roasting plant. The advantages of such procedure are—
Lime in the roaster charge appears to assist the thoroughness of the roast, whilst an incipient slag formation is commenced owing to the juxtaposition of basic oxides and silica, in the hotter parts of the roaster furnace.
2. Rapidity of melting is an indispensable feature of modern work. The conditions necessary for rapid melting have been reviewed above.
3. Use of Large Furnaces.—Reverberatory furnaces appear to have replaced the blast furnace in Great Britain somewhere about 1700, and by 1854 they were in general use in this country. At this period the usual dimensions were, for the hearth 13 feet by 9 feet, with a fire-box 4 feet by 4 feet, the furnace having a capacity of 12 tons per twenty-four hours. In Great Britain the size increased very slowly, and it was in the United States of America that the important increase in [Pg 89] dimensions and in enormous outputs were developed. The work was commenced systematically in about 1878 by Richard Pearse (a Swansea-trained metallurgist) at the Argo Smelter in Colorado. Table V. indicates the gradual improvements in practice resulting from these developments (see also Fig. 23, p. 90).
TABLE V.—Development in Size of the Reverberatory Furnace.
| Year. | Fire─box Dimensions. |
Hearth Dimensions. |
Stack | Capacity. | Tons Ore per Ton Coal. |
|---|---|---|---|---|---|
| 1878, | 4' 6" × 5' | 9' 8" × 15' | 2' 9" | 12 tons. | 2·4 tons. |
| 1882, | 4' 6" × 5' | 10' 4" × 17' 10" | 2' 9" | 17 " | 2·43 " |
| 1887, | 4' 6" × 5' 6" | 12' 8" × 21' 2" | 3' 0" | 24 " | 2·67 " |
| 1891, | 4' 6" × 6' | 14' 2" × 24' 4" | 3' 0" | 28 " | 2·8 " |
| 1893, | 5' × 6' 6" | 16' × 30' | 3' 6" | 35 " (43)‡ | 2·7 " (3·3)‡ |
| 1894, | 5' × 6' 6" | 16' × 35' | 4' 0" | (50)‡ | (3·7)‡ |
| 1903, | 5' 6" × 10' | 20' × 50' | 5' 5" | (70)‡ | (3·1)‡ |
| 1910, | 8' × 16' | 19' × 116' | .. | (275)‡ | (4·66)‡ |
| ‡ The charges of calcines were fed whilst still red hot. | |||||
This practice has been continued in modern smelter work, the developments being in the direction of attempting to melt the largest possible quantity of charge in one furnace as rapidly as possible. This has been found to depend upon the rapidity with which the fuel is burned, and the enlarging of the fire-box had a specially important influence in effecting this rapidity of combustion.
Then, with the size of grate fixed and the most efficient burning of the fuel arranged for, the capacity of the furnace depends simply on increasing the area of the hearth to as great an extent as the heat generated is capable of maintaining at the desired temperature.
The breadth of the furnace is however, limited by—
The maximum width so far found satisfactory is about 19 feet, so that this dimension being fixed, the furnace capacity is enlarged by increasing the length, and this is limited only by the distance from the fire-box to which the flame can maintain the temperature necessary for keeping the charge in a state of perfect fluidity. For many years the length was regarded as limited to 50 feet, smelting about 2·7 to 3·0 tons of charge per ton of coal, but E. P. Mathewson, at Anaconda, finding the escaping gases still very hot, gradually increased the length of the hearth, first to 60 feet, then to 80 feet, and finally up to 116 feet, when the furnace smelted 4·83 to 5·0 tons of charge per ton of coal. The gases then left the furnace at a temperature of about 950° C., and contained sufficient heat to fire two Stirling boilers, each of 375 H.P. Every furnace thus provided about 600 H.P. from this waste heat, and the gases finally escaped at a temperature of 320° C. [Pg 90]
[Pg 91] The capacity of these large furnaces is about 270 to 300 tons of charge per day, and in addition to the economy and efficiency resulting from the treatment of such large quantities of material at once, there are the further great advantages in that—
About 110 feet appears to be the practicable maximum for furnace length, and reverberatories of this size are being constructed wherever circumstances permit, several new smelters having erected such furnaces—there are eight at Anaconda, Mont.; two at Garfield, Utah; five at Tooele, Utah; four at Cananea, etc. The length of the hearth is naturally dependent upon the character of the fuel, particularly the length of flame given out on burning. Bituminous fat coals are the most suitable for this purpose, and in localities where such fuel is not available, the use of liquid fuel has now been successfully adopted.
4. Maintaining a Heated Matte Pool in the Furnace.—This is probably the most important and beneficial advance made in reverberatory practice.
In certain stages of the old Welsh process, a store of matte was retained in the furnace after skimming off the slag, but the object was to collect a sufficiently large quantity of matte in the furnace for convenient tapping out.
The modern practice has several objects and possesses enormous advantages—
(i.) It assists efficient settling.
(ii.) It conserves the heat inside the furnace.
(iii.) It presents a highly heated surface for the fresh charge to fall upon, and thus greatly increases the rapidity of melting, by ensuring that the charge is heated both from above and from below.
(iv.) It prevents the sticking of half-fused charges to the furnace bottom, the removal of which masses would necessitate much labour, and occasion cooling of the furnace by the opening of working doors.
(v.) It preserves the furnace bottom.
Liquid matte has practically no action on the siliceous material of the hearth, and so presents an inert mass between the bottom and the charge. This charge consists of calcines (mainly oxides of iron), which would, during the process of melting down, slag with and corrode the furnace hearth were it not protected by the matte layer. [Pg 92]
(vi.) It allows of continuous charging and withdrawal of materials, and of continued high temperature in the furnace, thus protecting the furnace lining from much wear and tear. Nothing damages furnace linings more than exposure to changes of temperature, on account of the continual expansion and contraction of the brickwork and the low thermal conductivity of the silica. Furnace linings wear out much more from such action than from long exposure to continued high temperature.
(vii.) There is effected an enormous saving of time, fuel, and labour by maintaining a constant high temperature, instead of having to heat the furnace up again after each tapping and charging, as was the case with the older methods of working.
(viii.) The levelling of the charges in the furnace is greatly facilitated. The charges would otherwise pile up under the charging hoppers, and form heaps which are not only difficult to melt down, but which tend to stick to the furnace bottom, requiring time and arduous labour for their removal. In modern practice, charges in quantities of 10 to 15 tons at a time maybe dropped in, these merely spread themselves out on the bath of molten material and float down in a thin stream towards the skimming door at the end, and they generally melt and disappear when half-way down the furnace.
By this means, the working doors at the side need practically never be opened for manipulating the fresh charges.
5. The Charging of Hot Calcines.—This improvement was also introduced by Pearse, and possesses very many advantages; he was able to increase the furnace output by 23 per cent. with the aid of this device.
Instead of allowing the materials from the roasters to cool down, they are taken straight from the roaster bins to the hoppers which feed the reverberatory furnace, where they retain much of their heat until charged into the furnace, being then still red hot as a rule. Much time and fuel is thus saved owing to the charge requiring less heating up, and the cooling action of charging is diminished.
A charge of 15 tons is completely melted within an hour.
6. Regulation of Furnace by Draft Pressure.—It has already been pointed out that rapid combustion of fuel, and consequently rapid melting, is greatly assisted by good draft through the furnace. In modern practice, where the factors, such as charge composition, nature of fuel, and furnace proportions, have been satisfactorily arranged for independently, the actual working of the furnace is regulated by the draft pressures. These are registered automatically by water-manometers [Pg 93] arranged at various points. One usually communicates with the furnace, above the fire-bridge; another is connected to the down-take flues. The indications of these instruments enable a record to be kept of the various operations, and of the charging of the furnace, as well as of the condition of the fire. The usual draft pressure worked with corresponds to about 0·8 inch of water, registered above the fire-bridge.
On opening the hopper for charging, the pressure drops almost to zero; the opening of any doors causes a reduction in pressure; the charging of coal is also rendered noticeable by a drop in the record. Reduction of pressure also indicates “airing” of the furnace by an excess of air entering through channels in the bed of coal; draft-pressure thus acting as a check on the firing and also on the grating, since the formation of excessive clinker in the fire-box is indicated by an increase in the pressure.
Corresponding to such record over an 8-hour shift, as shown on fig. 24, Offerhaus noted the following furnace manipulations, illustrating how accurately the operations are checked by this method:—
| a.m. | |
| 7.00–7.14 | Skimming (coal charged during this period). |
| 7.16–7.16½ | Side door opened. |
| 7.28–7.31 | Coal charged. |
| 7.52–7.57 | Charged. |
| 8.05–8.15 | Tapped. |
| 8.15 | Coal charged. |
| 8.40 | Coal charged. |
| 8.54–8.59 | Grating. |
| 9.05 | Side door opened. Charged. |
| 9.27 | Coal charged. |
| 9.49 | Coal charged. |
| 10.07 | Charged. |
| 10.25 | Coal charged. |
| 10.41 | Coal charged. |
| 10.45–10.58 | Skimming. |
| 11.04 | Coal charged. |
| 11.16 | Charged. |
| 11.16–11.35 | Some grating. |
| 11.36 | Coal charged. |
| 12.03 p.m. | Coal charged. |
| 12.04 | Charged. |
| 12.37–12.48½ | Tapped, 1½ ladles (about 11 tons). |
| 12.45 | Coal charged. |
| 1.00 | Charged. |
| 1.11–1.45 | Grating. |
| 1.26 | Coal charged. |
| 1.44 | Charged. |
| 1.51 | Coal charged. |
| 2.18 | Coal charged. |
| Total charges during shift,16 coal, 7 calcines. | |
[Pg 94] The draft record is placed close to the charging platform, in order to be in a convenient position for the guidance of the workmen. The draft in the main flues is 1·7 to 1·8 inches water pressure; this is similarly recorded in the foreman’s office.
7. Continuous Working of the Furnace.—The continuous working of the furnace is a most important factor in modern practice, and is naturally inseparably bound up with the principle of maintaining the heated matte-pool in the furnace, which allows of the continuous charging of hot “calcines,” and the continuous or regular withdrawal of slag and of matte when required.
The matte (which can be efficiently settled, owing to the prevailing high temperature and the large mass of heated material in the furnace) is stored there until required at the converters, when the desired quantities are tapped out. The slag which is produced by the smelting action gradually accumulates, and at regular intervals most of it is run out (rather than skimmed). This usually takes place every four hours. The slag accumulates until it reaches a level some 3 or 4 inches above the skimming plate at the end of the furnace, and the quantity [Pg 95] which is run out at each “skimming” amounts to some 60 or 80 tons, the contents of the furnace being lowered to such an extent that a fresh accumulation of material may proceed during the next four hours. No pulling of the slag is required as in the older methods of working, since the material is so very hot and fluid that it simply pours out of the furnace, and twenty minutes usually suffices for the whole of the 60 or 80 tons to run off, the rabble being used chiefly to regulate and control the stream, and to keep back siliceous crusts or floaters. The slag is run out until the matte is seen underneath, on flapping back a thin layer, or until the level of the skimming plate is reached, and its removal is such a short and simple operation that there is very little interference with the regular and continuous running of the furnace. Similarly, the tapping of as much as 50 to 100 tons of matte from the store of 250 tons of hot fluid material has little influence on the continuous working. Charging of coal and calcines is performed at regular intervals, and the charges of 15 tons of “calcines” fed in at a time, readily melt down and settle. Practically the only interference with continuous running is the necessity for claying and repairing, and the use of the matte pool on the hearth has lessened the frequency for this to a large extent, the hearth bottom itself being protected from corrosion, owing to the sulphides exerting no action upon it, whilst the oxides in the charge which would be capable of attacking the siliceous bottom are slagged off before they get an opportunity of reaching it. The hearth bottom, if properly put in, is practically permanent.
The portion of the furnace most subject to corrosion is at the slag line, where deep channels are gradually cut out. Every four to six weeks the furnace is tapped dry, repaired, and fettled, as much as 20 tons of fettling sand being often required for this purpose. The sand is thrown in and patted into place by long rabbles, the operations occupying about eighteen hours. Every nine months or so the furnace is repaired more fully, 20 or 30 feet of brickwork near the fire-bridge being taken down, and the great cavities in the side walls repaired by masons, using silica bricks. The employment of higher temperatures in modern work allows of more siliceous slags being produced, which lessens the tendency to the eating away of the walls.
The feeding of siliceous copper ores through a series of small hoppers situated in the roof, near to the walls, has lately been introduced with a view to protecting the furnace sides from the corrosive action of the slag, and to exposing a suitable siliceous flux to this material. This appears to have fulfilled its purpose to some extent, but various difficulties have been encountered in practice, especially [Pg 96] the tendency for the cold added material to form floaters, which require limestone additions in order that they may be fluxed off; and the cooling effects and leakages through the openings have also given trouble.
8. Modified Constructional Details.—In addition to the increased size of fire-box, hearth, and flues, and to the necessity for very heavy staying in order to keep the enormous arch in permanent shape, which are characteristic of modern practice, the construction of modern furnaces involves the building of a suitable hearth to carry the heavy burden of hot and fluid matte which is stored in the furnace.
It was formerly considered correct practice, in the smaller types of furnace, to construct the hearth over a vault, in order to keep the underside cool and thus prevent the corrosion and eating away of the siliceous bottom by the oxidised charges, during the process of melting down. In modern practice it is absolutely essential to work with a perfectly solid structure.

Fig. 25.—Skimming Reverberatory Furnace, Anaconda.
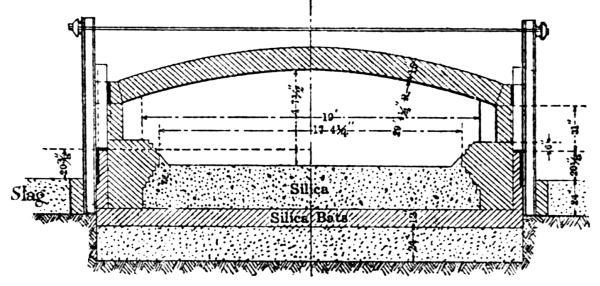
Fig. 26.—Transverse Section of Modern Reverberatory Furnace,
Anaconda, indicating Foundations, Hearth, and Bracing.
(a) Because the hearth must be kept as hot as possible, so as to ensure rapid melting of the charge and maintain the products in a perfectly fluid condition. Any circumstance tending to cool the hearth is rigorously avoided, this being the contrary of the older practice. The protective influence of the heated matte-pool in modern work preserves the bed from the corroding effects of fresh oxidised charges, and in consequence, the maximum degree of heat can with safety be maintained on the furnace hearth.
(b) The enormous weight of charge and the heavy arch and walls demand the strongest possible foundations and support.
[Pg 97] In building modern reverberatories, the foundation for the hearth is constructed of solid masonry or brickwork, or as at Anaconda, of a solid bed of slag, some 24 inches in depth, run in from an adjacent furnace. The I-beams used for carrying the bracing are erected in a surrounding trench, and a further quantity of slag (4 feet thick by 2 feet deep) is run in, thus yielding a perfectly rigid and impervious foundation (Fig. 26). On the top of this slag-foundation is built a layer, 12 inches thick, of silica bricks, and upon this, the actual working bottom of the furnace is constructed.
This bottom is now put in also in a manner different to the older practice, and excellent results have accrued from the change.
The old method of constructing sand bottoms consisted of putting in the beds of sand, layer by layer, and thoroughly fritting each one before the addition of the next: in modern practice, it is found that proper consolidation is not attained with beds of the enormous area now employed, when the bottom is constructed in such layers.
The present method of working the reverberatory furnace is not to drop the charge on to the sand hearth at all, but into the deep pool of matte, and the sand-hearth is regarded more as a convenient foundation for the support of this liquid working-bed, on account of its constituting a cheap non-conducting and fire-proof material which is unaffected by the materials resting upon it. It was found, however, on commencing this matte-pool practice, that the older method of putting in the bottom in successive sand layers was not suitable for this work; after a little wear, the beds became raised in layers, this being especially the case if any holes happened to be eaten through in places. Moreover, the large weight of matte tended to find its way down between the layers and raise them up bodily, or else it worked down at the edges of the hearth and side walls, and either broke out underneath the former or through the latter. When it was ascertained that liquid matte itself had no corrosive action on the siliceous hearth if the latter be kept constantly covered, and that the causes of breakouts were principally due to mechanical weaknesses, it required only improvements in design and construction in order to avoid them. This is now attained by constructing the bed in a compact and perfectly massive form, and is best accomplished by putting in the whole layer of 26 inches of sand at once, and firing as hard as it is possible for the brickwork to stand. The method has met with exceptional success in practice, rigid and impervious hearths are obtained; it being found that less than 1 inch has worn off the bed after two years’ working. [Pg 98]
Large Reverberatory Furnaces: Details of Construction.—The large furnaces at Anaconda were the first of the modern type to be constructed, they have met with enormous success in practice and constitute the standard form. Similar furnaces are now in operation or under construction at many of the large modern camps, and are of similar design and construction.
The hearth is 102 to 116 feet long by 19 feet wide.
Grate, 16 feet by 8 feet = 128 square feet grate area.
Ratio of hearth to grate area is 16 : 1.
Distance from hearth to level of fire-bridge, 26 inches; hearth to crown of arch, 6 feet 5 inches. Walls are 26 inches thick. Roof is 15 inches thick (except for 4 to 6 feet over the fire-bridge, where it is 20 inches). The bracing of the furnace is necessarily particularly strong (see Fig. 29). Lined inside with silica brick, said to be the finest in the world. The bed is of the finest Dillon sand (97·5 per cent. silica), ground to pass ¼-inch mesh; the bed has a slope of 8 inches towards the tap-holes, of which there are two. During the construction of the large furnace there are left in the roof ten expansion openings of 3 inches each, which by the time the furnace has attained its working temperature, become closed up (see Fig. 30). The conker plate which runs through the fire-bridge is 14 to 15 feet long, and is made thicker near the furnace side, where it is 3 inches thick. The air space through the plate is 2 feet 3 inches by 9 inches, and serves the purpose of keeping the fire-bridge cool; air passes through it continuously, and if the plate shows signs of becoming hot, a blast of cold high-pressure air is sent through it. Still further heating of the plate and signs of red heat are an indication that the 2 feet of silica of the fire-bridge wall are being burnt through.
Working of the Reverberatory Plant at Anaconda.—The plant consists of eight large furnaces, built parallel to one another, seven being usually at work whilst the eighth is undergoing repair. Each furnace treats 300 tons of hot calcines and flue-dust daily.
Charging.—The furnaces are charged every 65 to 70 minutes with 15-ton charges, and as soon as one charge is melted, another is added; with average running, 150 charges are worked in the seven furnaces daily. The charge train, consisting of an engine and three cars, each of which carries 5 tons of charge, travels from the roasters and enters the reverberatory building by an overhead track running above the charge bins of the furnaces. It discharges through hoppers into the bins which extend across the entire width of the hearth. Bins were formerly arranged at intervals all the way down the furnace, but now only the two bins nearest to the fire-bridge are employed. Into the back bin, 10 tons of charge are placed, and into the other, 5 tons. Each of these bins discharges through two hopper discharge openings, [Pg 100] feeding the furnace through holes in the roof (Figs. 29, 30), which are closed, when not in use, by round firebrick tiles 20 inches in diameter and 2½ inches thick; these are moved in and out of position by means of levers operated from the fire-box platform.
The temperature maintained in the furnace is high, approximating to 1,500° C., and just previous to dropping in a fresh charge, a workman, by means of a rabble, feels about the hearth below the charging hopper in order to ensure that all of the previous charge has been melted, and that none of it is sticking to the furnace hearth. By employing only the comparatively small quantities of 15 tons, this sticking is avoided, since such charges are not heavy enough to sink unmelted through the 8 inches of slag and 8 inches of matte in the furnace. The former practice of feeding charges amounting to 45 tons through hoppers situated all the way along the furnace had given serious trouble in that respect, and had consequently to be discarded. When the examination of the hearth is completed, the time occupied being very short, the side door is closed, and sealed with sand; the covers to the holes in the roof are now withdrawn, the gates closing the hoppers pulled back, and first the 5-ton, then the 10-ton charge is dropped into the furnace. The whole operation, including the preliminary opening of the door to test the furnace bottom, occupies five minutes.
Very little hand labour is required round these enormous furnaces, except for the grating of the fires, for the charging of coal and calcines every hour by the operation of levers from the fire-box platform, for the skimming of slag at intervals of four hours, and for the tapping of matte when required. The whole of this work is conducted by the skimmer and two helpers to each furnace, one of the men also looking after the boilers.
As soon as the charge has been dropped on to the pool of molten material, the mass appears to spread out over the surface and float towards the skimming door, in a thin slow-moving stream which disappears when about half-way down, being usually melted within one hour. The former 40-ton charges required as much as eight hours for melting.
Owing to the great heating effect of the large bath of hot material below, and of the intense flame above, there is but little cooling action on adding the fresh charge; whilst with this length of furnace, practically all the dust is settled, and very little is carried into the flues.
Coaling.—The quantity of coal employed amounts to 20 to 25 per cent. of the charge, or about 50 to 60 tons per day per furnace, 1 ton of coal smelting rather less than 5 tons of calcines.

Fig. 29.—Fire-box End of Reverberatory Furnace, showing massive
Bracing, Charge Bins, and Charging Levers—Anaconda.

Fig. 30.—Interior of Reverberatory Furnace (looking towards Skimming Door),
showing Expansion Spaces in Roof, and Charging Holes—Anaconda.
[Pg 101] Coal is charged every 40 minutes in quantities of 1½ tons at a time, from bins which extend across the entire width of the fireplace, feeding through four hoppers into openings 1 foot square in the roof of the fire-box, and the withdrawing of the gates is operated by means of levers at the platform. Over the fire-bridge are two rows of air-holes used for regulating the length and character of the flame in the furnace; the flame, however, plays a subordinate part in the smelting reactions. The coal employed is from Diamondsville, Wyoming, and gives a flame 125 feet in length, the appearance of which is gauged through the window fixed in the off-take flue, this being visible from the fire-box platform. The coal is run-of-mine quality, and considerable slack is used. It possesses a high calorific power and a large proportion of volatile constituents, but clinkers rather badly, and a clinker grate is worked with.
Grating.—The fire rests upon 3-inch round bars placed at 4½ to 6-inch centres, and is maintained at a depth of about 27 inches. Grating requires to be conducted at fairly frequent intervals, usually twice per shift, in order to keep the fire free and to prevent channelling, which is indicated on the draft gauge by a drop from 0·75 inch to 0·50 inch, due to airing. It serves further to prevent clinkering, which, when taking place in the fire, causes a rise of from 0·75 up to 1·0 inch on the gauge. The operation of grating usually occupies about half-an-hour; the work is arduous, and the heat to which the workman is exposed is itself very trying.
Coke Recovery.—A constant stream of half-burnt fuel and ashes falls through the bars, and during the clinkering operations large quantities are dropped. The material all falls down a bank inclined at 45°, into a channel where it is met by a stream of water which washes it along launders and through a grizzle, to a settling tank. The settled products are subsequently jigged, the recovered coke being washed over the tail-board to a trommel, and by this means 10 per cent. of the fuel charged into the furnace is recovered in a useful form. This coke is used up as a constituent of the briquettes. [Pg 102]
TABLE VI.—Daily Report—Reverberatory Furnaces.
August 17th, 1908 (Good Day).
| Charge. | ||||||||
|---|---|---|---|---|---|---|---|---|
| Furnace No. |
Coal | Total Smelted |
Calcines | Macdougal Flue-Dust |
Blast Furnace Flue-Dust |
Main Flue-Dust |
Extras | Residues |
| Tons | Tons | Tons | Tons | Tons | Tons | Tons ‡ | Tons | |
| 1 | 60·6 | 288·8 | 279·2 | .. | 8·9 | .. | 0·7 | .. |
| 2 | 57·2 | 277·7 | 262·7 | .. | 2·9 | 11·8 | 0·3 | .. |
| 3 | 64·1 | 286·7 | 253·2 | 12·0 | 8·9 | 11·8 | 0·8 | .. |
| 4 | 60·5 | 278·7 | 264·7 | .. | 2·6 | 3·9 | 0·2 | 7·3 |
| 5 | 57·3 | 245·9 | 221·7 | 12·0 | 11·2 | .. | 1·0 | .. |
| 6 | 57·3 | 273·1 | 264·4 | .. | 7·9 | .. | 0·8 | .. |
| 7 | .. | .. | .. | .. | .. | .. | .. | .. |
| 8 | 57·4 | 278·7 | 266·8 | 11·9 | .. | .. | .. | .. |
| Total | 414·4 | 1929·6 | 1812·7 | 35·9 | 42·4 | 27·5 | 3·8 | 7·3 |
| ‡ = Fine lime rock. | ||||||||
| Delays. | ||||||||
|---|---|---|---|---|---|---|---|---|
| Furnace No. |
Copper Material Smelted per Ton of Coal |
Cost of Coal per Ton of Metal Melted |
Waiting for Coal |
Waiting for Calcines |
Miscellaneous | Total Delays |
Boilers Working |
Ladles of Matte in Furnace at End of Day. |
| Tons | $ | Hours | Hours | Hours | Hours | Hours | ||
| 1 | 4·77 | 0·95 | —— No delays. —— | 24 | 10 | |||
| 2 | 4·85 | 0·94 | 24 | 10 | ||||
| 3 | 4·47 | 1·02 | 24 | 10 | ||||
| 4 | 4·61 | 0·99 | 24 | 10 | ||||
| 5 | 4·29 | 1·06 | 24 | 10 | ||||
| 6 | 4·77 | 0·95 | 24 | 10 | ||||
| 7 | .. | .. | .. | .. | ||||
| 8 | 4·85 | 0·94 | 24 | 10 | ||||
| Total | .. | .. | .. | .. | .. | .. | 168 | 70 |
| Draft, 1·7 inches. | Number of furnaces running, | 7·00 |
| All furnaces working slow. | Number of charges, | 140 |
| Furnace No. 5, one bad charge. | Ladles matte tapped, | 34 |
| Cupriferous material smelted per furnace, 275·6 tons. | ||
| DAILY REPORT—REVERBERATORY FURNACES. AUGUST 19TH, 1908. | ||||||||
|---|---|---|---|---|---|---|---|---|
| Charge. | ||||||||
| Furnace No. |
Coal | Total Smelted |
Calcines | Macdougal Flue-Dust |
Blast Furnace Flue-Dust |
Main Flue-Dust |
Extras | Residues |
| Tons | Tons | Tons | Tons | Tons | Tons | Tons | Tons | |
| 1 | 55·4 | 143·0 | 143·0 | .. | .. | .. | .. | .. |
| 2 | 55·4 | 246·4 | 240·1 | .. | .. | .. | .. | 6·3 |
| 3 | 62·1 | 250·7 | 236·9 | .. | .. | 13·8 | .. | .. |
| 4 | 58·9 | 262·7 | 262·9 | .. | .. | .. | .. | .. |
| 5 | 62·2 | 247·8 | 247·8 | .. | .. | .. | .. | .. |
| 6 | 59·1 | 241·9 | 241·9 | .. | .. | .. | .. | .. |
| 7 | .. | .. | .. | .. | .. | .. | .. | .. |
| 8 | 55·1 | 252·9 | 252·9 | .. | .. | .. | .. | .. |
| Total, | 408·2 | 1645·6 | 1625·5 | .. | .. | 13·8 | .. | 6·3 |
| Delays. | ||||||||
|---|---|---|---|---|---|---|---|---|
| Furnace No. |
Copper Material Smelted per Ton of Coal |
Cost of Coal per Ton of Metal Melted |
Waiting for Coal |
Waiting for Calcines |
Miscellaneous | Total Delays |
Boilers Working |
Ladles of Matte in Furnace at End of Day. |
| Tons | $ | Hours | Hours | Hours | Hours | Hours | ||
| 1 | 2·58 | 1·76 | .. | .. | 8·00 | 8·00 | 22 | 2 |
| 2 | 4·45 | 1·02 | .. | .. | .. | .. | 24 | 8 |
| 3 | 4·04 | 1·13 | .. | .. | .. | .. | 24 | 6 |
| 4 | 4·46 | 1·02 | .. | .. | .. | .. | 24 | 6 |
| 5 | 3·98 | 1·14 | .. | .. | .. | .. | 24 | 8 |
| 6 | 4·09 | 1·11 | .. | .. | .. | .. | 24 | 8 |
| 7 | .. | .. | .. | .. | .. | .. | .. | .. |
| 8 | 4·59 | 1·19 | .. | .. | .. | .. | 24 | 8 |
| Total, | .. | .. | .. | .. | 8·00 | 8·00 | 166 | 46 |
| Draft, 1·7 inches. | Number of furnaces running, | 6·67 |
| Furnace No. 1 delayed 8 hours tapping and claying. | Number of charges, | 118 |
| Furnace No. 7 down for repairs. | Ladles matte tapped, | 47 |
| Bad coal on all furnaces. | Cupriferous material smelted per furnace, 246·7 tons. | |
[Pg 103] Tapping the Furnace.—Matte is usually withdrawn from these large stores upon such occasions as it is required for the converters, though sometimes when the supply has got ahead of the converters’ demands, the matte is tapped and run outside the reverberatory building, being cast into large matte-beds. The tap-holes are situated between the second and third doors, and between the fourth and fifth; and each consists essentially of a copper plate 2 inches thick and 25 inches square, which at first stands back 9 inches from the outside of the wall. Through this plate a 1-inch hole has been drilled. The tapping bar is maintained inserted up this hole, being passed through the conical clay plug which closes it. At the back of the plate is 21 inches of lining material through which the tapping-hole passes. When the copper plate shows signs of a red heat, it is an indication of the lining tending to burn through; this part of the furnace is then cooled, the plate taken out, a 9-inch layer of sand is rammed into position, and the plate is thus moved forward a corresponding distance. Such a tap-hole plate lasts for about five months.
The reverberatories are usually not tapped until they contain about 250 tons of matte. The operation of tapping is performed by withdrawing the rod by means of a wedge and ring, when the matte flows along the launders leading to the ladles for the converters; two ladles of about 8 tons capacity each are usually filled at once, each ladleful being sampled at the runner. The tap-hole is then stopped with a cone of clay, and the tapping-rod driven through it again.
Typical daily reports of the furnaces are appended in Tables VI. and VII., and a monthly report on Table VIII.
TABLE VII.—From Daily Assay Report—Reverberatory Furnaces.
August 19, 1908.
| Furnace Number. |
Per Cent Copper in Slag. | ||
|---|---|---|---|
| Shift 1. | Shift 2. | Shift 3. | |
| 1 | 0·30 | 0·30 | 0·30 |
| 2 | 0·30 | 0·35 | 0·25 |
| 3 | 0·30 | 0·30 | 0·35 |
| 4 | 0·45 | 0·30 | 0·25 |
| 5 | 0·30 | 0·40 | 0·35 |
| 6 | 0·30 | 0·20 | 0·20 |
| 7 | .. | .. | .. |
| 8 | 0·35 | 0·25 | 0·30 |
| Average in slag, | 0·35 | 0·30 | 0·30 |
| Composition of calcines | SiO2, | 29·5 | per cent. | |
| FeO, | 37·3 | " | ||
| S, | 7·7 | " | ||
| CaO, | 2·7 | " | ||
| Copper, | 8·6 | " | ||
Composition of slag, |
SiO2, |
39·4 |
per cent. |
|
| FeO, | 40·7 | " | ||
| CaO, | 4·3 | " | ||
Copper in matte, |
38·6 | "[Pg 104] |
TABLE VIII.—Monthly Report—Reverberatory Furnaces.
Total Charge—All Furnaces.
| Charge. | SiO2. | FeO. | |||
|---|---|---|---|---|---|
| Tons. | Per cent. | Tons. | Per cent. | Tons. | |
| Calcines and lime rock | 50,054 | 27·20 | 13,616 | 39·40 | 19,721 |
| M‘Dougal flue-dust, | 977 | 30·50 | 298 | 21·90 | 214 |
| Blast flue-dust, | 1,639 | 35·90 | 588 | 22·00 | 361 |
| Converter flue-dust, | 132 | 1·90 | 2 | 6·60 | 9 |
| Main flue-dust, | 1,034 | 30·2 | 312 | 17·80 | 184 |
| Total, | 53,836 | .. | 14,816 | .. | 20,489 |
| Matte to converter, | 10,950 | .. | .. | 36·70 | 4,019 |
| Matte chips to B.F., | 74 | 8·20 | 6 | 38·10 | 28 |
| Slag chips to B.F., | 609 | 39·50 | 241 | 37·40 | 228 |
| Deduct from above total, | 11,633 | .. | 247 | .. | 4,275 |
| Leaves for slag, | .. | .. | 14,569 | .. | 16,214 |
| Lime. | Sulphur. | Copper. | ||||
|---|---|---|---|---|---|---|
| Per cent. | Tons. | Per cent. | Tons. | Per cent. | Lbs. | |
| Calcines and lime rock, | 2·30 | 1,150 | 8·40 | 4,205 | 8·266 | 8,274,799 |
| M‘Dougal flue-dust, | 1·30 | 13 | 14·00 | 137 | 7·884 | 152,295 |
| Blast flue-dust, | 4·30 | 70 | 6·70 | 110 | 5·698 | 186,782 |
| Converter flue-dust, | .. | .. | 12·10 | 16 | 68·743 | 181,482 |
| Main flue-dust, | 2·10 | 22 | 8·80 | 90 | 7·128 | 147,405 |
| Total, | .. | 1,256 | .. | 4,558 | 8·305 | 8,942,763 |
| Matte to converter, | .. | .. | 26·40 | 2,891 | 38·209 | 8,367,872 |
| Matte chips to B.F., | 0·20 | .. | 21·80 | 16 | 32·811 | 48,560 |
| Slag chips to B.F., | 2·30 | 14 | 2·20 | 13 | 35·597 | 43,357 |
| Deduct from above total, | .. | 14 | .. | 2,920 | .. | 8,459,789 |
| Leaves for slag, | .. | 1,242 | .. | .. | .. | .. |
| Analysis. | ||||
| Slag Calculation─ | Calculated. | Actual. | ||
| SiO2 | in slag, | 14,569 ÷ 38,538 | 37·8 | 37·1 |
| FeO | " | 16,214 ÷ 38,538 | 42·1 | 43·2 |
| CaO | " | 1,242 ÷ 38,538 | 3·2 | 2·8 |
| ——————— | ——— | ——— | ||
| 32,025 at 83·17 = 38,538 | 83·10 | 83·10 | ||
| ══════════ | ════ | ════ | ||
Fuels for Reverberatory Furnace Work.—The chief requirements of the fuel for good reverberatory work will now be apparent, particularly with regard to length of flame. This depends to a large extent upon the proportion of volatile hydrocarbons, but also on the conditions under which they are given off. For instance, a coal which rapidly parts with its hydrocarbons and leaves in the grate a dense layer of slow-burning coke would be unsuitable for reverberatory work, though some caking is necessary in order that the fuel should not burn away too rapidly, as it should yield a good bed of the required depth.
The great success of large reverberatory furnaces worked under suitable conditions, has had the tendency to tempt smelters in different parts of the world to erect furnaces of similar size independently of the character of the available fuel, and in several cases results have been unsatisfactory, at least in the earlier stages.
These preliminary failures have, however, served the purpose of developing the adaptation of other fuels for this work, and from the employment of oil for the purpose, important extensions in practice will undoubtedly develop in the future of reverberatory furnace working.
The device of using pulverised coal as a fuel has attracted attention at several smelters where the local coal as mined was proved to be unsuitable for use. In practice, however, the method has, up to the present, given unsatisfactory results, for although a longer flame and higher temperature have been obtained in the furnace, difficulties in working have arisen which appear to bar its use. One of the chief drawbacks has been due to the fine ash from the fuel, which is deposited in the flues in large quantities and even causes considerable slagging in them, impeding the working of the furnace and preventing the recovery of heat from the furnace gases. Further difficulty, though not quite so serious, was caused by the dust being blown upon the charge and tending to settle upon it; forming a non-conducting blanket which retarded the melting of the material by the flames. The method does not appear at present to offer much promise of extended application to copper smelting.
