
Title: The age of the Earth
Author: Arthur Holmes
Release date: March 19, 2026 [eBook #78241]
Language: English
Original publication: London: Harper & Brothers, 1913
Other information and formats: www.gutenberg.org/ebooks/78241
Credits: deaurider and the Online Distributed Proofreading Team at https://www.pgdp.net (This file was produced from images generously made available by The Internet Archive)
Harper’s Library of Living Thought


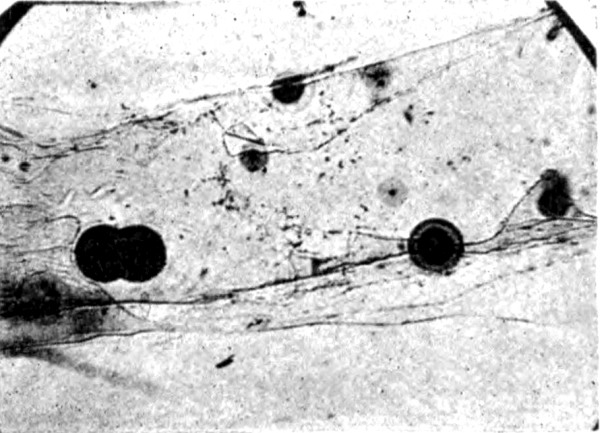
Fig. 1.—Haloes in cleavage flake of Biotite. ×70.

Fig. 2.—Radium Halo (lower part of field) and Thorium
Halo (upper part of field) in Biotite cut across cleavage. ×100.
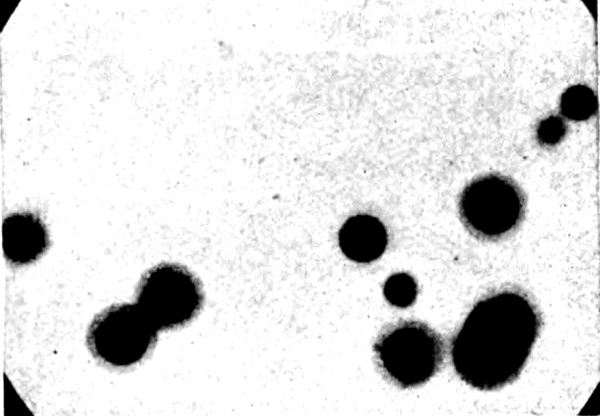
Fig. 3.—Haloes in cleavage flake of Biotite. ×85.
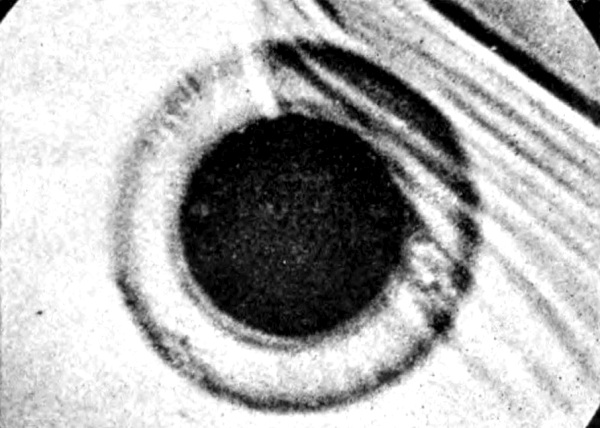
Fig. 4.—Haloes due to Ra. emanation (inner disc),
Ra. A and Ra. B. ×450.
PLEOCHROIC HALOES
BY
ARTHUR HOLMES
B.Sc., A.R.C.S.
MEMBER OF THE IMPERIAL COLLEGE
FELLOW OF THE GEOLOGICAL SOCIETY OF LONDON
FELLOW OF THE ROYAL GEOGRAPHICAL SOCIETY
ILLUSTRATED WITH TWENTY FIGURES AND DIAGRAMS
LONDON AND NEW YORK
HARPER & BROTHERS
45 ALBEMARLE STREET, W.
1913
Published March, 1913
TO
PROFESSOR THE HON. R. J. STRUTT
AND
PROFESSOR W. W. WATTS
IN GRATITUDE
FOR EARLY TRAINING
AND ADVICE
[Pg ix]
It is perhaps a little indelicate to ask of our Mother Earth her age, but Science acknowledges no shame and from time to time has boldly attempted to wrest from her a secret which is proverbially well guarded. On January 30th, 1911, I placed before the Natural History Society of the Royal College of Science a brief account of some of these attempts, and out of that paper this little book has gradually developed. In the present survey of the subject—the measurement of geological time—I have endeavoured to give as full and complete an account of the chief methods of attack as space would allow. My particular object has been to draw attention to their respective limitations and to test the validity of the various assumptions on which they are based. So far, the most remarkable feature of the problem lies in the extraordinary discrepancy between the conclusions drawn from the two most prominent methods of dealing with it. I have sought to mitigate the severity of this disagreement by discussing the possibility and consequences of reconciliation and by suggesting a path along which reconciliation may be found. If my treatment should stimulate a greater interest in the time problem, or provide material for further discussion, it cannot fail to bring nearer the ultimate [Pg x] solution of a question which for more than a century has had an unrivalled notoriety for provoking controversy.
I desire to acknowledge my gratitude to Prof. J. Joly and the Council of the Royal Dublin Society for permission to use the admirable series of micro-photographs which constitute the Frontispiece; to Prof. P. Lowell for his photographs of the Polar Caps of Mars; and to Sir Wm. Crookes and the Council of the Royal Society for the photograph and radiograph of a piece of pitchblende.
To a number of friends who have helped me in revising the proof-sheets, and to others who have turned my rough sketches into finished diagrams, I wish to tender my thanks.
Finally, I have a special pleasure in expressing my gratitude for the valuable assistance which has been afforded me by my friend Mr. R. W. Lawson, of Armstrong College, Newcastle-upon-Tyne. While the manuscript was in preparation I had the benefit of his suggestions and friendly criticism, and at a later stage he kindly verified many of my calculations. I am indebted to him in particular for the mathematical treatment of the final section of Appendix A (dealing with the uranium time-average) and for the two diagrams with which it is illustrated.
Geological Department,
Imperial College, S.W.
December 1st, 1912.
[Pg xi]
CONTENTS
| page | ||
| Preface | ix | |
| List of Illustrations | xii | |
| chap. | ||
| I. | The Time Problem and its History | 1 |
| II. | Time and Tide in the Solar System | 22 |
| III. | Astronomical Considerations | 32 |
| IV. | The Work of Denudation | 48 |
| V. | The Salinity and Age of the Oceans | 61 |
| VI. | Sedimentation and Geological Time | 76 |
| VII. | Radioactivity | 91 |
| VIII. | The Thermal Energy of the Sun | 110 |
| IX. | The Thermal Energy of the Earth | 122 |
| X. | Radioactive Minerals and their Ages | 137 |
| XI. | Review of the Evidence | 166 |
| Appendix A | 177 | |
| Appendix B | 184 | |
| Index | 191 | |
[Pg xii]
LIST OF ILLUSTRATIONS
| Figures printed as plates | ||
| Fig. 1 | Pleochroic Haloes | Frontispiece |
| Fig. 2 | ||
| Fig. 3 | ||
| Fig. 4 | ||
| facing page | ||
| Fig. 5 | Spiral Nebulæ | 28 |
| Fig. 6 | ||
| Fig. 7 | Polar Caps of Mars | 39 |
| Fig. 8 | ||
| Fig. 12 | Photographs of Pitchblende | 92 |
| Fig. 13 | ||
| Figures printed in text |
||
| page | ||
| Fig. 9. | Glacial Movement in S. Scandinavia | 40 |
| Fig. 10. | Upper Cretaceous Strata of Colorado | 45 |
| Fig. 11. | Section illustrating the Formation of | |
| Sediments on Continental Shelf | 83 | |
| Fig. 14. | Apparatus for Estimating Radium | 106 |
| Fig. 15. | Pleochroic Haloes of the Uranium Family | 108 |
| Fig. 16. | Temperature Gradients | 127 |
| Fig. 17. | Geological Time Scales | 172 |
| Appendix A— | ||
| Graph I | 180 | |
| Graph II | 181 | |
| Table of Radioactive Elements | 190 | |
[Pg 1]
THE AGE OF THE EARTH
Primitive races and their creation myths—The chronologies of Eastern sages—The demands of geology opposed by theological prejudice—Bishop Ussher’s date of the creation of the world—The Deluge and the Doctrine of Catastrophism—Ludicrous conceptions of fossils—The rise and eventual success of Uniformitarianism—Leonardo da Vinci—Steno—Generelli—Desmarest—Hutton—Scrope—Lyell—Ussher’s chronology rejected—Geological time no longer limited—The hour-glass of denudation and deposition—Darwin’s excessive drafts on time—The conservation of energy—Geology limited once again—Thomson on the age of the earth and sun, 1862—The great controversy—Huxley, 1869—Perry and the correspondence in Nature, 1895—Sir G. Darwin and the birth of the moon—Clarence King’s estimate of the age of the earth—Time implied by the stratified rocks—Joly on the salinity and age of the oceans—The significance of radioactivity—Emission of energy by radium and its widespread distribution—Radioactivity provides a new measure of time—Controversy again—Methods of dealing with the problem—The inadequate testimony of biology.
The most primitive races of mankind, strenuously engaged in the daily struggle for existence, appear to have given little thought to the antiquity of the world on which they lived. Even at the present day there exist barbaric tribes to whom it has never occurred that the [Pg 2] earth may have had a beginning. The conception of creation, of the production of order from chaos, indicates a marked intellectual advance, but into the myths and legends of which this idea was so often the inspiration, the element of time did not usually enter.
The East African natives have traditions of the upheaval of mountains and of the freeing of the earth from bondage, legends which may have had a foundation in the phenomena exhibited by volcanoes and glaciers. If the question “When?” is asked, the natural reply is merely a look of astonishment, and persistent enquiry elicits nothing more definite than a vague “Long ago.” Anything else, of course, could not be expected from a people whose conceptions of the development of the universe are limited to a recognition of the alternation of seasons and the insistent mysteries of life and death.
The priests and philosophers who flourished during the ancient civilizations of the East, speculated in greater detail and devoted some attention to the elaboration of a chronology of earth history. The Chaldeans had a well-deserved reputation for astronomy, and their known observations go back for more than 6000 years. Cicero relates that their venerable priesthood had records of stellar observations stretching back for 470,000 years; a fanciful period which tallies with the date assigned by the Chaldeans to the origin of Man. According to the same remarkable system, the earth had already existed for 215 [Pg 3] myriads[1] of years. The Persian sages, led by Zoroaster believed that the total duration of the world’s existence was limited to 12,000 years. The Hebrew chronological tables are familiar to everyone, and the restricted interpretation which was placed upon them during the Middle Ages, when they affected European beliefs so powerfully, will be presently referred to. Opposed to these ideas of a definite beginning were the more abstract conceptions of Indian philosophers, who regarded Time and the Earth as eternal.
To determine the age of the earth, or to express the actual problem with more accuracy, to measure the duration of geological time, became a definite scientific aspiration only during the last century. The ultimate purpose of Geology is essentially to elucidate the history of the earth, a record of which is imperfectly written in the stratified and igneous rocks to which access is possible. As the characters and sequence of the various formations gradually became better understood, it was found that the story they disclosed was one of successive changes of life and scene of the most impressive kind. The immensity of time which seemed to be indicated was at first a fruitful source of confusion and prejudice, for it brought geology into disrepute at an early period, owing to the widely prevalent idea that the writings of Moses fixed the antiquity of the universe beyond dispute. It is indicated by a marginal reference in most English Bibles that the creation of the world took place in the year 4004 B.C. This famous estimate, which probably represents the most limited period ever assigned to the past duration of our planet, was put forward in 1650 by [Pg 4] Bishop Ussher. Some such date as this had been generally believed in during the Middle Ages as marking the epoch of transition from chaos to an ordered world. The corresponding Byzantine date was 5509 B.C. The whole of geological history had therefore to be squeezed into about six or seven thousand years, and this limitation naturally demanded some extraordinary hypotheses to uphold it. As Prof. Sollas says, “In the days when Geology was young, it found a careful foster-mother in Theology, who watched over its early growth with anxious solicitude, and stored its receptive mind with the most beautiful stories which the young science never tired of transforming into curious fancies of its own, which it usually styled ‘Theories of the Earth.’”
At the time of the revival of learning at the close of the 15th century, men’s ideas of the earth’s past history were largely dominated by the exaggerated effects ascribed to the Noachian Deluge. This devastating catastrophe was the type of a succession of destructive cataclysms which was believed to have preceded it. Supported by Jewish cosmogony, and in harmony with the scriptures, this view prevailed as the Doctrine of Catastrophism until well into the 19th century. It was believed by some writers that all sediments were deposited during the Flood; others, impressed by the succession of different types of deposits found far from the present sea-level, and often containing fossils, considered that one flood was insufficient. As far back as the 9th century Rhabanus Maurus, in explaining this phenomenon, called to [Pg 5] his aid three great universal floods, the two later ones being contemporaneous with Jacob and Moses respectively, but these floods apparently were not as convincing as that of the time of Noah, for they appear no more in geological literature. Marine sedimentation of antediluvial times was believed by some to have been extraordinarily active, and this was added to the effects of the Deluge to relieve the latter of the enormous amount of work for which it was held responsible.
Fossils were regarded with suspicion and jealousy, and most of the early naturalists resolutely set themselves against the obvious deduction to be drawn from them. About 300 B.C., Theophrastus had failed to see in them the evidence of past life, and according to this philosopher a “plastic virtue latent in the earth” was supposed in some mysterious way to have given them birth. The process was occasionally explained as being akin to crystallisation, but other authors, gifted with more fascinating powers of imagination though with less philosophic insight, called to their aid the occult powers of “lapidifying juice” and the obscure consequences of “tumultuous movements of terrestrial exhalations.” There were even those who thought that fossils were the work of the devil, subtly designed to draw believers away from the faith.
Nevertheless, there were, from time to time, more rational thinkers to whom such ideas were both repugnant and untenable. Endowed with a keener perception than their fellows, and with a more critical temperament, they felt compelled to regard fossils as organic remains. [Pg 6] Leonardo da Vinci (1452-1519) and Steno (1631-1686) were amongst the most illustrious of these early observers. The work of Steno, published in 1669, shows how seriously he was handicapped by the orthodox belief that only a few thousand years had elapsed since the beginning of the world. Any ideas which demanded longer periods were contrary to revealed truth and were therefore bitterly opposed. Steno demonstrated not only the true nature of fossils, but also the orderly succession of stratified deposits. He traced the structure of the rocks to their subsequent movements, and their surface features to the destructive agencies of denudation. These remarkable investigations distinguished Steno as an observer far in advance of his age. The revelation of a tiny chapter of the earth’s past history which was thus afforded him must surely have suggested the necessity of calling more liberally upon time. However, whether or not he recognised how hopelessly inadequate was the period at his disposal, he remained the victim of theological prejudice, and cautiously avoided speculations as to the antiquity of the earth.
Gradually, as the result of careful and patient work like that of Steno, the foundation stones of modern geology were laid in the face of a bitter antagonism. Generelli in Italy in 1749, and Desmarest in France in 1777 established the importance of the slow but ever active processes at work in the evolution of the earth’s surface features. It was found no longer legitimate to evoke forces more intense, upheavals more violent, or catastrophes more devastating than those of present experience. [Pg 7]
Closely following these courageous authors, and independently of their influence, came in 1785 the Theory of the Earth of Hutton. In this epoch-making work the principle was defined which made dynamical geology possible and which has proved to be of the greatest assistance in wresting from the rocks their history. It was asserted that the activity of the agencies which had moulded the earth’s surface in the past and brought it to its present condition still remained undiminished in intensity. In existing causes lay the key with which to unlock the secrets of the past.
The doctrine of Uniformitarianism, as it came to be called, naturally implied that the earth’s age should be restricted no longer by dogmatic obstinacy. To Hutton time scarcely presented a difficulty. He found it at his unlimited disposal both in past and future, and he concluded his enquiries with the assurance that he found “no vestige of a beginning—no prospect of an end.” He did not, however, infer that the world had neither beginning nor end, a view for which some of his critics held him responsible. On the contrary, he carefully pointed out that in tracing back the course of events we are at last limited in our retrospect, and that beyond the dim horizon of those early times stretches an unknown past. Concerning this past, Hutton preserves an open mind. It is not time which fails but data; and as he says “to reason without data is nothing but delusion.”
Hutton’s convictions were regarded with righteous horror by the official leaders of the day, most of whom combined the study of theology [Pg 8] with that of their favourite science, and demanded in the latter a harmonious agreement with the scriptures. From their point of view Catastrophism had the advantage, and they were firmly persuaded of its truth. Fifty years had yet to elapse before the superiority of many of Hutton’s opinions came to be generally recognised, and even then the fallacies underlying the earlier doctrines were but grudgingly admitted.
The opening years of the 19th century were signalised by the work of Lamarck and Cuvier in palæontology. Lamarck recognised that fossil shells were most commonly of marine types and that in the gently accumulating sediments of the sea-bottom they were buried and preserved. In the succession of faunas which he studied, and in his belief that by some law of development they were all related, he found a cogent argument in favour of the great antiquity of the earth. Cuvier, however, was led by his researches to extend the conception of great world convulsions. Time after time the earth’s inhabitants had been destroyed and entombed, only to be replaced by the creation of fresh types after the force of the cataclysm had subsided. The Deluge was the last of these upheavals, and a scientific proof of its widespread effects was drawn from the superficial deposits found in so many parts of the globe.
Until the barren ideas of Catastrophism had been abandoned, the past provided little more than an exercise for the imagination. But the immature days of geology were passing away. The solid work of William Smith prepared the way for an accurate historical development of the [Pg 9] science. Scrope drew attention to the importance of studying geological processes actually in operation, and arrived at conclusions essentially the same as those of Hutton. Finally, during the years 1830-1833, the first edition of Lyell’s Principles was issued, a work which set flowing the full tide of Uniformitarianism. Ten years previously Lyell had felt compelled to renounce the unsound doctrines of his teachers, and with the publication of these carefully reasoned volumes he became the champion of the rival position. He denied the former existence of catastrophes of an order of magnitude different from those of the present. In their place he demanded only time. A short but convulsive past was to be exchanged for a longer one, uniform and comparatively tranquil throughout.