Oil Fuel in Reverberatory Practice.—The successful application of oil as a fuel marks a useful advance in reverberatory practice, particularly in connection with the working of large furnaces.
On several of the smaller plants, oil fuel has been in use with considerable success for some time, but within recent years the building of large-sized furnaces without having at hand suitable coal resources has led to attempts to employ oil in its place, and the preliminary difficulties appear to have been to a large extent successfully overcome. The work at the Cananea Smelter with oil fuel, and the discussion on Ricketts’ first report of his experience, afford [Pg 106] valuable indications of the possibilities of this method. Working on charges consisting to a large extent of flue-dust, several thousand tons of material have been smelted in furnaces yielding 245 tons daily output, at a cost which compares very favourably with that of ordinary practice. This success is particularly noteworthy in view of certain features in the preliminary system of working which will doubtless be altered at no very distant date, and of the fact that flue-dust is sometimes a difficult material to melt in a reverberatory furnace, even when good coal is available as a fuel.
The chief difficulties in working appear to have been largely in connection with the regulation of the flame and the management of the oil-burners. In endeavouring to obtain the requisite high temperature over the entire length of the furnace-hearth, an intense local action was caused near the place where the oil in the form of a spray entered [Pg 107] the furnace, resulting in the burning out of the roof-arch on several occasions. These difficulties will doubtless be overcome with further experience in the design and management of the burners constructed for this class of work.
At Cananea, four oil burners of the Shelby type are employed on each furnace, and this form is stated to project the flame further into the furnace, and to prevent its impinging on the roof, more successfully than the other types tried. The waste heat fires three Stirling boilers of 664 H.P. Less than one barrel (42 gallons, or 310 lbs.) of oil is consumed per dry ton of charge, and of this quantity 0·43 barrel is chargeable to steam-raising under the boilers. The manner of working the charges, and the furnace construction in other respects, follow very closely the methods of operation already described.
Costs of Oil-fired Reverberatory Working.—Ricketts has contributed a useful analysis of the costs of reverberatory work using oil as fuel, under the conditions prevailing at Cananea, Mexico. He noted that the use of too much oil should be avoided. This precaution led to a decrease in the amount of repairs necessary. 550 barrels of oil were required to get the furnace into fairly good condition, and 8 barrels per furnace per hour to keep it going well. It is hoped ultimately to reduce the oil consumption to 0·8 barrel gross per ton of charge.
Analysis of Oil-fired Reverberatory Furnace Costs—Cananea—
February to July, 1911, inclusive.
Furnace Days, 312·5.
| TONNAGE CHARGED— | Dry Tons. | Per cent. of Total. |
||
| Flue-dust, | 21,019 | 34·99 | ||
| Calcines, | 35,533 | 59·15 | ||
| Ores, | 3,040 | 5·06 | ||
| Limestone, | 479 | 0·80 | ||
| 60,071 | 100·00 | |||
| ══════ | ══════ | |||
| DISTRIBUTION OF COSTS— | Amount. | Per Dry Ton. |
||
| Operating expenses, | $ 111,687·17 | $ 1·8593 | ||
| Slag and matte expense, | 5,111·07 | 0·0851 | ||
| Boiler-house, | 11,468·77 | 0·1909 | ||
| General expense, | 4,218·58 | 0·0702 | ||
| Cost of flux, | 817·46 | 0·0136 | ||
| $ 133,303·05 | $ 2·2192 | |||
| Steam credit, | 48,861·86 | 0·8134 | ||
| Operating cost, | $ 84,441·19 | [Pg 108] | $ 1·4057 | |
| ════════ | ══════ | |||
ANALYSIS OF COSTS— |
||||
| (1) | Operating— | Amount. | Per Dry Ton. |
|
| Labour, | $ 17,829·42 | $ 0·2968 | ||
| Power, | 592·36 | 0·0099 | ||
| Fuel oil, | 88,028·99 | 1·4654 | ||
| Coal, | 243·61 | 0·0041 | ||
| Water, | 91·68 | 0·0015 | ||
| Transportation, | 380·45 | 0·0063 | ||
| Sundries, | 315·64 | 0·0053 | ||
| Flux, | 817·46 | 0·0136 | ||
| $ 108,299·61 | $ 1·8029 | |||
| ════════ | ══════ | |||
| (2) | Repairs— | |||
| Labour, | $ 11,063·93 | $ 0·1842 | ||
| Supplies, | 12,425·30 | 0·2068 | ||
| Shop expense, | 1,514·21 | 0·0252 | ||
| $ 25,003·44 | $ 0·4162 | |||
| Total, | $ 133,303·05 | $ 2·2191 | ||
| Steam credit, | 48,861·86 | 0·8134 | ||
| Net total, | $ 84,441·19 | $ 1·4057 | ||
| ════════ | ══════ | |||
Gaseous Fuel.—The proposal to employ gaseous fuel in copper smelting dates from the introduction of this method of furnace-firing by Siemens 50 years ago. It is, however, not in general use, although at several smelters gas-firing is employed in furnaces for the refining of the metal.
The chief difficulties have been in connection with the control of the flame, burning-out of the roof having been a not infrequent occurrence when employing gaseous fuel, and the method has been tried and given up at the Great Falls Smelter in Montana, and at several other works.
The practical difficulties ought not, however, to be insuperable should gas-firing be otherwise found most practicable for the particular conditions at the smelter, although there appear to be certain physical characteristics of such flames which may be responsible for some of the difficulties met with in employing this type of fuel for the working of very large reverberatory furnaces.
The Condition of the Charge for Good Reverberatory Work.— The considerations which decide the advisability or otherwise of installing at a smelter, any particular types of furnace, whether reverberatory or blast furnace or both, cover a very wide field, and will be more apparent when blast-furnace practice has been reviewed in detail. It is clear that the blast furnace is unsuited for the direct smelting of fine materials as such, and that the reverberatory form of furnace is [Pg 109] best fitted for their treatment when large quantities of this material require to be dealt with. Actual practice has shown, however, that the reverberatory does not give equally satisfactory results on all classes of fines, and that there are certain physical and chemical conditions of the charge which appear to be necessary for the most successful and rapid smelting. When such conditions are not adhered to, less satisfactory working has resulted. Recent experience has, to some extent, defined more clearly the nature of these requirements, and has indicated the procedure which is necessary in order to avoid an undue supply of the less suitable material for the reverberatory charge.
It is usual to smelt in the reverberatory furnaces, where such are available, the greater portion of the dust which accumulates in very large quantities in the flues at the smelter. The reverberatory is the only type of furnace in which such material could be treated directly, under the present conditions of working. In practice, however, it has been found in several instances, though not universally, that such dust is considerably more difficult to treat in the furnace, and entails considerably more expense in smelting than does the ordinary roasted concentrate. It is estimated by Ricketts that this extra cost is practically equivalent to the expense of roasting an equal weight of concentrate.
Flue-dust, as a rule, consists mainly of material in a minute state of division, in which condition, as is well known, a much higher temperature is required for its fusion than if it were in the form of coarser particles. This is largely due to the poor conductivity for heat which generally characterises such dust, and to the insulation by the air envelopes surrounding the individual grains, which thus prevents the heat passing from particle to particle, and retards their clotting, even when the prevailing temperature would otherwise be sufficient to cause fusion. The particles of flue-dust moreover, have been blown from the surface of the charge, especially in the blast-furnace process, and are thus rapidly and often almost completely oxidised in passing through the oxidising atmosphere which prevails above the charge and in the flues. Such oxides clot only with the greatest difficulty, and are characterised by comparative infusibility and poor conducting power, and hence are found to melt with considerable difficulty when treated in the reverberatory furnace.[10]
Roasted fine concentrate, on the other hand, constitutes an ideal material for the reverberatory furnace charge, and the system of passing [Pg 110] both the concentrate and the flux through the roasters has been shown to possess numerous advantages. In addition to the thorough mixing and the preheating of the furnace charge, it was found that its chemical and physical conditions were particularly well suited for the subsequent reverberatory furnace treatment. The particles of concentrate, being gradually heated and constantly stirred in the presence of the small proportion of flux usually required, roast well, and lose the desired quantity of sulphur without an undue amount of preliminary clotting which would otherwise interfere with the operation, whilst any residual sulphide in the product is uniformly distributed through the roasted charge. In addition, at the higher temperatures which prevail in the later stages of the roasting process when almost as much sulphur as was desired has been driven off, the materials are raised to a point approaching incipient fusion and slagging. The heat in the reverberatory furnace is sufficient to complete this effect, and enable the necessary chemical combinations and physical separations to be readily accomplished.
The roasted concentrate should therefore form the main proportion of the reverberatory charge, working in with it, in moderate quantities, such flue-dust as is made at the smelter. Of this flue-dust, it is naturally desirable to produce as small an amount as possible, not only on account of the difficulties in subsequent treatment, but also on account of the actual losses in the economy of the furnace processes and the cost of rehandling, etc. In modern smelting, naturally, every effort is made to reduce the quantity of dust to the lowest practicable limit.
The greater portion of the dust results from the treatment of unsuitably fine material in the blast furnace, and by decreasing the quantity of this constituent the flue-dust problem will be largely overcome. The smelting of fine concentrate in the blast furnace has up to the present been considered judicious where circumstances have rendered imperative the addition of sulphides to the charge irrespective of their physical condition (either to act as a base for the matte, or on account of their fuel values), though naturally the proportion of fines has been kept as low as possible.
The recent developments in sintering processes, however, suggest the possibility of the future successful treatment, after preliminary agglomeration, of fine concentrate in the blast furnace, and if it be found possible to conduct the sintering by utilising the heat of oxidisation of the more free sulphur atom of the pyrites, and thus leave the bulk of the iron-sulphide fuel values in the sintered product, as suggested by Peters, the difficulties in connection with [Pg 111] excessive flue-dust production from the above causes will be largely overcome, and the reverberatories will thus be relieved of this difficult constituent of their charge.
It therefore appears desirable, when circumstances permit, either to agglomerate fine concentrates and then treat them in the blast furnace, or else to roast them and smelt the product in the reverberatories.
So far as present experience has gone, it appears that—other circumstances being equally favourable—the correct scheme of treatment depends almost entirely upon the composition of the concentrate, there being for each process a particular class of fines for which it is best suited. The sintering process deals most satisfactorily with one class of concentrate, whilst the roasting process seems more particularly suited for a different type of material.
Thus the higher the iron and sulphur values, and the lower the silica content, the more successful, cheap, and efficient is the roasting process—the Anaconda material for example roasts well, requires practically no external fuel or heating, and with the added flux, works very successfully in the reverberatories.
As the silica content increases, however, and the iron and sulphur contents diminish, there is a consequent decrease in the natural fuel values of the material, and as a result, the roasting is neither so efficient nor so cheaply operated, owing to the need of external fuel for giving the required roasting temperatures. On the other hand, it appears to be just this class of material which is best suited for blast-roasting.
It is found in actual working practice that material which does not contain a certain proportion of silica does not work well in the blast-roasting or sintering processes, the resulting product being found to be more irregular in composition and more difficult to operate in the sintering plant. It would therefore appear that a certain class of fine concentrate higher in silica and lower in iron and sulphur contents, which is not quite so suitable for ordinary roasting (owing to the necessity for external heating, due to lower fuel values) is eminently suited for blast roasting or sintering processes, yielding lump products very suitable for subsequent blast-furnace treatment.
The reverberatory furnace thus deals most successfully with fine table concentrates high in iron and sulphur, moderately low in silica; roasted, with its required flux, to the necessary extent, and then charged whilst still red hot into the furnaces. To relieve the reverberatories of the greater bulk of the blast-furnace flue-dust, [Pg 112] which it treats with more difficulty, fine concentrates, as such, require to be kept out of the blast-furnace charge, either by subjecting the more siliceous material to a preparatory sintering process, or by reserving the highly pyritic variety for roasting and subsequent reverberatory treatment.
Peters, E. D., “Principles of Copper Smelting.”
Offerhaus, C., “Modern Reverberatory Smelting of Copper Ores.” Eng. and Min. Journ., June 13, 1908, pp. 1189–1193; June 20, 1908, pp. 1234–1236.
Ricketts, L. D., “Experiments in Reverberatory Practice at Cananea, Mexico,” and discussion, Trans. Inst. Min. and Met., vol. xix., 1909–10, pp. 147–185.
Ricketts, L. D., “Developments of Cananea Practice.” Engineering and Mining Journal, Oct. 7th, 1911, p. 693.
Functions of the Furnace—As Melting Agent—Reduction Smelting—Oxidation in the Furnace—The Pyritic Principle—Features of Modern Practice: Water-Jacketing, Increase in Furnace Size, External Settling—Constructional Details of the Furnace.
The Functions of the Blast Furnace.—The functions of the blast furnace may be considered from three points of view:—
In modern copper smelting practice, the blast furnace is under ordinary circumstances never employed in the capacity of a reducing medium, but is used for a variety of work in which its operations range from those of a melting furnace to those more particularly of an oxidising medium, as its oxidising functions are becoming developed to a gradually increasing extent.
In the older processes of copper smelting, when working on oxidised charges, the melting and reducing functions of the furnace were exercised simultaneously; when, at a later stage, sulphides were smelted in the charge, the directly reducing function was utilised to a very much smaller extent. In the reducing atmosphere then maintained inside the furnace, the sulphides liquated and melted down without causing much concentration of the copper in the product, elimination of sulphur being effected mainly by the direct action of heat on the pyritic constituents of the charge, and by the interactions between the sulphides and the oxidised compounds of copper present.
When, however, increasing quantities of sulphide ore became available, modifications in blast-furnace smelting practice were introduced with a view to increasing the concentration of the copper, this being attempted either by preparatory roasting or by the addition of oxidised [Pg 114] cupriferous materials to the charge, sulphur being thus eliminated and some concentration resulting in consequence. In such work the furnace chiefly exercised its melting function, allowing, as in the case of reverberatory working, of the formation and thorough fusion of sulphide matte and silicate slag from the mixture of oxides and sulphides in the charge. In the latest developments of practice, the oxidation has been carried out to a continually increasing extent by the air blast at the tuyeres of the furnace.
1. The Melting Functions of the Blast Furnace.—The blast furnace is under ordinary circumstances, usually regarded as the cheapest of melting agents. Compared with the reverberatory, the heat in the blast furnace is utilised more efficiently. Reverberatory working involves the passing of a flame over the surface of the charge, and the transference of this heat through the mass depends upon the conducting power of the material itself, which is, however, usually poor. Although the modern reverberatory practice of melting thin layers of preheated charge both from above and from below has greatly increased the efficiency of the furnace in this respect, the closer contact of charge and fuel in the blast furnace allows of a more thorough communication of the heat.
The principal features of blast-furnace working which tend to make it the cheaper and more efficient agent for the treatment of cupriferous materials—with the exception of fines—are those of construction, working, and fuel economy.
(a) The construction of the furnace is comparatively simple, and it is not excessively expensive to erect; furnaces and accessory plant can be purchased complete and easily set up and taken down again when required.
(b) The furnace is elastic in its operation, especially where the supply of material varies from time to time, involving changes in the composition of the charge.
(c) The furnace is readily started, shut down, and restarted at will, and without much difficulty or additional expense.
(d) The operation and smelting are rapid and cheap, the capacity can be made enormously large; all classes of material—except fines—such as ores, slags, and residues, which accumulate to a considerable extent round a smelter, can be conveniently dealt with directly, whilst fines can now, where necessary, often be prepared into a suitable form for blast-furnace treatment.
(e) The heat is more efficiently communicated to the individual parts of the charge, in consequence of the more intimate contact of charge and fuel. [Pg 115]
(f) The fuel consumption is low, the natural fuel values of the iron and sulphur on the charge can be utilised, and the degree of oxidation (and consequent concentration) can be controlled in the furnace operation.
(g) The furnace works continuously (in modern practice the reverberatory furnace is also continuous in its action).
Owing to the great elasticity in blast-furnace operation, and its capability of dealing with practically every class of copper-bearing material in lump form, modern practice is of the most diverse character.
2. The Blast Furnace as a Reducing Medium.—In modern smelting practice, with but a few exceptional instances, a distinctly reducing atmosphere is avoided as far as possible. This arises largely from the fact that the material available in modern work usually demands oxidation in order that satisfactory concentration may be effected.
In the early days of copper smelting, however, the reducing action was the chief function which was exercised, mainly because at that time oxidised ores constituted an important part of the charge, and a reducing action was required to obtain marketable products from such material. At a later stage in the development of blast-furnace practice, the sulphide ores which became available were roasted, and the resulting oxidised products were subjected to reduction smelting, in order to extract the metal. On such oxidised charges, blast furnaces were almost universally employed, using carbonaceous fuel either in the form of coke or charcoal, this material fulfilling the double purpose of fuel and reducing agent, the excess carbon causing the reduction of the metal from the oxidised ore.
This operation was known commonly as “black-copper smelting.” At the present time such oxidised ores are rarely met with in sufficient quantity by themselves to be worked by this method, which involves also very serious losses in operation. Further, such oxidised materials are in many cases valuable for smelting along with sulphide charges, greatly assisting the concentration, and it is usually advantageous to employ them in this manner.
The losses and difficulties in “black-copper smelting” are, however, of interest in so far as they apply to certain analogous problems in modern work. These difficulties in reduction smelting arose largely from three causes:—
[Pg 116] (a) In the case of reduction smelting where sulphides are not present in any appreciable quantity, the losses of copper may be either
(i.) Sulphur is the natural protector of the copper in the furnace charge, as, owing to their powerful affinity, a fusible, fluid and dense product is formed, which is very slightly soluble in slag; and on this account, a ready separation of the copper from the earthy materials can be effected. So long as sulphur is present in moderate quantity there is little chance of copper entering the slag as silicate.
In reduction smelting, however, and especially in black-copper smelting where sulphur is lacking, such losses are liable to occur, since copper oxide is itself strongly basic, and readily fluxes off with silica at high temperatures, yielding silicates. These products are less dense, and are markedly soluble in the other silicates which constitute the slag; moreover, the copper oxides themselves are likewise partly soluble in, and are readily carried in suspension by, the silicate slags.
In order to prevent such losses as much as possible, the reducing conditions in the furnace must be increased by the employment of more coke, so as to ensure the reduction of the copper oxides and silicates. These reducing conditions must not, however, be too drastic, especially if the temperature of working be high, on account of the great tendency to cause (b) a reduction of metallic iron, which results in the formation of bears and scaffolds, with their attendant difficulties of removal and their interference with working.
Between these opposing causes of loss and difficulty, a careful balance has to be observed in the smelting operations. (In modern practice, losses of copper as silicate and oxide, for reasons such as those detailed above, occur to a marked extent in those operations where the sulphur is present in small proportions only, and particularly where the reactions are intensely oxidising, as in the furnace-refining operations and the later stages in the converter process. The slags in such cases usually carry considerable quantities of copper in the form of silicate and oxide, not infrequently to the extent of 20 to 30 per cent., or even more. The quantity of this slag is, however, kept as small as possible, and copper in the material is readily recovered by the addition of these slags to the blast-furnace charge.)
(ii.) Losses of copper as metal also, were formerly serious in black-copper smelting, the metallic copper held in suspension in the slag being indeed the chief source of loss in this method. The efficient [Pg 117] separation of copper from slag, especially in the small quantities formerly operated, was therefore of importance. Satisfactory settling was, however, difficult of application, since the behaviour of metallic copper is very different from that of sulphides. It is much less fusible, much less fluid, and the small globules, as reduced, do not readily coalesce, whilst the high temperatures favourable to good fluidity of the products and to good settling, promote copper losses from the other causes noted above.
Moreover, the high melting point of the metal and its great conductivity added to the difficulties in providing suitable arrangements for settling, since the copper not only tended to chill readily in any external settler, but it was also very liable to do so in the crucible of the ordinary form of water-jacketed blast furnace, such masses being exceedingly difficult to remove, whilst the working of the furnace was necessarily much interfered with.
In order to conduct the necessary internal settling, the older type of blast furnace was required, in which water-jacketing near the hearth was dispensed with, a large crucible bottom of non-conducting brasque or brickwork being employed instead. Such a form of furnace is not adapted to the modern methods of smelting where enormous capacity and output are essential, whilst such a system of working interferes with the rapid and continuous smelting of large quantities, to a greater extent than if the whole of the molten products are run out of the furnace continuously and the settling performed in an external vessel.
3. The Blast Furnace as an Oxidising Medium: Sulphide Ores in the Blast Furnace.—In modern blast-furnace practice, the oxidising function of the furnace is the principal feature of working. Sulphide ores now constitute the chief source of copper, and the smelting operations involve the oxidation of the accompanying constituents and the elimination of the resulting oxidised products.
Such ores when smelted in the blast furnace with carbonaceous fuel, and under the reducing conditions characteristic of the older methods of working, would yield a product showing low concentration of the copper, since the reducing conditions would largely retard the oxidation of sulphur which is an essential for the enrichment of the matte. Except for the sulphur eliminated from the pyritic constituents by the direct action of heat, and a certain quantity by the interactions with oxides as already indicated, the loss of sulphur would be slight. The furnace under such circumstances would thus tend mainly to exercise its melting function, and the result of such working would be the melting down and subsequent separation of the sulphides and slag, with even less [Pg 118] tendency to concentration than occurs in the reverberatory furnace, where the atmosphere is less distinctly reducing.
The modern method of smelting sulphide ores being essentially an oxidising process, it is necessary that oxygen be added to the charge with the object of promoting the elimination of the sulphur and iron, and the consequent concentration of the copper.
This oxygen may be added in one of three ways:—
A. Addition of oxygen to the charge previous to the blast furnace smelting operation
(Roasting).
B. Addition of oxygen to the charge during the smelting operation itself.
i. By adding oxidised materials to the charge (Blast-furnace smelting with carbonaceous fuel).
ii. By using the air blast of the furnace for oxidising the iron and sulphur, thus at the same time utilising these elements as fuel and proportionately diminishing the amount of carbonaceous fuel required (The pyritic principle of smelting).
A. Roasting practice has already been discussed, and the reasons for avoiding the operation where practicable, on account of the expenses of an extra process, the losses involved, the fineness of the product, and the loss of fuel values, have been indicated (Lecture IV., pp. 66–80).
B. i. Addition of Oxidised Charges in the Blast Furnace.—The tendency for oxidised cupriferous materials to interact with sulphides finds useful application in copper smelting, since it assists the concentration of the copper in the resulting mattes. The principal reactions involved in this method are—
2CuO + Cu2S ➡ 4Cu + SO2
2Cu2O + Cu2S ➡ 6Cu + SO2
CuSO4 + Cu2S ➡ 3Cu + 2SO2
whereby copper is produced and sulphur is eliminated as SO2. The liberated copper interacts with the excess of iron sulphide usually present in the furnace charge, and enters the matte as sulphide, whilst the iron which is thus set free is oxidised and carried into the slag as silicate, the ultimate reactions being indicated approximately by the equation—
2Cu + FeS + xFeS ➡ Cu2S . xFeS (matte) + Fe (oxidised and enters slag).
[Pg 119] Copper silicates readily interact with iron sulphides in the charge, producing copper sulphides and iron silicates, thus—
Cu2O . xSiO2 + FeS ➡ Cu2S (enters matte) + FeO . xSiO2 (enters slag).
6(CuO . xSiO2) + 4FeS ➡ 3Cu2S (enters matte) + 4(FeO . xSiO2) + 2xSiO2 (enters slag). + SO2.
All the above reactions lead to an enrichment of the matte in copper contents, and at the same time, to the transference of iron from the matte to the slag, and although the conditions in the more reducing atmosphere of the coke-fed blast furnace are not so favourable to the fullest operation of these reactions as are the more neutral conditions of the reverberatory, the addition of oxidised materials constitutes a valuable means of increasing the concentration in this method of smelting.
The blast furnace is thus also particularly suited for the recovering of the copper from the oxidised residues, such as converter slags and scrap, “calcine-barrings,” and the like, which accumulate in very considerable quantities at a smelter, and which by reason of their carrying much copper as oxide or silicate, not only add their quota of copper to the products, but materially assist the concentration and the furnace operation generally.
B. ii. The Pyritic Principle in Blast-Furnace Smelting.—This is the most important principle introduced into modern blast-furnace smelting practice.
It has been evolved by the application of the results of experiments conducted from two different points of view—one series mainly on a laboratory scale, the other from actual industrial practice.
Starting from theoretical considerations, John Holway demonstrated by experiment that the heat of oxidation of the iron and the sulphur of pyritic copper ores was so great as to make their smelting a self-supporting operation under suitable conditions. On the other hand, within comparatively recent years, smeltermen as a result of working practice, have found that an increase of sulphides on the furnace charge has led to less and less carbonaceous fuel being necessary for the smelting operations, providing that the conditions in the blast furnace be sufficiently oxidising.
In utilising these results for general blast-furnace practice, the extended and successful application of this pyritic principle has led to marked advance in modern working.
The results obtained in a series of trials at the Keswick smelter, California, are typical of such experiments on a practical scale, and in spite of the two anomalous instances, the general effects of the increase of sulphides in the charge are strongly marked (see Table IX., p. 120). [Pg 120]
TABLE IX.—Effect on Coke Consumption of
Increased Sulphur in the
Furnace Charge
(Keswick Smelter, Cal.).
| Sulphur in Charge. | Coke Consumption. | ||
|---|---|---|---|
| 6·8 | per cent. | 15·7 | per cent. |
| 7·7 | " | 16·3 | " |
| 13·6 | " | 10·2 | " |
| 17·0 | " | 7·7 | " |
| 19·5 | " | 8·5 | " |
| 22·8 | " | 7·1 | " |
| 24·5 | " | 6·8 | " |
Recent practice at Anaconda affords another instance of the utilisation of the pyritic principle. A large quantity of the ore available (known as second-class ore) requires wet dressing before it can be treated most profitably at the furnaces, and the operation thus produces considerable quantities of sulphide concentrate, of which a moderate proportion is coarse—well suited for blast-furnace treatment. The charge if submitted to reduction smelting with carbonaceous fuel, would yield a matte too low in copper contents for immediate converter treatment, since there is not available a sufficient supply of oxidised cupriferous material to effect a high enough concentration for the direct production of a converter-grade matte. Instead of roasting so as to reduce the sulphur contents to the required degree, and then smelting with the usual amount of carbonaceous fuel, the pyritic principle has been utilised to the fullest possible extent, by smelting the raw charge containing as much of the coarse concentrate as is available, with a strongly oxidising blast, thus effecting the desired concentration, and occasioning the use of a lower coke proportion than would otherwise have been necessary. By gradually increasing the sulphide on the charge until the sulphur proportion reached 8 to 9 per cent., the coke consumption was reduced to about 11 to 12 per cent. During the past two or three years the advantages of introducing more and more sulphide have become so apparent, that increasing quantities of ⅜ inch concentrates are being included in the charge, and although such material is exceedingly difficult to deal with in the blast furnace, the advantages arising from its use outweighs the trouble it causes in actual working. By this further increase of the sulphur [Pg 121] proportion, from the former 8 to 9 per cent. up to 11 to 12 per cent., the coke consumption has been steadily reduced until it now amounts to about 9 per cent. only.
The fuel value of the iron and sulphur is augmented at a rate much greater than their actual increase in numerical proportion would suggest, on account of the much higher calorific intensity of large and massive quantities of fuel burned at once than that resulting from smaller amounts disseminated throughout a mass of inert material such as gangue.
The practical application of the pyritic principle to blast-furnace practice thus involves the employment of the furnace as a medium for conducting the required oxidation of the charge, as a result of which, the heat of this combustion proportionately reduces the amount of carbonaceous fuel required for the smelting and separation of the products, whilst at the same time the desired concentration is also effected. The basis of such working is, therefore, the powerful oxidising action within the furnace itself, and the fullest utilisation of the heat resulting from this oxidation of the sulphides.
In order to supply the heat necessary for the reactions and fusions of smelting, a definite quantity of fuel is essential in the furnace. In those cases where the proportions of sulphide are not sufficient to supply the required amount, a supplementary quantity of coke fuel becomes requisite.
The extent to which coke is necessary for the smelting operations decides whether the process may be termed “true pyritic” or “partial pyritic” smelting. In the former case, the coke allowance may be reduced to such small proportions that its influence in the smelting zone of the furnace is practically negligible.
In partial pyritic smelting, coke is necessary to the extent of supplementing the heat derived from the sulphide fuel, and the proportion employed in modern work is reduced to the lowest possible quantity. Not only is economy in coke allowance one of the chief essentials in furnace management, but the presence of a larger amount than is absolutely necessary decreases the efficiency of the smelting operations, since, owing to its reducing action and its consumption of the oxygen in the air blast which is to be utilised for the combustion of the iron and sulphur, the concentration of the copper in the resulting matte would be decreased.
The extent to which the pyritic principle may be operated in actual working depends in the first instance upon the nature of the charge itself, especially upon the relative proportions of copper, iron, and sulphur, and on the quantity of gangue. Since these vary in the ore [Pg 122] supply of different localities, the extent to which the principle may be applied and the coke consumption be reduced, will be subject to alteration accordingly.
Thus in the case of an ore which contains such proportions of these constituents as would on simple melting yield a matte of converter grade, the pyritic effect in the furnace would necessarily be very small, and the smelting would be almost entirely a melting operation requiring from 10 to 15 per cent. of coke on the charge, even though the sulphur contents of the charge be high. Ores and charges of such a composition are, however, rarely met with in modern practice, the ratio of copper to iron sulphides usually being low.
On the other hand, in the case of an ore consisting largely of iron sulphides with but little copper—i.e., a massive low-grade pyritic ore—the pyritic effect in the furnace might reach a maximum, and the coke required on the charge be reducible to very small proportions. Such material is well suited for true pyritic smelting.
Hence modern practice ranges from the true pyritic smelting, where pyritic fuel is principally employed, through varying degrees of partial pyritic smelting, where the pyritic fuel is supplemented to the required degree by coke, to reduction smelting, relying mainly on carbonaceous fuel for the necessary heat supply.
In all cases, the object of the operation is to oxidise inside the furnace so much sulphur and iron as is necessary to yield a matte product of converter grade, utilising the natural sulphide fuel values of the material so as to reduce to the lowest possible proportion the quantity of coke required.
Features of Modern Practice.—Apart from the applications of pyritic smelting, which will be considered separately, three features of great importance have been introduced into modern blast-furnace working. These involve:—
[Pg 123] A. The Practice of Water-jacketing.—The evolution of the blast furnace from the primitive hole-in-the-ground form to the modern type may be rapidly sketched. In its early stages, the development was carried out mainly on the Continent of Europe, following the course of the enclosing of the charge in shafts which became of gradually increasing height, the introduction of blast through tuyeres near the bottom of the shaft, and the arrangements for collecting the molten materials in the hearth, and for tapping. By the year 1850 a typical form of furnace was represented by the Mansfeld pattern, which consisted of a rectangular firebrick shaft enclosed by massive stonework. At the lower extremity was a hearth constructed of refractory material, usually of brasque—a mixture of fireclay and coke—well tamped down. The dimensions were from about 2 feet to 2 feet 6 inches broad, 14 feet to 16 feet high, with two tuyeres of 1½ to 2 inches diameter, supplying blast at 4 to 10 inches water pressure; the capacity of such a furnace being about 4 tons per twenty-four hours. It is of interest to note that this form of furnace possessed arrangements both for internal or external settling of the products, the usual practice being, however, to allow the smelted material to collect and settle in the hearth. In endeavouring to increase the capacity of the furnace and the rapidity of working, as well as to ensure efficient settling of the products, it became necessary to maintain a high temperature in the lower parts; but in consequence of the excessive heat and the corrosive nature of the molten materials, the most refractory brasquing available was rapidly attacked, and the necessity for adopting means to prevent the destruction of the furnace linings became apparent.
The use of water-jacketing for this purpose had long before been applied to certain branches of cast-iron refining, and in 1875 the Piltz water-jacketed blast furnace was introduced for the smelting of lead ores. This form of furnace was circular in horizontal section, and the boshes consisted of two concentric shells between which a stream of water circulated. This principle was quickly adopted for the purposes of copper smelting furnaces, although modifications were found to be necessary in certain particulars before perfectly successful working was achieved. Owing to the higher temperatures prevailing in the furnace, the height to which the water-jackets were carried required to be increased, and it was chiefly when the rectangular form of furnace was introduced that the thoroughly successful application of water-jacketing was accomplished. This feature in blast-furnace work was rapidly and very successfully developed by the American copper smelters when the new establishments in the West were opened up, and the substitution of the older form of lining by metallic water-cooled jackets, which in comparison are practically indestructible, immediately led to an enormous improvement in smelting practice.
The modern blast furnace is essentially a water-jacketed shell from charging floor to base plate, rectangular in plan, and completely sectionised.
Many of the advantages of such a furnace construction are apparent, and have been referred to in discussing the furnace as a melting agent. The salient features of the modern water-jacketed furnace are:— [Pg 124]
(i.) Water-jacketed furnaces are planned, constructed, and erected simply and with ease.
(ii.) The first cost of the furnace, making allowance for excavation and foundations, is not unfavourable to the water-jacketed furnace, whilst the ease of fitting and the interchangeability of parts due to sectioning, reduce the costs of erection.
(iii.) The convenience and simplicity in operation of the water-jacketed furnace are very marked, whilst the permanence in the shape tends to greater uniformity of working and to ease of management.
(iv.) Accretions and the general difficulties of working are readily dealt with and controlled, barring and other operations being more conveniently conducted.
(v.) The repairing of water-jacketed furnaces is rendered very simple, cheap, and rapid in operation, the principle of sectionising, allowing of the ready removal or replacement of the jackets for repairs; the saving in time, labour, and general expense being particularly marked.
(vi.) The elasticity of the furnace, both as regards size and management, has been enormously increased, and the successful extension and working of the large modern furnaces have only become possible with the adoption of this feature.
(vii.) Water-jacketing has allowed of the rapid driving of furnaces, leading to an enormous increase in the output per square foot of hearth area, by permitting intense heating inside the furnace, and rapid withdrawal of the molten products.
The chief consideration affecting the adoption of water-jacketing in any locality might be the scarcity or unsuitability of the water supply, which may necessitate a choice between the employment of brick furnaces, or the crushing, roasting and reverberatory treatment of the ore. In cases where the water supply is not well suited for jacketing purposes, settling or other preliminary treatment of the water might be required.
The former objection to water-jacketing on the assumption of valuable heat being carried away by the jacket water, thus involving a waste of fuel, has proved to be groundless in practice; with good management such heat losses are smaller in amount and less damaging in effect than those due to radiation from highly heated brick walls, quite apart from the actual necessity for such jacketing in modern furnace construction, even had such losses been marked.
B. The Development in Furnace Size.—The blast furnace increased but slowly in size during the nineteenth century up to 1850, and the dimensions of the most advanced type did not exceed 4 feet by about 2 feet 6 incites internally at the tuyere level, the capacity being about 4 tons per day. Furnaces at this period were usually square or circular in section.

Fig. 33.—Blast Furnaces under Construction, showing Fixing of Jackets,
Bottom Plate,
Method of Support, Sectioning, etc. (T. E. Co.).
[Pg 125] The size of such furnaces was largely dependent on the penetrating power of the blast, and a slight increase in cross-section resulted gradually, as improvements in the mechanical contrivances for producing blast were developed. This, however, soon reached a limit, owing to the difficulties in making the blast penetrate to the centre of the charge in the wider furnaces, and to the disproportionate costliness and increased working difficulties attendant on such practice. It was further found that the high pressure required in order to force the blast through an increased width of charge produced an intense local heating effect against the tuyeres, resulting in high slag losses and low concentration on smelting, whilst the consumption of fuel was much increased.
An important modification in blast-furnace design was introduced in 1863, when the principle of increasing the size of the furnace in direction of its length, whilst maintaining the width which had been found best suited to economical working, was applied by Rachette. This was first intended for the purposes of lead smelting, but the principle was quickly recognised as having important applications to copper smelting practice, and was readily adopted and developed. It has become the basis of all subsequent modern copper blast-furnace design, and the gradual increase in dimensions up to the enormous blast furnaces with huge outputs of the present day has been made by extending the length whilst maintaining a relatively small width.
For some time development proceeded along these lines slowly and with much caution, chiefly owing to the difficulties anticipated in the management of such large units. Up to 1885, the largest blast furnace (at the Parrott Smelter, Butte) was but 8 feet long by 36 inches wide; by the year 1900 the dimensions had reached 10 feet by 42 inches. Subsequently, under the direction of the remarkably enterprising management of the Washoe Smelter at Anaconda, a wonderful era of furnace extensions was commenced, and is indeed, still undergoing development. [Pg 126]
Here in 1902, blast furnaces 15 feet long by 56 inches wide were erected, the plant eventually consisting of seven such furnaces built in a straight line, and situated 21 feet apart from each other. A largely augmented ore supply subsequently coming to the smelter for treatment, an increased furnace capacity was required, for which only a very limited suitable space was available. Mr. E. P. Mathewson, the smelter superintendent, determined upon attempting the revolutionary idea of joining up two of the 15-foot furnaces by bridging over the 21-foot space between them, and continuing the vertical side [Pg 127] water-jackets across this space, thus forming a furnace 15 + 21 + 15, or 51 feet in length. No work on such a large and boldly conceived scale had ever been attempted before, and many difficulties in construction and operation were anticipated.
Mathewson first conducted a series of constructional trials, and found in the first instance that by taking suitable precautions, it would be possible to carry out these changes whilst the furnaces themselves were running. It was found that it was possible to remove or replace single jackets without shutting down the furnace, by the device of forming a crust against such a jacket, of sufficient thickness to bear the weight of the charge for the short period of time during which the change was being made. Such a crust is readily obtained by shutting off the tuyeres in the particular jacket and in its neighbours, and maintaining a rapid stream of cold water through these jackets. Further, it was found that any desired portion of the sides or hearth of such a long furnace could be well barred and cleaned whilst the rest of the furnace was in operation, whereas such barring and cleaning on a small furnace seriously interrupted the working, and reduced the capacity.
The preliminary tests being satisfactory, the necessary constructional work was carried out whilst the two furnaces were in blast; the inner end jackets of these furnaces were taken down, and in a short time the new 51-foot furnace was in regular operation, and proved so remarkably successful that two other pairs of furnaces were similarly joined up. In the following year a still further great extension was made by joining up in a like manner the end 51-foot furnace to the last remaining 15-foot furnace, by again bridging over the intervening 21-foot space, thus constructing a furnace of the enormous length of 51 + 21 + 15, or 87 feet.
It was at one time intended to carry this progress still further by joining up the other two 51-foot furnaces, so as to make a single one 123 feet in length, but certain difficulties in the matter of bringing coke supplies to the two sides, under the special conditions of available floor space, and the disastrous effects of the financial panic of October, 1907, stopped all extension work for the time. Such extensions would however, present no real difficulties either in construction or in subsequent furnace management or operation.
Figs. 35 and 36 indicate in plan and elevation the arrangement of the plant and accessories for these extended furnaces. Each 15-foot furnace had its own settler situated in front, and these have been retained without any change of position or any further additions. The hearth of the newly bridged portion slopes from the middle of the bridge to the tap-holes of the old furnaces, which still serve this purpose for the larger ones, and from which a continuous stream of matte and slag flows through a slag spout to the settler in front. The side water-jackets of the old furnaces remain, being built up in two sets of panels, each 7 feet 6 inches wide, whilst the new bridge portions are constructed of three sets of jackets, each 7 feet wide. [Pg 128]
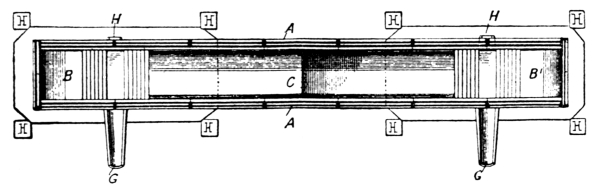
Fig. 35.—Plan of 51-foot Blast Furnace, Anaconda, indicating Position of Crucibles,
Spouts, and Connecting Bridge between Old Furnaces.
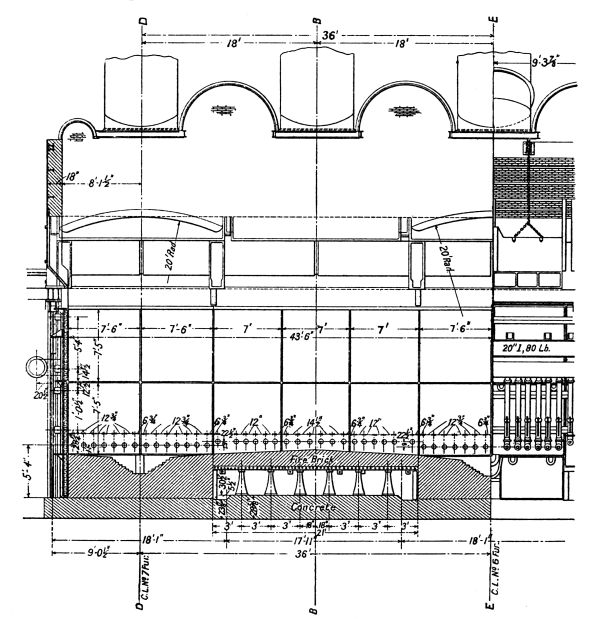
Fig. 36.—Longitudinal Section and Part Elevation of 87-foot Blast Furnace,
Anaconda, indicating Crucibles of Old Furnaces, Bridge, and Jacketing.
[Pg 129] The furnaces in their lengthened form have proved a tremendous success, far indeed beyond the anticipation of the designers and managers. This is largely due
(a) To the increased efficiency and economy of replacing a number of smaller furnaces situated end to end by a single large furnace;
(b) To the increased intensity of heat and reactions owing to large massed quantities of fuel burned at once, and to large masses of material being smelted and in a state of chemical activity.
The advantages which result from such lengthening of blast furnaces are:—
(i.) Gain in hearth area without extension of the blast-furnace floor and building.
(ii.) Increase in smelting or hearth area and in consequent capacity, at a rate very much superior to the extra water-jacketing involved. Thus, in the 51-foot furnace, the capacity has been increased in the proportion of 3·8 to 1, the jacketed surface has increased only at the rate of 2·4 to 1. The output has increased at a much greater speed than was actually anticipated from the additional hearth area.
(iii.) A very marked saving of fuel. The amount of coke required for similar charges has been reduced by one-tenth; more than 11 per cent. was required formerly on a charge, only 10 per cent. was necessary under the new conditions.
(iv.) The rapidity of working of the furnace has increased owing to the effect of the narrow width and small crucible dimensions as compared with the length. This has caused a more rapid flow through the furnace slag-holes, thus preventing the formation of obstructions, and tending to wash out any which might threaten to stick.
(v.) Higher furnace temperatures result, and both slag and matte are hotter than in smaller furnaces. In consequence more siliceous slags can be run, thus saving the cost of the fluxes which might otherwise be necessary.
(vi.) Marked decrease in incrustation. Crusting is most likely to occur at points where the smelting activity is lowest, and in the cooler parts of the furnaces, such conditions being usually prevalent at the corners, where the shape also assists in the holding up of material. Crusting is one of the chief troubles to be prevented and overcome in operating the blast furnace.
The elongated furnace of 87 feet length practically takes the place of five shorter ones, representing no less than 20 corners and 10 end jackets; the new furnace thus reduces the opportunities for crusting at [Pg 130] least five-fold. In this way the hearth area has been very greatly increased, with still but two ends to hold crusts. The long furnace-walls with their ends so far apart, in addition, offer much less opportunity for the formation of crusts than do the side walls of shorter furnaces, accretions obtain little support, and often tend to break down under their own weight, whilst they can be more readily removed by barring, on lowering the height of the furnace charge for a time.
(vii.) The elasticity of the furnace operations has been much increased. In short furnaces, cleaning and barring for the removal of obstructions, etc., necessitate the shutting down of the unit, often a complete taking down of the furnace-walls and their subsequent replacement, followed by a re-starting of the furnace work. The ideal in modern work is continuous running of the unit. The larger furnaces allow of such practice, since they can be kept in operation whilst a particular portion is undergoing cleaning or repair. As stated above, the elongation of the furnaces themselves was conducted whilst the older 15-foot portions were working. Leaky or worn-out jackets or spouts are readily removed without serious interference with the working of the rest of the furnace, and this operation usually requires a few hours only.
(viii.) The charge may be varied in different parts of the furnace to suit special requirements, without interfering with the general operations. Thus, suitable additions for the smelting out of crusts, or variations in the charge to reduce corrosion near the 21-foot bridge, can be effected whilst the furnace is running as usual.
(ix.) Increased flow of material through the settlers is effected without decreasing the efficiency of the settling. Each settler now serves 25 feet of furnace-hearth length, instead of the 15 feet of the smaller furnaces, and in spite of the more rapid passage of the materials, the settling is actually better and the resulting slag cleaner, owing to the higher temperatures of working and the consequent greater liquidity of the products, whilst the settler is also hotter. Thus the greater output of material has required no extra labour or construction on the tapping floor, though tappings are now more frequent.
(x.) The labour costs per ton of furnace capacity are greatly reduced, as are also the operating and management costs, since such labour and control are to a large extent dependent on the number of units comprising the plant.
(xi.) The initial cost, per ton of furnace capacity, is also much reduced. In the elongated furnace, the settlers have not been added to, [Pg 131] the old slag notches only are required to do duty as before, and the older equipment for bracing and trussing provides for much of that required in the extensions whilst the original building itself served for the housing of the increased furnace area.
(xii.) Further extension of the furnace length is readily possible if desired.
The older 15-feet furnaces had a smelting capacity of 5·6 tons per square foot of hearth area per day, those of 51 feet length smelt on an average 6·72 tons per square foot daily, whilst the output of the 87-foot furnace amounts to 3,000 tons of material daily, corresponding to 3,000 ÷ 87 feet × 4 feet 8 inches, or about 7·5 tons per square foot of hearth area. Whilst this particular smelter is of course unique in the dimensions, equipment, organisation and management of its plant and the magnitude of its operations, and though at most modern smelters the ore supplies and smelting conditions do not admit of the introduction of such enormous units; at the same time the principles which underlie the great advantages of the longer form of blast furnace have had an important influence on blast-furnace equipment and design generally. The constructional details of these large furnaces are, for the most part, common to all modern blast furnaces; it is mainly the size and capacity which are exceptional. The usual length adopted at smelters with more modest output varies from about 15 to 25 feet, with a smelting capacity of from about 400 to 800 tons per twenty-four hours, depending naturally on the working conditions.
C. The Practice of External Settling.—In connection with modern blast-furnace practice, the feature of external settling is of much importance, its adoption having had a marked influence on:—
(a) The function of the blast-furnace plant is the concentration of the values into a matte of correct grade for further treatment, and the production of a slag which is sufficiently clean—that is, free from copper and other values—to allow of its being disposed of as waste, immediately. Numerous factors decide the copper contents of the slag which is economically the cleanest—the general average is about 0·25 to 0·35 per cent. of copper. The actual condition of the copper in the slags is a matter of some uncertainty, and it does not appear improbable that very small quantities of sulphides may actually be in [Pg 132] solution in the silicate slags. The general consensus of opinion, however, favours the view that much of the copper which is present exists in the form of minute shots of the matte, actually held in mechanical suspension, and this is certainly the case when the copper contents exceed the limits stated above. In consequence, it is frequently noted in practice that the copper in the slag increases with the grade of the matte. The question has been reviewed by L. T. Wright who suggests some actual solubility of matte-products in the slag. Wright’s curve indicating the connection between matte-grade and slag values is reproduced in Fig. 37. This connection might however, possibly result from the fact that the individual shots of matte are themselves higher in copper contents, since it may be assumed that in fairly clean slags practically the same number of shots are held up, owing to the forces of capillary attraction and surface tension, and that the increased density of the higher grade mattes would influence but slightly their downward settling when in such a fine state of division.[11]
[Pg 133] The molten products of the blast-furnace operation are separated by the settling of the matte and slag under the action of gravity, and the production of the economically cleanest slag depends upon the fulfilment of those conditions which allow of the most perfect downward settling of the small particles of matte. The three main requirements for efficient settling, apart from the composition of the slag, are:—
In each of these essentials, the method of external settling, as now conducted at modern smelters, best satisfies the conditions required for successful work.
The present practice is to make no attempt to conduct settling in the blast furnace, but to run the products through and out of the furnace with the greatest speed attainable, and to allow the matte and slag sufficient time and opportunity to settle and separate in some independent and external vessel, which stores the matte and allows the clean slag to run straight away to waste.
The former method of inside settling gave rise to many difficulties in practice, but objections were urged against the external settler, to the effect that heat might be wasted by the abstraction of hot materials from the furnace to an exterior vessel, and that the settling would not be efficiently conducted outside, as in the very hot interior of the smelting furnace. Modern practice has proved conclusively that both objections are groundless. Such heat as is carried away by the continual stream of molten material can usually be well spared in the modern plant, which is driven so rapidly that an abundant supply of exceedingly hot matte and slag pass through to the settler, whilst the results of every-day working demonstrate the efficiency of the external settler, which cannot be equalled, far less surpassed, by any method of inside settling, under modern smelting conditions. Thousands of tons of slag pass daily through the settlers, clean enough to discharge straight to the dump, the copper contents rarely exceeding 0·40 per cent.
(b) The modern conditions of rapid working and large output render the use of external settlers practically essential, owing to the double work of smelting and separating being no longer confined to one and the same vessel. The aim in present practice is to exercise the smelting function only of the furnace, and to do so to its fullest capacity, smelting for matte of the desired grade as rapidly as possible, and therefore running the products through the furnace in a constant rapid [Pg 134] stream and allowing them to settle quietly outside. Under these circumstances the furnace itself smelts most economically and efficiently.
It will be recalled that present-day practice involves the subsequent treatment of the fluid matte—product in the converter, so that whilst the former methods of working might have possessed certain advantages for the settling and storing of matte in the small furnaces, and then tapping out and casting into cakes for subsequent treatment, such methods have practically no application to modern systems of working.
Internal settling almost invariably leads to the accumulation of debris, of chills and of any infusible masses of material which may be produced in the furnace, occasioning delay in the operations, waste and difficulty in working, and so interfering seriously with the speed and continuity of the smelting, as well as decreasing the output of the furnace. On the other hand, a rapid flow of hot molten material through the furnace not only tends to prevent this formation of chills or accretions, but greatly assists in the dissolution or removal of such as might be formed. Should the production or collection of such masses be transferred to the settler instead, they are more readily attacked and remedied without interfering with the continued operation of the furnace.
Further, the nature of the hearth which would be most satisfactory for internal settling is not at all suited for modern smelting conditions. The ordinary water-jacketing would have too marked a cooling effect on the hearth for the materials to remain sufficiently hot and fluid to allow of proper settling, whilst a brasque or similarly lined hearth suitable for such settling would, under the present conditions of rapid driving and intense reactions, be unable to withstand the highly corrosive and abrasive action to which it would be subject, so that breakouts, necessitating delays and repairs, would constantly occur. Water-jacketing in this portion of the furnace is indeed an essential for modern conditions, and consequently rapid driving and quiet internal settling in the same area are quite incompatible. The modern fore-hearth, on the other hand, is accessible and easy of repair, and in the event of any trouble occurring therein, the furnace itself can continue its smelting activity to the full, since other suitable arrangements can readily be made for temporarily dealing with the products.

Fig. 38.—Water-Jacketed Blast Furnace (48 inches by 240 inches). Lower Portion,
indicating Air and Water Connections, Bottom Supports,
End Slag Spouts, etc. (P. & M. M. Co.).
[Pg 135] (c) The functions of the blast furnace in the modern smelting scheme are particularly dependent upon the employment of the external settler in conjunction with it. The work of the furnace plant is to produce as rapidly as possible, a supply of suitable grade matte for the converters; large quantities of hot fluid matte must be available at a moment’s notice, and such demands are often very erratic, being dependent on the working of the converter plant and the refining furnaces. It is essential to the successful operation of the blast furnaces that the manager should be in a position to work his furnace as rapidly and continuously as possible, which is best attained by making the output independent of irregular tappings of matte just when required by the converter department. The settlers, in exercising the function of reservoirs for matte, from which the converter department may draw at will, allow of regularity of working and rapidity of output in a manner possible in no other way. The only alternative, using internal settling, would consist of tapping out matte at regular intervals and casting such material when it is not immediately required, a wasteful and unnecessary practice incompatible with modern ideas of smelting work.
During the early stages of the development of smelter plant, the use of reverberatory fore-hearths received considerable attention, the principle being to build a fire-box in communication with the settler, so as to ensure a sufficient supply of heat in the vessel for efficient settling. Modern furnaces however, usually supply a large enough quantity of very hot and fluid matte and slag as to allow of very efficient separation without the use of extra heating, providing the position and construction of the settler is suitably planned, as will be described in due course.
Dimensions.—The modern blast furnace is a long, narrow, water-cooled shell, rectangular in plan. The dimensions, particularly the length, vary greatly, being regulated according to the anticipated output of the furnace-unit. The size is generally expressed in terms of the internal dimensions at the tuyere level, which represents the smelting area. The width of the modern furnace varies usually from 44 to 56 inches, according to the blast pressure, method and speed of working, concentration to be effected, and so forth. The length in many cases is between 15 and 25 feet, when the furnace may be conveniently worked in connection with one large settler. The capacity of such a unit naturally depends on the conditions of working; it may be taken roughly as from 4 to 6 tons of material per square foot of hearth area per twenty-four hours.
Foundations.—The furnaces are built upon a foundation which is necessarily very strong, being usually either of solid rock or of concrete. [Pg 136]
Bottom Plate.—The bottom plate of the furnace usually carries part of the weight of the lower tier of water-jackets as well as the furnace burden, and is supported, some distance above the ground, on screw-jacks leaving an air-space below the furnace, which allows of convenient access for repairs or adjustment. The height of the construction is thus raised to a convenient distance for adjustment to the discharge to the settlers. The bottom plate should consist of sectionised water-cooled cast-iron plates bolted together, with a thin layer of brickwork placed above, to protect them from the corrosive influences to which they are subject. There is a slight slope towards the slag-notch. The actual working bed of the furnace is however, a chilled crust of material which sets on this bottom owing to radiation below, and which, when suitable precautions have been taken, usually adjusts itself naturally whilst the furnace is in operation, by what may be termed automatic radiation. Thus, apart from the water-cooling devices, if the working bottom wears down towards the metal plates, the loss of heat by radiation through the thin layer of material causes a chilling effect which leads to a thickening of the crust. Should the crust thicken unduly and so threaten to interfere with the discharge, the radiation is decreased owing to the thickness; and the high temperature which prevails upon this layer causes a partial melting so that it gradually becomes thinner again—thus regulating itself for the most part automatically.
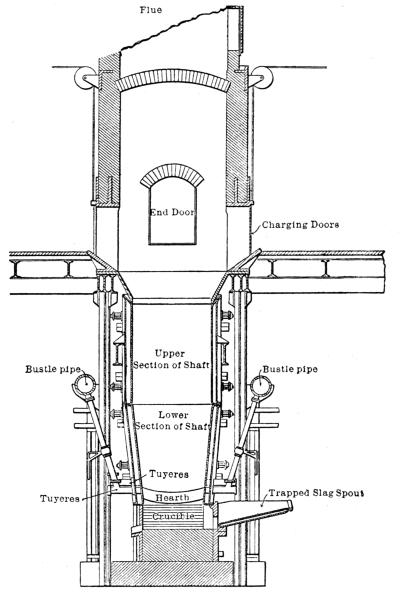
Fig. 41.—Transverse Section through Modern Blast Furnace,
showing Arrangements of Boshed Lower Jackets, Upper Jackets
and Plates, Stays and Supports, etc.
[Pg 139] Water-Jackets.—The usual height of the modern furnace, as reckoned from tap-hole to charge floor, is roughly from 14 or 16 feet up to 20 feet, water-jacketed all the way. The sides and ends of the furnace are constructed of sectionised water-jackets arranged horizontally in tiers and vertically in panels. There are usually two, occasionally three, tiers, suitably stayed and supported. The practice as regards the shape and arrangement of the jackets varies greatly. It was formerly not uncommon to work with three tiers of jackets for the sides; of these the lower tier extended only from the sole-plate to the level of the slag-notch, forming practically the crucible jackets, the height varying from 2 feet 6 inches to 4 feet. These were most used when the discharge to the settler was situated at the side wall of the furnace. Above these jackets was situated the second tier through which the tuyeres passed; these build up the boshes of the furnace, and are termed the “bosh” or “tuyere” jackets. In most modern furnaces these two tiers of lower jackets are replaced by one set of panels of from 7 to 10 feet in height, the jackets being given a slight slope towards each other at the bottom, so as to form a very small bosh angle; the contraction is about 8 inches. This improvement does away with a good deal of the jointing otherwise necessary near the hottest parts of the furnace, and thus lessens the danger of leakage at these points. The water-cooled breast-plate containing the opening for the escape of the products is now put in position as a separate piece, well secured to the rest of the jacketing (Fig. 39). Above the lower tier of jackets is placed the upper series, often from 7 to 9 feet in height, which carries the walls of the furnace up to within a few feet of the charging platform. These jackets are parallel, and no bosh is given (see Fig. 41).
The end jackets are usually built in two tiers only, the upper, 7 feet to 7 feet 6 inches, as a rule, and the lower, 8 feet to 9 feet 6 inches, according to circumstances; in the smaller furnaces the end wall may sometimes consist of a single jacket only. They are vertical, no end bosh being allowed. The end jackets are each single panels, whilst the side walls are built up in panel sections, the width of which vary, but are often 7 feet to 7 feet 6 inches wide, the panels being bolted or clamped together and strongly stayed.
The water-jackets are constructed of flanged steel plate, the inner sides of which are 5⁄16 to ⅜ inch thick, the outside ¼ to 5⁄16 inch. The seams are flanged outwards, so as to prevent joints, etc., being exposed to the inside of the furnace. The water space between the two plates of the jacket is from 3 to 4 inches.
It is usual to support the weight of these jackets on I-beams carried by the upright columns; very strong bracing and tieing is also necessary in order to prevent the side walls from bulging by the great pressure to which they are subjected. In order to protect the jackets themselves from buckling by the forces acting upon them, they are strengthened inside the water space by a series of ┻ bands, which run vertically downwards between the plates, and are rivetted to the outer side—this device is found not to interfere unduly with the proper circulation of the water. Leakage between the joints of the separate jackets is prevented by asbestos packing. In spite of the strong binding and bracing of the walls in this manner, the connections are so devised as to allow of their being unfastened very easily, so that jackets may be readily disconnected and taken down when it becomes necessary to do so.
Arrangements for the water supply to the jackets vary considerably. In localities where a plentiful supply is available, each jacket has its independent outlet and inlet pipes; in other cases it is common to arrange an independent feed to each set of panels, water being supplied [Pg 140] first to the jackets of the lower tier, and being discharged from them to the jackets situated above. The supply pipes for the various jackets branch from water main pipes running at the sides of the furnace.
The tuyere or bosh jackets are pierced horizontally at intervals of about 1 foot, with a line of 5-to 7-inch holes for the fitting in of the tuyere pieces. These are formed of steel thimbles, of ⅜-inch metal, which have a slight taper, fitting secured against the inner plate and rivetted to the outer one, thus allowing of ready replacement when necessary (see also Fig. 40). Above the side jackets of the furnace there is usually a heavy mantel-plate, 2 feet to 2 feet 6 inches high, with a sloping front, and surmounting this are apron plates, 1 foot 6 inches to 2 feet high, inclined at 45°, constituting a hopper which directs the charge towards the centre of the furnace in such a way as to keep the fines nearer to the middle line, and thus leave the sides of the charge more open, in order to ensure more regular working.
Superstructure.—The jacketing, together with the apron and mantel plates carry the structure up to the charging floor. Above this is the superstructure with the arrangements for taking off the furnace gases, and for the feeding of material for the charge. In many cases the general practice still prevails of constructing the walls of this portion of brickwork, often about 14 feet high, surmounting this with a hood of metal from the top or sides of which large off-takes carry the furnace gases to the dust chambers, and thence to the flue system and stack. Modifications in the design of the blast-furnace superstructure have been, however, in course of progress at many works, particularly in connection with the employment of automatic or mechanical charging appliances and the taking-off of the gases below the feed-floor level. This is specially the case at plants operating the pyritic process and where the gases are to be utilised for acid manufacture, as well as in connection with the treatment of smelter fume. Several furnaces are also at work using either metallic water-cooled or air-cooled tops, from which the removal of accretions is stated to be very readily effected.
Some of the most recent developments in the design of blast-furnace superstructure have been described by Emmons in reviewing the experiments at the Copperhill Smelter, Tennessee. The gases here are used for acid-making, and are sent to Glover towers under some pressure. The furnace top consists of cast-iron corner-posts and dividers, the walls and ends laid up with brickwork, surmounted by a tubular top of the Shelby type from which the gas off-takes lead. The horizontally pivoted doors open inwards and fit tightly. These arrangements are stated to be very satisfactory.

Fig. 43.—Showing Upper Jackets, Apron and Mantel Plates,
and Superstructure of Blast Furnace, Anaconda.
[Pg 141] The charging platform, suitably supported on vertical columns, runs at the upper level, being provided, on either side of the furnace, with tracks of rails for the charge cars. The charging doors usually correspond in position to the panels of water-jackets, and are situated along the whole length of each side furnace-wall, the bottom of the charging opening being flush with the floor. They are generally moved up and down in the grooved guides of the upright columns between them, and are of sheet steel suitably strengthened, from 6 to 7 feet wide and 4 feet 6 inches to about 5 feet high, supported by wire-rope and chains, and operated by compressed air cylinders.
The Air Supply to the Blast Furnace.—The quantity of air required by the blast furnace varies very widely with the class of work, rapidity of output, character of charge, and general smelting conditions. It may be stated roughly as being from 300 to 500 cubic feet of air per minute per square foot of hearth area, at a pressure of about 40 to 50 ozs. per square inch.
The rotary blower of the Roots or Connersville type is very well suited for the supply of these enormous quantities of air at moderate pressures, but for blast at higher pressures the air leakage becomes excessive, and piston-driven blowing engines become almost a necessity. Such improvements have, however, been made in rotary-blowing appliances within recent years that most blast-furnace plants are equipped with blowers of the rotary type, which are found highly satisfactory. The air is brought along blast mains of considerable size—about 30 inches diameter—to the furnace building, thence to the bustle pipes of 24 inches diameter, which surround the furnace, from which branch off the pipe connections (5 or 6 inches diameter) for the tuyeres. The practice of equipping each furnace with its own blowing unit is fairly general, making the necessary reserve connections in case of temporary breakdown; many smelters, however, adopt the system of delivering the air from all the engines into one large common air main, making the necessary connections from this to each separate furnace. The importance of avoiding leakages is recognised, and the requisite valves for regulating and controlling the air supply are arranged for.
From the bustle pipe the air passes down the pipe connections which are attached by flanged joints, thence to the tuyere pipes, which are of cast-iron, the blast being regulated by valves. The actual form of tuyere employed varies considerably, each smelter usually having its [Pg 142] own special devices for the convenience of repair, renewal, and fixing, as well as for valve regulation and punching. The tuyere is held against the face of the jackets by bolts, leakages being prevented by asbestos packing.
The tuyeres are usually 4½ to 5 inches in diameter, and are generally [Pg 143] placed about 12 inches apart. Air is supplied only through the side jackets, and not at the ends of the furnace.
Heating the Air Blast.—The advisability of heating the air-supply for copper blast-furnace smelting has been the subject of very considerable discussion, the question requiring consideration both with respect to its influence on the rationale of the smelting operation as well as from the economic standpoint. The matter is dealt with more fully in connection with pyritic practice, from which point of view Peters has reviewed the subject exhaustively. It may be here stated that there appears to be no advantage in preheating the air when the true pyritic process is operated, and actual trial has resulted in the rejection of the method at the smelters practising this work.
Where, however, coke fuel to any considerable extent is employed on the charge, a supply of heated air through the tuyeres may result in an increased rapidity of smelting, as well as in the production of hotter and more fluid slags. Especially in partial pyritic smelting and more particularly when working charges which contain but little sulphide and where the employment of much coke is not advantageous, the use of preheated blast may be economically very useful. In such cases, the heat production in the furnace is not so fundamentally bound up with the thermo-chemical reactions of slag formation as it is in true pyritic smelting, and therefore the enhanced intensity of combustion of coke-fuel at the tuyere-zone by the use of hot air may exert an important influence in improving the furnace operation and in decreasing the amount of coke-fuel required. In many such instances indeed it has been chiefly the economic factor with reference to the cost of installing and operating suitable devices for warming the air-supply which has determined the question of adopting this system. As is well known, the use of a supply of heated air causes a largely increased calorific intensity from the combustion of coke, resulting in higher temperature at the tuyere-zone, under which circumstances the charge materials are smelted more rapidly, and the resulting products are more fluid, whilst slags of higher silica content (sometimes economically advisable) can be conveniently worked with.
The devices employed for the preheating of the blast vary considerably—cheapness, capacity, simplicity in design and operation being the main essentials.
The utilisation of the waste heat from the smelting furnaces or products would suggest itself as an economical method for accomplishing the warming of the blast, but in practice several difficulties are encountered in efficiently making use of this heat. Heat is available [Pg 144] from two sources, either from the furnace gases or from the hot slag. The very successful operation in cast-iron smelting, of hot-blast stoves worked by the “waste gases,” cannot, however, be applied to copper blast-furnace smelting, since the gases in this case do not possess similar calorific value owing to the small proportions of carbon monoxide present. Further, the temperature of these gases is not sufficiently high to allow of the effective application of the regenerative principle using brickwork chambers. In consequence, the use of metal pipe-stoves offers the only method of utilising the heating values of the furnace gases, but their comparatively low temperature does not afford sufficient heat for the warming of the large quantities of air which are required at the tuyeres.
The much higher temperature of the reverberatory furnace gases offers, however, much greater scope for their utilisation in this respect, if both classes of furnace happen to be in operation at the plant and if they are conveniently situated for the purpose.
At several smelters, blast furnaces have been equipped with hot-blast “tops” for the purpose of preheating the air supply, the air-heating pipes being exposed to the gases in the upper portions of the furnace. The Giroux blast-heating device has been installed on furnaces at smelters in Mexico and Arizona, whilst at others in the same localities, the Mitchell system of baffle passages has been successfully used. The Kiddie system of running the blast pipes through the dust chambers has been tried at Tyee, B.C. The advantages of thus utilising the heat of waste gases have generally, however, been found to be more than balanced by the extra costs involved.
Efforts have been made to use the heat contained in molten slag for warming the air, but owing to the low conducting power of these materials, and the difficulty of bringing extended surfaces in close contact, the method has not proved itself very efficient. Blast is occasionally warmed by passing the air through tunnels in which bogies of molten slag are allowed to remain for some time.
When methods of utilising waste heat from the furnace products fail, the fuel-heated iron pipe-stove is generally employed. Since the temperatures required are comparatively low, and the margin of profit involved by the use of hot blast is usually small, the use of the cheapest class of fuel available is imperative; but many classes of fuel unsuitable for other purposes may find useful application for this work.
The stove is of the usual ![]() cast-iron pipe form, designed to give the maximum exposing surface,
suitably strengthened and protected from direct action of the fire.
[Pg 145]
Much valuable information on the advantages, disadvantages, and
appliances for blast heating was afforded by the smeltermen who
contributed to the symposium on “Pyrite Smelting,” which Rickard edited
for the Engineering and Mining Journal.
cast-iron pipe form, designed to give the maximum exposing surface,
suitably strengthened and protected from direct action of the fire.
[Pg 145]
Much valuable information on the advantages, disadvantages, and
appliances for blast heating was afforded by the smeltermen who
contributed to the symposium on “Pyrite Smelting,” which Rickard edited
for the Engineering and Mining Journal.
Mathewson, E. P., “The Development of the Modern Blast Furnace.” Eng. and Min. Journ., May 27, 1911, p. 1057.
Wright, Lewis T., “Metal Losses in Copper Slags.” Bulletin Amer. Inst. Min. Eng., 1909, Sept., No. 33, p. 817.
Shelby, Geo. F., “Cananea Blast Furnaces.” Engineering and Mining Journal, April 25th, 1908.
Emmons, N. H., “Copper Blast-Furnace Tops.” Bulletin Amer. Inst. Min. Eng., Feb., 1911, p. 119.
“Heating Blast.” Engineering and Mining Journal, June 16 and Sept. 15 and 29, 1906.
“Pyrite Smelting,” T. A. Rickard.
Also the Authors already referred to, Austin (p. 80), Gowland (p. 17), Peters (p. 80).
Charge Calculations—Charging—Working—Disposal of Products—Pyritic Smelting—Sulphuric Acid Manufacture from Smelter Gases.
Charge Calculations.—Modern practice aims at the production of a matte of converter grade, containing usually from 40 to 50 per cent. of copper, and preferably in a single smelting operation; except in true pyritic work.[12]
Full analysis of the whole supply of material available at the smelter is essential, as well as a report on the quantities of each separate constituent.
The first step in the charge-calculation is the computation of the total weights of copper, iron, and sulphur available for the smelting campaign; from these quantities the losses of copper and sulphur to be allowed for during the operation itself, as based on previous experience, are deducted. The balance indicates the quantities of these elements from which the matte and slag can be produced. The copper is transformed into matte, in which product it may be regarded as existing in the form of copper sulphide, Cu2S, and the sulphur required for this combination with the copper is calculated from the relation—
| Cu2S = Cu2 : S | :: | 2 × 63·5 | : 32 |
| :: | 127 | : 32 | |
| :: | 4 | : 1 approximately. |
Thus every unit of copper combines with one-quarter of its own weight of sulphur.
A matte of converter grade containing, say, 44 per cent. of copper is constituted as follows:—Copper, 44 per cent. + sulphur, 11 per cent., or copper sulphide, 55 per cent., the remaining portion of the matte being iron sulphide, which amounts to 100 − 55, or 45 per cent.
Assuming as a first approximation that this iron sulphide has the formula FeS,[13] the proportions of iron to sulphur in this material are
| Fe : S | :: | 56 | : 32 |
| :: | 7 | : 4 |
hence 7⁄11 of the remaining 45 per cent. of the matte is iron and 4⁄11 is sulphur—that is, the matte contains in addition, iron 28 parts, sulphur 17 parts. Hence the composition of the converter matte is approximately—Copper 44 parts, iron 28 parts, and sulphur 11 + 17 = 28 parts.
The amount of copper for the matte is fixed by the available ore supply; the quantity of sulphur is controlled by the furnace operation and charges, as judged from previous experience—the oxidation being so regulated that the proper grade of matte is produced. The iron required for the matte is next considered. Every 44 parts of copper require 28 parts of iron for the production of a matte of the correct grade. If the quantity of iron in the materials available at the stock-bins be not sufficient to furnish the amount required, as just calculated, ferruginous material must be added as flux, if, on the other hand, there is a superabundance of iron available in the charges for this purpose, the excess must be fluxed off.
In this manner the amounts of the constituents for the matte production are determined, and the composition and making up of the slag-forming constituents are next considered. In this connection the local conditions with respect to proximity and cost of suitable flux, as well as experience with the previous working of the furnace and ore charges are important factors in determining the type and composition of the slag to be made, whilst in true pyritic practice the special conditions of working fix certain limits to the composition of the slag, as will be indicated later—the pyritic furnace “tending to make its own slag.”
In partial pyritic smelting, the coke allowance and the furnace conditions allow of fairly wide latitude in making up the charges for the production of suitable slags with which the furnace can deal efficiently, since the heat production is not dependent on the formation of any particular slag. It is always possible to add extra coke for the purpose of melting the slag desired.
The scientific principle governing the calculations for slag composition is the proper proportioning of acid and basic constituents. This is based upon the oxygen ratio—i.e., the proportion of oxygen [Pg 148] in the acid constituents compared with that in the bases. With the doubtful exception of alumina in certain cases, silica constitutes the entire acid portion of most copper-smelting slags.
The requirements for a satisfactory slag are that it shall be—
It is well known that within certain broad limits of silica content, slags will fulfil these conditions to a greater or less extent, whilst the most suitable and economic slag under any particular circumstances is decided, as stated above, by the composition of the charge, the quantity and character of the available fluxes, and the previous experience with the furnace. The limits of the silica content for suitable slags as just indicated are fixed by several well-known general properties of the silicates.