Lyell’s views did not find many ready adherents among the older geologists. Buckland at Oxford and Sedgwick at Cambridge had long taught a system of geology which they believed to be in accordance with the first few chapters of Genesis. Eventually, however, they found that under more careful examination the evidence of a universal flood faded away, and with them and their generation the tendency to demand a literal agreement between geological theory and the writings of Moses gradually disappeared. A new fraternity of geologists arose whose ideas were limited by fewer prejudices, and who found the time barriers raised against them no longer.
Geology was now able to stand firm on its own evidence, and to insist without fear of contradiction that long periods must have elapsed during [Pg 10] the slow accumulation of strata. The burden of reconciliation now fell upon the theologians. Happily, the days of malice and persecution had gone by, and in the light of a more broad-minded criticism it was found that the Church had nothing to lose by the rejection of Bishop Ussher’s chronology. The first three words of the Bible, “In the beginning,” were interpreted afresh, and from the indefinite lapse of time which they seemed to imply the geologist was allowed to draw at will.
From this period until 1862, when Thomson (Lord Kelvin) attacked the problem in an entirely new way, there was no necessity to apologise for the most extensive drafts on the bank of time, and no further restraint was felt in estimating the antiquity of the earth. But the data were still inadequate, and many mistakes were made. Most of the estimates put forward were afterwards regarded as wildly extravagant, and naturally, they have no intrinsic value to-day. The stratified rocks, representing the aggregate accumulation of material denuded from the lands, afforded a valuable time-index. It was recognised that the processes of denudation and deposition, like a gigantic hour-glass, had been recording time since land and sea were first defined. But the record was perplexingly difficult to read, and the time units, based on estimates of present rates of erosion and sedimentation, were little more than guesses, uncertain and even misleading.
The only interpretation worthy of serious consideration was that of John Phillips, who concluded in 1860 that the time required for the [Pg 11] deposition of the complete succession of strata lay between 38 and 96 million years. As an example of one of the more extravagant demands, mention may be made of Darwin’s estimate in 1859 of the time elapsed since the latter part of the Cretaceous period. From the rate of chalk erosion in Kent, he deduced that the excavation of the Wealden valleys had required a period of 300 million years. Referring to this estimate, Jukes gave his opinion that while it might be a hundred times too great, it was equally probable that it was a hundred times too small. Evidently 30,000 million years, in the absence of more reliable data, was not considered an absurdly excessive period for the erosion of the Weald. It should be noticed, however, that after the publication of Thomson’s views, Darwin withdrew this estimate from the Origin of Species.
We must now return to consider the problem as it appeared to the leading physicists of half a century ago. In 1842 Mayer, and during the following year Joule, had placed the conception of the conservation of energy on a firm experimental basis, and started the principles of thermo-dynamics on their brilliant career through the sciences. Twenty years later, Thomson invaded the domain of Geology, hoping to reform its speculations and bring them into accordance with the doctrines of the conservation and degradation of energy, doctrines which were apparently set in defiance by the orthodox tenets of the science.
As early as 1852 he had shown that under the laws to which matter and energy are subject, the earth within a finite period of past time must [Pg 12] have been unfit as a habitation for life. This general conclusion he afterwards worked out in detail, determined to protest against what he considered the immoderate application of the principle of Uniformity. So profoundly did he influence geological opinion, that for nearly fifty years the question of the earth’s antiquity centred around his name. A short account of his contributions to the physical aspect of the subject, and of the famous controversy which they aroused, cannot therefore be dispensed with.
In 1855 Thomson indicated the way in which observations of underground temperatures might be applied to the determination of absolute dates in geological history. Already the origin and maintenance of the sun’s heat had been discussed by Mayer and Helmholtz, and during the years 1860 and 1861 Thomson dealt with their respective suggestions. During the following year, 1862, came the epoch-making papers On the Age of the Sun’s Heat, and On the Secular Cooling of the Earth.
The age of the sun’s heat evidently restricts geological time in a very decided way, and Thomson drew attention to the necessity of the fact that unless the sun were a perpetual miracle, it could not have continued to radiate heat-energy for an unlimited period. His guarded conclusion was that the sun most probably has not illuminated the earth for 100 million years, and almost certainly not for 500 million years.
Regarding the earth as a globe which had gradually cooled down, he tried to show that the principles of thermo-dynamics had been [Pg 13] disregarded by geologists. From the earth’s present store of heat, as revealed by underground temperature gradients, he calculated that the consolidation of the crust took place about 100 million years ago. Owing to the uncertainty of much of the data on which this estimate was based, he allowed wide limits. Had a solid crust formed permanently less than 20 million years ago, underground heat should be greater than is actually observed. Had it formed at a date more remote than 400 million years ago, then the temperature gradient near the surface should have been notably less than it is.
In 1865 appeared a remarkable little paper in which attention was drawn to the earth’s thermal history, and particularly to its more energetic youth, the object again being to refute the doctrine of Uniformity. During the next year Thomson delivered the Rede Lecture on The Dissipation of Energy, and showed the importance of the tides in terrestrial dynamics. All over the ocean the effect of the friction set up by moving water is either to accelerate or to retard the earth’s rotation. The final result was, he asserted, to retard the earth as though a gigantic friction brake were being slowly applied. The ultimate tendency was towards a state when relative motion between earth and moon should be no more.
In 1868, in an address on Geological Time, Thomson gathered together his three limiting criteria, and further developed the application of tidal friction to the question. In his discussion the earth’s figure was supposed to be a survival from the period of consolidation; a permanent record of terrestrial conditions at that [Pg 14] time. This being so, the rate of rotation could be computed under which that form would be in fluid equilibrium.
The three maximum time limits which Thomson drew from his arguments were respectively 500, 400, and 1000 million years, with a lower limit of 20 million years. The final conclusion was “that the existing state of things on the earth ... must be limited within some such period of past time as one hundred million years.”
The reply to this series of attacks on what was supposed to be orthodox geological opinion, came from Huxley in 1869. He did not deny the consequences of the dissipation of energy in the cases to which attention had been directed, but decided in favour of the geological evidence, which indicates, as time has more and more substantiated, that “they have made no practical difference to the earth during the period of which a record is preserved in the stratified rocks.”
Thomson returned to the attack a month or two later, protesting once more against this attitude. At the close of his address, he said: “A large proportion of British popular geologists of the present day have been longer contented than other scientific men to look upon the sun as Fontenelle’s roses looked upon their gardener. ‘Our gardener,’ say they, ‘must be a very old man; within the memory of roses he is the same as he has always been; it is impossible he can ever die, or be other than he is.’”
There were no further contributions to the problem until 1876, when Thomson revised the former conclusion which he had based on the [Pg 15] distribution of underground heat, and narrowed his limits to 50 and 90 million years. In later years he reduced both these limits still further, though he was always more prudent than Tait, who with little justification cut down the time allowance in a most alarming way. Tait wrote in 1875, “Ten million years is about the utmost that can be allowed from the physical point of view for all the changes that have taken place on the earth’s surface since vegetable life of the lowest known form was capable of existing there.” Assertions such as this were among the most embarrassing circumstances that geologists had to face. The late Sir George Darwin was more kindly disposed, although his contributions to the question were considered to support the physical arguments. Believing that the moon had been derived from the earth by the separation of an enormous tidal wave, he calculated that since this stupendous event at least 56 million years must have passed.
The physical evidence appeared at first to be irrefutable, and the estimates based upon it equally certain. Yet earth history could not comfortably be squeezed into less than 100 million years. The stratified rocks were there in undoubted succession; mile after mile of thickness with no indication of more rapid accumulation than that of modern deposits. In spite of this, however, a prejudice in favour of short estimates was gradually aroused, and some attempt was made to hurry up geological activities in the past in renunciation of the principles of Uniformitarianism. But many geologists refused to give way and vigorously attacked the physical stronghold, searching out and [Pg 16] exposing all the assumptions, and noting with satisfaction the uncertainty of much of the data and its doubtful applicability. A few physicists themselves denied that the limited estimates were securely founded.
In 1895 the controversy was re-opened by the publication in Nature of a correspondence initiated by Prof. Perry. While Perry had previously accepted Kelvin’s conclusions, he now challenged the validity of all three. The strongest criticism was directed against Kelvin’s assumption of a homogeneous earth. It was shown that if the rocks of the interior, under high temperatures and pressures, were to conduct heat ten times as well as those near the surface, then Kelvin’s figures would need to be increased fifty-six times. To settle this point adequate data were not then forthcoming, but Perry stated his belief that, if geologists had sound reasons for demanding long periods of time, he saw nothing which denied them four times the greatest (1000 million years) of Kelvin’s estimates.