Speaking broadly, and from the point of view of the more or less ferruginous silicates constituting copper-smelting slags, the more basic silicates—such as the subsilicate class (oxygen in acid : oxygen in base < 1 : 1)—are generally characterised by high formation-temperature, and by being very fluid, thin and fiery, dense and corrosive. On the other hand, the more acid silicates, such as those of the multi-silicate class (oxygen in acid: oxygen in base > 2 : 1) are characterised by lower formation-temperature and low density, and by being thick and viscous.
As the silica content within this range of silicates increases, the melting point is lowered and the specific gravity is reduced, features which are very advantageous from the point of view of the production of clean slags. Their fluidity, however, decreases, and a very high temperature is thus required in order to render them sufficiently limpid to run freely from the furnace. On this account the highest proportions of silica usually considered feasible in a slag, correspond to the bisilicates of the representative composition, MO. SiO2. With high temperature conditions in the furnace and rapid working, such slags can be dealt with successfully, and if the charges are necessarily highly siliceous, it may be advantageous from the economic point of view to work with this class of slag.
In proportion as the silica content gradually decreases and as they become more basic, the silicates are more and more corrosive and fiery, and especially in the case of the iron silicates, they gradually attain such a high specific gravity that efficient settling of the matte is not [Pg 149] possible. In addition, the more basic the silicate the greater is its dissolving power for sulphides, hence high copper losses in the slags result from these combined causes. Such basic silicates possess, however, the advantage of marked liquidity, and of flowing from the furnace in a thin limpid stream. The high density and the solvent power of basic slags thus fix a limit to the composition which is considered economically suitable, and the lowest proportions of silica usually worked with correspond to the mono-silicates represented by the formula 2MO. SiO2. Slags containing a greater proportion of base (usually iron) possess too high a density to permit of clean settling. In practice, therefore, the majority of slags are mixed silicates of a composition ranging between the limpid but somewhat dense mono-silicate and the lighter but more viscous bisilicate, corresponding to silica contents of from 30 to 48 per cent. of silica, and within the limits of 35 to 45 per cent. of silica most copper blast-furnace slags will be found. The composition roughly corresponds in a large number of cases to that of the sesqui-silicates of the general formula 4MO. 3SiO2 (oxygen in base : oxygen in acid :: 4 : 6 :: 1 : 1½).
As is well known, mixed silicates—i.e., silicates of two or more bases—are generally characterised by the properties of increased fusibility, and often of increased fluidity, and their employment is usual and generally advantageous in smelting practice. The relative proportion between the various bases in such mixed silicates is largely a matter depending upon the prevailing conditions at the smelter.
In modern smelting, particularly where partial pyritic work is conducted, and where fairly siliceous charges are worked, a slag running about 40 per cent. SiO2 is aimed for, iron and earth oxides constituting the remaining 60 per cent. or so. In cases where this quantity of iron is present in the charge, the slag may be constituted chiefly of iron silicate, but even in such instances the advantages of lime additions are marked. When iron is not available in sufficient quantity, the extra fuel costs and working difficulties of running with more siliceous slags would render their production undesirable, and the purchase of limestone or similar earthy flux is particularly advantageous. The purely iron silicates are usually dense, and thus tend to hold up copper values both in mechanical suspension as well as in solution; the addition of lime, which has a marked effect in reducing the specific gravity, permits of more basic slags being worked with, where necessary, without such heavy losses in the slag.
The presence of lime silicate with the iron silicates has a marked influence on the fluidity of the slags, even when they are more highly [Pg 150] siliceous, whilst on account of the lower atomic weight of calcium, lime will, weight for weight, flux off a greater quantity of silica than will ferrous oxide. In forming a slag of similar oxygen ratio, thus—
hence for the production of a slag of the same oxygen ratio, less weight of lime would be required to flux off the same weight of silica; in other words, the replacing values of the two oxides are as 112 to 144, or 7 to 9.
Of the other bases which are occasionally present in slags, the proportions of the oxides of magnesium and zinc are sometimes considerable, the calculations being analogous to the previous cases. The case of alumina is anomalous, and its behaviour in slag production is not definitely understood. Many experienced workers hold the view that it tends to act either as acid or base, according to the proportions of silica. Thus, in a very siliceous slag, alumina in moderate quantity behaves as a basic oxide, forming aluminium silicates, and in very basic or low silica slags the alumina appears either to neutralise some of the excess base, acting as an acidic oxide, or to dissolve as such in the slag, whilst in intermediate cases it possibly behaves partly as an acid and partly as base. This view has recently been questioned, and it has been suggested by Shelby that alumina always acts as an acid in the formation of slags. The matter is thus one which requires further considerable investigation.
Usually neither alumina nor zinc oxide behave very satisfactorily in the furnace when present in large quantities, tending to thicken the slags and to promote viscosity.
Anaconda Practice in Charge Calculations.—An example of some of the practical considerations which enter into the calculation and making up of charges is well illustrated in certain particulars of the practice as conducted at Anaconda. Details of the materials charged over a period of one month are indicated in Table X. The important charge constituents available in large quantity include:—
| Cu. | SiO2. | Fe(O). | S. | |
|---|---|---|---|---|
| % | % | % | % | |
| First-class smelting ore, | 8·6 | 54·0 | 13·6 | 14·0 |
| Concentrates, | 10·9 | 26·0 | 32·0 | 32·0 |
| Briquettes, | 5·0 | 50·0 | 13·0 | 13·0 |
| Lime-rock (flux), | .. | .. | .. | .. |
| Old converter slags and residues, | .. | .. | .. | .. |
TABLE X.—Blast-Furnace Charge
Calculations—
Total Charge, all Furnaces.
| Tons of Charge. |
SiO2. | FeO. | CaO. | ||||
|---|---|---|---|---|---|---|---|
| % | Tons. | % | Tons. | % | Tons. | ||
| First-class ore, | 28,646 | 52·80 | 15,125 | 14·90 | 4,268 | 0·50 | 143 |
| Second-class ore, | 1,913 | 53·50 | 1,023 | 15·79 | 300 | 0·60 | 11 |
| Lining ore, | 52 | 83·71 | 44 | 4·16 | 2 | 0·67 | 1 |
| B. and B. slag, | 6,667 | 35·98 | 2,399 | 47·27 | 3,152 | 1·11 | 74 |
| B. and M. slag, | 481 | 42·92 | 206 | 42·14 | 203 | 0·12 | 1 |
| Precipitates, | 333 | 8·00 | 27 | 12·40 | 42 | .. | .. |
| Precipitates from old works, | 41 | 2·70 | 1 | 15·40 | 6 | .. | .. |
| Slimes from old works, | 19 | 56·60 | 11 | 65·0 | 1 | 0·80 | .. |
| Coarse concentrates, | 14,083 | 25·27 | 3,558 | 32·96 | 4,642 | 0·45 | 63 |
| Calcine bearings, | 232 | 9·50 | 22 | 57·00 | 132 | 0·80 | 2 |
| Briquettes, | 27,560 | 48·77 | 13,441 | 15·16 | 4,177 | 0·65 | 179 |
| Reverberatory matte, | 146 | 4·30 | 6 | 37·50 | 55 | 0·80 | 1 |
| Reverberatory slag, | 687 | 43·10 | 296 | 39·60 | 272 | 4·00 | 27 |
| Converter cold matte, | 552 | 13·60 | 75 | 29·50 | 163 | 4·90 | 27 |
| Converter slag, | 9,999 | 31·30 | 3,129 | 55·90 | 5,589 | 0·70 | 70 |
| Converter cleanings, | 7,891 | 30·53 | 2,437 | 36·55 | 2,917 | 0·79 | 64 |
| Lime-rock, | 61,794 | 6·90 | 4,264 | 0·50 | 309 | 48·80 | 30,155 |
| Coke, 18,766·235 tons, at 14·21 per cent. ash, | 2,667 | 45·28 | 1,208 | 12·21 | 326 | 6·31 | 168 |
| Total charge, | 163,853 | 28·85 | 47,272 | 16·21 | 26,556 | 18·91 | 30,986 |
| Total production, | 18,447 | 6·38 | 1,191 | 29·36 | 5,486 | 1·57 | 293 |
| To slag, | .. | .. | 46,081 | .. | 21,071 | .. | 30,693 |
| Tons of Charge. |
Sulphur. | Copper. | |||||
|---|---|---|---|---|---|---|---|
| % | Tons. | % | Pounds. | ||||
| First-class ore, | 28,646 | 15·50 | 4,440 | 6·641 | 3,804,555 | ||
| Second-class ore, | 1,913 | 14·60 | 279 | 5·476 | 209,447 | ||
| Lining ore, | 52 | 1·45 | 1 | 3·834 | 3,988 | ||
| B. and B. slag, | 6,667 | .. | .. | 0·797 | 106,325 | ||
| B. and M. slag, | 481 | .. | .. | 1·919 | 18,450 | ||
| Precipitates, | 333 | .. | .. | 58·853 | 392,352 | ||
| Precipitates from old works, | 41 | .. | .. | 68·344 | 56,607 | ||
| Slimes from old works, | 19 | 7·50 | 1 | 4·203 | 1,637 | ||
| Coarse concentrates, | 14,083 | 32·10 | 4,521 | 10·782 | 3,036,802 | ||
| Calcine bearings, | 232 | 4·50 | 10 | 9·321 | 43,230 | ||
| Briquettes, | 27,560 | 15·32 | 4,223 | 4·928 | 2,716,299 | ||
| Reverberatory matte, | 146 | 23·30 | 34 | 35·752 | 104,651 | ||
| Reverberatory slag, | 687 | 1·10 | 8 | 1·566 | 21,501 | ||
| Converter cold matte, | 552 | 18·20 | 100 | 42·675 | 470,839 | ||
| Converter slag, | 9,999 | 1·10 | 110 | 3·018 | 603,429 | ||
| Converter cleanings, | 7,891 | 6·60 | 528 | 16·840 | 2,688,024 | ||
| Lime-rock, | 61,794 | .. | .. | .. | .. | ||
| Coke, 18,766·235 tons, at 14·21 per cent. ash, | 2,667 | .. | .. | .. | .. | ||
| Total charge, | 163,853 | 8·70 | 14,255 | 4·357 | 14,278,136 | ||
| Total production, | 18,447 | 21·13 | 3,898 | 39·483 | 14,567,376 | ||
| To slag, | .. | .. | .. | .. | .. | ||
| Analysis. | |||||
| SiO2 in slag, | 46,081 ÷ 110,810 tons slag | = | Calc. 41·59 % | Actual 41·30 % | |
| FeO in slag, | 21,071 ÷ 110,810 " | = | 19·01 " | 19·00 " | |
| CaO in slag, | 30,693 ÷ 110,810 " | = | 27·70 " | 28·00 " | |
| Total, | 97,845 tons, at 88·30 per cent. = 110,810 tons slag. | 88·30 " | 88·30 " | ||
| Coke consumption, 10·63 per cent. wet weight = 10·96 per cent. dry weight. | |||||
[Pg 152] The other constituents used in the charge comprise varying quantities of materials which accumulate round the works, and which, being rich in copper values, it becomes useful and essential to clean up. For the calculating of the furnace charges, the amounts of cupriferous material available at the stock-bins are reported to the blast-furnace department. The quantities decided upon are divided among the number of charges which are considered likely to be worked off during the day, this number averaging about 1,100. The result of this calculation indicates the amount of each kind of material to be weighed for the separate charges; the analysis of each constituent being naturally known. The materials available for smelting are highly siliceous in character, the first-class smelting ore, of which large quantities are treated, giving a strongly acid composition to the charge; copper-bearing basic materials suitable for fluxing are not available in large quantity, and this necessitates the purchase of barren lime-rock, this item being the largest of the blast-furnace charge. In making up the charge sheet, as large a quantity of concentrate as possible is included, since this constituent is not only high in copper values, but owing to a high iron and sulphur proportion, it increases the fuel value of the charge, the influence on the coke consumption being very marked. The concentrate further forms a base for the matte, and introduces iron, of which there is a shortage, into the slag, thus reducing its too-siliceous character and lessening the quantity of lime which it would otherwise be necessary to procure for the purpose.
The briquettes are next worked in to as great an extent at possible, since by this means the large stocks of settling-pond slime and of screened fines are reduced and their 5 per cent. of copper is extracted. The whole stock of old slags and residues is used up on the charge, these materials introducing considerable amounts of copper, whilst being irony, they further help to reduce the acidity of the slag, thus saving the employment of the lime-rock otherwise required for fluxing. The total quantity of copper, iron, and sulphur available being then calculated, and the allowances for sulphur elimination and for the copper loss on smelting (2 to 7 per cent.), as based upon previous experience, being deducted, the amount of iron required to constitute the 45 per cent. copper matte is estimated. From this figure the FeO remaining for slag production is determined. The silica introduced by the above materials is also known, and the amount of lime-rock required to produce an easily running slag is next calculated. The slag which is found by experience to give the most satisfactory running has a composition of about— [Pg 153]
| SiO2 | 41 per cent. | |
| FeO, | 19 " | |
| CaO, | 29 " |
Variations from this composition, especially as regards higher silica contents, immediately introduce difficulties, increasing the expense of furnace running, by requiring more fuel and care in working, reducing tonnage, and producing a slag which runs far less freely. So that although the large quantity of siliceous material at hand might tempt the management to work with a more siliceous slag, and so save the procuring of such large amounts of barren lime-rock, the cost of this material is much more than compensated for by the advantages which result from the working with a slag which contains only about 40 per cent. of silica.
The quantities of the charge constituents thus calculated, divided by the likely number of charges to be worked, are entered up on the charge sheet, which is handed over to the charge foreman.
The Charging of the Blast Furnace.—The method of “hand charging,” as employed in the older processes of working, when using small furnaces of small output, possessed several theoretical advantages, but it is essential in modern practice, where at least 300 tons of charge, and often much larger quantities, are fed into the blast furnace daily, to employ mechanical means for charging. At many smelters, however, the coke is added separately, from barrows.
Care in the charging is now recognised as being of special importance for successful blast-furnace operation, especially for the purpose of procuring the correct distribution of coarse and fine material. The principle of keeping the sides more open by distributing the coarser materials against the jackets and keeping the fine parts nearer to the [Pg 154] centre is often favoured, since this device reduces the tendency to crusting by the finer sulphide particles against the walls. It is partly with this object in view that the mantel and apron plates are arranged in the hopper form, whilst at the same time the distance between the top of the charge and the feed-floor level is maintained at such a height that this desired distribution of the fresh charges is obtained.
The practice still commonly employed is to feed the materials from side-dumping cars (of very varied design) brought along in a train drawn by locomotives and travelling along tracks running at each side of the furnace. A form of car frequently used has a V-section, and it is secured in a vertical position whilst in transit by some form of catch-pin device, which is readily released when it is required to tilt the car for charging.
Another form, employed at Anaconda, has a ![]() shaped section, the sides of which are pivoted and admit of being very
readily secured or unfastened as desired. The car bottom itself is
tilted by connecting it with a compressed air lift by means of a hook
situated at the side of the car remote from the furnace. The material
is thus discharged along the inclined chute so produced.
shaped section, the sides of which are pivoted and admit of being very
readily secured or unfastened as desired. The car bottom itself is
tilted by connecting it with a compressed air lift by means of a hook
situated at the side of the car remote from the furnace. The material
is thus discharged along the inclined chute so produced.
An interesting method is employed at the Granby Smelter, where the Hodge car and the end-feeding method are in use. The cars, which have a double-hopper discharge, are divided into four compartments by vertical plates. These cars enter at the ends of the furnace through suitable openings at the level of the feed-floor, and run by small wheels on tracks which are built inside the furnace along the side of each vertical wall. In this manner a straight vertical fall for the charge is arranged, and this affords the best control of proper distribution. The furnace holds three cars at a time, and there are patent openers and closers for manipulating the end doors of the furnace, as well as for releasing the hopper-bottoms of the cars.
A particularly ingenious and successful device is in use at the Ducktown Smelter of the D.S.C.I. Co.,[14] Tennessee, where the pyritic process is operated. Careful charging is here held to be one of the great essentials for successful working of the process, especially in the narrow furnaces in use, where the dangers of crusting are greatly increased. The principle of working is, that by dropping the charges vertically downwards, having previously arranged the materials in the desired order across the furnace, they will fall into the position, and be distributed just as desired. The Freeland charger is a kind of conveyor belt made of overlapping steel plates, which is exactly the length and width of the furnace, so that when the machine is brought over it, the furnace opening is entirely covered. The conveyor is carried on a frame mounted on wheels, and this is moved forward and backward by a motor in the front, near which is seated the chargeman who is also the motorman. An independent switch and gearing causes the belt to move round and thus deposit its charge over the end. In front of the frame is a strong catch, fitting into a recess on the cover of the furnace, which is water-cooled and mounted on wheels, so that as the conveyor is brought into position the cover is moved back. All these run along a track which extends below the stock feed bins in the same straight line. The furnace gases are drawn off below the feed-floor. [Pg 155]
[Pg 156] The method of working is to bring the charger under the bins and to drop the various materials for the charge—weighing 2 tons—on to the belt. By deflectors on the ore chutes, the charge can be directed to any desired position across the belt, and material is thus deposited near the outer or inner side as desired—in falling into the furnace it is found to take the same position that it had on the plates. The charger moves forward and reaches the furnace top, the catch is fastened, and as the charger now advances the cover is pushed back, the conveyor thus taking its place until in its turn it covers the top of the furnace. The motion is now reversed, the conveyor gradually recedes, bringing the cover along with it; meantime the chargeman has set the belt-conveyor gearing working independently, and the belt thus travelling round and over the end pulleys, discharges its burden into the furnace. The disposition of the charge along the length of the furnace can be altered at will by increasing or reducing the speed of the frame. When the conveyor has at last traversed the furnace, the cover is in its place—the charger is now disconnected, and goes back for a fresh load. The furnaces are charged eight times per hour with 2 tons of material. The operations are fascinating to observe, and the control over the disposal of the charge is quite complete, whilst the conditions for the operator are not exceptionally arduous. Many other suitable devices are in use at different works.
At the Cananea smelter is operated an ore-bedding system, the store-bins feeding the charge down hoppers through which it falls directly into the furnace. A similar feeding system is in use at Garfield, Utah.
The lay-out of the plant to allow of the most efficient charging is so arranged as to locate the stock-bins at a high level, so that ore is fed directly from the discharge chutes into the cars of the charge trains which run on tracks underneath, and these tracks are situated at [Pg 157] such a level that the trains are readily and conveniently hauled to the charging platforms of the blast furnaces.
The charge foreman receives from the blast-furnace department his charge sheets which inform him of the amounts of the various materials to be loaded on to each car—calculated in the manner already indicated. Proceeding to the stock-bins, the gates and chutes of which are automatically controlled, he sets the scale of the weigh-bridge which is situated under each bin to the desired weight. At the same time an electric-light indicator is switched on in front of the particular bins from which material is to be withdrawn, thus assisting in spotting the cars and checking the weighing-out. The charge train is brought along the tracks running underneath the bins, and into each car [Pg 158] is dumped the correct amount of charge, usually to within 50 lbs., with rapidity and ease. The train then passes to the furnace building, where the charges are dumped or otherwise emptied into the furnace.
The Coke Allowance.—As has been already indicated, the coke allowance depends largely upon the nature of the charges and the individual experience at the smelter. The main principle involved is to reduce the coke consumption as much as possible by applying the pyritic principle to the fullest possible extent, working as much sulphide material into the charge as is economically practicable.
In partial pyritic smelting, where the coke may constitute from 5 to 10 or 12 per cent. of the total charge, it is usual not to feed it in with the rest of the materials from the cars, but to charge it into the furnace separately. The charge foreman puts it in just when and how he considers it necessary, and he is encouraged to use as little as possible, consistent with proper running of the products at the slag spout. In pyritic smelting proper, the small amount of coke is fed on to the top of the charge-material in the charge-cars.
Working of the Blast Furnace.—The top of the charge, which is usually some 3 to 5 feet below the level of the feed-floor, appears fairly uneven, there being a tendency for it to sink along the middle. It is moderately hot, showing practically a black heat except where red-hot patches near the side appear in positions corresponding to where the tuyeres are situated below. There is not very much fume at the feed-floor level if the chimney draft be good, nor excessive agitation at the top, unless much fine material is being worked. Sulphide fines tend to the formation of accretions near the top of the charge and occasionally lower down, also to a considerable extent against the walls of the brick superstructure—this is said to be lessened considerably by the use of water-jacketing at these parts, which also greatly assists the barring down of the masses.
A considerable amount of barring is sometimes necessary when much fine concentrate is worked, otherwise a well-managed furnace runs smoothly and satisfactorily under favourable conditions. Trouble may arise occasionally by leakages occurring in the jackets or spouts, but by the modern methods of sectional construction and by the devices for time-saving in making the necessary connections, working is usually not seriously interfered with for a very long period. Even for the removal or replacement of a slag spout, the slag-hole is plugged, and the repair is completed within an hour and a-half, by which time slag is again running freely over the replaced slag spout.
The tuyeres are punched regularly two or three times per shift, and a steady stream of material issues from the slag notch and over the spout to the settlers. [Pg 159]
Disposal of the Furnace Products.—Under ordinary circumstances, the products resulting from the blast-furnace operations include—
The Matte and Slag.—In modern practice, as already indicated, the fluid products of the blast furnace are run out of the furnace as rapidly as possible, and flow continuously, as they are formed, through a trapped slag notch. So important has this principle of rapid removal of the fluid products become, that the hearth or crucible portion is being made smaller and smaller. The slag notch, is, in addition, placed so low that only so much molten material remains in the furnace bottom as is necessary for the regulation of the temperature for maintaining perfect fluidity of the materials during their discharge, and for avoiding crust formation on the hearth. The depth of material remaining in the bottom—that is, the distance from the hearth bottom to the slag notch—is from about 8 to 12 inches, depending on the conditions just indicated.
The discharge of the furnace products takes place through the trapped slag notch of the furnace, an opening constructed in the tapping-breast [Pg 160] or tap-jacket, which is usually a small special jacket-portion constructed and kept in position separately on account of the great local wear at this point (see Fig. 39). The trapping device is an important and essential feature in connection with the modern practice of rapid and continuous running, the principle being to arrange a sufficient height of molten material at the outer side of the slag opening to overcome the inside blast pressure, and thus prevent the escape of blast with its attendant inconveniences and danger. The flow of liquid material can thus proceed quietly and uninterruptedly. The blast is trapped by the construction of a dam in the form of a slag spout around the slag opening, of such a shape and secured to the tap-jacket in such a manner and position, that the molten material before overflowing at the end, fills the spout and thus covers the discharge outlet of the furnace, trapping the blast so that as fast as the molten products form, a constant stream overflows into the settlers (see Fig. 52).
The slag spouts are often of sheet steel, sometimes of copper or of bronze, and are from 3 feet 6 inches to 5 feet in length, being separately water-cooled units. The discharge at the end is from 12 to 18 inches higher than the centre of the slag notch in the tap-jacket through which the molten material issues from the furnace. The spout is secured to the tap-jacket, being arranged so as to admit of ready replacement where necessary. Usually it is bolted to the jacket and is securely wedged up against it, being supported at the discharge end by the wall of the settler, and the joints are made perfectly tight by very careful asbestos packing and claying. The spout lasts for several months, the greatest wear being at the end over which the molten stream issues, but the life has been considerably lengthened, with greatly increased convenience of furnace working, by providing the spouts with separate easily replaceable water-cooled nose-pieces of cast-iron which are bolted to the ends, thus taking up most of the wear and tear, and allowing of a very ready removal and replacement without disturbing the slag-spout connections to the furnace itself. These are indicated in Figures 52 (A) and 59. The slag spout is protected along its entire length by a hood of clay, by which means the stream of matte and slag running down it is maintained hot and fluid. [Pg 161]
[Pg 162] The position of the outlets from the furnace, connecting to the settlers, is largely affected by the available floor space and the general lay-out and arrangements of the plant. Under suitable conditions, and especially with long furnaces, the arrangement of the settler in front of the furnace works very advantageously, leaving the alignment of the blast furnaces free, and allowing plenty room for working around the settlers. The settlers are then arranged in the middle line of the crucible portion of the furnace, so that working is conducted evenly from both ends of the furnace towards the discharge in the centre, and the smelting is thus regular and allows of good control. At many smelters the discharge of products takes place from spouts at the ends of the furnaces, the settlers thus being in alignment with them. This plan, under suitable conditions, has several advantages, permitting of ready access to the sides of the furnace, even working of the furnace by discharge at both ends, and ready co-operation between adjoining furnaces and settlers.
Settlers.—The modern type of settler is often circular in section, about 16 to 18 feet in diameter and 5 feet in height, storing about 40 tons of matte. Other forms, rectangular or oval, are, however, also employed.
The outer shell is of ½-inch steel plate bound together by band-bolts, the lining is often 9 to 15 inches in thickness, with an inside layer of looser stuff. The lining material employed varies greatly, according to the grade of matte, character of slag, and working conditions. The wearing out of the lining depends very largely on the class of material passing through the settler, the most rapid wear being occasioned by the fiery and corrosive low-grade mattes and basic slags, whilst high-grade mattes and more siliceous slags give little trouble in this connection. The more corrosive the products, the more refractory and hard-wearing must be the lining, and consequently the materials employed for the purpose range from chromite, silica brick and firebrick down to loam, according to the requirements; the chief duty is that of being non-corrodible and of protecting the outer shell. It is not an uncommon practice to thicken the walls close to the tap-holes, where they are subjected to most wear, and often chromite is used at these points owing to its power of withstanding the forces of erosion. On the other hand, at the Copperhill Smelter of the Tennessee Copper Company the settlers have been found to give as satisfactory service on fairly low-tenor matte, when lined throughout with good firebrick as with the more expensive materials formerly used, whilst still more recently, siliceous copper ores have been successfully employed as lining material instead of bricks. [Pg 163]
There is usually a spray of water from a circular pipe which surrounds the settler near the top—this playing against the steel sides keeps the outside cool and protects the lining. The settler is roofed over with slag, except at the back where the stream of matte and slag enters, and also at those points where the slag overflows. The slag escapes over short launders attached to the top of the steel casing. The position of these discharges depends largely on the arrangement of tracks, size of furnace, temperature of working, and quality of products. Under modern conditions of high temperature and rapid working, they are situated as far away from the entrance as possible, thus giving fuller opportunities for very quiet settling in a large pool and affording gentle overflow of slag with little abrasive action on the linings. These outlets may be situated opposite to the entrance or at the sides. The discharge spouts for slag may be one or two in number, usually of cast-iron coated with thin clay, and often roughly hooded over with clay. They have replaceable cast-iron nose-pieces to facilitate repair after wearing down. The continual gentle stream of [Pg 165] slag runs along launders, where it is either discharged into slag bogies and dumped, or much better, is met by a strong stream of water which immediately granulates it, and washes it along flumes to the dumps.
[Pg 166] The matte tap-holes are generally two in number, situated close to the bottom of the settler, and usually at an angle of 120° from each other and from the entrance spout.
The hole through the brick wall for tapping is about 1½ inches in diameter, and the matte is discharged through a tapping piece of cast-iron, 6 inches in diameter and 3 inches thick, perforated by a 1-inch hole. This iron disc has, cast around it, a copper tapping-plate about 1 foot in width and 2 feet high, which is recessed into the steel sheet of the settler. In the iron tapping-piece is a conical recess, into which the conical clay plug is rammed when closing the tapping-hole. These iron tapping-pieces withstand the action of converter grade matte fairly well, and are conveniently replaced when necessary—about once a month. They are illustrated in Fig. 58.
The tapping-plate is fixed into position in a special section of the shell, known as the launder casting, to which the matte launder is secured, whilst a newer form of settler has the tap sections also removable, so that these can be taken out and the brick renewed during the campaign of a furnace, being as readily removable as a furnace jacket. The matte launder is of cast-iron or of steel, thickly coated with clay or suitable material (slime-pond product, etc.) to protect it from corrosion. In modern work the steel tapping bar is always rammed through the conical plug and tapping-hole until it just reaches the matte, so that its withdrawal by ring and wedge is readily performed when the matte is to be tapped whilst by this means the tap-hole is securely closed.
The workers are protected from shots of matte, etc., during tapping or closing, by means of a slotted sheet-iron hood which can be swung back when not required, a convenient and useful as well as necessary precautionary device. Matte is tapped from the settlers into ladles as required by the converters; such ladles are constructed of thick steel plate, washed with clay, and often lined with a hull of chilled material. It is sampled at the runner with each tapping. The tap-hole is closed by a clay plug on the end of a dolly which is rammed home, and a warm pointed steel bar is then driven through until it reaches the matte, being knocked in occasionally as the end is very slowly eaten away. Several of the features named in the previous sections are well indicated in the photograph (Fig. 59) of the tapping platform at the Anaconda Smelter.
The “Gaseous” Products of the Furnace.—Great variation is to be found in the arrangement at different works for the disposal of the gaseous products of the furnace. Reference will be made later to the methods employed in connection with pyritic work, and where the gases are to be utilised for the production of sulphuric acid. Formerly the general method, even at the large modern plants, was to lead the gases from the top of the superstructure to the off-takes and large dust-catcher flues, thence to the stack.
[Pg 167] With the introduction of automatic and mechanical charging methods, now being inaugurated to a considerable extent in place of dumping from cars alongside the furnace, the method of withdrawing the gaseous products just below the level of the feed-floor is being adopted.
The off-take flues of the modern furnace are of steel, 4 to 6 feet in diameter—lined or unlined according to circumstances—and leading to very large dust chambers of varying design, sometimes rectangular, often of large circular section, or of balloon-shaped section, etc. In all cases these flues are provided with hopper discharge openings at suitable intervals, under which cars run on tracks, for the collection and conveying of the dust. Arrangements for the further settling and collection of the flue-dust are essential in connection with modern blast-furnace plants, where blast pressures of from 40 to 50 ozs. per inch are employed and where it is often found economical to work with [Pg 168] as much fine material as possible, either as such or in an agglomerated form; where too, the dropping of charges from some height and the agitation caused by the blast are practically unavoidable. Rarely less than 2 per cent. of flue-dust is made in any modern blast furnace, whilst 5 per cent. is by no means uncommon, and even larger quantities are often produced. Such dust is, moreover, often somewhat higher in copper contents than the original charge, owing to the brittleness of copper sulphide minerals, which, being more readily broken up, are carried over in the form of fine particles. Hence the economic aspect of the recovery of values, in addition to legislative requirements, call for efficient collection of these products.
The gaseous products of the furnace carry solid matter in two forms. As a rule, under the usual conditions of copper-smelting charges, the larger portion of the solid matter thus carried is in the form of very fine particles of charge material itself, mechanically suspended and carried over in the current of the escaping gases. This is the flue-dust. In addition, values in the form of volatilised metallic products are also conveyed by the gases, particularly when lead, zinc, arsenic, etc., are present in the furnace charge, and these are carried forward in the form of fume. They tend to solidify as the temperature of the gases becomes lower, although their settling is very greatly impeded owing to the exceeding minuteness of their particles and also to their dilution; the problem of separating and collecting them is in consequence attended with great difficulty.
Chambers of enormous capacity are required in order to give the fine solid particles an opportunity of settling by decreasing the velocity of the gases and by cooling them down, whilst for the settling of fume, capacious flues in which are suspended wires or similar devices for assisting the process must be adopted. Where large quantities of lead, etc., are present some bag-house system of fume filtration is necessary, especially if silver be present, since this metal tends to be carried over in the leady fume. At the majority of copper smelters such extreme refinements are rarely necessary, although modern legislative requirements make severe demands on the managements for the freedom of the gases from injurious constituents.
Dry settling methods and filtration are in general use where such separation is required and the use of high-tension electricity has been successfully tried at Californian smelters. Wet methods have so far not proved economically successful.
The flue-dust from the flues is dealt with in a number of ways, according to the conditions at the smelter. It may be smelted with the “roaster-calcines charges” in the reverberatory furnaces, although excessive quantities have proved difficult to deal with in certain [Pg 169] instances, it may be included in the charges for sintering or briquetting processes, and it has been very successfully incorporated with the matte in beds when it has been necessary to cast low-grade matte into cakes previous to re-concentration in the blast furnace, at a smelter employing the pyritic process.
Still more recently, the East Butte Copper Mining Company has installed and successfully operated a sintering plant on the Dwight-Lloyd principle for the treatment of the flue-dust preparatory to blast-furnace smelting. The capacity of the plant is 100 tons per day. The material is rendered more or less cohesive by the effects of heat alone, but the operation is not yet perfect. (See Mining Journal, Jan. 6th, 1912, p. 21.)
The freed gases finally pass along series of long and capacious brick main-flues connecting with all the branch flues, furnished with discharge hoppers at intervals, gradually rising and discharging into a wide stack of such a height that damage to vegetation in the district is entirely prevented.
Pyritic Smelting.—Modern blast-furnace practice, as has been stated, is conducted according to two main systems of working:—
The term Pyritic Smelting (or pyrite smelting) is thus applied to that class of practice in which the whole of the heat required in the smelting zone is obtained by the combustion of the ore or matte charge itself; it implies the application of the pyritic principle to the extreme limit, the use of carbonaceous fuel being reduced to a minimum.
Ideal working is to feed unroasted ore or matte, together with the requisite fluxes, into the blast furnace, and by the action of an adequate air blast, to burn out part of the sulphur and iron, the former escaping with the furnace gases, the latter being slagged off, whilst the copper in the charge is concentrated in the matte product of the operation.
This type of smelting is conducted at a number of large modern works, and though up to the present time the use of coke on the charge has not been entirely eliminated, research and practical experience have [Pg 170] demonstrated that the small quantity which is employed is not utilised as fuel by combustion in the air blast at the tuyeres, but that it is, in fact, oxidised in another manner at some considerable height in the furnace.
History.—The idea originated with John Holway, of London, who sought to extend to the smelting of copper the principles so brilliantly applied by Bessemer to steel manufacture, and who, in a work which was published in 1879, suggested and demonstrated the process of utilising the heat of oxidation of the iron and sulphur constituents of copper-bearing materials for the smelting and extraction of the copper. That work is to-day recognised as one of the most masterly expositions of the principles underlying pyritic smelting and converting, and many of the most important and recent developments in these branches of work are proceeding on lines forecasted by him. Holway’s experiments, conducted on a considerable scale, proved the feasibility of the principles underlying the process, which was to prepare metallic copper from sulphide ores in one combined series of operations in a single furnace unit. Owing, however, to mechanical troubles and difficulties of operation, as well as to the ultimate withdrawal of financial support, he was unable to carry the process to a commercial success, and the single-stage process is at present regarded as being beset by almost insuperable difficulties, although the latest phases in modern practice are tending towards a realisation of Holway’s scheme of working. His paper and published results deserve the closest study.
Inspired by the pamphlet, an English Company in 1887–8 attempted the practice at a smelter at Toston, Montana, and showed the possibilities of the method, although the plant available did not lend itself to completely successful operation. L. S. Austin, who took a leading part in this work, patented the process in the United States, and developed the practice, and in 1891 Dr. Peters conducted a very full enquiry into the conditions of working, which placed the system on a definite practical basis. From that time the method has developed coincidently with the more empirical practice at many works of replacing coke fuel by sulphides to as great an extent as possible. T. A. Rickard focussed scientific and practical opinion on the subject in the symposium on “Pyrite Smelting,” which he called forth and edited, and many celebrated smeltermen have contributed to the progress of pyritic smelting practice. At the Copperhill Smelter of the Tennessee Copper Company and at the Ducktown Sulphur, Copper and Iron Co.’s Smelter at Isabella, Tennessee, remarkably good pioneer work was done by Parke [Pg 171] Channing, Freeland, and others in developing the process. Enormous service has been rendered within recent years by the masterly researches and brilliant exposition of Robert Sticht, in which latter work Peters has worthily seconded him.
Pyritic smelting is at the present time being very successfully practised at Mt. Lyell, Tasmania; at Tennessee; Tilt Cove, Newfoundland; and other districts, whilst the smoke problem alone has prevented for a time a number of other smelters from successfully operating the process.
The Mechanism of the Process.—The mechanism of the changes involved in the pyritic process is now fairly well understood in general outline. One of the most important steps in elucidating the matter was made by Sticht’s discovery that the oxidation area of the furnace in pyritic smelting was confined to a narrow zone situated just a little higher than the tuyere level; by actual experiment it was found that scarcely any free oxygen existed above this narrow tuyere zone.[15] It thus became evident that the first series of changes near the top of the charge were those mainly caused by the effects of heat alone, and that only by a second series of changes lower down at the tuyere zone were the reactions of rapid and intense combustion and oxidation of the sulphides being effected. Finally, at the bottom of the furnace, the molten matte and slag collected and ran out. Thus the furnace operations proceed in two main stages; preparation (liquation of the sulphides from the charge) in the upper portion, and oxidation and fluxing (bessemerising of the liquated sulphides) in the oxidising tuyere zone or focus.
The usual and typical ore charged into the furnace in pyritic smelting is impure chalcopyrite (essentially a copper-bearing pyrites, FeS2). When heated in an atmosphere free from oxygen, this pyrites loses some of its sulphur and approaches pyrrhotite in composition. On further heating in a neutral atmosphere more sulphur is evolved and the material approaches FeS in composition, whilst at very high temperatures and under favourable circumstances, a still further quantity of sulphur is liberated, resulting in the production of the well-known fusible iron sulphide, which is the eutectic of the iron : iron-sulphide series of alloys, melting at 970° C., and containing about 85 per cent. of FeS. Thus in the pyritic furnace, free sulphur is liberated as such at the upper levels, and passes up the furnace unchanged until it meets free air above the surface of the charge, when [Pg 172] it there burns to SO2. The residual fusible sulphide melts, trickles down, and becomes the true pyritic fuel of the furnace. The copper sulphide constituents of the charge are practically unaffected in composition by heat alone, and they pass down the furnace with the rest of the charge unchanged until the hotter zones of the furnace are reached, when these sulphides also liquate out, become dissolved in the melting iron sulphides, and are thus carried down to the oxidising zone. Until the sulphides meet free oxygen, no further reactions proceed, since they are without action on silica at even the highest furnace temperatures.
When, however, they reach the blast of air which enters the furnace at the tuyeres, an intense action proceeds as the sulphides become bessemerised. The heat of oxidation of iron sulphide has long been known to be very great, and Holway pointed out that this heat corresponds to the large quantity of heat which is developed by the free roasting of heavy sulphides, compressed into the space of a few moments, and thus results in an exceedingly great intensity with consequent high temperature. Sulphur is burnt out to SO2, iron is converted to the oxide which instantly combines with the white-hot silica skeleton that is present and forms an iron-silicate slag, evolving still more heat. This slag, with the enriched matte, melt thoroughly at the prevailing temperatures, and issue from the slag spout of the furnace.
The work of Sticht and Peters thus allow of the mechanism of the processes being followed during the passage of the materials through the furnace.
At the Mount Lyell Smelter, where Sticht operated, the charge extends about 12 feet above the tuyeres. In the upper 6 or 7 feet, elemental sulphur is driven off from the pyritic materials by the effects of heat alone, and the furnace gases in this zone consist chiefly of nitrogen, SO2 (from the bessemerising), sulphur vapour, a little CO2, but practically no free oxygen. About half-way down, the temperature is sufficiently high to melt out the fusible sulphides from the charge; these liquate and trickle unchanged through the still solid masses of gangue and silica-flux, until they meet with free oxygen of the air blast, when they are oxidised and burnt up with great rapidity and with the evolution of intense heat. This bessemerising zone extends from a short distance above the tuyeres to a point where all the oxygen is used up by the iron and sulphur. The distance is variable, but is probably some 2 feet or so. At this level the ferrous oxide produced is instantaneously seized by the white-hot particles of free silica with the production of a silicate slag, the composition of which corresponds to the silicate whose formation temperature is equal to that prevailing in this bessemerising zone. [Pg 173]
Control of the Operations.—It has thus been established that the oxygen of the air blast entering the furnace through the tuyeres is practically all expended in this bessemerising of the liquated sulphides in the narrow bessemerising zone, and that it does not operate at all by any roasting reactions in the upper part of the furnace, as had been formerly supposed.
From this knowledge it therefore becomes possible to indicate the essential factors which control the successful operation of true pyritic smelting. The degree of bessemerising depends upon the amount of air supplied for the oxidation of the sulphides, and upon the quantity of siliceous flux present to slag off the iron oxide produced.
The actual smelting takes place at the focus where the liquated sulphides are instantaneously bessemerised, and the more rapid this oxidation, the more intense are the reactions and the higher the temperatures which result.
For successful pyritic smelting it is, therefore, essential that there shall be present—
(a) The supply of heat required for the smelting of the charge and the thorough fusion of the products depends entirely on the intense combustion of the iron and sulphur constituents, and the greater the proportion of these materials oxidised per minute, the higher is the temperature. As has been already noted, such heat intensity increases at a rate greater than the mere arithmetical increase in the fuel proportion, by reason of well-known thermo-chemical laws regarding mass effects. Indirectly, too, the higher the proportions of sulphides present, the smaller is the quantity of inert or useless matter which requires to be heated and slagged off in the furnace—apart from the question of the necessary flux material. Hence the higher the iron and sulphur contents of the ore, the more successfully may true pyritic smelting be applied to it. True pyritic smelting may be said to cease when carbonaceous fuel requires to be burnt at the tuyere zone in order to supplement the heat derived from the sulphides, and broadly speaking, from about 28 per cent. of iron and about 30 per cent. of sulphur are necessary in the charge for good pyritic work under present [Pg 174] conditions. At Tennessee, with about these proportions, the coke consumption on the charge is reduced to about 3 to 4 per cent.; at Mt. Lyell, where the ore runs from 40 per cent. of iron with a corresponding quantity of sulphur, the coke consumption amounts to only about 1·25 per cent. None of this coke probably reaches the bessemerising zone at all.
(b) Being supplied with enough sulphide fuel, the requisite quantity of air for the rapid and sufficient combustion of this iron and sulphur is essential. The oxygen is used up entirely in the bessemerising of the sulphides at the tuyere zone of the furnace, and in consequence, not only the heat supply, but also the concentration depends upon the amount of oxygen furnished at this point, since the greater the quantity of oxygen which is used up, the greater is the amount of sulphur eliminated and the amount of iron oxidised and slagged off, and in consequence, the higher is the proportion of copper in the resulting matte. In other words, the oxygen supply largely controls the concentration effected in the smelting process, and consequently an adequate quantity is of the utmost importance. The amount of air theoretically required per minute is readily calculated from the estimated capacity of the furnace and from the charge analysis. Liberal allowances are required for losses, leakages, blower efficiency, etc.; and the volume necessary at the furnace amounts to something like 5,000 cubic feet per minute per 100 tons of sulphide.
(c) Sufficient siliceous flux is required for the satisfactory slagging of the iron oxides produced. The presence of the requisite silica on the charge is exceedingly important. The iron of the sulphides, upon oxidation by the air blast, is converted into iron oxides, primarily FeO. This oxide is incapable of existing by itself, but possessing when nascent a powerful affinity for silica at high temperatures, it produces ferrous silicates, which are, in the main, fusible slag-like products. This action is particularly evident in the tuyere zone of the pyrite furnace, where the silica is present in a white-hot condition. If sufficient silica be not present to combine with the iron oxide produced, the ferrous oxide which is exceedingly unstable, finding itself without the necessary flux, is converted under the continued oxidising effect of the blast into higher oxides of iron such as ferric oxide or magnetic oxides, materials which are practically infusible, and this results in the production of an infusible sinter which leads to the choking of the furnace. On the other hand, if excess of silica be present in the charge, highly siliceous and unworkable products result, which will not run out of the furnace. Any further excess of silica simply remains unfused and unattacked, and causes the ultimate stoppage of the furnace operations. [Pg 175]
The silica for fluxing is consequently an important factor in controlling the running of the pyritic furnace, and the provision of the requisite quantity, as nearly as possible, is essential, since otherwise the presence of adequate sulphide and air blast is not in itself sufficient to ensure satisfactory working.
The actual quantity of silica required is determined by the factor known as the formation temperature of the silicates. Every silicate has a definite formation temperature—i.e., a definite mixture of iron oxide and silica requires a definite temperature in order that complete combination may occur and a chemical compound silicate be formed. Conversely, at any definite temperature, only those silicates having a corresponding formation temperature to this degree of heat can be produced. In consequence, if the oxidation of the sulphides at the tuyere zone produces any particular temperature, that particular silicate whose formation temperature corresponds to this will tend to be formed, and the required quantity of free silica must be present to yield this definite silicate with the whole of the iron oxidised. Only a limited quantity of silica can thus be taken up for any definite rate of oxidation of iron sulphide, and the presence of either more or less silica does not greatly affect the composition of the slag. Thus the concentration (sulphide oxidation) is primarily dependent on the oxygen supply, which determines how much iron shall be burnt, but the success of the operation depends upon the presence of the correct amount of silica to flux off this iron oxide. This proportion is fixed by the temperature attained at the tuyere zone, which restricts the silicate produced to such a composition that its formation temperature coincides with this degree of heat. Hence the general law has been deduced and has been confirmed in practice, that “a pyritic furnace produces a slag corresponding in composition to the silicates whose formation temperature equals that prevailing at the tuyere zone,” accounting for the well-known observation “that the pyritic furnace tends to make its own slag.” If the smelting operation is to proceed satisfactorily, slag approaching this composition will be produced, and assuming the air supply to be adequate for the purpose, the absence of the requisite silica on the charge affects the quantity rather than the character of the slag. The amount of iron sulphide oxidised depends largely upon the presence of silica to combine with the iron oxide produced; so much will be oxidised as the silica can deal with, and in consequence, if the free silica supply is deficient, a smaller quantity of slag is formed, whilst the matte will be larger in amount but of lower grade. [Pg 176] An addition of silica to the furnace charge under such circumstances would thus raise the grade of the matte by encouraging the slagging of more iron, and would produce slag of approximately the same composition as before, though in larger quantity.
Deficiency of silica also results in the production of over-fire, owing to the fact that the air blast, being unable to bessemerise any more iron sulphide at the tuyere zone, passes to the higher portions of the furnace and gradually roasts the ore there, thus consuming the sulphide fuel of the furnace which might otherwise be most effectively used for bessemerising in the tuyere zone. This over-fire, resulting from the heat of roasting which is given out in the upper part of the furnace, is very disadvantageous in true pyritic smelting, and successful control of the process depends on using up the whole of an adequate air supply at the bessemerising zone, and on supplying sufficient siliceous flux to combine at once with the whole of the iron oxide produced. For fluxing purposes it is only the free silica in the charge which is effective, since any silica existing as silicate is already in a state of combination and thus is not free to act as flux. The combined silica, except for its adding to the fusibility of the charge by admixture, is very disadvantageous, consuming heat and space, diluting the reaction intensities by presenting an inert substance among the active constituents, and increasing the quantity of slag which requires to be melted.
The three requirements—iron sulphide, oxygen supply, and fluxing silica—thus bear an intimate relationship to one another in true pyritic smelting, and alteration of any one factor requires simultaneous adjustment of the others for the production of the same grade of matte and slag. The speed and degree of oxidation primarily depend on the air supply. The more iron burnt up, the greater is the heat production and the higher the temperature at the tuyere zone, and since the more basic slags are known to have the higher formation temperatures, the basicity of the slags increases with the speed of oxidation and consequent concentration.
Ores suitable for true pyritic smelting are not commonly met with in practice, and the presence of earthy bases other than iron is not desirable. Whilst the advantages of polybasic slags from the point of view of reduced formation temperature, increased fusibility and liquidity are very marked in ordinary smelting practice, their presence is not so advantageous in true pyritic smelting, since they consume silica which is required for the iron oxide at the instant of formation, and thus tend to decrease the speed of oxidation and concentration. Polybasic slags have a lower formation temperature, and in consequence the production of the highly ferruginous slags of high formation temperature which it is desired to make by the oxidation of as [Pg 177] much iron as possible is retarded. In addition, the presence of other earthy bases in the charge dilutes its fuel value; they may even consume valuable heat by requiring decomposition, as in the case of carbonates. These considerations are not so important in partial pyritic smelting, where the required heat balance can be adjusted by coke.
The Advantages of Pyritic Smelting.
(1) The possibility of direct and immediate treatment of highly pyritic raw ore in the blast furnace, thus saving all the costs of preliminary treatment and handling.
(2) The saving of the costs of roasting heavy sulphides.
In former smelting practice, high sulphide contents in a copper ore were particularly disadvantageous, since the higher the sulphur contents of the charge the lower was the grade of the resulting matte, when smelted directly in the blast furnace. In consequence, the higher sulphur content necessitated a more complete roasting of the ore in order to ensure a high-grade matte on smelting.
With pyritic smelting the conditions are completely reversed, and the charge becomes more suitable for direct furnace treatment as its sulphide contents increase, so that the most suitable ores for pyritic smelting are those in which the greatest saving is effected by their not requiring a preliminary roasting operation.
As has been already indicated, this saving includes labour, plant, handling, time, and interest on capital tied up in the roast yards, as well as the avoiding of all the mechanical and other losses connected with such preliminary treatment. Thus at Ducktown, Tennessee, the material economies effected by the substitution of pyritic smelting for the processes involving preliminary roasting amounted to no less than 3 to 4 cents per pound of copper produced, in addition to the later advantages derived from the recovery of values from the gases, and from the improved conditions of life in the district.
(3) The cost of coke is saved.
Fuel is one of the main items of expense in blast-furnace smelting, and by the substitution of the cost-free natural-sulphide fuel for coke, the proportion of the latter required on the charge is reduced from the 9 to 10 per cent. formerly employed with roasted materials to about 3 to 5 per cent., and in certain special cases to very much smaller amounts.
Difficulties of the Process.—That the technical difficulties in applying the process on a practical scale are considerable, under present conditions of working, will be understood from the nature of the operations. [Pg 178]
(1) The pyritic process works on a narrow margin of heat, and allows of but little flexibility in the conditions of working, since there are few factors which can be altered should difficulties in operating arise, as compared with the circumstances when a free use of supplementary carbonaceous fuel may be employed. The only source of heat energy at the smelting zone is in the sulphide charge itself, and small variations in the working conditions may readily disturb the delicate equilibrium upon which successful working depends. Irregularities, stoppages, and variations in grade of matte may therefore arise, unless the operations are regulated with exceeding watchfulness. In true pyritic smelting the employment of coke for restoring the balance or for producing heat required at the tuyere zone is not permissible or practicable, since, as will be indicated later, such coke addition would altogether destroy the equilibrium in the process; the grade of matte and the composition of slag would be altered, the reactions disturbed, and a restoration to normal pyritic smelting conditions rendered almost impossible.
Difficulties in operation have therefore to be overcome along the lines of pyritic action—that is, in the further adjustment and manipulation of blast, sulphide or silica supply, or in charging methods, etc.—and in practice such careful “doctoring” is resorted to when the furnace shows signs of working unsatisfactorily.
It is very often possible by such careful attention to gradually bring a furnace back to smooth running. It occasionally happens, however, that the conditions gradually become worse, and the furnace commences to show signs of “gobbing.” This is indicated at the top of the charge by the formation of crusts round the side and end walls, whilst from the slag spout below, there issues a much reduced quantity of thick siliceous slag, together with an abundant stream of thin low-grade matte. The furnace gradually ceases running, and it becomes necessary to stop its working, to take down the furnace jackets, bar out the debris, and restart operations. This is usually not so objectionable a procedure as it might appear, and indeed, within certain definite limits, such a course may economically be sound policy. In the modern operation of pyritic practice it often pays better to risk the occasional gobbing up of a furnace and clear out the debris, than to work with so large a quantity of coke as would avoid such a necessity. Not only is the modern furnace so designed and constructed as to entail but comparatively little trouble in cleaning out in this manner, but such practice, even if temporarily a necessary evil, may, in places where coke is expensive, and where conditions for pyritic smelting are otherwise favourable, be, within certain definite limits, actually the [Pg 179] most profitable. It is by the taking of these risks, combined with further experiment and working experience in manipulation, such as in charging methods, blast conditions, and the height and distribution of charges, etc., that the ultimate continuous and successful working at still lower costs may be attained and the true pyritic process be worked as continuously as ordinary smelting practice. Short campaigns are not, therefore, unusual under the present conditions of true pyritic smelting, and the cleaning out of the generally fairly loose debris is accomplished with moderate ease, from 24 to 36 hours being the usual time required to take down, clean out, and restart a furnace, whilst the cost of such an operation (chiefly in labour) is not, under the circumstances, excessive. At Tennessee, hard driving and short campaigns result in lower costs and greater tonnage.
(2) The composition of the slag often prevents high concentration.
It has been indicated that the thermal conditions in the bessemerising zone of the pyritic furnace tend to the production of highly basic slags, which, though hot and limpid, are characterised by high density. Such slags are not conducive to good settling and separation of mattes, and they tend to occasion high copper losses, because—
(a) The difference in density of slag and matte is not sufficiently great.
(b) The solubility of sulphides in the slag increases with its basicity.
The greater the concentration effected by the smelting operation, the higher is the grade of the matte produced; at the same time, the actual weight of matte is smaller. On the other hand, since more iron is oxidised from the charge and slagged off, the quantity of slag produced increases proportionately. Contrasting then, the likely losses of copper which would result from the association of a small quantity of high-grade matte with much slag, compared with those resulting from the association of a considerable quantity of low-grade matte in the presence of but little slag, the former condition is obviously the more productive of heavy loss, for not only will many more shots of matte be held in suspension, but each shot of high-grade matte represents a larger quantity of copper.
It is found in practice that it is most economical to make a fairly low-grade matte on the first or “green-ore” smelting, and to re-concentrate this matte pyritically up to converter grade by a second smelting operation. The extra cost of casting the low-grade matte, of breaking up, rehandling it, and resmelting, with all the extra charges on capital, etc., involved, is less than the losses which would be incurred if higher-grade converter matte were made at the first smelting, although there is no difficulty at all in producing such [Pg 180] mattes so far as the actual furnace operations are concerned. It is entirely a question of the slag losses involved.
Under ordinary smelting conditions (not truly pyritic), when using some coke for fuel, it would be readily possible to alter the density of the slag by adding suitable constituents, such as limestone or additional silica, but in pyritic smelting this is not practicable. The furnace chooses to make at the tuyere zone its own slag, and that a highly basic one. High concentration and a slag low in iron content cannot be obtained together in true pyritic smelting, since high concentration means rapid oxidation of iron sulphide, and this necessitates high temperature and produces a highly ferruginous slag in consequence. Additional silica added to the charge could not alter the slag composition markedly and still yield the same grade of matte. The silica content of the slag depends on the temperature at the tuyere zone, and this is governed by the rate of oxidation of the iron sulphide. If the slag is to be more siliceous it must be produced at a lower temperature, which would be obtained by oxidising the iron less rapidly. This would lead to the production of low-grade matte, and probably would so reduce the furnace activity that there would not be sufficient heat to keep the slag molten.
If extra silica be added to the charge, it would probably be unattacked unless more iron were oxidised in order to flux it off. In such a case the blast would have to be increased in order to produce iron oxide more rapidly, the temperature would in consequence be raised, a still more basic slag would be produced in larger quantity, whilst the matte would be increased in grade and reduced proportionally in weight.
The addition of sufficient lime to the charge, in order to produce a sufficiently low-gravity slag, is also impracticable in true pyritic work, because—
(a) The extra lime consumes silica, and interferes with the desired reactions at the bessemerising zone, tending to lower the concentration. It also absorbs heat.
Lime has a very powerful affinity for silica, more strongly marked than that of iron oxide, its replacing value is higher, its more siliceous silicates are readily formed and they have a lower formation temperature, all of which factors tend to an undue consumption of silica which is urgently required by the iron if the rate of oxidation is to be maintained. The marked tendency for lime in the charge to consume the silica tends to retard the oxidation of the iron sulphide, which proceeds most satisfactorily when free silica is available for the nascent iron-oxide, and in consequence concentration is decreased [Pg 181] and the heating effect in the furnace reduced. In addition, the larger bulk of calcareous slag carries considerable heat from the smelting zone of the furnace. Lime silicates and the polybasic lime slags have a markedly lower formation temperature than the normal ferruginous slags of true pyritic smelting, they are hence formed readily without requiring so much oxidation activity at the tuyere zone. In consequence less iron is oxidised, and the resulting concentration in the matte is proportionately reduced.
(b) The lime is introduced in the form of limestone, and the carbon dioxide liberated from this material in the furnace is found to have a deleterious effect on the furnace gases if the manufacture of sulphuric acid from them is intended—this being a consideration of great economic importance in connection with many modern pyritic smelters.
Hence, in practice, pyritic smelting is at present generally conducted in two stages for the production of a matte of 30, 40, or 50 per cent. converter grade. The “green ore-matte,” or first matte, runs usually from 8 to 13 or 14 per cent. of copper, depending upon the copper ore available, which is usually very low grade—2 to 3 per cent. copper contents; the second or concentrated matte assays 28 to 40 per cent. copper. Special care is taken to ensure good settling of matte from the basic and irony slags, and by these means the copper losses in the slags are reduced to the comparatively moderate proportions associated with normal practice.
It does not appear improbable that with the developments of basic converter practice, involving eventually the continuous converting of low-grade mattes, the necessity for this second pyritic smelting and re-concentration may be avoided. The removal of this feature from pyritic smelting practice would add enormously to the potential economies arising from the method.
In spite of the difficulties connected with the process, as detailed above, the method has proved itself an exceedingly profitable one on a large scale, and the experience of the companies financially interested, as well as the opinions of managers of the plants in practical operation, leave no doubt as to the economic success of this application of scientific principles to a practical problem on a very extended scale.
Special Features of Pyritic Smelting.— Several points of particular interest have given rise to much discussion in connection with pyritic smelting practice. These include the question of the coke proportion required on the charge, and the advisability or otherwise of employing heated blast for the furnace.
Coke Proportion.—Whilst ideal pyritic practice involves the entire absence of supplementary carbonaceous fuel, it has not been found practicable, up to the present, to ensure satisfactory working over any [Pg 182] reasonable period of time, unless a minimum of about 1·25 per cent. of coke is incorporated with the charge. The function of this coke has been a matter of much speculation, but the investigations of Sticht already referred to, now permit the tracing, with some considerable accuracy, of its function and of its action in the furnace.
It is found that in true pyritic smelting the coke does not reach the bessemerising zone at all, but that it is completely consumed in the regions above this point. It is, moreover, not burned by the oxygen of the air, none of which exists above the tuyere zone, since all this oxygen is consumed by the combustion of the sulphide. It appears that the coke is oxidised by the SO2 which results from this sulphide combustion. The examination and analysis of samples of the gases withdrawn from different parts of the furnace have confirmed this view, and have elucidated the probable reason for the apparent necessity of a certain small proportion of coke in the process, under the present conditions of working. The heat generated from the oxidation of the coke by the SO2 is of much value in preheating the materials of the charge for the removal of excess sulphur and the liquation of the sulphides. The amount of heat which is available for this operation is small, being practically all derived from that carried upwards by the hot gases leaving the smelting zone, and none is obtainable by the usual processes of coke or sulphide oxidation in the upper regions of the furnace, since no available oxygen is believed to get past the bessemerising zone and reach these upper areas. It is indeed necessary for the success of pyritic smelting that such oxidation or roasting of sulphides in the upper part of the furnace should be prevented, since every available particle of iron sulphide is required for heat production at the smelting zone, by its combustion there, and any oxidation elsewhere not only deprives this zone of fuel, but spreads the heat over too wide an area for sufficiently intense combustion.
Thus, by supplying an additional amount of heat to the upper parts of the furnace, where heat is needed to assist in the preparation and liquation of the sulphides, the extra coke, in being oxidised by the SO2 without robbing the tuyere zone of fuel or air, just fulfils its useful purpose at the required place, in such a way as to keep the smelting operation running smoothly.
The presence of more coke than is absolutely necessary for the fulfilment of this purpose is, in addition to its extra cost, of no advantage, and in true pyritic smelting none should reach the tuyere zone, since it introduces a reducing influence where the most marked [Pg 183] oxidising effect is required. By consuming oxygen for its combustion, it deprives the iron sulphide of this material, less iron is, therefore, oxidised, and the matte is consequently increased in quantity and lowered in grade, whilst the amount of iron carried into the slag is decreased.
1·25 per cent. of coke is about the minimum quantity with which it is found practicable to maintain satisfactory working of the furnace under present conditions, 0·5 per cent. has been worked with occasionally, and none at all over certain short periods of time. The average quantity employed is from 2 to 3 per cent., and when about 5 per cent. is used, coke reaches the tuyere zone and the process ceases to be truly pyritic—the reactions and smelting conditions become entirely changed.
It does not seem unlikely that, as knowledge of these conditions increases and as the mechanism of the process becomes more generally understood, modifications in furnace design and blast conditions may lead to the successful operating of the pyritic process entirely independent of the use of coke fuel.
Heating of the Blast.—For true pyritic smelting it has been shown in practice that the use of heated blast possesses no advantages; many smelters operating the process have tested the effects, and have usually given the method up, whilst the work of Sticht and Peters affords valuable evidence and close argument as to the reasons for its unsuitability. Success in true pyritic working depends upon the intensity of oxidation of the sulphides, and upon the localisation of the resulting heat at the narrow bessemerising zone situated just above the tuyeres. The greater the quantity of iron which is there oxidised per minute, the better is the concentration, the greater is the smelting and fluxing intensity and the higher is the resulting temperature. Since the character and composition of the slag vary in accordance with these conditions, depending largely upon the temperature in the tuyere zone, the furnace works most rapidly and satisfactorily when slags of high formation temperature are being produced. These can only be formed if much iron is being oxidised, because iron is the chief fuel in the process. The addition of extra heat by warming the blast appears to allow of the formation of silicate slags possessing a lower formation temperature, such slags are less basic, and consequently less iron need be oxidised and slagged off per minute in order to produce them. Less iron sulphide fuel is, therefore, burned, and the reaction intensity at the tuyere zone is reduced, so that the necessary heat margin for satisfactory smelting may not be attained. The extra heat carried in by the warmed blast may not be [Pg 184] sufficient to compensate for that which is lost owing to this decrease in oxidation intensity; the furnace consequently tends to work cold, whilst the excess air supply leads to the production of over-fire, by the oxidising of sulphides higher up in the charge.
These features are specially interesting, as they afford one of the most marked distinctions between true and partial pyritic smelting. In the latter process, the fuel value in the adjustable supply of coke at the tuyeres allows of the ready production of any extra heat which might be required. The slag composition is, in consequence, more independent of the furnace conditions, since the heat required for the smelting operation does not depend so much on the formation of slag of any particular composition. Sufficient heat is always obtainable by coke additions when smelting for any special slag which may be desired. Neither is localisation of the heat at the narrow tuyere zone so essential in partial pyritic smelting. Warm blast produces a greater combustion intensity when employed in oxidising carbon, so that it may present advantages, both economic and operative, in partial pyritic work, whereas it is distinctly disadvantageous in the true pyritic method.
Pyritic Smelting Practice in Tennessee.—The pyritic process is operated in Tennessee at two smelters; that at Copperhill under the Tennessee Copper Company, and at Isabella by the Ducktown Sulphur, Copper and Iron Co. The ore averages from 2 to about 2½ per cent. copper, 31 to 37 per cent. iron, 20 to 30 per cent. sulphur, 10 to 25 per cent. silica, the remainder being earths, including lime about 6 per cent., magnesia 2 per cent., zinc 2 per cent., and alumina—i.e., a heavy sulphide ore with but little excess of free silica available for the fluxing of iron.
Copperhill.—The process is conducted very much according to the principles just considered. The Copperhill plant operates seven furnaces of the ordinary rectangular water-jacketed type—the general features of furnace design being little different at present, whether true or partial pyritic practice be conducted. Several important devices in detail have been introduced with successful results, and the management is distinguished for its pioneer work and experimental enterprise in connection with the process. The furnaces were formerly all 56 inches wide; three of them are 180 inches long, the other four being 270 inches. The height of charge is from 10 to 12 feet, the capacity of the smaller furnaces 375 to 400 tons of charge daily, and a blast of 19,000 cubic feet of air per minute at 50 ozs. pressure is supplied to each. The larger furnaces have a capacity of 500 to 600 [Pg 185] tons daily. Many trials have been made to determine the best shape for the water-jacketed sections, both broad and narrow panels having been employed. In one of the furnaces, curved end-jackets were tried, with the object of lessening the production of crusts which tend to form at the corners, owing to coldness and reduced furnace activity at these points. The advantages expected have not been realised, the tendency to crusting has not been lessened, and although barring has been rendered easier, the disadvantages of rounded corner-jackets and their greatly increased cost of construction outweigh their advantages, and their use has now been given up.
An important modification in the form of the tuyeres has been introduced with the object of furnishing more effectively the necessary large volume of air at suitable pressure, and of increasing the efficiency at the tuyere zone. Instead of supplying the air to the furnace at a number of separated points, it was felt that the closer these could be brought together the better. A narrow slot all round the furnace for air admission has been held to be the most perfect method, but hitherto it has been thought impracticable, though a recent form of furnace (not at this plant) has been devised on this system. The improvement here has been the use of slotted tuyeres, 12 inches long by 4 inches wide, each of which replaces two of the older tuyeres of 3¼ inches diameter. These have proved very successful, the furnace thus equipped handling a much larger tonnage, and it has been decided to adopt the new form on all the furnaces. [Pg 186]
Charging is by side-dumping V-shaped cars, and great care is taken in the handling and distribution of the charges. The furnaces are fitted with tops of special design, and with elaborate dust-catching devices which have been the subject of long and numerous experiments; the special purpose being to allow the taking off of the gases below the feed-floor, and to reduce the height of the superstructure to the smallest possible proportions, so as to prevent excessive dilution (by air) of the furnace gases, which are used for sulphuric acid manufacture. The furnace tops were originally of the standard form—brick walls supported by steel frame-work. It was, however, necessary to damper down the flues in order to obtain sufficient pressure to force the gases through the Glover towers, and the heat has caused the steel work to warp badly. A low top was tried, using a brick-lined flue at the end for taking off the gases below the feed-floor. This was found to be good for charge-dumping and general convenience, but it allowed the escape of too much smoke and flames, which greatly interfered with the furnace manipulation. In consequence the tubular top was used, gradually raised until a suitable height was reached. This form has been described on p. 140.
The present practice at Copperhill is to smelt the ore pyritically for a 9 to 10 per cent. matte, passing the products through the 16-foot settlers which are now lined with siliceous copper ore, then tapping the matte into ladles which empty it into beds of flue-dust. Alternate layers of matte and dust are thus incorporated, and yield a porous material convenient for the concentrating pyritic smelt which follows. This re-concentration is now conducted in a furnace narrowed to 44 inches, which has been found specially well suited for the work; the furnace runs fast, smelting sometimes over 800 tons of charge per day. The system of working is that of hard driving so long as the furnace smelts rapidly. As soon as it slows down, the furnace is tapped out and started afresh. The re-concentrating charge contains some limestone in order to reduce the copper losses in the slag, the saving effected by this feature being equivalent to 2 lbs. of copper per ton of ore smelted. The resulting matte is bessemerised.
The furnace gases are utilised for sulphuric acid manufacture, the acid plant being the largest in the world, with an ultimate capacity of 400 tons per day.
Ducktown.—It was at the Ducktown Company’s smelter that the first work on pyritic smelting in the district was carried out, and the successful development of the process generally, owes much to Freeland’s early pioneer work, the remarkable results of which led Parke-Channing to adopt the process at the Copperhill plant. [Pg 187]
TABLE XI.—Typical Charging Tables at Pyritic Smelter.
| B.F. No. 3—Night Shift. Typical Green Ore Charges. |
|||||
|---|---|---|---|---|---|
| I. | II. | III. | IV. | ||
| Lbs. | Lbs. | Lbs. | Lbs. | ||
| Coke, | 180 | 240 | 240 | 400 | .. |
| Ore A., | 5,000 | .. | .. | .. | .. |
| Ore B., | .. | 5,000 | .. | .. | .. |
| Ore C., | .. | .. | 5,000 | .. | .. |
| Slag, | .. | .. | .. | 4,000 | .. |
| Lime rock, | .. | .. | .. | .. | .. |
| Green ore (low grade) matte, | .. | .. | .. | .. | .. |
| Flue-dust, | .. | .. | .. | .. | .. |
| Quartz (for flux), | 950 | .. | .. | .. | .. |
| Total weight of charge, | 6,130 | 5,240 | 5,240 | 4,400 | .. |
| Hours of Charging— | No. of Charges |
No. of Charges |
No. of Charges |
No. of Charges |
Total per Hr |
| 6–7, | 2 | 2 | .. | .. | 4 |
| 7–8, | 2 | .. | 2 | 2 | 6 |
| 8–9, | 2 | 2 | .. | .. | 4 |
| 9–10, | 2, 2 | .. | 2, 2 | .. | 8 |
| 10–11, | .. | 2 | .. | 2 | 4 |
| 11–12, | 2 | .. | 2 | .. | 4 |
| 12–1, | 2, 2 | 2 | .. | .. | 6 |
| 1–2, | 2 | .. | 2 | 2 | 6 |
| 2–3, | 2 | 2 | .. | .. | 4 |
| 3–4, | 2 | .. | 2 | .. | 4 |
| 4–5, | 2 | .. | .. | 2 | 4 |
| 5–6, | 2, 2 | 2 | 2 | .. | 8 |
| Total No. of charges daily, | 28 | 12 | 14 | 8 | 62 |
| B.F. No. 5—Day Shift. Typical Concentrating Charges. |
||||
|---|---|---|---|---|
| I. | II. | III. | ||
| Lbs. | Lbs. | Lbs. | ||
| Coke, | 150 | 400 | extra 700 | .. |
| Ore A., | .. | .. | .. | .. |
| Ore B., | .. | .. | .. | .. |
| Ore C., | .. | .. | .. | .. |
| Slag, | .. | 4,000 | .. | .. |
| Lime rock, | 700 | .. | .. | .. |
| Green ore (low grade) matte, | 3,500 | .. | .. | .. |
| Flue-dust, | .. | .. | .. | .. |
| Quartz (for flux), | 1,050 | .. | .. | .. |
| Total weight of charge, | 5,400 | 4,400 | 700 | .. |
| Hours of Charging— | No. of Charges |
No. of Charges |
Total per Hr |
|
| 6–7, | 2, 2, 2, 2, 2, 2, 2 | .. | .. | 14 |
| 7–8, | 2, 2, 2, 2 | 2 | .. | 10 |
| 8–9, | 2, 2, 2, 2, 2 | .. | .. | 10 |
| 9–10, | 2, 2, 2, 2, 2 | .. | .. | 10 |
| 10–11, | 2, 2 | 2, 2, 2, 2 | .. | 12 |
| 11–12, | 2, 2, 2, 2, 2 | .. | .. | 10 |
| 12–1, | 2, 2, 2, 2, 2 | .. | .. | 10 |
| 1–2, | 2, 2, 2, 2, 2 | .. | .. | 10 |
| 2–3, | 2, 2, 2 | 2, 2 | .. | 10 |
| 3–4, | 2, 2, 2, 2, 2, 2 | .. | .. | 12 |
| 4–5, | 2, 2, 2, 2, 2 | .. | .. | 10 |
| 5–6, | 2, 2, 2 | 2, 2, 2, 2 | .. | 14 |
| Total No. of charges daily, | 120 | 12 | .. | 132 |
It will be observed that the concentrating furnace works twice as quickly as the green ore matting furnace, and hence one furnace only is required for the concentration of the matte product from two of the matting furnaces.