Kelvin wrote that he would rather know the date of the consistentior status than that of the Norman Conquest, so interesting did he find the subject. After remarking that 100 million years were ample to satisfy Geikie, he said, “I should be exceedingly frightened to meet him now with only 20 million in my mouth.” However, Kelvin carefully examined the data referring to the conductivities of rocks and decided that they were not favourable to Perry’s suggestion. Thus fortified, he lent his support to the independent estimate of 24 million years which had been arrived at in 1893 by Clarence King. [Pg 17]
Kelvin’s last pronouncement of his views was in 1897, when he delivered an address on The Age of the Earth as an Abode Fitted for Life. He then narrowed down his earlier estimates to 20 and 40 million years. To most geologists these limits were seriously in conflict with the requirements of their science. A feeble attempt was made to force an agreement, but it was generally held that the sedimentary succession implied a period three times as long. An independent mode of reckoning geological time was developed by Prof. Joly in 1899. He made a careful study of the amount of sodium annually removed from the land by solvent denudation, and calculated the time during which the total quantity of sodium now held by the oceans could have accumulated. The period closely approached 100 million years, and thus further support was added to the testimony of the rocks, for with the birth of the ocean their deposition must have commenced.
Among many of the more optimistic geologists there was a prevalent expectation that some flaw would ultimately be found in the physical arguments. Their anticipation was realised ten years ago in the most surprising and unexpected way. In 1903 came the discovery by Curie and Laborde that radium maintains a temperature above that of its environment owing to the spontaneous evolution of heat involved in its disintegration. Other investigators found that radium and its radioactive associates were widely distributed in the earth’s surface materials. In all waters and gases of natural origin, and all rocks and soils, traces of these elements have been detected. We owe a great deal [Pg 18] to the careful researches of Prof. Strutt, which have shown conclusively that the earth can no longer be regarded merely as a cooling body. A newly recognised source of heat must now be taken into account, and indeed, so relatively abundant is the supply, that our present difficulty is to understand why the earth is not hotter than we actually find it.
With these discoveries the long controversy was finally buried, and Kelvin’s treatment of the problem was proved to have been fallacious. Kelvin lived just long enough to know something of the first attempts to utilise the principles of radioactivity in solving the vexed problem of time. The discovery of radium did not only destroy the validity of the older thermal arguments; but also, it led directly to the elaboration of a new and more refined method. As we shall see in the sequel, every radioactive mineral can be regarded as a chronometer registering its own age with exquisite accuracy. The record is not always completely preserved, but a few attempts have been made to read it, and in the more favourable cases, periods of enormous duration have been revealed. Indeed, if our interpretation is correct, some of the oldest Archean rocks must date back 1600 million years.
Not many years ago geologists were dissatisfied with the shortness of their time allowance; to-day they are confronted with an embarrassing superabundance. Certainly, it has been dogmatically denied that radioactive minerals can have the great ages which have been attributed to them, or that they can account for more than a small proportion of [Pg 19] the earth’s loss of heat. But such denials do not help to remove the difficulty; they rather tend to aggravate it. In the face of two rival and mutually inconsistent systems of earth chronology, neither of which can be rejected with impunity, it becomes essential to examine most carefully the fundamental assumptions underlying each method, in the hope of detecting the subtle errors which are responsible for so glaring a disagreement. In this way alone can a means of reform be indicated and the road made clear for the construction of a time-scale which will be acceptable to all.
It is obvious that as yet we cannot measure the earth’s absolute age if by that expression is meant the time which has elapsed since our planet first existed. Kelvin’s work most nearly approached this desideratum, but, as we have seen, it was doomed to ultimate failure, though not indeed, to discredit. Sir George Darwin’s calculation of the lapse of time since the birth of the moon stands apart; his results are limited only by a definite minimum, and otherwise are sufficiently elastic to meet any reasonable demand. But this convenience, added to the doubtful validity of the hypothesis on which his estimates were based, detracts from their value. It would be unfair to expect too much of them, for they were only put forward in support of a hypothesis which would have been immediately disproved if ridiculously long or short time periods had been involved.
As we shall see in the following chapter we are still far from understanding the sequence of events which led up to the origin of the [Pg 20] earth. All we can hope to do is to fix the dates of critical periods of its history and assign its origin to a point still more remote. The different methods which have been elaborated to deal with the problem are all based on a common principle. The rates of certain changes at the present day are determined as accurately as possible, and in imagination, the respective processes are traced backward in time, until limiting conditions are arrived at. Thus, Kelvin takes us back to a time when the earth was not yet a solid globe; Darwin traces back the moon’s history until he finds it revolving close to the earth; Joly bids us imagine the oceans in their original freshness, free, or nearly so, from salt; Geikie finds an end at last to the long succession of stratified rocks and seeks to estimate the time they represent. Last of all, and most brimful of promise, there lies in the mechanism of radioactivity an elegant method for assigning a date to the period of crystallisation of every igneous rock in which suitable minerals can be found.
The testimony of biology scarcely calls for consideration here, for as Huxley pointed out, biology must take its time-scale from geology. The procession of life forms shows broadly that the time involved must have been very great; and, moreover, where we first meet it in the Cambrian rocks, it is already far on its journey. This is usually taken to signify that pre-Cambrian time must be at least as long as that which has elapsed since. Such a statement can only be regarded as very conservative. [Pg 21]
Prof. Poulton considers that the process of evolution must have required much longer periods of time than those estimated by the geologists. Sollas, on the contrary, is satisfied that 26 millions of years would be ample to meet all the demands of biology. It is obvious that a numerical estimate cannot be derived from the succession of organic forms, for the rate of change of species is as yet known only relatively. The influence of changing environment is complex beyond the possibility of exact analysis, and any method of chronology based on the development and ultimate extinction of past types would necessarily give results of very unequal value. The little shell Lingula has retained its individuality with but little deviation from its original form throughout the time represented by the fossiliferous strata, but the same record is eloquent of the gradual unfolding of fauna after fauna, culminating at last in the highly specialised organisms of to-day.
[Pg 22]
Early ideas of cosmogony—The Laplacian hypothesis—Tidal friction in the earth-moon system—Its application by Kelvin to the question of the earth’s antiquity—Its application by Sir G. Darwin to the history of the moon and the date of its birth—Difficulties in the way of the Laplacian hypothesis—The Planetesimal hypothesis—The origin of the ancestral solar nebula and its transformation into the solar system—Early stages of the earth’s history.
Speculative fancies concerning the origin of the world form the subject matter of many of the earliest writings on record, and throughout the intellectual history of mankind the problem has proved to be one of supreme fascination. It was not, however, until quite recent times that the efforts of imagination gave place to reasoned hypotheses, tempered by a more sober regard for physical probabilities. At first, on having attained the status of a science, geology steadfastly refused to consider seriously the cosmogonic fantasies then current. It was Hutton, who by advocating the direct observation of nature in place of the old scholastic arguments, first delivered geology from the inevitable wranglings that would necessarily have arisen from so premature a discussion of the beginning of things. Cosmogony, in spite of this, continued to receive attention from workers in other sciences, [Pg 23] and while to-day we are still unable from geological facts alone to trace back with confidence the details of the earth’s beginning, yet the uncertainty which justified Hutton in entirely disregarding the earth’s genesis no longer exists. Astronomy, physics, and chemistry have all contributed to the elucidation of what may be called the prehistoric period, and have done much to remove our modern ideas from the dangerous quicksands of speculation.
It is becoming more and more evident that many of the fundamental problems of geology can be solved only with reference to the processes involved in the making of the earth and in its subsequent evolution. The dynamic agencies at work to-day are genetically the outcome of the ancestral forces which first moulded our planet, and theoretically, the tectonics and constitution of the earth’s crust should lead back to a more complete understanding of its initial condition. But the mind of man is impatient of delay; shrewd guesses are made and gradually adjusted to known facts, with the result that many geological doctrines are founded not on observation alone, but also in part on fundamental hypotheses.
During the 19th century scientific thought was powerfully influenced by the achievement of Laplace, for it was he who first presented the famous Nebular hypothesis in a consistent and acceptable form. Previous thinkers, notably Buffon in 1745, and Kant in 1755, had put forward similar generalisations, but they fell into certain errors which Laplace escaped, and their expositions lacked the completeness and [Pg 24] authority which his mathematical genius guaranteed. In the Système du Monde, published in 1796, Laplace attempted to trace the gradual evolution of the solar system from a spheroidal nebula, under the normal operation of natural laws. He started with a hot gaseous nebula of lens-like shape extending beyond the present orbit of the outermost planet and rotating slowly in the same direction as that of the sun. The average density of such a nebula would be about ¹/₂₅₀,₀₀₀,₀₀₀ that of ordinary air. As Laplace explicitly states that the sun was already foreshadowed in the nebula as a strongly condensed central nucleus, the outer atmosphere must have been of inconceivable tenuity, a condition maintained only by intense heat. Through the loss of this heat by radiation, and under the influence of its own gravitation, the nebula would slowly contract. In accordance with the laws of dynamics, contraction would necessarily be accompanied by a compensating increase in the rate of rotation. At a certain stage, the centrifugal force at the Equator would balance the attraction of gravity, and a ring of gas would be left behind. The same process of ring separation would afterwards be repeated from time to time as contraction progressed, and each ring being inherently unstable, would rupture, ultimately forming a spheroidal mass with the requisite directions of rotation and revolution. The Asteroids, a group of tiny planets revolving in the zone between Mars and Jupiter, probably represent a ring which failed to coalesce into a single planet. In their newly-born condition the planets were still hot and gaseous, and by the operation of the same [Pg 25] mechanical agencies on a smaller scale most of them detached a second generation of rings, and these having aggregated in turn became the satellites. The great central nucleus, continuing to contract after the last planetary ring had been abandoned, became the sun.