[Pg 188] The Isabella smelter comprises two furnaces of moderate size, 17 feet by 3 feet 4 inches at the tuyeres, having a joint capacity of 500 to 600 tons daily. The furnaces are about 9 feet high, and are water-cooled. Air at only 20 to 30 ozs. pressure is supplied through 3-inch tuyeres. The smelting scheme is somewhat analogous to that adopted at Copperhill, the first smelting producing a 20 per cent. copper matte from the 2 per cent. ore, whilst the re-concentration results in a converter-grade matte assaying 50 per cent. The coke proportions are somewhat similar to those used at Copperhill, being 5·0 per cent. for the first smelting, and 3·5 per cent. for the second. The furnace management at this small plant is exceedingly efficient, and the campaigns are long, it being claimed that the furnace operations have never had to be completely stopped on account of crusting or gobbing. This is held to be due to the results of special care in feeding and charge distribution, the ingenious Freeland charger already described being used. The charge is kept low (6 to 8 feet above the tuyeres), and is evenly red hot all through. The slags assay 35 to 36 per cent. silica, 38·8 per cent. iron, and 8·0 per cent. lime—with moderate copper losses. The annual output is equivalent to about 3,000 tons of metallic copper. An acid-making plant is also attached to these works.
The Manufacture of Sulphuric Acid from Pyritic Furnace Gases.—Modern legislative requirements make severe demands upon the managements of smelter-works where sulphury ores are dealt with, by reason of the disastrous effects of the sulphurous gases upon the conditions of life generally in the vicinity. In other cases, litigation by neighbouring farmers and others impose restrictions on the amount and character of the gases which the smelters are allowed to emit from their furnace stacks. So serious has the problem become that several smelters have had to cease operations altogether, others have been mulcted in enormous costs by law suits, by claims for compensation, or by the installation of plant and processes which they have been compelled to adopt for dealing with the gases. These matters have become subjects of historical importance in the development of smelter practice.
As has been the case in analogous circumstances elsewhere, when interference with the uncontrolled dispersion of then-considered waste products has often proved of ultimate benefit and a source of much profit to their producers, the enforced treatment of highly sulphurous furnace gases has in several instances resulted in considerable gain to the copper smelters. [Pg 189]
Among the methods which are at present economically practicable for dealing with the smelter gases, those of dilution, and of utilisation for acid manufacture are the most important.
The considerations which decide the best course of treatment depend on the numerous economic and local factors which are always of such prime importance in connection with industrial undertakings demanding large capital outlay. The installation of a plant for making sulphuric acid from the gases largely depends on—
(a) For the successful operation of acid-making plant, as at present developed, it is necessary that the proportions of sulphur dioxide in the gases shall not fall below a certain minimum, and further, that the gases shall not contain more than certain limiting proportions of other interfering constituents, such as, for instance, CO2. It is for this reason that the blast furnace operating the true pyritic process furnishes gases of the type most suitable for acid manufacture, since by this process the sulphur-dioxide is obtained in the gases in the most concentrated and the least contaminated form possible under smelting conditions. Even under these circumstances the gases are not in the least of an ideal composition for treatment, owing to their dilution with nitrogen, etc., and the development of the acid-making plants and processes adopted for the successful utilisation of copper blast-furnace gases furnishes a record covering many years of very slow and costly experiment, marked by many preliminary failures and disappointments. These difficulties have now been overcome, as the working of the successful plants attached to both of the Tennessee copper smelters affords conclusive proof, and the sulphur which formerly cost money to dissipate by roasting, now not only acts as fuel, but furnishes a very profitable bye-product.
The requirements for the gases are chiefly the presence of sufficient SO2 and oxygen, and of as little CO2 as possible—factors which depend largely on the proportions of sulphide in the charge. The gas for the acid plant must be supplied in regular and continuous amount, at a specific temperature, and this calls for special care in the smelting operation, furnace manipulation and blast supply, supplementary air admission, etc.
About 3·5 to 4 per cent. of SO2 in the gases delivered at the chambers is the minimum proportion for satisfactory working; CO2 should not exceed about 5 per cent., and about 6·0 per cent. or more of oxygen is also necessary. [Pg 190]
(b) In addition to the capital charges involved in the acid-making installation and the costs of adapting the furnace plant and operations to the process, the problem of putting the acid upon the market on a satisfactory economic basis is important, particularly in view of the competition from other sources. The districts which offer a consuming area for the large and regular supply of acid from the smelters are not unlimited in number, and are probably readily accessible to other sources. In view of the costs of production, the distance of the smelter from the market is a serious consideration, since freight charges on sulphuric acid are high, involving special regulations with respect to the form of car and conditions of traffic, and they may readily exceed all possible profits resulting from the sale of the product.
In Tennessee the companies were forced to install acid plants. That at Copperhill is the largest in the world; commenced in 1906, acid manufacture began about two years later, after much experimenting, and further units have gradually been added. The plant now includes two Glover towers, 30 feet across and 50 feet high, 64 cooling chambers about 11 feet × 11 feet × 70 feet high, eight cooling chambers 11 feet × 24 feet × 70 feet high, twelve old chambers 50 feet × 50 feet × 70 feet, six new chambers 50 feet × 50 feet × 75 feet, eight new chambers 23 feet × 50 feet × 80 feet, eight Gay-Lussac towers, with complementary tanks, etc.—producing at the rate of 168,000 tons of 60° B. acid per annum.
The Ducktown Company’s plant was installed in record time, and, like the Copperhill plant, comprises elaborate dust chambers and flues, with Glover and Gay-Lussac towers of special design and construction, and enormous acid-making chambers with complex valves and fittings. The plant is designed to produce about 160 tons of 60° B. acid daily. The analysis of the gases supplied to the towers varied during the early working of the plant; under fairly normal conditions the average analysis of the gases delivered is SO2 3·5 per cent., CO2 3·5 per cent., SO3 trace; the oxygen in the mixture being about 8·0 per cent. The temperature is also apt to vary. Full details on these points are not yet available for general service.
The management of both companies have been successful in obtaining particularly satisfactory contracts for the purchase of their acid by fertiliser corporations. [Pg 191]
Peters, E. D., “Principles” and “Practice of Copper Smelting.”
Blast-furnace Manipulation.
Shelby, Geo. F., “Alumina in Blast-Furnace Slags.” Eng. and Min. Journ., 1908.
Offerhaus, C., “Copper Blast-Furnace Smelting at Anaconda.” Eng. and Min. Journ., 1908, Aug. 7, pp. 243–250.
Sackett, B. L., “The Granby Smelter Equipment.” Mines and Minerals, 1910, April, p. 524.
“Operations of the Tennessee Copper Company.” Official Annual Reports of the General Manage.r
Walker, A. L., “The Metallurgy of Copper in 1910.” Eng. and Min. Journ., 1911, Jan. 7, p. 39.
Austin, L. S., “Review of Metallurgy in 1910.” Met. and Chem. Ind., 1911, Jan. 11, p. 40.
Rice, Claude T., “Handling Copper Smelting Gases.” Eng. and Min. Journ., 1911, Mar. 25, p. 614.
“Cottrell’s Fume Smelter.” Min. and Scient. Press, Aug. 26, Sept. 2, 1911.
Herrick, R. L., “Boston and Montana Co.’s Smelter at Great Falls.” Mines and Minerals, 1909, Dec., p. 257.
Harvard, F. T., “Condensation of Fume and Neutralisation of Furnace Gases.” Bull. Amer. Inst. Min. Eng., No. 44, 1910, Aug.
“Mineral Industry.” Annual.
Pyritic Smelting.
Holway, John, “A new Application of Bessemer’s Method of Rapid Oxidation, by which Sulphides are utilised for Fuel.” Journ. Society of Arts, Feb. 1879.
Rickard, T. A., “Pyrite Smelting.”
Sticht, Robert, “Ueber das Wesens des Pyrites Verfahrens.” Metallurgie, Nov. 22, Dec. 8, 1906.
Wintle and Alabaster, “Pyritic Smelting.” Trans. Inst. Min. and Met., 1906, vol. xv., p. 269.
Nicholls, F. S., “Pyrite Smelting in Tilt Cove, Newfoundland.” Eng. and Min. Journ., 1908, Sept. 5, p. 462.
Wright, L. T., 44 “Pyritic Smelting without Coke.” Min. and Scient. Press, 1906, Sept. 29.
Sulphuric Acid Manufacture.
Falding, F. J., and Channing, J. P., “Pyrite Smelting and Sulphuric Acid Manufacture.” Eng. and Min. Journ., 1910, Sept. 17, p. 555.
Freeland, W. H., and Renwick, C. W., “Smeltery Smoke as a Source of Sulphuric Acid.” Eng. and Min. Journ., 1910, May 28, p. 1116.
Development of the Process—The Converter—Converter Linings—Grade of Matte—Operation of the Process—Systems of Working.
In modern copper smelting practice, matte of “converter grade,” containing from 30 to 50 per cent. of copper, is bessemerised for the production of metallic copper. Successful practice depends upon a regular and continuous output of matte from the furnace plant being available, and upon a capitalisation and resources on a sufficiently large scale for continuous operation of the whole of the smelting plant.
Development of the Process for Bessemerising Copper Mattes.—The success of Bessemer’s process, which was applied in 1856 to the production of steel by blowing air through molten cast-iron, led to a suggestion for its application to copper mattes and to some experiments on the subject by Semenikow, a Russian engineer, ten years later. It was not until 1878 that any further work was conducted on a practical scale. In that year John Holway suggested and worked out the scheme already referred to, the principles of which as outlined by him, form the foundation of the pyritic and converter practice of the present time. Air was blown through heated Rio Tinto pyrites in an ordinary Bessemer steel converter and the experiments met with considerable success. The apparatus was, however, not deemed convenient, as the process worked very intermittently and large quantities of slag were produced which required to be poured off at intervals, whilst the position of the tuyeres in this form of converter was found to be unsatisfactory. There are many practical difficulties in employing the same kind of apparatus for the converting of copper mattes as for the bessemerising of cast-iron into steel. In the first instance, the final steel product differs but little in weight or bulk from the original charge, whilst the process produces but little slag, owing to the comparatively small proportions of silicon and manganese which require to be oxidised—whereas in copper converting, the quantity of slag produced is almost equal in weight to the amount of matte originally [Pg 193] charged, whilst the resulting copper product amounts to less than one-half of this weight. Further, in bessemerising cast-iron, the blow is of very short duration; in copper matte converting, it occupies more than two hours, and the relative heat losses are, in consequence, markedly different. Finally, the lining of the steel converter chiefly serves to protect the shell; its function in the copper converter was to act also as flux for the iron oxides produced on blowing.
In Holway’s final form of apparatus for the pyritic smelting of copper ore to metal, the introduction of siliceous material as a flux for the iron oxide and the use of basic lining were arranged for, with the object of overcoming the difficulties caused by the corrosion of the siliceous lining which acted as flux.
Though several years elapsed before the pyritic treatment of ore was successfully conducted, the process of bessemerising the fluid matte to metal was successfully applied on a commercial scale by Manhès in 1880, although it was not until the following year that David’s device of placing the tuyeres horizontally and at such a height above the bottom as not to interfere with the metal which is obtained, solved the final difficulties of operation on a practical scale. In 1883–4 the Manhès converter was introduced into the United States, and at about the same time the barrel form was designed by Manhès and David, and was also readily adopted. Both forms developed in size, increasing in capacity from 1 ton to that of 7 to 10 tons.
Until comparatively recent years, the chief modifications in practice were concerned with operating and constructional details rather than with radical changes in the principles of work. Experiments and research have meanwhile been in constant progress with the object of overcoming several of the grave defects connected with the apparent necessity for the destruction of the siliceous converter-lining by using it as flux, which was due to the difficulties of causing the iron oxide to flux with silica when introduced in any other way.
The most vital improvement introduced into converting practice, and that with which the future developments are most closely bound, is the successful adaptation of basic material for the purpose of lining the converter. This achievement, together with recent success in the introducing of siliceous flux, promises to solve many of the difficulties connected with the bessemerising of low-grade matte by a continuous process.
Suggested by Holway, basic linings were tried at the Parrott Smelter, Butte, in 1890, by Keller and others, but under the conditions of working at that time they were found to be unsuccessful when operated [Pg 194] on an industrial scale. Valuable pioneer work was undertaken by Baggaley in Montana, and after many trials, his method was successfully operated for some months at the Pittsmont Smelter under Heywood’s direction in 1906. Visits of inspection to this smelter in 1908 proved disappointing, it being found that most of the plant which had promised the solution of such difficult problems had been dismantled, largely owing to economic difficulties connected with its operation, and the works were in process of re-organisation for the older system of working. Meanwhile, since 1903, Knudsen, at Sulijtelma, Norway, has successfully employed a small basic-lined converting furnace for the combined pyritic smelting and converting of heavy sulphide ores. The process consists usually of pyritic liquation of the sulphides, followed by a further concentration of the matte up to ordinary converter grade by bessemerising, the higher grade matte being then transferred to a silica-lined vessel and blown to metal in the usual way.
The successful operating of the basic-lined converter on the large scale and under the conditions of working at great modern plants was first established by Smith and Pierce at the Baltimore Copper Company’s Smelter, and the method has since been installed and worked with success at Garfield, Utah (five converters in operation, one in reserve); at Perth Amboy, N.J.; at the Washoe Smelter at Anaconda—where the whole plant is being adapted for basic-converting—and at several other works.
A recent and promising development has been the reported successful blowing of fine siliceous concentrates through the tuyeres of converters at the Garfield Smelter, a method by which it might be possible to effect the rapid and efficient extraction of values from fine material otherwise difficult to deal with, affording at the same time a means of conveniently supplying siliceous flux in a manner possessing many advantages.
Principles of the Bessemerising Process.—The principles underlying the converter process are those which form the basis of pyritic smelting practice—of which bessemerising is but a phase. The reactions involve the very rapid oxidation of iron and sulphur under practically ideal conditions, and the fluxing by silica of the iron oxide so produced. The heat of oxidation keeps the materials in a thoroughly molten state, and maintains the temperature well above that required for slag formation and perfect fluidity. The heat derived by the combination of oxygen with the iron and sulphur and that of the iron oxide with silica is developed so rapidly and in such quantity, owing to the large masses now worked with, as to cause a reaction-activity sufficient to make the process independent of heat from external sources. [Pg 195]
It will be noted how markedly the more recent developments of copper smelting have taken advantage of the factors of the time element and mass influence in obtaining enormous heat intensities and consequent high temperatures, by conducting oxidation of sulphides as rapidly and in as large mass as possible. The same absolute quantities of heat per unit weight of charge were liberated in the older smelting methods involving roasting, but the more leisurely manner of operating allowed the dissipation and dispersion of much of this heat, thus necessitating the employment of supplementary carbonaceous fuel.
The Converter.—The converter is a lined steel vessel in which the molten matte is contained, and which allows of air being blown through the material by means of tuyeres which pass through the walls.
The early form of converter was bottom-blown, and similar to that invented by Bessemer, but it was not successful in operation on the small quantities of copper matte worked with, owing to the chilling effect of the cold air on the copper, which, when produced, sank to the bottom and set above the tuyeres, stopping the air blast, and causing much loss of metal in the slag.
The later form of converter was barrel-shaped, with a horizontal row of tuyeres situated at some distance above the bottom so as to allow the copper to settle, protected from the action of the blast, and also to allow of the punching of the tuyeres as required.
The modern forms of converter comprise both the vertical and the barrel types, modified largely as regards size and constructional details, and although the vertical form is still in use and is even preferred at several smelters, it has been largely superseded at most plants by the barrel-shaped variety, whilst the possibilities of greatly enlarged vessels using basic linings are likely to favour this replacement still further.
1. The Upright Bessemer Vessel is used, and found satisfactory at Great Falls and at Mt. Lyell. The general size has been 8 feet diameter and 16 feet height, with a capacity ranging from 5 to 12 tons, according to the condition of the lining, though at Great Falls converters of 12 feet diameter with corresponding capacity are now in use. The advantages of the vertical form are, that, owing to the greater depth of matte through which the air passes, the oxidation is more rapidly conducted, the lining is more efficiently supported, and the wear by abrasion upon the lining is found to be considerably less in amount and to be more uniformly distributed.
On the other hand, the greater depth of matte necessitates a greater blowing pressure in order to force the air through the material, whilst [Pg 196] control over the operations becomes a matter of greater difficulty.
[Pg 197] 2. The Barrel Form of Converter is the type in common use. Among the advantages claimed for this form are those which accrue from being able to operate the same weight of matte in more shallow layers, as compared with the upright form—thus requiring lower blast pressures. Another advantage is the greater ease of regulating the depth of material blown through, by tilting the converter and thus altering the relative position of the tuyeres.
Owing to the successful adoption of the basic lining, the barrel type of converter has now to be divided into two classes, since the basic converter differs from the silica-lined type in constructional details, and is usually of much larger dimensions. Its operation is also conducted on somewhat different lines.
(a) The silica-lined barrel converter varies somewhat in size, the Anaconda converters were, however, representative of the most convenient dimensions.
The shell consists of ¾-inch boiler plate, 8 feet in diameter, and 12 feet 6 inches long. The converter is constructed in two portions, the body and the hood, in order to facilitate removal, relining, and general repairs. The ends are lined with 9 inches of firebrick, and the body with 4 inches; it is then rammed with lining material to a thickness of about 18 inches in all parts. There are 16 1-inch tuyeres placed horizontally, and in the latest forms of converter, the air is [Pg 198] supplied by individual tuyeres which are connected to the blast box, and which are provided with ball-valves to prevent leakages and back-running during the necessary punching. The cavity is about 8 feet × 4 feet by 6 feet deep when first made, and the converter then holds conveniently about 7 tons of matte. The weight of lining is about 16 tons, and it lasts six to nine blows. The blast-pressure used is 16 lbs. per square inch.
The hood is bolted on to the body, and is furnished with conical safety-pieces to give notice of the wearing through of the lining. The converters tilt upon rails, which are strapped round the body, and which travel upon rollers. Motion is communicated to the converter either by connection with an electrical drive, or very often by hydraulic power connecting through a rack to a pinion attached to one of the trunnions. The air supply is usually from piston-driven blowing engines, communicating through a blast pipe to the hollow supporting trunnion of the converter, from which the air passes to the blast box.
(b) The Basic-lined Converter.—The adoption of basic linings is of such recent date that although the present form appears to have given satisfaction, later developments in basic practice may cause further modifications in design. R. H. Vail gives the following details:—
As at present operated, the basic-lined converters are long barrel-shaped vessels consisting of a ¾-inch steel shell, 23 feet long and 10 feet in diameter, lined with magnesite materials so as to leave a cavity about 20 feet × 7 feet × 6 feet. Air is supplied from thirty-two 1¼-inch tuyeres, each separately connected with the blast box and controlled by a valve. Provision has to be made for the marked expansion of the basic lining-material by leaving the top of the steel shell open, joining-up the free ends by tie-rods (13, Fig. 65), whilst the tuyere-pipe connections are flexible. The main opening or throat, [Pg 199] for the charging of matte and flux, is situated in the arch at one end of the converter; it is 40 inches in diameter, and surmounted by a short chimney-cap of iron, which is 30 inches high and lined inside with clay. The vessel is charged through this opening. Metal and slag are poured from the converter through an opening in the side opposite the tuyeres, which is kept closed by bricks during the operations. An oil-burner is provided at one end, for the purpose of supplying such extra heat as might be required, in consequence of undue cooling of the copper towards the end of the blow or for heating up the lining after repairs. The converter is supported as in acid practice, though a tilting device employing wire ropes attached to hydraulic plungers is now being introduced in place of the rack and pinion method.
Converter Linings.—The question of the lining has been the most important consideration in copper matte converting-practice.
The functions proper of the lining material are—
The employment of the lining material as a provider of suitable siliceous flux for the iron oxide, though until recently of vital importance for the practical operation of the bessemerising process, has been a necessary evil in many cases, and although it might have been a source of considerable profit under certain conditions, this function is unlikely in the future to be the consideration of greatest moment.
The vital requirements in modern converter practice are permanence of the lining and efficient means of effecting the fluxing of the iron oxide produced in the converting operation. The necessity for the frequent relining of converters involves not only heavy direct expenses, but it occasions waste of heat in the old linings, waste of [Pg 200] material, loss of time, interruption of the processes, liabilities to outbreaks from the converters, and necessitates much heavy machinery for the conveying of vessels for relining, as well as large capital outlay in relining shops, plant, and appliances. In consequence, the employment of siliceous lining material as flux is usually a most expensive method of supplying the requisite silica; and so much is this the case, that an arbitrary limit to the iron contents of the matte has been rendered necessary, in order to prevent too much of the lining material being used up at a single blow. It was found cheaper to use other means of concentrating low-grade matte to a suitable grade for bessemerising—i.e., to flux off the excess of iron by means of silica in the blast-or the reverberatory-furnace processes.
Siliceous Linings.—Until recently, the only method for fluxing the iron in bessemerising, found practicable on a commercial scale, has been by the destruction of the siliceous lining, minimising the dead losses as much as possible by employing for the purpose siliceous materials from which values in the form of gold, silver, or copper could be simultaneously extracted and collected in the products of the operation.
Numerous attempts were made to effect combination of the iron oxides with silica introduced by some other method, but none met with success. Manhès blew sand through the tuyeres, and obtained as result a spongy unfused mass in the converter—whilst silica introduced in the form of lumps rose to the surface unchanged. In each case what silica was required for flux, was taken up from the siliceous lining. Experiments of a similar nature, in which basic linings were worked with, resulted in the fluxing silica being unabsorbed as before, whilst the iron which was in process of oxidation, not finding a suitable flux, became super-oxidised, resulting in the production of very infusible masses of magnetic or ferric oxides which rendered the process unworkable. Baggaley and others in Montana devoted much attention to experiments on different methods for introducing silica which would flux successfully, methods such as superheating or introducing silica held in suspension in fused silicates being tried, but without marked success, and for many years siliceous linings were necessarily worked with.
Owing to the large quantities consumed, the siliceous material must be obtainable cheaply and in abundant quantities. It should be high in free silica contents, since this constituent alone is effective as flux; it should have the property of binding well with clay or other material, so as to yield a rigid and impervious lining; and most important of all from the economic standpoint, it should carry values, [Pg 201] since by this means only, could its destruction become an actual source of profit. At first barren quartz and barren clay were largely used for linings, but practice gradually developed in the direction of employing more profitable materials, and especially those from which the extraction of the values might present difficulties, in treatment by ordinary smelting methods. The practice as followed until recently at Anaconda is typical of such progress. Until 1908 the lining was chiefly made from highly siliceous ore obtained from Snowstorm, Idaho, carrying 80 to 85 per cent. of SiO2, 4 per cent. copper, as well as gold and silver, and a little iron and sulphur. This ore was crushed in mills and mixed with sufficient slime from the slime ponds of the concentrating plant to make a binding mixture. The slime, which carries about 60 per cent. of silica and also 2·5 per cent. of copper has excellent binding properties, owing to its clayey consistency. The proportions employed were 3 of siliceous rock to 1 of slime—no water was used, the mixture being almost dry to the touch. Since May, 1909, instead of employing ore obtained from outside sources, siliceous second-class Butte ore, which was formerly concentrated, has been very largely incorporated in the mixture used as lining material, it contains 65 per cent. silica, about 3·5 per cent. copper, a little gold and silver, and also iron and sulphur. The lining mixture consisted of 2·9 parts of this material with 1 part of slime. It was thought at first that owing to the greater proportion of sulphides and the lower silica content of the Butte ore, this lining mixture might prove inefficient compared with the former material, but with somewhat greater care in lining, it was found that very little more ore was required, and that tested by comparative silica contents it was more effective. Thus, where the former linings lasted for an average of six 7½-ton charges, equal to 20½ tons of copper per lining, the new ones last 5¼ such charges, equivalent to 17¾ tons of copper per lining, showing that although the efficiency per lining was reduced to 90 per cent., yet, calculated on comparative silica content, the new lining proved to be the more efficient.
The operation of lining is conducted with much care; the old lining is knocked away where necessary, rods are placed through the tuyere holes, and lining mixture is dumped in; 6-inch layers of material at a time being stamped down hard by means of an Ingersoll-Sargent tamping machine, until the lining reaches within 6 inches of the tuyeres. The wooden mould for the cavity, made up of a number of jointed pieces, is then placed in position, and the ramming of layer after layer round the sides is continued as before. The hood, inverted, is lined in a similar [Pg 202] manner, it is then placed in position on the converter body and bolted down, a joint being made of moistened lining material. The whole operation takes about 1½ hours. The converter is then slowly dried by a wood fire, coal being subsequently added and kept burning under the action of a low blast for five or six hours; it is conveyed to the stand when required, dropped into position on the trunnion bearings, and the connections and adjustments very readily made.
The manipulation of relining at the Tennessee smelter is conducted in a very similar manner.
Basic Linings.—The all-important feature of the basic lining is its permanence, which, rendering the frequent relining of the converter unnecessary, allows of many economies in connection with capital outlay on plant and in operating costs. Further, owing to the lessened need for lining repairs, the frequent hauling of converters to the repair-shops situated at the further end of the buildings is avoided. This allows the employment of much larger converter units, with obvious attendant advantages, whilst it increases the ultimate possibility of continuous operation. Thus, the size at present employed, though the process has been in operation but a short time, is 26 feet by 12 feet, with a capacity of 35 to 45 tons of matte, and a daily output of 33 tons of copper from 40 per cent. matte. Such a converter, lined with 9 inches of basic material, will operate for 2,000 to 3,000 tons of copper before requiring repairs.
Keller’s report on basic linings in 1890 stated that they could not be employed successfully, because (a) basic material, being a good conductor, caused the outside of the converter to become too hot and the inside too cold; (b) such material broke up easily and so was unsuitable for use in permanent linings; and (c) even when basic linings were employed, the silica which was added as flux, refused to combine with the iron oxides. These views were very generally accepted for some years, until Baggaley’s persistent efforts and finally those of Pierce and Smith showed that by perfecting the constructional methods and details, by preventing heat losses as much as possible, and by operating on very large masses of hot material, the above difficulties could all be overcome and the basic lining successfully employed. The lining is of magnesia brick, and is 9 inches in thickness, except at the tuyeres, where the bricks are 18 inches thick. In the bottom of the converter and extending to within 18 inches of the tuyere level is placed a filling of ordinary firebrick, which is 13½ inches thick in the middle and 4 inches thick at the sides. The magnesite bricks are laid in dry magnesite powder, except near the [Pg 203] tuyeres, where a mixture of magnesia and linseed oil is used. Expansion cushions of wood are inserted at intervals along the side of the fresh linings which are then “seasoned” with molten copper.
The required quantity of siliceous flux, as calculated, is now successfully introduced by dumping it into the converter, and pouring the matte charge upon it.
The Grade of Matte for Converting.—The grade of matte which is economically the most profitable to treat in the converter is a factor of great importance, since, if limits be fixed, the preliminary smelting stages for matte production are made less flexible, whilst in order to obtain matte of the correct grade, the smelting operations may require to be conducted at greater cost, or else additional smeltings for further concentration of the first matte may be necessitated—as is the case, for instance, in pyritic smelting at present.
The grade of a matte is usually expressed in percentages of copper, but from the standpoint of the practical converter operations, the proportion of iron is the factor which decides the suitability or otherwise of the matte for treatment, and since mattes may be regarded as mixed sulphides of iron and copper, a matte rich in copper is correspondingly low in iron contents, whilst a low-grade matte is high in iron.
The importance of the iron contents of the matte from the viewpoint of converter practice is due to iron being the chief source of heat in the operations, and to the fact that the iron oxide produced from it is the constituent which requires a supply of flux in order that the reactions may proceed and the process be successfully operated. The economic limit to the grade of matte suitable for the converter process is reached when it becomes less costly and more profitable to supply the required siliceous flux for the iron in the ordinary smelting furnace rather than in the converter. So long as the destruction of the lining was practically the only medium by which silica could be efficiently supplied, the limit to the iron contents of the matte was fairly rigid.
The bessemerising of a low-grade matte (low in copper contents, high in iron) entails the great advantage that a high temperature is obtained, owing to the fuel-value of the iron. On the other hand, however, grave disadvantages attend such practice, especially when working with the comparatively small quantities of material usually operated, and when employing siliceous linings. These disadvantages include the factors that—
[Pg 204] In bessemerising a high-grade matte, the heat production is much smaller, owing to the decrease in the quantity of iron, which is the chief fuel of the process, and the limiting grade is quickly reached above which the bessemerising operation upon the matte ceases to be self-supporting.
In consequence, up to a comparatively recent date, a compromise has necessarily been effected, and the grade of matte operated upon has been such as to cause as much heat production as possible, together with the smallest practicable amount of fluxing action.
On these grounds, a matte containing from 40 to 50 per cent. of copper (equivalent to 32 to 22 per cent. of iron) has been found generally the most suitable. At several smelters, lower-grade mattes of from 32 to 40 per cent. copper-contents are converted most profitably, owing to such special circumstances as the profits resulting from the destruction of lining material, or in consequence of the fact that greater operating costs would be involved in concentrating the matte to a higher grade by the ordinary furnace-smelting methods.
In this connection, the successful adaptation of the basic lining by permitting the supplying of flux by means other than from the linings, has very important application and possibilities.
Owing to the frequent relining of these converters being then no longer necessary, mechanical difficulties of conveying the converter bodies to the relining shops are lessened, and larger converter units can now be employed, treating, even at the present stage of development, between six and seven times as large an amount of matte as formerly. By operating on such big charges, pouring off slag as produced, and adding fresh matte and flux without fear of destroying the lining, the difficulties attending the converting of low grade mattes have been successfully overcome.
The limit to the grade of matte economically suitable for the process will depend, in the future, chiefly upon the comparative costs of effecting the required concentration up to any desired grade, in the blast-or reverberatory-furnace, or in the converter.
The modern smelting scheme appears, therefore, likely to develop into the preliminary smelting of the ores by the cheapest method available, for matte of a grade best suited economically to the running of the [Pg 205] furnace, the grade being independent of any rigid limit for the subsequent converting operations—the matte being then bessemerised as usual.
The Converting Process—Acid Lining.—There are two main stages in the converting of copper mattes. The first is essentially elimination of iron sulphide; the second, elimination of the remaining sulphur.
The product of the first main stage is a white metal, practically pure copper sulphide, the iron of the matte having been slagged off in the form of silicate, and the corresponding sulphur eliminated as SO2. The reactions during this stage are well known: the oxygen of the air blown in, yields oxides of iron and of sulphur, as well as some copper oxide. The latter, immediately reacting with iron sulphide which still remains, re-forms copper sulphide, with the production of more iron oxide. The iron oxides are fluxed by the siliceous materials present, forming ferrous silicate slags. The iron oxidation is productive of the greater part of the heat in the operation, and high temperature usually marks this stage of the process, which may be termed “the slagging stage.”
The flame which issues from the converter during this period is usually characterised by a green colour, caused apparently by the formation of iron-silicate slag.
When this stage is completed and the slag poured off, the white metal is blown up to blister copper—this constituting the second main stage of the process. The chief reactions are those of sulphur elimination and the production of metallic copper, caused by the action of some of the copper oxide first produced, upon the copper sulphide still present.
The flame during this period is small, thin, and fairly non-luminous, usually of a red-purple to bronze purple colour.[16]
The progress of the blowing from copper matte to white metal and thence to blister copper is usually indicated and controlled at the smelter by the appearance of the flame which issues from the nose of the converter during the first periods, and by the character of emitted shots during the later stages. This is particularly the case with mattes of moderate purity worked in the silica-lined converter. The successive changes in these indications are gradual, but are easily followed by the experienced skimmer, who is thus able to judge readily as to the manner in which the blow is progressing, and also as to the temperature, composition, and nature of the metal in the converter. [Pg 206]
TABLE XII.—Changes in Composition during Bessemerising.
| Time. | Composition. | |||||
|---|---|---|---|---|---|---|
| Hrs | Mins | Cu. | Au. | Ag. | ||
| % | Oz. | Oz. | ||||
| 11.52 am | Charged No. 1 blast matte, | .. | .. | 46·08 | 0·15 | 31·50 |
| 11.54 am | Blow commenced. | |||||
| 12.04 pm | Sample No. 1 blowing, | .. | 10 | 46·02 | 0·17 | 31·80 |
| 12.14 pm | Sample No. 2 blowing, | .. | 20 | 51·46 | 0·18 | 35·80 |
| 12.18 pm | Punched 3 minutes. | |||||
| 12.24 pm | Sample No. 3 blowing, | .. | 30 | 53·27 | 0·20 | 37·80 |
| 12.25 pm | Punched 7 minutes. | |||||
| 12.34 pm | Sample No. 4 blowing, | .. | 40 | 56·29 | 0·21 | 40·80 |
| 12.40 pm | Punched 2 minutes. | |||||
| 12.44 pm | Sample No. 5 blowing, | .. | 50 | 59·90 | 0·22 | 43·70 |
| 12.45 pm | Punched 2 minutes. | |||||
| 12.54 pm | Sample No. 6 blowing, | 1 | 00 | 62·67 | 0·23 | 44·90 |
| 12.55 pm | Punched 6 minutes. | |||||
| 1.04 pm | Sample No. 7 blowing, | 1 | 10 | 67·89 | 0·25 | 49·20 |
| 1.07 pm | Punched 3 minutes. | |||||
| 1.12 pm | Blow stopped. | |||||
| 1.13 pm | Skimmed. | |||||
| 1.14 pm | Blow resumed. | |||||
| 1.14 pm | Sample No. 8 blowing, | 1 | 20 | 73·97 | 0·27 | 54·90 |
| 1.17 pm | Punched 2 minutes. | |||||
| 1.21 pm | Blow stopped. | |||||
| 1.22 pm | Skimmed. | |||||
| 1.25 pm | Blow resumed. | |||||
| 1.25 pm | Sample No. 9 blowing, | 1 | 30 | 77·82 | 0·28 | 57·30 |
| 1.34 pm | Sample No. 10 blowing, | 1 | 40 | 74·16 | 0·26 | 54·30 |
| 1.44 pm | Sample No. 11 blowing, | 1 | 50 | 81·72 | 0·15 | 57·60 |
| 1.54 pm | Sample No. 12 blowing, | 2 | 00 | 98·50 | 0·78 | 107·70 |
| 2.02 pm | Punched 1 minute. | |||||
| 2.04 pm | Sample No. 13 blowing, | 2 | 10 | 98.57 | 0·40 | 81.60 |
| 2.05 pm | Punched 2 minutes. | |||||
| 2.08 pm | Blow stopped, test for Cu. | |||||
| 2.09 pm | Blow resumed. | |||||
| 2.10 pm | Blow finished. | |||||
| Converted copper, | 2 | 16 | 99.08 | 0·38 | 83·80 | |
| Total time punching, | .. | 28 | .. | .. | .. | |
| Total time of blow, | 2 | 16 | .. | .. | .. | |
| Actual time of blow, | 2 | 09 | .. | .. | .. | |
| Time. | Composition. | ||||||
|---|---|---|---|---|---|---|---|
| Hrs | Mins | Insoluble | Fe. | S. | As. | ||
| % | % | %. | %. | ||||
| 11.52 am | Charged No. 1 blast matte, | .. | .. | 0·15 | 24·30 | 24·70 | 0·22 |
| 11.54 am | Blow commenced. | ||||||
| 12.04 pm | Sample No. 1 blowing, | .. | 10 | 1·30 | 23·70 | 22·95 | 0·07 |
| 12.14 pm | Sample No. 2 blowing, | .. | 20 | 0·30 | 20·50 | 23·10 | 0·06 |
| 12.18 pm | Punched 3 minutes. | ||||||
| 12.24 pm | Sample No. 3 blowing, | .. | 30 | 1·10 | 18·70 | 22·15 | 0·06 |
| 12.25 pm | Punched 7 minutes. | ||||||
| 12.34 pm | Sample No. 4 blowing, | .. | 40 | 1·30 | 16·20 | 21·85 | 0·06 |
| 12.40 pm | Punched 2 minutes. | ||||||
| 12.44 pm | Sample No. 5 blowing, | .. | 50 | 0·90 | 13·70 | 21·95 | 0·06 |
| 12.45 pm | Punched 2 minutes. | ||||||
| 12.54 pm | Sample No. 6 blowing, | 1 | 00 | 1·30 | 11·40 | 21·35 | 0·06 |
| 12.55 pm | Punched 6 minutes. | ||||||
| 1.04 pm | Sample No. 7 blowing, | 1 | 10 | 0·65 | 7·60 | ||
| 1.07 pm | Punched 3 minutes. | ||||||
| 1.12 pm | Blow stopped. | ||||||
| 1.13 pm | Skimmed. | ||||||
| 1.14 pm | Blow resumed. | ||||||
| 1.14 pm | Sample No. 8 blowing, | 1 | 20 | 0·25 | 3·40 | 20·10 | 0·05 |
| 1.17 pm | Punched 2 minutes. | ||||||
| 1.21 pm | Blow stopped. | ||||||
| 1.22 pm | Skimmed. | ||||||
| 1.25 pm | Blow resumed. | ||||||
| 1.25 pm | Sample No. 9 blowing, | 1 | 30 | 0·15 | 0·90 | 19·60 | 0·04 |
| 1.34 pm | Sample No. 10 blowing, | 1 | 40 | 3·30 | 2·60 | 16·60 | 0·04 |
| 1.44 pm | Sample No. 11 blowing, | 1 | 50 | 0·25 | 0·20 | 15·35 | 0·04 |
| 1.54 pm | Sample No. 12 blowing, | 2 | 00 | 0·017 | trace | 0·78 | 0·050 |
| 2.02 pm | Punched 1 minute. | ||||||
| 2.04 pm | Sample No. 13 blowing, | 2 | 10 | 0·052 | 0·01 | 0·78 | 0·033 |
| 2.05 pm | Punched 2 minutes. | ||||||
| 2.08 pm | Blow stopped, test for Cu. | ||||||
| 2.09 pm | Blow resumed. | ||||||
| 2.10 pm | Blow finished. | ||||||
| Converted copper, | 2 | 16 | 0·017 | trace | 0·01 | 0·033 | |
| Total time punching, | .. | 28 | .. | .. | .. | .. | |
| Total time of blow, | 2 | 16 | .. | .. | .. | .. | |
| Actual time of blow, | 2 | 09 | .. | .. | .. | .. | |
| Samples taken each 10 minutes from beginning of blow until finished. | |||||||
[Pg 207] In general character, this colour sequence, during the bessemerising of the ordinary class of copper mattes—i.e., those consisting largely of iron, copper, and sulphur, with but moderate quantities of impurity—does not vary very markedly, but the body and luminosity of the flame depend to a great extent on the nature of the charge and on the working conditions. The colours are intensified by very hot metal, large charges, heavy blast, and rapid working, and particularly by the presence of secondary constituents, such as zinc, lead, or arsenic, which liberate dense white fumes, and so increase the luminosity of the flame.
There are generally four main variations in the appearance of the flame from the acid-lined converter:—
At commencement of blow, |
Oxidation of secondary constituents, |
Dark reddish-brown flame. |
||
| Burning of iron, sulphur, and coal, |
Accompanied by much smoke. |
|||
| Slagging stage, | Iron-sulphide oxidation, | Apple-green flame. | ||
| White metal stage, | Copper oxidation in presence of slag, |
White-blue flame. | ||
| Blowing to blister copper, |
Sulphur oxidation, | Thin red-purple flame. | ||
The changes in composition of the charge during a converter blow have been traced by Mathewson, who assayed samples during the various stages; some of these results are indicated in Table xii. and in Fig. 66. For full record see Trans. Amer. Inst. Min. Engineers, 1907.
In general, of the constituents present in the matte, iron and sulphur are removed very readily, 96 per cent. of the former and 53 per cent. of the latter in the slagging stage of the blow, whilst the elimination of the injurious impurities is high, bismuth and arsenic being removed to the extent of upwards of 90 per cent., and of the antimony, selenium, and tellurium, from 40 to 70 per cent. are eliminated (see p. 217).
Working of a Typical Charge in Silica-lined Converter.—The Anaconda converter plant is now being operated with basic linings. The former practice at this works was representative of the best type of acid-lined working, and the following description, based upon this practice, is typical of the method in general use. There were in operation twelve converter stands of the dimensions previously given. Normal working was to convert the 45 per cent. copper matte to white metal, to pour off slag, blow to blister copper, and pour the resulting metal—in regular sequence.
The colour-changes in the flame during bessemerising are indicated in the colour-photographs reproduced in the frontispiece. [Pg 208]
Seven to 8 tons of matte at an average temperature of 900° C. are charged into the converter, which is in an upright position with the blast on (16 lbs. per square inch). The operation of charging occupies three minutes. A few lumps of coal are thrown in, a vigorous action commences, copious and heavy white fumes and smoke and a full red to red-brown flame being emitted. The converter is now turned slowly back, so as to bring the tuyeres more completely under the charge and ensure more rapid and efficient oxidation, and the blow proper then commences. The flame drops for a time, continuing to be of a red to red-purple colour for two to eight minutes, after which, green commences to show in the red smoky flame (A), indicating that the first or slag-forming period of the blow is beginning. The green colour becomes more prominent and continues for 40 to 45 minutes (B). A preliminary pouring off of slag is then usually made, owing partly to the danger of violent or even explosive interaction which might otherwise occur between matte and slag, and also with the object of keeping down the copper losses in the slag by removing the greater portion of the latter at as early a stage as possible. The blowing is then continued. Flashes of blue now occasionally appear in the flame, and gradually increase in number until the flame becomes blue-white (C), which indicates that most of the iron has been slagged off and that the white metal stage is reached. The blue-white colour of the flame is to be attributed to the production of copper silicate, owing to the tendency of the copper oxide formed by the air blast at this stage, to flux off, and to produce the silicate rather than attack the copper sulphide. This formation of copper-silicate is particularly liable to occur in the [Pg 210] presence of much slag and at high temperatures, factors which are well known to encourage this selective combination, and which prevail at this stage.
The blowing up to white metal takes about one hour.
Slag is then poured off again, until an iron rabble held under the stream commences to show signs of “metal” which give an appearance of spots of grease on the blade. The charge is then usually “doped.” “Dope” consists of highly cupriferous scrap, cleanings, slags, residues, also some siliceous material, added partly for the purpose of cooling down the charge which tends to become overheated at this stage. The converter is turned up again and the blowing is resumed in order to convert the white metal to blister copper.
The main reaction which now proceeds is represented by the equation
Cu2S + 2Cu2O ➡ 6Cu + SO2.
This stage of the blow also occupies about one hour or more, according to circumstances. It commences with a vivid red flame accompanied by smoke, but this soon dies out and a thin purple, almost colourless, flame results, which continues practically unchanged for the remainder of the blow (D). The temperature of the white metal is to some extent judged by the appearance of the flame, a red-brown colour indicating the correct temperature. If the colour be too red, the metal is too cool, and coal is thrown in; if the tint be too orange, the temperature is too high, and dope is added. Constant punching of the tuyeres by long steel chisels is required during this stage of the blow, owing to the lessened heat production due to diminution of iron, and also to the marked tendency for the liberated copper to chill round the tuyeres. The end of the blow is most difficult to judge, and although the size and colour of the flame offer some criterion, the usual and most important guide is the emission of small shots of copper which no longer stick to the hood situated above the converter throat, but which rebound from it. This is the stage where the skill and judgment of the skimmer are most tried.
When the blow is considered satisfactory, the character of the metal is further tested by pouring a small quantity on to the floor—a rugged and uneven surface indicating satisfactory metal. If poured too soon, the copper is coarse and impure; if poured too late, heavy losses in the slag result, owing to excessive oxidation of the metal.
The copper is then poured into a ladle, and conveyed to the refining and casting furnaces.
The whole operation for a straight run occupies about two hours, but the time required in general naturally depends upon the rapidity of [Pg 211] working, and particularly on the grade of matte, and the volume and pressure of the blast.
The slags during the early part of the blow generally carry about 2 per cent. of copper, after the white metal stage is passed, they are usually much richer, on account of the intensely oxidising atmosphere which prevails, and the decreasing quantity of protecting sulphur. These later slags often contain upwards of 20 per cent. of copper, and in consequence as much slag as possible is poured off during the early stages of the blow, and the quantity towards the close is kept at a minimum.
The subsequent treatment of the converter slag depends very much upon the conditions of work at the smelter; at Anaconda, the iron contents of this slag are very useful in the blast-furnace charge, as there is a shortage of suitable basic flux for the silica of the rather siliceous charges. The slag is poured from the converters into ladles, and conveyed to a slag-casting machine, consisting of a conveyor belt carrying cast-iron moulds which are sprayed with cold water, the slag being thus cast into cakes suitable for the blast-furnace charge.
At Tennessee, the pyritic-smelting slags are already too ferruginous for any addition of irony converter-slags in the blast-furnace charge to be desirable, and the only metallurgical treatment for which these are suited is that of recovering from them the large amount of copper which they carry. The molten converter slag is, therefore, poured directly into the blast-furnace settlers, and by this means, the slags are cleaned and the values recovered.
At the new Tooele Smelter, under Mathewson’s organisation, the molten converter slags are poured directly into the reverberatory furnaces, there being no blast-furnace or settler plant, and the cleaning and settling are thus very satisfactorily conducted.
Systems of Working: Acid-lined Converter.—The “normal” system of working—i.e., blowing a matte-charge first to white metal, then to blister copper—is not always practicable nor economically the best practice, and the system of operating the charges depends largely upon the working conditions, which are subject to much variation at different smelters. Even at the same plant, the procedure has to be varied according to the attendant circumstances.
Conditions which may influence the system of working include:—
[Pg 212] As instances of the way in which some of these circumstances affect procedure, the following examples may be quoted.
(a) When working with matte of low grade, especially in small quantities, as formerly operated, the loss of heat by radiation and by that carried away in the large quantity of slag produced, is very considerable, whilst towards the later part of the blow, the amount of sulphide fuel diminishes to such an extent that the maintenance of the desired temperature is difficult. The bulk of the final copper-product of the operation is very small and the metal is therefore liable to chill. In such cases, the system of “doubling” is useful. This consists of blowing the matte to the white-metal stage, pouring off the slag and adding a further charge of matte. This, on the resumption of blowing, restores heat and yields a charge of white metal sufficient to maintain the required temperature for the last stage of the blow, as well as affording a convenient yield of metallic copper.
(b) i. When working with a freshly-lined converter the charge is necessarily rather less than usual, owing to the smaller size of the cavity, and this results in a smaller yield of white metal, which is also colder. At the white-metal stage the slag is poured off, and the cavity having now become larger, owing to the fluxing action upon the lining, a fresh charge of hot matte is added, introducing fresh heat, further enlarging the cavity, and providing for a hot and plentiful supply of white metal for the blowing up to blister copper.
(c) ii. When the lining commences to wear thin, the converter may be retained solely for the purpose of blowing successive charges of white metal up to blister-copper, since owing to the very low iron content of white metal, there is little fluxing action on the lining during this stage, whilst the large quantity of white metal which can be operated in the enlarged cavity ensures a good supply of heat.
When linings burn through, the charge is transferred to another converter and the bessemerising finished there.
The management of the converters as thus indicated, and the distribution of the charges among the various converters are left to the head skimmer, who has control of the converter floor.
Working of the Basic-lined Converter.—The actual operations of bessemerising in the basic-lined converter differ but little from those where the silica lining is used. One important change has, however, been made, viz.: the introduction of the siliceous flux before the commencement of the blow. The lining having been heated up and “seasoned,” the charge of four or five ladles-full (30 to 40 tons) of [Pg 213] matte is poured into the upright converter through the throat, 3 to 4 tons of siliceous flux, which must be well dried, are added, and the blast is turned on gently (at 5 lbs. pressure), whilst the converter is slowly turned back—these precautions being necessary in order to prevent excessive blowing out of the dry siliceous fines at the commencement of the work. When the silica is fairly well incorporated, the blast-pressure is increased to about 10 to 12 lbs. per square inch, the blowing is continued for 30 to 45 minutes, and after the silica has been fluxed by the iron oxides—which is tested by feeling the charge with an iron rod inserted through an opening in the breast—the converter is turned over and the slag poured off. A fresh charge of matte and a further quantity of siliceous ore are added and the blowing is resumed, these operations being repeated several times until the desired quantity of white metal has been accumulated, which is then blown up to blister copper in the usual manner. During the early stages of the blow, the operation is largely controlled by judging the quantity of iron remaining in the matte, from the appearance of small samples which are ladled out of the converter from time to time, and from this, the quantity of siliceous material required for the further fluxing is deduced. This material must be quite dry, so as to flux evenly and not form floaters. One of the advantages of the basic process is that siliceous ores containing values (the extraction of which may be profitable) which might not be suitable for use in siliceous linings, can be conveniently employed as flux in conjunction with the basic lining, though naturally the best work is done with flux containing a maximum of free silica. The character of the slag is not very different to that produced in the silica-lined converter, though it is usually lower in silica contents, and owing to the methods of frequent pouring, it is lower in copper values.
Special Features of Basic-lined Converter Work.—The basic-lined converter tends to lose heat by radiation and conduction more quickly than does the silica-lined vessel, due to the walls being thinner and the lining material a better conductor. Owing, however, to the use of larger charges, to the increased fuel value of the low-grade mattes, and to the larger blast-volume used, heat is retained sufficiently well for the successful operation of the bessemerising process. The temperature is, however, generally lower than that obtained when using the siliceous lining, and constant punching of the tuyeres is necessary—two men being required per shift for this work. The great advantages of the basic lining are connected chiefly with the fact that the frequent relining associated with the silica-lined converter is avoided, hence an extensive relining plant is not required, smaller [Pg 214] building space and a lighter crane can be used. The use of basic linings further affords a means of extracting the copper and other values from siliceous ores which can be used as flux, but which might otherwise be difficult to treat, and it has made possible the cheaper treatment of low-grade mattes.
The disadvantages are chiefly those caused by
Converter Shop Organisation.—The introduction of the basic lining has, to a large extent, overcome the necessity for devoting so much shop space to the repair department, which formerly occupied a very considerable area. The converter stands are usually placed in alignment down one side of the building, the centre space is kept clear, and is commanded by the travelling crane for the conveyance of the ladles of matte, metal, or slag, to or from the converters. At Anaconda, the converters are charged from a train of matte-ladles mounted on bogies which run along a track behind the converters and situated some distance above them, the matte being poured down a launder which swings into position over the converter throat.
At Copperhill, Tenn., the converters are charged from ladles which are filled from the blast-furnace settlers situated at the other side of the furnace-building, whilst at the most modern large plant, at Tooele, Utah, the matte is run directly from the reverberatory furnaces to the converters along launders which are nearly 80 feet long and inclined at about 7 in 100. This method avoids all the handling of matte by cranes and ladles with the attendant troubles of skulls, breaks-down, spills, etc., and no difficulty has been found in keeping the channel free and open, nor in supplying matte at a sufficiently high temperature. At Anaconda and Tooele, the side of the converter-shop situated opposite to the converters is devoted to the refining and casting furnaces and to the slag-casting machines.
Modifications of Converter Practice.—(1) David’s Best Selecting Process.—David devised a special form of converter and suggested a method for conducting in the converter, instead of in the reverberatory furnace, the operations of the best “selecting process” on the principles of the old Welsh practice. The method embodied the converting of the matte somewhat beyond the white metal stage, by which means a small quantity of metallic copper was produced, in which the whole of the gold and silver values and most of the impurities collected, the remaining white metal being left tolerably pure. The metallic copper, thus obtained, was run into a side pocket in the lining and tapped from there, the rest of the pure white-metal was blown up to pure best-select copper.
[Pg 215] The method is, however, too specialised for ordinary commercial copper smelting, especially when electrolytic refining of the crude metal can be conveniently arranged for.
(2) The Haas Converter.—The Haas converter is spherical in form, and the tuyere holes through the lining are arranged at such an angle as to lessen the pressure required for the forcing of air through the metal. It is claimed for this form that it ensures better mixing of the materials and more even wear on the lining, by imparting a swirling motion to the bath.
Douglas, James, “Treatment of Copper Matte in the Bessemer Converter.” Trans. Inst. Min. and Met., 1899, vol. viii., p. 1.
Baggaley, “A Brief Description of the Baggaley Process.”
Heywood, W. A., “The Baggaley Pyritic Conversion Process.” Eng. and Min. Journ., 1906, Mar. 24, p. 576.
Knudsen, E., “Pyrite Smelting by the Knudsen Method in Norway.” Mineral Industry, vol. xviii.
Moore, Redick R., “Copper Converters with Basic Linings.” Eng. and Min. Journ., 1910, June 25, p. 1317.
Editorial, “Improvements in Copper Smelting.” Eng. and Min. Journ., 1911, Mar. 4, p. 450.
Schreyer, Fr., “The Question of the Basic Bessemerising of Copper Mattes.” Metallurgie, 1909, vol. v., No. 6, p. 190.
“Improvements at the Washoe Smelter.” Mines and Minerals, 1910, April, vol. xxx., No. 9, p. 520.
Vail, R. H., “The Pierce and Smith Converter.” Eng. and Min. Journ., 1910, Mar. 12., p. 563.
Moore, Redick R., “Basic-lined Converter for Leady Copper Mattes.” Eng. and Min. Journ., 1910, Aug. 6, p. 263.
“Recent Practice in Copper Matte Converting.” Eng. and Min. Journ., 1910, Sept. 3, p. 460.
Neal, Carr B., “Further Data on the Basic Converter.” Eng. and Min. Journ., 1911, June 13, p. 964.
Keller, E., “A Study of the Elimination of Impurities from Copper Mattes in the Reverberatory and the Converter.” Mineral Industry, 1900, vol. ix., p. 240. [Pg 216]
Mathewson, E. P., “The Relative Elimination of Iron, Sulphur, and Arsenic in Bessemerising Copper Mattes.” Bull. Amer. Inst. Min. Eng., 1907, Jan. 7, No. 13, p. 7.
Offerhaus, C., “Operation of an Anaconda Converter.” Eng. and Min. Journ., 1908, Oct. 17, p. 747.
Levy, D. M., “The Successive Stages in Bessemerising Copper Matte as indicated by the Converter Flame.” Trans. Inst. Min. and Met., 1910., vol. xx., p. 117.
Hixon, H., “Notes on Lead and Copper Converting.”
Semple, Clarence C., “Analyses of Converter Fume.” Eng. and Min. Journ., 1911, Mar. 11, p. 508.
Haas, Herbert, “The Vortex Copper Converter.” Eng. and Min. Journ., 1910, May 7, p. 972.
Preliminary Refining and Casting into Anodes— Electrolytic Refining—Bringing to Pitch, and Casting of Merchant Copper.
The further treatment of the converter metal depends to a large extent upon its composition, and the purpose for which it is intended. The matte-smelting operations on copper ores bring about the elimination of the greater part of the constituents accompanying the copper. The converter-grade matte may, however, in addition to the copper, iron and sulphur, also contain considerable proportions of easily reducible impurities of the ore, possessing a greater tendency to enter the matte than to be oxidised and eliminated in the slag. Such constituents may include gold and silver (practically all concentrated and retained in the cupriferous product), arsenic, antimony, bismuth, selenium and tellurium (retained to very considerable extent), as well as lead, zinc, nickel and cobalt (in much smaller proportions). The amount of these latter impurities ultimately retained in the converter matte depends very largely upon the proportions originally present in the ore, and upon the smelting conditions.
Under the strongly oxidising conditions of the Bessemer process the copper retains but small quantities of impurity, and those which do remain in ordinary converter metal may be broadly divided into two classes[17]—(a) those which are oxidisable with comparative ease, and (b) those which persist in the metal even under oxidising influences, unless treated by special means. The former include iron, sulphur, and zinc; the latter, arsenic, antimony, bismuth, selenium, tellurium, gold, and silver. Keller gives the following figures for the average elimination of the impurities in the converter:—
| Iron, | 99 | per cent. |
| Sulphur, | 99 | " |
| Zinc, | 99 | " |
| Cobalt, | 99 | " |
| Bismuth, | 97 | " |
| Lead, | 96 | " |
| Arsenic, | 81 | " |
| Antimony, | 71 | " |
| Selenium, | 47 | " |
| Tellurium, | 40 | " |
| Nickel, | 37 | " |
[Pg 218] Of the persistent elements, the retaining of the gold and silver in the converter-copper is a factor of much economic advantage, but the other impurities are curiously just those which are characterised by possessing most injurious effects on copper intended for electrical work—for which purpose most of the material is employed.
The demand for particularly pure metal in electrical and conductivity work therefore usually necessitates a further purification of the converter-copper (unless it be an exceptionally pure brand) and the production of metal specially free from the injurious constituents which persist to a small but sometimes very appreciable extent in the metal under the ordinary oxidising conditions. The presence of silver and gold in the copper may afford in many instances sufficiently good reason for a separating process independently of the market for the pure copper itself.
In modern practice, electrolytic methods are almost universally employed for the purification of the crude copper. By this means the large demands of the present day can be conveniently met, and the copper be obtained in a condition of remarkable purity. The frequent presence of gold and silver in the metal, and the convenience and completeness with which they are separated on electrolytic treatment of the copper are particularly advantageous features which recommend the adoption of electro-refining, and may in some cases be the reason for this procedure even though the metal might otherwise be already quite up to specification for electrical service. In the large majority of cases, these bullion-values constitute a welcome and independent bye-product, the returns from which may be set against the expenses of the refining operations on the copper, which might, in any case, be necessary.
The process may, therefore, be operated with one of the following objects—
Under the present industrial conditions, the electrolytic refineries are located at centres often at very considerable distance from the smelters. Situations for the refineries are chosen where the local conditions as regards power supply, technical resources, and particularly proximity to markets and distributing centres, allow of the operations being conducted under the most advantageous [Pg 219] circumstances, and it is customary for smelters situated in the remoter mining districts to ship the crude copper to these custom refineries, instead of conducting the process themselves. At Anaconda, the well-equipped electrolytic refineries have been closed down, and the anode metal shipped to the Eastern refineries for treatment.
Preliminary Refining of Converter Copper and Casting into Anodes.—For modern electro-refining practice, the crude metal must be prepared into anodes, which are usually in the form of plates about 2 feet 6 inches × 3 feet by 2 inches thick. It is found that the metal as produced in the converters, on being cast into such plates, does not as a rule yield anodes which work satisfactorily in the tanks. This is largely owing to the impure and crude condition of the metal, which results in the production of plates which are spongy, coarse, and exceedingly rough and uneven on the surface. In consequence the direct employment of such metal would occasion irregularity and difficulties in the operation of the tanks, giving rise to short circuits, uneven wear, breaking off in large pieces, and similar troubles. Furthermore, the tank liquors and slimes become badly contaminated if large quantities of impurity be present in the anodes, and the deposition of good clean metal is thus greatly interfered with. All these reasons render it advisable that the converter-copper should, as a rule, undergo a preliminary furnace treatment before being cast into anodes.
The Anaconda practice is representative of the manner in which these preliminary refining and casting operations are conducted, except that the enormous scale and organisation of the operations are practically unique. The principles involved and the general method of operation are in all essentials those of the old Welsh furnace-refining process.
The Furnace.—The finished metal from the converters is teemed into ladles, and from these is poured directly into one of three casting furnaces. Two of the furnaces are in constant use, one of them engaged in refining, one being filled, and one in reserve or repair. Two of the furnaces are 14 feet × 22 feet 8 inches hearth dimensions, with a capacity of 95 tons; the third has a 14 feet × 28 feet hearth, and a capacity of 110 tons—the fire-boxes being 5 feet 6 inches × 7 feet. The furnace bottom is constructed of the local silica brick (which is claimed to be the finest in the world) laid down in four beds, the three lower being each 12 inches thick, whilst the working bed is constructed of 20-inch bricks; brick being found to be better than sand in this class of work. The bottom is curved to a depth of 2 feet. These furnaces are, in consequence of their different function, constructed on somewhat different principles to the reverberatory smelting furnaces. [Pg 220]
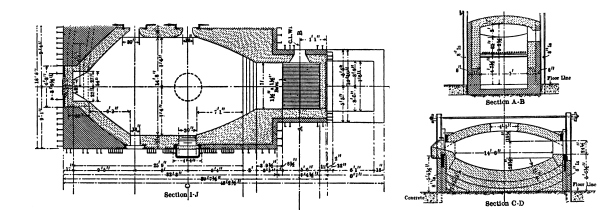
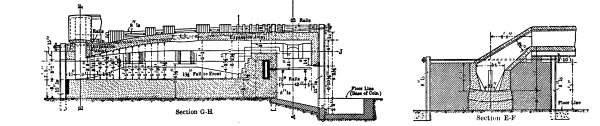
Fig. 69.—Sectional Plan, Elevation, and Transverse Sections of
Refining and Anode-Casting Furnace, Anaconda (Peters).
[Pg 221] Owing to the high conductivity of copper, and to the fact that the functions of the furnace are either largely as a medium for simple fusion or as a receptacle for molten metal, and further, that but little slag is produced, that no settling and separation of the fluid materials are required, and that there is no danger of dusting-losses, the furnace may conveniently be built with a deep hearth which need not be of very considerable length. The main requirements are refractoriness of the building materials, particularly careful construction so as to avoid breakouts, and very strong bracing indeed on account of the deep and heavy bath of material which is carried on the furnace hearth.
Operations—(a) Oxidation Stage.—The furnace is loosely filled with scrap copper which has accumulated round the works (8 to 12 tons), and converter metal (of composition say about 98·3 per cent. copper) is then poured in at the side door from ladles bringing it in quantities of about 5 tons at a time, as teemed from the converters. When the furnace is about half-filled, a blast of air at 90 lbs. pressure is injected through the metal by means of iron pipes, which at this stage just dip below the surface. These pipes are gradually eaten away by oxidation and slagging action, but as the end wears down, the pipe is pushed further in. The function of this air blast is to supply oxygen for the purpose of acting upon the small quantities of oxidisable impurity which remain in the metal after bessemerising, and which consist chiefly of iron and sulphur, in addition to the small quantities of metalloids. The oxygen partly acts directly on these constituents, but as already indicated, the scouring action is to a great extent performed by copper oxide which is produced and which is itself a powerful oxidising agent. The iron appears to be one of the first elements to be removed, and then a little sulphur, but this is chiefly eliminated after the iron has been oxidised. The interaction between the copper oxide and the sulphides liberates metallic copper and yields SO2, which bubbles up through the metal and gives to it an appearance of “boiling,” by which name this stage is known. Too rapid an oxidation during the early stages is dangerous if much sulphur be present, owing to the evolution of sulphur-dioxide assuming a degree of explosive vigour. Up to this point, the oxygen has been utilised in removing iron, sulphur, etc., which are eliminated as oxides, so that but little of the oxygen is retained in the metal, but after the boiling stage is passed, oxygen is actually absorbed, the copper now [Pg 222] becoming oxidised, and the oxygen contents of the metal rapidly increase. As in the analogous instance of steel bessemerising, it appears essential to introduce some excess of oxygen into the metal in order to ensure the complete removal of the oxidisable impurities, so in copper-refining, an excess which amounts to about 0·7 per cent. of oxygen (equivalent to about 6 per cent. of Cu2O) must be introduced.
In the refining practice as conducted by the Welsh process, much of this aëration took place during the slow melting down of the crude blister copper, and subsequently during the flapping operations with the rabble; but the use of the air-blast hastens this oxidation considerably, especially as the metal is now often directly poured into the furnace in a molten condition, so that oxidation during melting is not possible. It is essential to defer this final oxidation and elimination until it can be conducted at the refining furnaces rather than to attempt it in the converter, since the refining furnace allows of the operation being performed much more gradually and under better control, whereas if conducted in the converter, the necessarily vigorous action would occasion unduly heavy losses of copper in the slag and probably excessive oxidation of the metal.
During the aëration, the furnace contents are continually added to, by additional ladles-full of metal, and usually by the time the furnace is filled the air-blast has oxidised most of the impurities from the metal. These have entered the slag, and the copper has become “dry,” owing to the necessary super-aëration. If this stage has not been reached, the sample often shows “sprouting” (also known as “spewing” or “throwing a worm”), which is caused by the escape of SO2, and indicates that all the sulphur has not been eliminated. In that case the blowing is continued until small samples ladled out from the bath exhibit the characteristics of dry copper, viz.: the depression down the middle line of the ingot, brittleness of the metal, and a purplish brick-like fracture.
These preliminary operations may occupy some three or four hours or more.
(b) Poling and Bringing to Pitch.—The oxidation having proceeded to the required stage, the highly cupriferous slag is skimmed off, after being first thickened with ashes from the fire-grate, and the poling of the metal is then commenced. This operation is conducted by immersing poles of timber, three to six at a time, in the metal, holding them well under the surface and pushing them further in as the ends burn away. It is essential that the timber should be green and not dry, and preferably it should be hard wood, such as birch, beech, or oak. The poles are usually as long as possible, and are from 6 to 8 [Pg 223] inches thick at the butt end. The function of the wood, particularly during the early stages is, to a great extent, mechanical, and any chemical changes effected are by indirect action.
The poling operation really consists of two stages, the first of which is the final elimination of SO2 retained by the metal, and the last, the actual reduction of the excess oxide and the “bringing of the metal to pitch.”
The green timber, when inserted into the copper, liberates large amounts of moisture and reducing gases which agitate the bath considerably and “shake” the gas out of the metal more or less mechanically, replacing, at the same time, some SO2 by CO and hydrocarbons which copper possesses the power of absorbing. When the SO2 has been satisfactorily eliminated, the reduction stage is arrived at, and this is conducted in a manner similar to the familiar poling operation of the Welsh process. The surface of the bath is completely covered over with a layer of coke, anthracite, or charcoal, and more poles are inserted. The exact mechanism of the operation has not yet been definitely traced, but the action of the wood at this stage is partly of a mechanical and partly of a chemical nature. The reducing gases liberated by the charring and destructive distillation of the wood have themselves a reducing action on the oxides which are dissolved in the dry copper, but an important feature of the action of these gases is the agitation and splashing which they occasion, thus bringing the molten metal into close and vigorous contact with the layer of reducing carbonaceous material maintained upon the surface of the bath.
Poles are inserted usually two or three at a time, and samples are constantly taken and examined for surface indications and for fracture. This preliminary refining operation usually has for its chief object the preparation of a fairly pure metal which will yield a sound, clean, and even anode casting, and which is not required at this stage to pass the rigid mechanical tests essential for the market product. In this case it is therefore usual to carry the poling operation only to such a degree that the samples ladled out and cast into small ingots solidify with the even, smooth surface desired and which is characteristic of “tough-pitch copper”—irrespective of any special mechanical properties of the metal. If the test is satisfactory, the metal is ready for casting.
The poling occupies some hours, and usually from 40 to 50 poles of wood are used up before the metal is in a suitable condition for casting. During these operations the coal fire in the grate is manipulated in a [Pg 224] manner best suited to the various stages of the process; there may thus be an oxidising flame during the early part of the refining, but the flame must be of a reducing character whilst poling is in progress.
Casting.—Until comparatively recent years, the size of the refining furnace has been necessarily limited to small dimensions, owing to the difficulty in emptying the furnace of large charges. The practice, as conducted hitherto, has been based on the familiar method of the old Welsh process, viz., that of ladling out the metal by small hand ladles. This involves so much hand labour, and requires such a long period of time for its operation as to make practically impossible any attempt to deal with large quantities of metal, or to lead to any considerable increase in furnace capacity. The chief difficulties to be overcome when operating on large charges of copper by this hand-lading method are those of maintaining the metal at the correct pitch during the lengthy period of ladling; whilst the large amount of time during which the finished copper has to remain within prevents the furnace being used for its chief purpose, that of refining more metal.
The method of hand ladling was employed for so many years on account of the difficulties of controlling the stream of metal and of tapping the furnace in the usual way—i.e., through a tap-hole at the lowest point of the bath. These difficulties were due to the very high working temperatures, to the great weight of metal behind the stream, which forced it out under great pressure, and to the high melting point, conductivity, and tendency to chill of the copper, which was apt to cause setting of metal in the tap-hole, and led to the latter becoming rapidly closed up and useless. Regulation of the stream of metal to a gentle flow was impossible under such conditions.
With the introduction of casting machines by Walker, and the improvements in the methods of tapping by the adoption of the vertical tapping-slot, these difficulties have been removed, and the casting of 100 tons of metal from one of the modern large casting furnaces presents, to experienced workers, little practical difficulty.
The modern casting machine brings a series of moulds continually under the supply of metal which issues from a large ladle fed continuously from the tapping-slot of the furnace.
The method of tapping now used is to allow the copper to gently run out of the furnace, by gradually lowering the level of a temporary retaining wall which is constructed in a narrow vertical slot in the tapping side of the furnace. [Pg 225]
This slot, which is ![]() -shaped
in plan, extends from the lowest point of the hearth to well above the
highest possible level of the liquid metal. It is about 3 feet high and
4½ inches wide, and whilst the furnace is working it is kept rammed
with a mixture of loam and anthracite, this filling being supported by
a series of short transverse bars, 16 inches long and 1 inch square in
section, which are set 3 inches apart and rest upon lugs fixed to the
iron plates which strengthen the furnace-wall. During the operation
of casting, this hard filling can be readily cut away as required and
the level of the dam thus gradually lowered at will, permitting the
gentle and continuous overflow of the molten metal. The stream is also
regulated by inserting a pole of wood in the opening, should the flow
become too rapid, and by this means it is kept under absolute control.
The molten metal flows along a spout which feeds a small suspended
[Pg 226]
ladle of about 800 lbs. capacity, the supply being so regulated that
this ladle is filled sufficiently slowly as not to get ahead of the
moulds. The ladle is supported hydraulically, and is pivoted so that
it can be brought forward and tilted for pouring, and then lowered and
moved a slight distance backwards, to allow the next mould to come into
position.
-shaped
in plan, extends from the lowest point of the hearth to well above the
highest possible level of the liquid metal. It is about 3 feet high and
4½ inches wide, and whilst the furnace is working it is kept rammed
with a mixture of loam and anthracite, this filling being supported by
a series of short transverse bars, 16 inches long and 1 inch square in
section, which are set 3 inches apart and rest upon lugs fixed to the
iron plates which strengthen the furnace-wall. During the operation
of casting, this hard filling can be readily cut away as required and
the level of the dam thus gradually lowered at will, permitting the
gentle and continuous overflow of the molten metal. The stream is also
regulated by inserting a pole of wood in the opening, should the flow
become too rapid, and by this means it is kept under absolute control.
The molten metal flows along a spout which feeds a small suspended
[Pg 226]
ladle of about 800 lbs. capacity, the supply being so regulated that
this ladle is filled sufficiently slowly as not to get ahead of the
moulds. The ladle is supported hydraulically, and is pivoted so that
it can be brought forward and tilted for pouring, and then lowered and
moved a slight distance backwards, to allow the next mould to come into
position.
On tilting the ladle, the metal flows gently and without splashing through a three-hole grid in the front—which keeps back slag or cinders—and runs into the mould, which is rapidly filled. In order to prevent the metal overflowing in the mould, and also to rapidly cool that portion which forms the lugs of the anode-plate, a hollow water-cooled block 2 feet 6 inches long and of 6 inches square section, situated opposite the ladle, is brought forward hydraulically into such a position that it rests on the mould just against the edge of the lugs.
At many smelters the circular form of anode casting machine introduced by A. L. Walker is employed. This apparatus consists of a horizontal wheel which can be rotated slowly, carrying a series of arms at the end of which the moulds are supported, so that they form a broken ring. By the rotation of the machine, one mould after another can be brought under the ladle and filled. The moulds are pivoted, so as to allow of tilting, and when the metal has set, the ingot is thus dropped into a cold water bosh, whence it is carried to the yards by a conveyor. At Anaconda, the casting machine consists of a series of moulds carried on a platform conveyor which is operated hydraulically—the moulds are attached by bolting them on to the belt through lugs fixed underneath. The moulds are constructed of 1 inch cast-iron, and allow of the production of anode ingots 2 feet 6 inches × 3 feet by 2 inches thick, provided with lugs at the corners of one end for the purpose of supporting the plates in the tanks.
Each mould holds about 560 lbs. of metal, and when the anode has been cast, the ladle is dropped back into position and the mould is moved forward by means of the conveyor belt. After traversing a distance equal to three times its own length, the ingot becomes fairly solid, and at a point corresponding to this position the conveyor base inclines slightly upwards. The cake is sprayed gently during its passage over a distance of about 8 feet, the conveyor belt then passes over a pulley-wheel, and when in a vertical position, the anode is forced out of the mould by a crowbar and falls into a water bosh, from which it is carried by another conveyor on to a platform. Here it is wheeled to stacks, examined for flaws, and weighed. Sample anodes are placed on one side, and the others are packed for shipment to the Eastern refineries.