On this hypothesis the earth was originally a hot fluid globe with a heavy atmosphere consisting of the vapours of its more volatile constituents. As it contracted it rotated faster and faster, until a limiting velocity was attained which allowed the separation of the ring which ultimately condensed to form the moon. At a later stage the earth began to solidify and the crust first made its appearance. The lighter igneous rocks, the granites and corresponding lavas, presumably formed the outer layers, while beneath this zone the more basic magmas arranged themselves in the order of their density. When cooling had sufficiently progressed, the waters condensed to form the oceans, and with the emergence of the lands, denudation began for the first time and the earth’s historical period was inaugurated.
At the time of the birth of the moon the rotatory velocity of the earth must have been very rapid compared with that of the present day, and an obvious deduction is that the earth has gradually slowed down in the course of its history—in spite of the accelerative effect of contraction—and is probably still being retarded. From a comparative study of ancient and modern eclipses, Dunthorne demonstrated in 1749 that the earth appeared to be losing time. More than a century later, Adams showed from a revision of all the data, that the loss amounted to [Pg 26] 22 seconds in a century, although he was careful to point out that the assumptions on which his calculation was based were themselves not securely founded. It was Kant who started the idea that the ceaseless operation of tidal friction would tend to bring about this result. Long afterwards Kelvin indicated the way in which tidal retardation might be applied to estimate the date of the earth’s solidification. He supposed that the form then assumed by the earth had survived in its essential features throughout geological history. Subsequent diminution of the rate of rotation was not considered to be attended by a corresponding change of oblateness. Granting this assumption, the present figure of the earth ought to inform us of the rate of rotation under which it would be in fluid equilibrium at the time of consolidation. After making these concessions to speculation, Kelvin safeguarded himself by assigning wide limits to the earth’s age. His maximum estimate was a liberal 1000 million years, but he further stated his opinion that if a much higher antiquity than 100 million years were demanded, the polar flattening and equatorial bulging should be more marked than we find them.
These conclusions were necessarily somewhat vague, and based as they were on a very questionable assumption, they were readily susceptible to damaging criticism. It is known from the concordant evidence afforded by seismic, geo-physical, and astronomical phenomena that the earth is essentially solid throughout with a rigidity more than twice that of steel. So far our conception of the earth is similar to [Pg 27] Kelvin’s, but the postulate that such a body could resist rotational changes without modification in shape, cannot now be granted. As a whole the earth tends to conform to the laws of fluids, though its response may be tardy and the alteration of form may lag considerably behind the ultimate cause. This being so, it would be more correct to refer Kelvin’s time limits to the period of “lag” rather than to the period of the earth’s history as a solid globe.
Sir G. Darwin’s departure from the ring conception of Laplace in the case of the moon, and his alternative theory of the moon’s origin and history are well known. Tracing back to their logical conclusions the effects of tidal friction in the earth-moon system, he has developed one of the most fascinating romances in the domain of cosmogony. Unfortunately it is impossible to apply any definite dates to the critical stages of this history, if for no other reason than the imperfection of our present lunar and tidal theories. His lower estimate of the time which has elapsed since the disruptive catastrophe lies between 50 and 60 million years, but the actual time would certainly be much greater. Indeed, Darwin himself, referring to the problem of cosmical time after the advent of radium and the overthrow of the older standards, wrote, “I feel with some degree of confidence that if the tidal theory shall ultimately be condemned it will not meet its execution on the score of lack of time.” A recent discussion of the problem by Prof. Chamberlin and his colleagues is unfavourable to Darwin’s view. After a careful study of the rate of tidal retardation they show that the lengthening of the day is of the order of one second [Pg 28] in 500,000 years, and that if the physical condition of the earth has remained essentially as at present, at least 220,000 million years must have elapsed since the day occupied 20 of our present hours and the month 29 such days. Yet just after the birth of the moon, as outlined by Darwin, day and month were alike in duration, each being equal to about three of our present hours. It must be confessed that, after all, we know but little of the moon’s history with certainty.
At the time of its announcement to the world the Laplacian theory was remarkably in accordance with the knowledge of the time. But as further discoveries were made, numerous inconsistencies and contradictions arose. Not only were certain of the movements of the planets and satellites found to be in a retrograde direction, but, besides these awkward facts, at every stage of the postulated development of the solar system insuperable mechanical difficulties appear which render the truth of the hypothesis highly improbable. Finally, in the dynamics of the system there are so many discrepancies between the requirements of theory and the actual circumstances that Prof. Moulton and Dr. See both insist that it could not have originated in the way that Laplace imagined. The heavens have been diligently searched for nebulæ of the Laplacian type, but no certain case has been observed. The vast majority conform to the spiral type and to a nebula of this kind appeal has been made in the recently developed Planetesimal hypothesis of Chamberlin and Moulton.

Fig. 5. Symmetrical Nebula in Piscium, M 74.

Fig. 6. Whirlpool Nebula in Canes Venatici, M 51.
SPIRAL NEBULÆ.
[Pg 29] The general structure of a spiral nebula is immediately suggestive of tidal action. From a central nucleus two spiral arms emerge at diametrically opposite points. Often they are beautifully defined (Figs. 5, 6), but seen from the side they appear as discs of misty light. In the arms nebulous knots and irregularities are generally apparent, the precursors, perhaps, of a system of bodies analogous in their distribution to the planets. To account for the existence of a spiral form, conditions of extreme tidal distortion are suggested. In the case of our ancestral sun such conditions would be brought about by the close approach of another celestial body. As the stranger drew near, the tidal stresses set up would gradually increase until explosive outbursts of matter from the sun were projected in the plane of attraction, one on the near side, another on the far side. The result of the combined attractions of the two suns on the ejected material would be to develop a spiral structure. An enormous number of tiny planets or planetesimals would thus begin to circulate about the sun, associated with scattered knots of larger dimensions which would form the nuclei of the future planets and satellites. These knots would at once begin to grow by coalescence at the expense of the vast numbers of planetesimals associated with them in adjacent zones. Dynamically the scheme is sound, and the many suggestive consequences which unfold themselves explain away most of the difficulties which proved so embarrassing to the Laplacian hypothesis. Into a discussion of these advantages space does not allow us to enter, but it may be said that of all the attempts to grapple [Pg 30] with the fundamental problem of the genesis of the solar system, the Planetesimal hypothesis appears to be the most successful. It is to be welcomed, apart from its many convincing features, if only because of its stimulation to the further study of the early stages of earth history.
The earth began on this hypothesis as a nebular knot, and it has since grown up to its present mass by the capture of outside planetesimals. It is very unlikely that it was ever in a molten condition. Internal heat arose in part from the condensation of the mass during the period of its growth. The temperature would slowly rise until the fusion point of certain of the constituents was reached and the liquid tongues and pockets thus formed would then tend to move away from the centre—the lighter and less viscous stony material being squeezed upwards relatively to a network of the heavier and more rigid metallic material. Once vulcanism had been initiated in this way the process would continue until a highly metallic nucleus had collected. Surrounding it there would gradually form a thick zone of silicate rocks, the differentiation from the original heterogeneous mixture of stony and metallic constituents being due to the selective fusion of the former. In dealing with the question of the earth’s heat in a later chapter, the value, and indeed the necessity of this conception will be realised.
The atmosphere and oceans must have been derived from the planetesimals themselves, and on analogy with rocks and meteorites, the planetesimals would not be lacking in the raw material from which to evolve them. Even now, the outer 70 miles of the earth’s crust would be competent to [Pg 31] supply all the nitrogen of the atmosphere, the water of the oceans and the vast quantity of carbon-dioxide represented by limestones and carbonaceous deposits. With the existence of an ocean and atmosphere, a new factor in surface differentiation arose. Mechanical and chemical denudation became possible, and the first sediments were deposited. Although the earth’s growth had not yet ceased, all the agents occupied in its subsequent development were now at work and its geological history may be said to have definitely commenced.
[Pg 32]
The Great Ice Age—Extension of glaciation—Croll’s ingenious hypothesis—Inadequacy of the explanation—The ice caps of Mars—De Geer’s discovery of the annual layers of glacial clay in Sweden—Application to the measurement of time—Sederholm’s observations in Finland—Cyclic sedimentation in the Cretaceous strata of Colorado—Its correlation by Gilbert with an astronomical time unit.
In the last chapter mention was made of Kelvin’s work on tidal friction and of Sir G. Darwin’s speculations as to the early history of the earth-moon system. Their conclusions depended very largely on a cosmogony which has failed to meet with general acceptance. A very different appeal to astronomical causes was made by Croll in his famous attempt to account for the anomalous conditions of the glacial period. Here we may also consider the correlation by Baron De Geer and Mr. G. K. Gilbert of certain unusual phases of sedimentation with the seasonal and climatic changes brought about respectively by the earth’s motion and its periodic fluctuations. All of these investigations find a common basis in their direct reference to astronomical considerations.
In the course of its history the earth has undoubtedly passed through [Pg 33] several periods of extreme cold, periods when Arctic conditions swept down from their polar strongholds and invaded the temperate zones and even the tropics. Ice and snow have left their traces in many a grooved and striated surface, and in gently rounded outlines the landscape often betrays their former presence. Erratics and perched blocks, terminal and lateral moraines, lakes and alluvial terraces, U-shaped and hanging valleys all bear witness to the wide extension of the vanished glaciers of the past. Relics of glaciation have been preserved in the Cambrian or pre-Cambrian rocks of Norway, China, Australia and South Africa. Still more remarkable are the records of a Permian ice age found in the rocks of South America, South Africa, India and Australia. No unequivocal evidence of glaciation in later periods is forthcoming until the advent of the Pleistocene. The Great Ice Age through which the earth has so recently passed was not, however, a single epoch of glacial extension. Four episodes in the climatic cycle can be recognised—a genial episode, a period of falling temperature and glacial advance, a glacial episode, and finally, a period of rising temperature and glacial retreat. That this cycle has been four times repeated is the testimony of the Eastern Alps as interpreted by Prof. Penck. In the British area the ebb and flow of temperature has not left so clear a record, and a more continuous and persistent glaciation appears to be indicated.
At the time of the maximum advance of the ice, all northern Europe lay buried beneath an immense ice sheet, which was fed by enormous glaciers [Pg 34] slowly creeping down from the uplands. Curiously enough, Siberia, which in parts experiences the coldest winter in the world, is not now and was not then covered by the ice-field. Farther south the ice collected in the Pyrenees, Alps, Caucasus and in the great Asiatic ranges and descended in all directions far below the level of the present snow-line. In Canada and the United States the ice mantle proceeded from three great centres—from Labrador along the Atlantic coast, from the Keewatin district by Hudson’s Bay and from the Cordilleras along the Pacific coast. In the tropics and in the southern hemisphere the story is the same. Down the slopes of Kenia, Kilima N’jaro and Ruwenzori, the towering peaks of Central East Africa, the glaciers descended 5000 feet below their present termination. Kosciusko in New South Wales bears conspicuous traces of a former ice-cap which completely shrouded all but the highest peaks of the plateau. From the highlands of Tasmania and New Zealand, and from the Cordilleras and Andes of Patagonia and Chili the evidences of a prolonged glaciation are equally clear.
No satisfactory theory of climate has yet been propounded which affords an adequate explanation of this universal lowering of the snow-line. Whether or not the temperature fluctuations were synchronous all over the earth is difficult to decide, but that a colder climate characterised the southern no less than the northern hemisphere cannot be doubted. The causes to which appeal has been made in accounting for climatic changes are of three classes,—astronomical, geographical and atmospheric. In general the various tendencies will be opposed to one [Pg 35] another and by their interference prevent the attainment of universal extremities of climate. Occasionally, however, circumstances may arise when their joint action will lead in a single direction. A wide extension of tropical or polar conditions would then be expected to follow.
Of the three contributory causes, only one, the astronomical, bears any determinable relation with time. The theory that secular variation of terrestrial climates results from the changing eccentricity of the earth’s orbit, was first proposed by Adhémar. In 1868 James Croll greatly elaborated and extended this theory, presenting it with such a formidable array of quantitative data and yet in such an attractive form that it exerted a considerable influence on the geological thought of the day. Not only did it seem to offer an acceptable means of escape from the maze of difficulties by which the problem was shrouded, but in addition it promised a faithful chronology fixing the date and duration of the Ice Age with almost ideal precision.
Laplace established the fact that the ellipticity of the earth’s orbit is subject to periodic oscillations between certain limits. Sometimes the path is nearly circular but at other times it becomes a more flattened ellipse. If the earth were the only planet, its orbit would suffer no change; that it does is due to the attractions of the sister planets. Nevertheless, the orbit is essentially stable, and the yearly journey always occupies the same time. Formulæ were devised by Leverrier from which it was possible to calculate with some accuracy, the actual value of the eccentricity at any given period in past or [Pg 36] future within a few million years of the present. Croll utilised these formulæ to compute the dates of maximum and minimum eccentricity for the past three million years. He found three important periods when that factor was specially high, betraying a type of orbit more than usually flattened. These were:
| (a) | from | 2,500,000 to | 2,600,000 | years | ago. |
| (b) | ” | 720,000 to | 980,000 | ” | ” |
| (c) | ” | 80,000 to | 240,000 | ” | ” |
It was to the last of these that the Glacial Period was assigned.
When the earth is at perihelion, i.e. in that part of its orbit which lies nearest to the sun, it enjoys a more generous radiation than falls upon it at aphelion, when it is farthest from the sun. But this alone does not determine the time of summer and winter. At present the northern winter and southern summer occur when the earth is in perihelion. As is well known, this is owing to the inclination of the earth’s axis and the blanketing effect of the atmosphere. In the northern hemisphere, the greater thickness of air presented to the sun’s rays during the winter keeps out more heat than is gained by the relative closeness to the sun. However, this condition is not permanent. As discovered by Hipparchus in the year 134 B.C., the positions of summer and winter and of the equinoxes on the ecliptic are subject to a slow forward movement. In 26,000 years they make the complete circuit, and so in the course of time the relation of the seasons to perihelion is slowly altered. [Pg 37]
The precession of the equinoxes was shown by Newton to be a dynamical consequence of the spinning of the earth about a tilted axis. While this change is progressing, the position of perihelion is also shifting, and the resultant period is thereby reduced on the average to 21,000 years. Thus, in about 10,000 years from now the northern hemisphere will enjoy summer at perihelion. At the same time, the northern winter will occur at aphelion. The cold season will then be longer and more severe than now, and the annual accumulation of snow correspondingly increased. If, in addition, we suppose the orbital eccentricity to approach its maximum value, the northern hemisphere would then be in the grip of winter for nearly four months of the year, and the cold would become still more bitter than before. As a slight compensation for the rigours of the winter, the summer, though short, would be very much hotter. It was extremes such as these, accompanied perhaps by favourable geographical conditions, that Croll postulated for his glacial period. He believed that the snow and ice which would collect during a long frosty winter would successfully resist the evaporative powers of the summer, and that permanent snow-fields would therefore arise. Once started, the snowy mantle would tend to continue. A great deal of heat could be absorbed without raising the temperature above freezing-point, and the result of evaporation would be the creation of a thick blanket of fog, an effective agent in guarding the ice against the ravages of the summer radiance.
A peculiarity of Croll’s hypothesis is that glacial epochs could not [Pg 38] exist in both hemispheres at the same time. South of the Equator the summer would be longer than it is at present, and the winter would not only be short, but also comparatively mild. The climate would be that of a genial interglacial period. The alternation of glacial epochs between north and south is a necessary consequence of Croll’s view, but when the facts are examined they are found to be unfavourable to this assumption. In Sweden the last period of extension of the ice appears to have been synchronous with that in New South Wales, the close of each being dated, if our present time-scale is reliable, at 15,000 to 20,000 years ago. Both date and coincidence are decisive against the theory. Although Croll’s hypothesis achieved a great popularity, geologists were not wanting who considered the suggested causes to be utterly inadequate to produce so radical a change of climate. It happened that during the penultimate period of extreme eccentricity, which began nearly a million years ago, the astronomical conditions were more favourable to glaciation than they have been since. The question therefore arose why no traces had been preserved in the deposits of that time. Indeed, since the Cambrian, many hundreds of glacial periods should have come and gone. Had this been the case, and corroborative evidence sufficiently convincing, the earth’s chronology would have been written boldly in its rocks. But Croll’s brave attempt to number the ages was unsuccessful, and his attractive theory no longer holds the field.
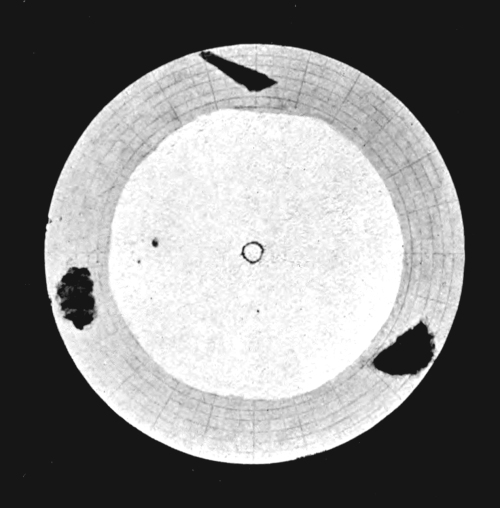
Fig. 7. At maximum: full extent of white.
At minimum: inner circle.
North Polar Cap.