[Pg 227] The furnace deals with one charge (usually of 100 tons, but occasionally much more) per eight-hour shift, and the casting machine yields 25 tons of anodes per hour.
Samples weighing from 4 to 6 ozs. are taken three times per shift from the stream of copper running into the moulds, by batting the metal into water with a wooden paddle. This method checks very well with drillings taken from the anode plates, the chief discrepancy feared having been with respect to silver contents, owing to the tendency of this metal to segregate. The assay of the anode metal at Anaconda averages copper 99·3 per cent., silver 80 ozs. per ton, and gold 0·5 oz. per ton.
Electro-Refining.—Electrolytic refining was introduced on a commercial scale by Elkington at Pembray in 1865, and with the general adoption of the dynamo for the production of power, dating from about 1870, the process was greatly developed. Most of the copper now placed on the market has passed through the electrolytic refinery.
System of Working.—The method of arranging the electrodes in the depositing tanks which is usually adopted at the great refineries at the present day, is that known as the parallel or multiple system.
In this method of working, the anodes are all connected to one pole of the circuit, and the cathodes, situated between them, are all connected to the other. In this way, each tank comprises in reality one large anode and one large cathode, and the voltage as measured between any two neighbouring electrodes will be the same. The system thus allows of currents at low voltage being employed, since the voltage is a factor of the number of electrodes in series, and in consequence danger of short circuiting is lessened. This allows of plates being placed closer together in the tank, with less danger from this source of trouble.
A large number of tanks are employed at the refineries, and they are usually arranged in series, the anode plates of one vat being connected to the cathode plates of the neighbouring one, the current thus passing from one vat to the other through the entire system.
Various other methods of arranging the electrodes have been favoured from time to time, and of these the series-system is the most important, this being still in use at several large refineries, though it has been generally superseded by the multiple method.
The plan underlying the series method was that of avoiding the trouble and expense of preparing and working with the special cathode sheets of pure copper as are necessitated by the multiple system. In the series-method, each anode was made to serve as a depositing surface for [Pg 228] the pure cathode copper produced by the operation, so that as impure anode copper was dissolved away on one side of the “anode”-plate, pure copper was gradually deposited upon the other side. This system appeared, therefore, to have several marked advantages to recommend it, but in practical operation many difficulties in working and several serious disadvantages were encountered. The chief points in favour of the series-system are—
(a) Smaller first cost of the installation, particularly in the matter of electrical connections, since the multiple system requires heavy leads running along each side of the tank, as well as close attention to the providing of good contacts, in order to connect all the anodes and all the cathodes together with a minimum of current leakage. In the series-method, the plates are readily connected one to the other.
(b) The great saving of the cost of preparation and arranging for specially pure cathode plates, this constituting a very important factor in the costs of the multiple process.
(c) The output of metal per vat is greater.
On the other hand, the disadvantages of the system, except under special conditions, are very serious.
(a) More scrap is produced and requires re-treating, owing to the difficulty of separating the new deposit from the remaining portions of old anode, which often adhere very firmly.
(b) Higher voltage through the tanks is required, owing to the large number of electrodes in series in the bath. Hence the danger of current-leakage and short circuiting is greatly increased, especially when impure anodes are used, since they tend to produce conducting layers of mud on the bottom of the vat.
(c) The anode plates have to be made particularly smooth and even on the surface, since in order to lessen the voltage required, the plates are brought as close together as possible, in consequence of which, any excrescences upon the surface greatly increase the danger of short circuiting. Preliminary furnace-refining and special straightening of the anodes are therefore essential in connection with the series method.
(d) Special tanks are required, as the protecting lead liner cannot be employed, since the danger of current-leakage through it is increased, owing to the higher voltages required. Hence special acid-resisting material, such as slate, is necessary, the expense of which is considerable.
(e) The cost of stripping the cathodes is high, and the operation is often difficult.
(f) The cost of maintaining the plant is greater.
[Pg 229] The special advantages of the multiple system are that—
It is, however, general to carry out this preliminary refining, which yields sounder anodes, keeps the electrolyte purer, and promotes the more regular working of the electrodes and electrolyte—although some smelters still cast the anodes direct from converter metal.
Summarising, it is found generally that—
Outline of the Process.—The electro-refining industry is a highly specialised one, and the methods of putting the comparatively simple underlying principles into practical operation have assumed great complexity and diversity in detail, concerning which Ulke has collected and published much valuable information.
The following details have more particular reference to the multiple system of working, as being the most representative of the electro-refining methods in general use.
The outlines of the process constitute the passing of direct electric current through tanks containing acidified solutions of copper sulphate, employing plates of crude copper as anodes, and depositing pure metal upon cathode-plates of specially-refined thin sheets of pure copper. The precious metals and most of the impurities of the anode metal are liberated as small insoluble particles which gradually settle to the bottom of the tanks in the form of mud, soluble constituents, such as iron and zinc, first passing into solution.
General Conditions—Anodes.—The usual dimensions of the anode-plates are 3 feet high by 2 feet 6 inches wide and about 2 inches thick; they are generally cast with lugs, so as to allow of suspension in the tanks. [Pg 230] The anode metal is usually brought by a preparatory operation, to as high a state of purity as is economically practicable—
(a) The necessity for the employment of solid and even anodes has already been indicated; it allows of closer suspension of the electrodes, lessens the liability of sprouting and unevenness on the deposits and the irregular wear and breaking up of the anode-plates before they are sufficiently worn away.
(b) The more free the metal is from iron, sulphur, zinc, nickel, etc., the purer remains the electrolyte, since these elements pass into solution at a greater speed than does the copper itself, and, gradually concentrating in the tank liquors, render them more and more impure—the purity of the metal deposited at the cathode being in consequence decreased. The preliminary refining and bringing up to pitch of the metal before casting into anodes, as already described, thus has for its object the preparation of electrodes in a suitable mechanical as well as chemical condition. The copper content is rarely less than 98 per cent. and is often more than 99 per cent. The gold and silver contents are not affected by this preliminary treatment, nor are, to any great extent, the proportions of arsenic, antimony, bismuth, etc.
The size of the anode-plate varies somewhat at different refineries, the usual standard dimensions being indicated above; the size depends to a large extent upon the facilities for handling the electrodes and on the circuit system operated. There is a tendency at several works possessing suitable facilities, to increase the size of the electrodes.
The Cathodes.—The copper is deposited from the electrolyte upon cathode sheets, which are usually thin plates of pure copper corresponding in size to the anodes. As these sheets cannot be conveniently provided with suitable lugs for suspension, they are usually made of somewhat greater length than the anodes, so as to allow of bending over the cross-conductors; otherwise they are furnished with metallic clips for attachment to these bars.
These cathode sheets are prepared by depositing layers of pure metal upon plates of refined copper of suitable surface dimensions and of about ¼ inch thickness. Each side of these plates, which are specially smoothed, is first slightly oiled so as to allow of the subsequent convenient stripping of the sheet when made, and it is then well coated with graphite in order to present a conducting surface on [Pg 231] which deposition can proceed. The cathode-sheets are deposited either in the regular tanks of the refinery or in vats specially devoted to the purpose, using in that case, pure electrolyte, and working in the usual manner. On attaining a thickness of about 1⁄25 inch, the sheet is stripped off, cleaned and clipped ready for use as a cathode.
The Electrolyte.—The electrolyte is essentially an acid solution of copper sulphate. The average proportions are from 15 to 20 per cent. of copper sulphate crystals, and from 5 to 10 per cent. of sulphuric acid—the usual density of the solution ranging from 1·12 to 1·25. The liquid under ordinary conditions of working, remains reasonably pure for a considerable time. It tends, however, to decrease in acidity and to increase in copper contents, partly owing to the presence of cuprous oxide in the metal, which passes into solution independently of the indirect transference of metallic copper from anode to cathode. The composition of the tank liquors must, therefore, be frequently checked. The gold and silver values do not pass into solution under ordinary working conditions, and the addition of a small quantity of common-salt or of hydrochloric acid to the vat effectually prevents any silver from remaining in solution in the liquors.
A considerable proportion of the arsenic in the tough-pitch anode-copper, existing as arsenate, is deposited with the mud residues, it being insoluble and non-conducting. Arsenic in a reduced condition is, however, soluble, and may gradually concentrate in the liquors and contaminate the cathode copper, unless suitable precautions are taken. Some of the reduced arsenic, moreover, tends to produce a slimy arsenite of copper, which, though insoluble, exists in a colloidal non-settling form. Addition of ammonium sulphate to the electrolyte prevents this formation, whilst combined aëration and heating promote the precipitation of arsenic as insoluble arsenates which settle with the tank slimes.
Some of the antimony and bismuth tend to first pass into solution, but for the most part they are precipitated as insoluble basic salts. Under suitable conditions with respect to the acidity and copper contents of the electrolyte, there is little tendency for deposition of these impurities with the copper, but deviation from the correct composition is liable to cause contamination of the deposited metal. These impurities, when in solution, tend to be oxidised by aëration, and this operation greatly encourages their precipitation with the mud.
Iron readily dissolves in the electrolyte, forming soluble ferrous sulphate which tends to gradually accumulate in the solution. This [Pg 232] contamination spoils the quality of the deposited metal, and interferes with the process of deposition, decreasing the conductivity of the bath and thus necessitating higher voltage. Aëration of the solution, especially when warmed, leads to the formation of basic ferric sulphates which are insoluble, and which therefore accumulate at the bottom of the tank.
Selenium and tellurium, which when present most probably exist as insoluble selenides and tellurides of copper or silver, are also precipitated, and thus do not find their way into the deposited metal.
The copper itself is deposited from the electrolyte on to the cathode-sheets by the action of the current, whilst at the anodes, the metal passes into solution, and the other constituents are either dissolved or precipitated. It follows that in an undisturbed solution, the liquid near to the cathode becomes gradually impoverished in copper, resulting in a decrease in the rate of deposition and necessitating greater electrical pressure, whilst in the neighbourhood of the anode, the liquor is proportionately stronger in copper and less acid in character. Should these conditions continue to any great extent, the working of the bath is seriously interfered with, since diffusion proceeds too slowly for uniformity to be restored, and in order to secure uniform composition of the electrolyte, it must be maintained in gentle motion by some system of circulation. This agitation and mixing is assisted by the aëration of the bath for the purpose of hastening the oxidation and precipitation of several of the impurities—this being effected by blowing through the liquid a gentle supply of air.
Temperature.—The electrical resistance of the solution decreases as the temperature rises, and in practice the bath is maintained at a uniform temperature of 45° to 50° C. By this means a useful increase in conductivity is obtained, the strength of the deposited copper being at the same time greatly augmented.
Electrical Conditions.—The electrical factors which mainly control the working of the electrolytic process are those of—
(a) Current Density.—The quantity of metal deposited from an electrolyte is proportional to the current which passes, and to the electro-chemical equivalent of the particular metal. Thus a current of one ampere will deposit from copper sulphate solution, 1·1832 grammes of copper per hour, and the total quantity deposited in any given time is determined by the product of the current, the time, and this electro-chemical equivalent (which is determined experimentally). [Pg 233]
In practical operation, a factor which is amongst the most important of those governing the working of a plant, is the current density, or current per unit of area of depositing surface, since from this factor the rate of deposition upon the cathode plates is determined, and from it the power requirements, accommodation, etc., for the plant are fixed. The current density is subject to wide variation, but, as a general rule, it ranges from 8 to 18 amperes per square foot of plate-area. Its value is largely dependent upon the speed of working, the cost of power, and the composition of the anode metal, the electrolyte and the desired product, etc.
In general, high current-density possesses many advantages, resulting from the fact that it occasions a more rapid deposition. It causes a proportionately greater output, consequently the stock of metal held back in the tanks is reduced, and hence there is less capital locked up in the form of metal undergoing treatment, and less plant and accommodation are required for the same output.
The current density permissible is, however, limited by the composition of the electrodes and the solution. High current-density causes rapid dissolution of the anodes, and if the plates are not particularly free from impurity, the electrolyte rapidly becomes contaminated, since its dissolving power on the impurities becomes greater with increased electrolytic action, and this affords less opportunity for the precipitation and settling of the injurious constituents. In consequence, the cathode copper is contaminated through this mechanical inclusion of impurities, whilst electro-deposition of some of these materials may also be encouraged. The presence of much silver in the anodes causes the rapid breaking-up of the plates, especially if the current density be high, and thus the separation of the values in the slimes is not so efficiently managed. With high values in the anode copper, it is necessary to reduce the current density to 8 or 10 amperes per square foot, whereas with purer metal a density of as much as 16 to 20 may be conveniently employed.
(b) Voltage.—Electrical pressure is required in order to force the depositing current through the electrolyte against the resistances in the circuit. The voltage required depends upon the current density, the composition and temperature of the electrolyte, the composition of the anodes, and also upon the general conditions of working. These being constant, the voltage necessary is largely a factor of the number of electrodes in series in the tank and of the distance apart of the plates. Under ordinary circumstances this voltage varies from 0·1 to about 0·3 volt. High voltage is to be avoided, owing to the danger of short-circuiting, especially in cases where the accumulation of mud in [Pg 234] the tanks, or the impregnation of metallic salts in the tank walls, or the growth of excrescences upon the plates, lead to the passage of the current through these conductors rather than through the electrolyte solution itself. Short circuiting naturally diminishes the output of the plant.
These electrical factors which form the basis of the power requirements of the refinery, call for careful observation during the progress of the operations in order to ensure successful working and a high efficiency of the plant.
The current from the dynamos is brought by heavy leads and is distributed through the sets of tanks in the manner best suited to the installation. At one of the newer works, dynamos producing about 6,000 amperes at 120 to 150 volts, supply sufficient current to operate a set of 400 vats working on the lines just indicated.
The Depositing Tanks.—The tanks are usually constructed of wood, such as strong pitch pine, and they are lead-lined. The cross-section is usually such as will allow a space of about 3 inches between the edges of the electrode and the wall, and a 6-inch space from the tank-bottom to the lower edge of the plate. The length of the vats varies considerably, according to the desired output and to convenience of working—10 to 15 feet being average dimensions. This size of vat will hold 15 to 25 anodes together with a corresponding number of cathodes (16 to 26). The tanks are arranged across the building in a number of rows which are usually stepped down in stages of about 2 to 3 inches each, so as to assist as much as possible the circulation of the electrolyte through the system by gravity, and the vats are set in pairs with aisle-ways between (see Fig. 72). [Pg 235]
Leads run along each side of the tank, the current being conveyed to all the anodes at once by resting one lug of each plate upon the lead which runs along one side of the tank, carefully insulating the other lug from the conductor situated at the opposite side, this being used for connecting up the cathodes. The cathode-sheets are suspended from metallic cross-bars, which rest upon their own conducting lead, and are carefully insulated from the anode lead at the other side. The solutions are heated by means of steam coils.
Distribution of the Electrolyte.—The necessary circulation of the electrolyte is effected as much as possible by the natural action of gravity. The tanks of the top row in the depositing-house receive a constant supply of fresh solution from upper distributing vats, whilst old electrolyte is drawn off from near the bottom of the tanks, and flows over to those on the next and lower level. Fresh solution thus enters at the top of the tank, old solution is drawn off from below, and thus a uniform density and composition is maintained. From the tanks situated at the lowest level, the solution passes to a well, and from there is pumped up to the store-tanks or, when necessary, to the purifying tanks; air-pressure pumps being often employed for this work.
In course of time—and under the modern system of working with moderately pure anodes, this period is of considerable duration—the gradual accumulation in the electrolyte, of the small quantities of [Pg 236] impurity which are dissolved from the anode, may render the liquid so impure, that a danger arises of contamination of the cathode copper to such a degree that it becomes unfit for conductivity work. It then becomes necessary to purify the solution. In present-day practice, this continued accumulation of impurity in the electrolyte is prevented by continuously withdrawing, for separate purification, a certain proportion of the electrolyte from the circuit—replacing it by a fresh supply of pure solution from the store-tanks. Constant regeneration, purification and circulation are thus effected, whilst uniform composition is maintained.
After considerable use, the electrolyte solution gradually tends to increase in copper contents, and the first stages in the scheme of treatment for the old solution is to recover this excess of copper, which is effected in tanks known as “liberating tanks.” These are similar in general features to the refining vats, except that lead plates are employed instead of the copper anodes, so that the excess metal is deposited without any addition of copper being made to the solution, from the anodes. In due course, the desired composition in the electrolyte is once more attained.
When the solutions have become too impure for further use in the tanks, the bulk of the copper sulphate is recovered by evaporation in large pans, followed by crystallisation in somewhat shallow vats of large dimensions. The crude blue vitriol is further purified by repeated crystallisation, and any copper which still remains in the solution is then precipitated on scrap iron, the cement copper being worked through the furnaces again. Excess acid is also often recovered on further evaporation of the liquors, and is employed in the subsequent treatment of the slimes.
Working.—In the large modern refineries, the anodes are carried to and from the tank-house by cars, and at the tank-room are suspended from frames which are conveyed over the baths by means of overhead electrical cranes of about 10 tons lifting capacity. These rectangular frames correspond in size to the dimensions of the tanks, and are constructed of steel girders. Under the longer sides of this frame a series of hooks project, upon which the lugs of the anodes rest, and the hooks are placed at distances corresponding to the eventual position of the plates in the tank, so that the whole series of anodes can be dumped into position at one operation.
The cathodes are placed in a second rack, and likewise brought into position, between the anodes. The solution is then turned into the tank, the current started, and the refining proceeds, with a steady flow of liquid circulating through the system. The operations of [Pg 237] changing electrodes, cleaning and reloading occupy about one hour, and, but for this manipulation, the process under normal working is continuous. In ordinary practice, about 20 to 25 lbs. of copper are deposited daily on each cathode. Constant examination is made as to the electrical conditions, and the composition, temperature, and density of the solutions.
The anodes usually remain in the bath for a period of about six weeks, and they are then removed from the tank, scrubbed, and sent back to the furnaces to be remelted and re-cast into fresh anodes, the quantity of such anode scrap under good working conditions amounting to about 9 or 10 per cent. of the original metal.
The cathodes remain in the tanks for about one week, by which time a deposit of from 150 to 170 lbs. of pure metal has been obtained upon each. The practice of frequently replacing the cathodes possesses, among other advantages, those of maintaining a more even current density over the plates, of preventing the growth of excrescences and the irregular dissolution of the anodes, and of lessening the danger of breakdown of the somewhat slenderly suspended cathodes, by putting less weight on the supports. The removal in one operation of the entire batch of cathodes from the bath is effected by means of the suspended hook-frame, as employed in charging. The plates are rinsed, the top edges are cut off and returned with the anode scrap, whilst the pure electrolytic copper passes to the refining and casting furnaces, where it is prepared for the market. [Pg 238]
Collection of the Slimes.—Depending upon the working conditions of the refinery, but usually at intervals of three months, the precipitated slimes are collected and the tanks are cleaned out. The quantity of slime deposited is generally not very large, from 15 to 25 lbs. per tank being a not unusual yield. The current and the supply of solution are cut off, the plates removed, the contents of the tank allowed to settle, the liquid siphoned off to within about 6 inches of the bottom, and the residues are swilled out through a trap at the bottom of the tank. The sludge passes through a sieve that separates the lumps of anode copper which have broken off and fallen to the bottom of the tank, the slime then passes to the special refinery for treatment. The processes adopted for recovering the gold and silver from this residue are highly specialised, and belong properly to the technology of refining of the precious metals.
Modifications of Electrolytic Refining.—Great success has not yet attended the attempts which have been made to employ copper matte in the form of anodes in electrolytic refining processes, and the method is not in operation at any of the great modern works. Marchésé, Hoepfner, Siemens-Halske, Keith, and others have introduced processes, but their practical operation is attended with very great difficulty and but little commercial success. Matte is exceedingly brittle and it readily breaks up, it is a bad conductor and necessitates the use of high voltage, the solutions become very foul, and the processes require very special apparatus and equipment.
Methods for the production, by electrolytic processes, of pure copper in forms ready for service, such as wires or tubes, have been introduced successfully by a number of workers, including Elmore, Thomerson, and Cowper-Coles. Several of these methods are now in apparently successful commercial operation, and the published results of the working of the processes and of tests on the deposited materials offer considerable promise for their future industrial application for special purposes, if not for general use. The attaining of the necessary compactness, toughness, and strength of the metallic product is aided by the employment of pressure during deposition, as by burnishers, or by very rapid rotation of the depositing surfaces in the solutions. Details of these processes and products may be found from the references subsequently given.
Bringing up to Pitch and Casting the Merchant Copper.—The final stages in the smelting process from ore to market-metal are those of “fining,” toughening, and casting the cathode copper, the object of these operations being to impart to the metal the chemical composition [Pg 239] and mechanical and physical properties which are required in order to fit it for the market, and also to prepare it into a suitable form for service. In addition to cathode copper, other forms of the metal, if of suitable composition, are also treated with this object.
For conductivity copper, however, these final operations are conducted on metal from which practically all the impurities have been removed, but which is not sufficiently tough and homogeneous or which is not in a suitable shape for immediate industrial use. The toughening operation consists almost entirely of adjusting the percentage of oxides in the metal, partly in order to overcome the influence of any traces of injurious impurity that might remain, but mainly to exercise the functions previously indicated, of imparting by its more or less direct action upon the metal, a definite toughening and strengthening effect. The mechanism of the action is not perfectly understood, but the recent work referred to in Lecture II., p. 28, affords useful evidence as to its possible mode of action.
The actual refining operation and the furnace employed for the process are exactly similar to those used in preparing the metal to ensure the casting of sound ingots, as already described. The operations consist of a preliminary aëration, by means of which any oxidisable impurity still remaining in the metal is oxidised out, mainly through the action of copper oxide which is formed during the process in some considerable excess.
After the copper has become “dry” or over-oxidised, which condition is characterised by brittleness, depressed surface, and brick-like purple-red fracture of the metal, it is reduced by poling and timbering operations to a definite point, viz.: until a sample ingot of the metal indicates a maximum of toughness, accompanied by level surface and bright salmon-coloured silky fracture—it is then of “tough-pitch” quality.
The furnace employed for the refining has already been described. One of the main features in which it differs from the ordinary modern reverberatory smelting-furnace is that owing to the exceedingly high heat-conducting power of metallic copper, and to the absence of an insulating layer of non-conducting slag, there is little danger of much chilling action occurring on the hearth of the furnace; the temperature may, indeed, often become too high rather than too low. In consequence, it is not so usual to construct the furnace with a very massive hearth foundation as for smelting, but to build it upon a vault or upon a series of piers. With this type of foundation, the very considerable, but practically unavoidable, absorption of metal in the hearth-material [Pg 240] is reduced to a minimum. It is usual to work a charge consisting of scrap and oxide in the furnace before the regular smelting campaign begins in order to “season” the hearth. This procedure allows the primary absorption of copper by the hearth-material, and assists its consolidation, whilst the action of the oxide promotes a surface glazing which lessens the tendency for further absorption of copper, and gives a good surface to the working bed. As has been already stated, the hearth is generally built of brickwork rather than of sand. The furnace is constructed to hold from 80 up to 200 tons of metal. The method of working differs mainly from that previously described, in that instead of pouring molten copper into the furnace, as is usual with converter-metal, the cathode plates must be charged in a different manner.
In order to deal with such a large quantity of charge in this bulky form, without occupying so much time as to make the whole operation too protracted, it is usual to employ some form of charging machine rather than to use hand labour for the operation. In some cases a small melting furnace is employed solely for the purpose of preparing the metal in a molten form for feeding into the refining furnace. The type of cathode-charger most used is very similar in operation to the Welman charger for steel furnaces, and by its means, 100 tons of material can be charged per hour.
Operation.—The refining and toughening process is conducted in the six stages of:—
(a) The charging is sometimes conducted in stages, this being indeed unavoidable when very large quantities of material are worked with, the bulk of which, when solid, would more than fill the furnace. Two-thirds or three-quarters of the material may be put in at first and just melted down slowly, after which the remainder is added.
Owing to the not infrequent presence of sulphur in the furnace coals, and to its ready affinity for copper, resulting in undesirable consequences for the commercial metal, contamination by this element is usually prevented, as much as possible, by giving to the cathodes a wash of lime previous to charging.
(b) The melting is generally conducted somewhat slowly, so as to allow some oxidation of the metal during this stage, which may occupy [Pg 241] some twelve hours. Skimming of slag as it forms, and subsequent blowing of the copper towards the end of the melting stage are frequently resorted to.
(c) The slag which accumulates, sometimes in considerable quantity, is skimmed off as occasion requires. When converter metal of such purity as not to need electrolytic refining is treated directly in the furnace, much of this slag is converter-slag introduced from the ladle, and requires to be skimmed off at an early stage. In the usual process of melting cathode-copper, slag is produced from the last traces of iron which may have remained in the metal. In order to render it sufficiently viscid to be pulled out by the skimmer, ashes from the fire-grate are thrown upon and rabbled into the slag. This skimming may continue for some time, and a very rich coppery slag is pulled off, from which the metal values are subsequently recovered.
(d) The oxidation of the small quantities of impurity still remaining in the metal is completed by the operation of airing, as already described, and the action is continued in order to produce a small excess of oxide until the copper is “dry.” The time occupied for this airing is now not very protracted, since most of the impurities have been previously removed from the metal.
(e) The copper is then brought up to pitch by “poling” in the manner previously indicated, except that at this final stage, the testing of the metal and the adjusting of the oxygen proportion are conducted with much greater precision than was necessary for the simple production of the sound anode plates. In the present instance, the character of the metal and its value as a commercial article largely depend upon the care and accuracy with which the correct “pitch” is reached and is maintained in the bath during the entire period of casting of the metal. The poling for the “shaking out” of the gases is rarely necessary with cathode metal, and the addition of the cover of carbonaceous material for the purpose of effecting the reduction of the oxides to the desired extent, is made either at the commencement of poling or else shortly afterwards. After some time, a series of small samples is taken at intervals, by means of ladles, and the surface of the ingot is examined. The depression characteristic of dry copper gradually becomes less marked, the brick-like fracture appears finer and finer until it becomes silky, whilst the colour eventually turns to a very delicate salmon-pink. Meanwhile the mechanical properties have gradually improved, signs of brittleness disappear, and somewhat larger samples of the metal, which are now taken and tested, are characterised by a very marked toughness and strength. This is the moment at which [Pg 242] the poling must cease. The residual copper-oxide has now reached the proportion which was necessary for the imparting of the best mechanical properties, and the metal is tough-pitch. The skill of the workman is now exercised to the highest degree, in maintaining the metal in this condition during the whole of the subsequent casting period. Oxidation must be avoided in order to prevent a reversion to dry copper, whilst any further reducing action removes some of the necessary oxide, and results in “over-poling.” The metal would then become brittle again, coarsely fibrous and possibly somewhat spongy in fracture and very pale in colour, whilst in setting it would show a ridge upon the surface. In that case it would be necessary to “air” the metal again until it became dry, and then to pole it back to the “tough-pitch” stage.
The copper, when of correct pitch, is therefore removed from the furnace and cast at once; this being readily conducted through the tapping slot, the level of which is gradually lowered. The metal then flows down the spout to the ladle, and is poured into the moulds attached to some form of mechanical casting machine; the ingots being finally dropped into a water-bosh, weighed, sampled, and stacked, and are then in a condition ready for the market.
Phosphorus is sometimes employed for giving soundness to the castings, being added to the bath in small quantities in the form of phosphor-copper containing about 10 per cent. of the non-metal. Although very little of this phosphorus is retained by the metal, being mostly eliminated as oxide, special caution is required in employing it for high-grade conductivity copper, since the effect of very small quantities has a deleterious influence upon the conducting properties.
Silicon also is used for a similar purpose, and causes a considerable increase in toughness.
(f) When intended for conductivity work, the metal is cast into the form of “wire-bars” of very varied shape and size, according to requirements; thus the 100-lb. bars are about 3 feet long by about 3 inches square section, the 500-lb. bars 7 feet long by about 4½ inches square. Furnace samples weighing about 1 lb. are drawn down gradually to about ⅛-inch wire, and are tested for conductivity, as well as for strength and toughness, occasional analysis being also undertaken, whilst samples of the wire-bars in market form are similarly examined.
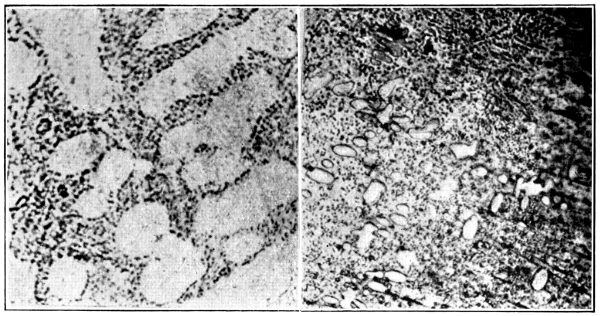
a b
Fig. 76.—Microstructure of Commercial Copper containing Oxygen (Hofman).
| a. Calumet and Hecla copper after 60 minutes’ poling. |
b. Calumet and Hecla “dry” copper before poling. |
| 0·22 per cent. oxygen = 1·98 per cent. Cu2O. | 0·64 per cent. oxygen = 5·76 per cent. Cu2O. |
Compare with Fig. 6, p. 28. |
|
| (By permission of American Institution of Mining Engineers.) [Pg 243] | |
Ulke, T., “Modern Electrolytic Copper Refining.” (With complete Bibliography).
Peters, E. D., “Modern Copper Smelting” (1905). (Chapter xviii. by M. Barnett.).
Mineral Industry. Annual Review.
Cowper Coles, S., “An Electrolytic Process for the Production of Copper Wire.” Proc. B’ham. Met. Soc., 1908–9, p. 5.
Schnabel and Louis, “Handbook of Metallurgy,” pp. 327–369.
Wraith, W., “Sampling Copper Anodes at Anaconda.” Trans. Amer. Inst. Min. Eng., March, 1910.
“The De Lamar Electro-Refinery at Chrome, New Jersey.” Eng. and Min. Journ., Jan. 13, 1906, p. 73.
Flinn, F. B., “Electrolytic Copper.” The Metal Industry, April, 1910, p. 112, vol. ii., No. 3.
Greenawalt, W. E., “The Greenawalt Electrolytic Process.” Eng. and Min. Journ., Nov. 26, 1910, p. 1062.
See also References to Johnson (p. 34), Hofman and others (p. 50), Keller (pp. 50, 215), and Peters (p. 80).
BELL AND BAIN, LTD., PRINTERS, GLASGOW.
Griffin’s Metallurgical Series.
Sixth Edition, thoroughly Revised
and considerably Enlarged. 18s. net.
An Introduction to the Study of
METALLURGY.
By Sir W. ROBERTS-AUSTEN, K.C.B., D.C.L., F.R.S., A.R.S.M.
Revised throughout by F. W. HARBORD, A.R.S.M., F.I.C.
“Professor Austen’s volume will be invaluable.”—Chemical News.
Fifth Edition, Revised, Considerably Enlarged,
and in part Re-written. With Frontispiece
and numerous Illustrations. 21s.
THE METALLURGY OF GOLD.
By T. KIRKE ROSE, D.Sc.Lond., Assoc.R.S.M.,
Chemist and Assayer of the Royal Mint.
“A comprehensive practical treatise on this important subject.” —The Times.
Sixth Edition, Revised throughout and Enlarged.
Pp. i-xx+538. With 314 Illustrations,
including 12 Folding Plates. 21s. net.
THE METALLURGY OF LEAD.
By H. F. COLLINS, Assoc.R.S.M., M.Inst.M.M.
“A thoroughly sound and useful digest. May with
every confidence be recommended.” Mining Journal.
Second Edition, Revised throughout and Enlarged.
At Press.
THE METALLURGY OF SILVER.
By H. F. COLLINS, Assoc.R.S.M., M.Inst.M.M.
Fourth Edition, Thoroughly Revised. Two Volumes. 36s. net.
With Additional Chapter on The Electric Smelting of Steel.
THE METALLURGY OF STEEL.
By F. W. HARBORD, Assoc.R.S.M., F.I.C.,
And J. W. HALL, A.M.Inst.C.E.
Third Edition, Revised and Enlarged.
With many new Plates. 16s. net.
THE METALLURGY OF IRON.
By Prof. THOMAS TURNER, Assoc.R.S.M., F.I.C.
“A thoroughly useful book, which brings the subject
up to date. Of great value to those engaged
in the iron industry.”—Mining Journal.
Frontispiece in Colours, and Beautiful Series of
Photo-micrographs. 12s. 6d. net.
ALLOYS and their Industrial Applications.
By EDWARD F. LAW, A.R.S.M.
“Concise and practical ... will be appreciated by student
and manufacturer alike.”—Foundry Trades’ Journal.
In Large 8vo. Handsome Cloth. Fully Illustrated. 12s. 6d. net.
ANTIMONY:
Its History, Chemistry, Mineralogy, Geology, Metallurgy,
Uses and Preparation, Analysis, Production and Valuation.
By C. Y. WANG, M. A., B. Sc.
“There is not, to our knowledge, any other complete treatise on antimony
among all the English text-books.”—Iron and Coal Trades’Review.
LONDON: CHARLES GRIFFIN & CO., LIMITED, EXETER STREET, STRAND.
Footnotes:
[1] This figure indicates thousandths of an inch.
[2] The term “impurity” might in several instances be replaced by the word “constituent,” since many so-called impurities are purposely added for conferring desired properties on the metal. It is here taken as implying elements other than copper.
[3] The temperatures given in Heyn’s diagram require revision in the light of later knowledge, and have been omitted here.
[4] The treatment of flue-dust is considered later.
[5] Though these actions represent with fair accuracy what occurs on smelting, there will be indicated later, in the proper place, some modification, due to interactions of certain oxides and sulphides, in the furnace.
[6] It might also be possible to assist the concentration in the matte by the addition of any available oxidised ores or residues.
[7] The chloridising roasting of copper ores is also sometimes employed in connection with wet processes.
[8] When copper combines with sulphur.
| 2Cu : Cu2S | :: | 2 × 63·5 | : | 2 × 63·5 + 32 |
| :: | 127 | : | 159 | |
| :: | 4 | : | 5 approximately. |
When sulphur combines with iron.
| S : FeS | :: | 32 | : | 56 + 32 |
| :: | 32 | : | 88 | |
| :: | 4 | : | 11. |
[9] Some flue-dust is also melted down.
[10] The East Butte Copper Mining Company has recently reported the successful sintering of its flue-dust by Dwight-Lloyd machines. (See Mining Journal, Jan. 6, 1912, p. 21).
[11] Later work on this subject has been published by W. Wanjukoff:—“Investigations on the Conditions governing the Entry of Copper into the Slags on Matte-Smelting, on the Chemical Form in which such Copper exists, and on the Lessening of the Copper Losses in Slags.” Metallurgie, 1912, Vol. x., Nos. 1 and 2, pp. 1–27, Jan. 8 and 22, 1912.
[12] This point will be dealt with in due course.
[13] The iron sulphide of a copper matte is probably the eutectic of the iron-sulphide : iron series containing about 85 per cent. of sulphide.
[14] Ducktown Sulphur, Copper, and Iron Company.
[15] These views have recently been controverted in an interesting paper by G. A. Guess. (“Notes on Pyritic Smelting,” Engineering and Mining Journal, 1912, Jan. 13th, p. 113). He defines Pyritic Smelting as the production of a Ferrous Silicate Slag from Iron Sulphide Ore.
[16] With nickeliferous mattes, the green colour in the flame is reported by Heywood to persist throughout the whole process.
[17] The case of nickel may be here treated as exceptional. It is eliminated with difficulty, the nickel and copper tending to oxidise together on bessemerising.
The cover image was created by the transcriber, and is in the public domain.
The illustrations have been moved so that they do not break up paragraphs and so that they are next to the text they illustrate.
Inconsistent hyphenation has been left as printed, except “firebox” has been changed to “fire-box” in the Index to match the spelling in the main body of the book.
Typographical errors have been silently corrected but other variations in spelling and punctuation remain unaltered.
In some tables “Number” has been abbreviated to “No.”, “United States of America” to “U. S. A.” and trailing punctuation has been omitted to make the rows shorter.
The advertisements at the beginning of the book have been re-located to the end of the book, just before the footnote section.