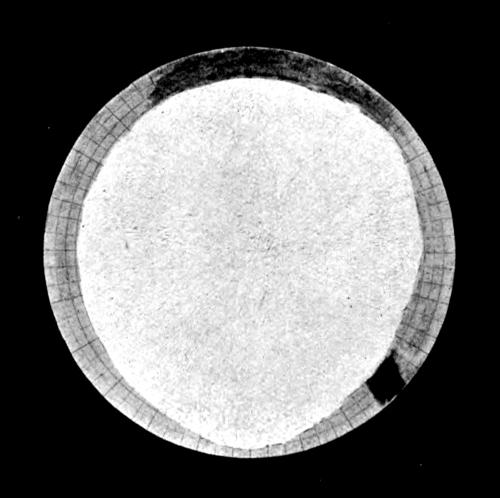
Fig. 8. At maximum: full extent of white.
At minimum: disappears entirely.
South Polar Cap.
MARS.
[Pg 39] A gigantic experiment illustrating the very conditions which Croll postulated has been recently shown to us by Prof. Lowell. For his working model we must look to the sky and carefully watch the changing seasons of Mars. The eccentricity of the orbit of Mars is much higher than that of the earth could ever have been, and moreover, the southern winter falls near aphelion. The conditions for a permanent ice-cap over the south polar regions are therefore ideal. The actual facts are surprising, and on the accompanying plate Prof. Lowell has depicted them admirably. During the winter a large snow-cap collects around the south pole, but in summer it is entirely dispersed. Around the north pole the winter snow-cap is less extensive, but all through the summer it never quite disappears. While the long southern winter undoubtedly makes possible a greater accumulation of snow, the hot short summer more than compensates by its superior powers of evaporation. A permanent and widely extended ice-field evidently could not originate. This demonstration of Croll’s hypothesis in actual practice does not present an altogether complete analogy to terrestrial conditions. The surface temperature of Mars and the thermal properties of its atmosphere may be widely different from ours, and the excessive rate at which the polar caps diminish in the spring clearly indicates the comparative thinness of the deposit. But in spite of these differences, the remarkable conclusion stands unassailed—that the evaporative power of the short hot summer of the south exceeds that of the long but cooler summer of the north. [Pg 40]
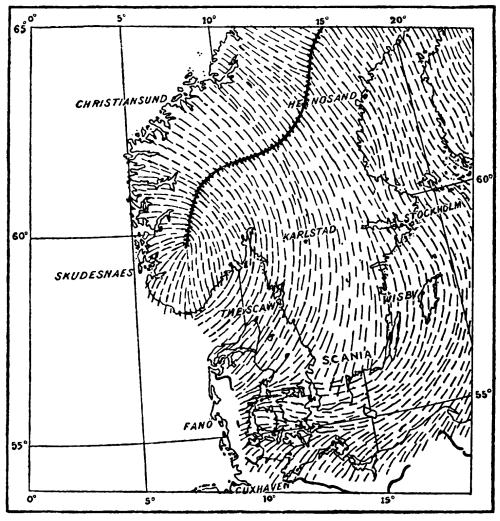
Fig. 9.
Directions of Glacial Movement in Southern Scandinavia.
It was stated above that since the culmination of the last glacial epoch more than 15,000 years have elapsed. Of the several methods which have been employed to determine this period only one can be dealt with here. The present genial climate has not greatly varied during the last 7000 years. As we trace back the record of temperature still farther a gradual fall can be discerned, accompanied by a wide extension of the [Pg 41] ice. Fig. 9 depicts the directions of movement of the last continental glacier of Scandinavia and its southern boundary across Jutland and the Baltic provinces. As it retreated, it left the terminal moraine known as the Baltic ridge, and its southern limit was gradually pushed back till it extended to what is now the coast of Scania. From this point its recession has been followed in great detail by De Geer, who has made a careful study of the deposits which, extending from the Baltic up to the Scandinavian ice-shed, mark the progress of its annual retreat. Upon his observations he has founded a system of geological chronology which is of the greatest importance in that it marks the first effort towards absolute accuracy.
Each spring and summer, as the glacier thawed, a great deal of sand and clay was set free and carried away in suspension by the numerous streams which flowed from under the melting ice. The coarser material, on reaching the sea, settled down almost at once, but the finest particles of clay, able to remain in suspension much longer, were not completely deposited. Then came the autumn and winter, and the freezing of the streams. The sea received no further supply of sediment, and the load of fine mud slowly settled on the sea-bottom to form a thin layer of pure clay, sharply differentiated from the coarser bed below. The following year the glacier retreated a few hundred feet to the north, and the material then liberated was sorted out as before and again deposited in two well-marked seasonal layers. As this process continued year after year the area of deposit moved northwards with the ice, and [Pg 42] the annual layers of sediment thus became superimposed one upon the other like wedge-shaped tiles on a roof. The width of each bed is generally less than thirty miles, for even the finest mud cannot be traced beyond that distance. This being the case, no vertical section contains all the layers, and it is rarely that more than a hundred can be counted in one place. The total thickness of the recessional deposit seldom exceeds thirty feet.
De Geer successfully attempted the difficult task of counting the annual bands of glacial clay deposited throughout the period of retreat from the Scanian coast. Fortunately, any given set of layers can be traced from one locality to another, and as each ribbon of sediment dies out the higher beds are followed up in the same way until the whole series has been examined from bottom to top. The late-glacial beds number about 5000, and the time which has elapsed since the ice border reached the eastern coast of Scania is therefore 12,000 years.[2] The time of recession from the Baltic ridge to the Scanian coast remains to be estimated. Much of the record is hidden beneath the waters of the Baltic, and in North Germany De Geer’s method has not yet been applied. It appears, however, that the withdrawal of the ice was not uniform. It began slowly and reluctantly, but towards the north became more rapid. In the region of Stockholm the retreat was five times as fast as in Scania. We may therefore assume that in the still earlier stages the time taken was considerably more than that required [Pg 43] for a retreat over an equal distance in Scania. The latter would have occupied rather more than 2500 years, and on this basis Sollas provisionally accepts 5000 years as the period during which the ice front was driven back to the south coast of Sweden. In this way the whole interval which has passed since the culmination of the last glacial episode is determined to be greater than 15,000 years, with 17,000 years as a probable value.
Amongst the Archean rocks of N.W. Finland, Prof. Sederholm has found in the Bottnian schists and phyllites primary characters which are strikingly similar to those of the banded glacial clays. Under the microscope their textures are distinctly clastic, and each composite stratum is sharply divided into two thin bands, the coarser one originally of sand and marking the beginning of a new year of deposition, the finer one originally of clay. Sederholm interprets the phenomenon on a uniformitarian basis as indicating that, even in those remote times, there was a marked difference in the seasons. By measuring the thickness of many thousands of annual layers, he finds their mean thickness to be about five inches. The total thickness of the banded phyllites amounts to 10,000 feet and the time they appear to represent is therefore only 24,000 years. This conclusion is probably far from the truth, for, as Sederholm particularly emphasises, it is difficult to know what is meant by the thickness of a deposit, so many arbitrary and misleading elements enter into its determination.
From De Geer’s work, it might have been said at first that a thickness [Pg 44] of thirty feet was deposited in a hundred years, but as the deposit was traced over the country, it became equally evident, that with no apparent thickening of the formation, deposition had gone on for 5000 years, and considering the growth northwards from the Baltic ridge, perhaps for 10,000 years. The whole difficulty lies in determining which parts of a formation are strictly contemporaneous. A measurement of thickness is significant only in relation to the immediate area of deposit and to the rate at which it moves landwards or seawards. The thickness of a formation and the maximum thickness of the layers formed in successive years may differ enormously, as De Geer’s researches have made so evident. For the same reason the term rate of deposit is loose and misleading unless it is clear to what it refers.
Another astronomical method of estimating time, though embracing much longer periods, has been applied by Gilbert to certain formations in Colorado. The basin of the Arkansas River is largely occupied by Cretaceous sediments, a succession of which is given in the adjoining diagram. At the four stages marked A, B, C and D the argillaceous shales give place to a calcareous type, and in these there is a regular alternation of thin layers of limestone and of calcareous shale. At A the average thickness of a pair of beds is 1·5 feet, and the number of repetitions is 15. At B the limestones are more massive, but the parting shales are very thin. Here again the average thickness of two adjacent beds is 1·5 feet. The limestones at C are less pure, and each, with its associated layer of shale, amounts on the average to about 2·7 feet. At D the succession is similar. [Pg 45]
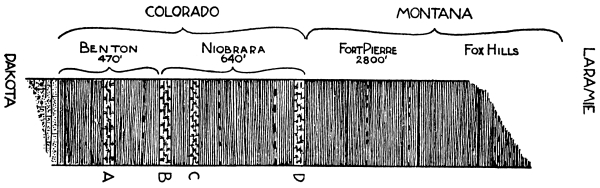
Fig. 10.
Vertical Section of the Upper Cretaceous strata of Colorado.
Below are the Comanchian or Lower Cretaceous beds;
above are the Transition beds to the Eocene.
[Pg 46] To explain the remarkably regular alternation of conditions which determined this uniform rhythm of sedimentation, purely terrestrial causes appear to be insufficient. Upheaval and subsidence of the earth’s crust, and the changing distribution of land and sea, are characterised rather by their irregularity than by any rhythmic sequence. While there is undoubtedly a rough periodicity in earth movements, yet it is discernible only on the broadest scale and is out of all proportion to the requirements of this case. Gilbert therefore suggests an astronomical cause. Of the several cyclic changes to which it is reasonable to appeal, the annual revolution of the earth, and the variation of the eccentricity of its orbit, demand periods which are in the first case too short (one year), and in the second too long (91,000 years). As we have already seen, the relation of the seasons to the position of perihelion repeats itself about every 21,000 years, and this astronomical cycle seems better adapted to meet the case. The climatic changes which accompany the precession of the equinoxes might influence the character of sedimentation in many ways. With a changing circulation of winds and currents, argillaceous material might be transported and deposited at one time and calcareous at another. On land, vegetation might predominate during part of the cycle, and the surface waters would then dissolve more calcium carbonate than during a period when vegetation became sparse. At the same time mechanical [Pg 47] erosion would be impeded in the first instance, but would be more active in the second.
It is evident that the shale was deposited more rapidly than the limestone, for when the principal deposit was calcareous the thickness is 1·5 feet, whereas, in the case of the less calcareous beds, it rises to 2·7 feet. In the normal shales a conservative estimate of the equivalent thickness would be 4 feet. Adopting the astronomical time unit of 21,000 years, the rate of deposit would then be of the order:
| Limestone, | 1 foot in | 14,000 | years. |
| Shale, | 1 foot in | 5,000 | years. |
On this basis the 3900 feet of shale in the Benton, Niobrara and Pierre formations represent about 20 million years, for in this example the term thickness seems to have a definite meaning. If the assumptions are correct, the duration of the whole Cretaceous period must therefore be considerably greater than this.
[Pg 48]
Transference of material from land to sea—The denudation ratio—Weathering of rocks—The work of chemical denudation—Summary of the data—Composition of the saline matter in the oceans and of that annually carried to the oceans—The work of mechanical denudation—Suspended and bottom loads of rivers—Mississippi not a good average case—Dole and Stabler’s work in the United States—Application to the whole land area—Total material removed and rate of degradation of land—Marine erosion—Types and quantities of sediments annually produced.
The purely geological methods which have been devised to investigate our problem are of two kinds. The first attempts to apply a time-scale to the sedimentary rocks and was, historically, the earliest to be proposed; the second, due to Joly, deals with the accumulation of salt in the oceans. The one is concerned with material carried away from the land mechanically; the other with the material removed in solution. The various agents of weathering, of which rain and frost are the chief, disintegrate the surface rocks and supply the rivers with their load of detritus. The turbid condition of rivers when in flood, heavily charged with alluvial matter, is a familiar and convincing proof that the effect of erosion in conjunction with the transporting power of running water must always be to wear down the land areas. In the dynamical [Pg 49] study of denudation and sedimentation the first essential is to know the rate at which the rivers are working. Measurements of their load of silt and dissolved salts and of their annual discharge to the sea make it possible to arrive at reliable estimates of their activity.
Incidentally, it is useful to determine the ratio which solvent denudation bears to the whole. The denudation ratio, as it may conveniently be called, is the ratio of the load of dissolved material to the total load carried both in solution and in suspension.
The amount of material removed in solution from the surface rocks is not quite the same as that which is carried to the oceans. A small proportion is abstracted from the over-ground circulation by the waters which sink below the surface, and while some of this is undoubtedly brought up again through the agency of springs, it seems possible, as Prof. Schwarz has boldly suggested, that certain constituents, such as iron and magnesium, may be permanently removed from the earth’s crust by downward migration. In the denudation ratio this possibility is left out of account as having no bearing on the study of sedimentation.
A rough estimate of the denudation ratio may be made by considering the weathering and decay of rocks in situ. Soluble constituents are withdrawn by leaching and a residue of the more stable minerals and alteration products is left behind. Weathering involves not only the abstraction of material but also the introduction of fresh material from external sources. Oxidation, hydration, and carbonatisation are [Pg 50] the most typical reactions, and they must be allowed for in determining the proportion of the original rock lost by solution. This can be done approximately by assuming that some element—aluminium being usually chosen—has remained invariable during the course of decomposition. From the analysis of a large number of fresh rocks and of their altered equivalents, it is found that on an average 30% is dissolved, leaving a residue of 70%. The denudation ratio ought therefore to be about 0·3.
The direct determination of the work of chemical denudation requires three distinct sets of measurements: (a) the annual discharge of rivers into the oceans; (b) the analysis of their waters; (c) the measurement of their drainage areas. Mellard Reade was the first to point out the importance of these factors in the study of dynamical geology. In 1879 he collected such information as was then available and deduced from it the quantity of rock material annually removed from the whole land area, his estimate being 5280 million tons. Sir John Murray’s corresponding estimate of 1887, which was based on analyses of the waters of nineteen of the world’s principal rivers, was 4975 million tons.
Until 1909 this figure could not be improved upon, but in that year there was published by the United States Geological Survey the results of the detailed and systematic work carried out by R. B. Dole and H. Stabler. For the first time an attempt had been made to measure the discharge, drainage areas, salinity, and suspended load of all the important rivers of a large continental area. The estimates represent [Pg 51] the averages of observations made daily for a year or longer, and in the case of the discharge, the measurements extended over at least seven years.
Seasonal variations and the effects of floods are apt to be misleading, and a long continued series of observations is necessary if their relative importance is not to be over—or under—estimated. The amount of material in solution varies but slightly from year to year, and the average of one year’s results is within 10% of the true mean value. The annual discharge varies much more than this, and still more inconstant is the load of suspended material, which, in certain years, may differ from the average value by 50%. These figures indicate how difficult it is to introduce exact measurements into geology with any hope of finality.
A summary of all the best data now available has recently been given by Dr. F. W. Clarke, and covers about 28 million square miles of the drainage areas of the earth. The details are given in the following table:
| Continent. | Drainage Area in sq. Miles. |
Solvent Denudation. Tons Annually Removed. |
|
|---|---|---|---|
| Per Sq. Mile. |
From Whole Area. |
||
| N. America | 6,000,000 | 70·5 | 423,000,000 |
| S. America | 4,000,000 | 45·5 | 182,000,000 |
| Europe | 3,000,000 | 90·0 | 270,000,000 |
| Asia | 7,000,000 | 75·0 | 525,000,000 |
| Africa | 8,000,000 | 40·0 | 320,000,000 |
| 28,000,000 | 61·0 | 1,708,000,000 | |
[Pg 52] The total land area of the globe is estimated by Murray at 55·7 million square miles, but of this 11·5 million square miles are areas of internal drainage, such as the Great Basin of the United States and the Asiatic depressions, which contribute nothing to the ocean. The circumpolar regions, representing 4·5 million square miles, must also be left out of account, and the remaining 39·7, or say, 40 million square miles, is that from which the oceans are fed. If the figures given above be accepted as typical, then the annual addition of material to the oceans by solution amounts to 2440 million tons. Some of this, however, is derived from the atmosphere—chiefly as carbon-dioxide. Applying the necessary correction of nearly 10%, there remains 2220 million tons as the amount actually derived from the rocks. Murray’s estimate, it will be noticed, is almost exactly twice that of Clarke.
The composition of the saline matter carried to the oceans may be found by suitably weighing each analysis of river water according to the discharge of the latter. The general mean of all such results is given in the table opposite, together with the total amount of each substance in the ocean. The small traces of elements other than those listed are quite insignificant. [Pg 53]
| Constituent. | Annual Addition of Saline Matter in millions of tons. |
Total Saline Matter of the Oceans in millions of tons. |
|---|---|---|
| SiO₂ | 284 | |
| Fe₂O₃Al₂O₃ | 67 | |
| Mg | 83 | 1,535,000,000 |
| Ca | 497 | 490,000,000 |
| Na | 156 | 12,616,000,000 |
| K | 37 | 454,000,000 |
| Cl | 138 | 22,800,000,000 |
| Br | 78,000,000 | |
| CO₃ | 857 | 80,300,000 |
| NO₃ | 22 | |
| SO | 299 | 3,172,000,000 |
| Total | 2440 | 41,230,000,000 |
| Annual discharge of river water into ocean | = | 24·3 × 10¹² tons |
| Volume of ocean water | = | 307,496,000 cubic miles |
| Density of ocean water | = | 1·026 (mean) |
| Mass of ocean water | = | 1,178,270 × 10¹² tons |
It is obvious from a comparison of these two columns that the annual increment of dissolved matter is not permanently retained by the oceans. The greater proportion is precipitated by chemical and organic agencies, and either becomes incorporated with detrital material or goes to form individual sediments. The chief substances produced are calcium and magnesium carbonate, gypsum, limonite, and silica. Rather more than one-third of the carbonates appear to associate themselves intimately with sands and muds. The greater part of the remaining two-thirds is deposited as limestone on the continental shelves (after being used by various organisms in shell-making) in waters which are comparatively free from terrigenous sediment. The abstraction of gypsum [Pg 54] from the ocean takes place at irregular intervals under suitable conditions of concentration. Of the limonite and silica, the chief precipitation takes place on the continental shelves where they associate themselves with the detrital sediments. The history of potassium is rather obscure, but on the contrary, that of sodium appears to be the simplest of all. That it is stored up in the oceans is an assumption which is granted as justifiable by most geologists.
The only new factor required in order to estimate the mechanical work of denudation is the load of material carried by the rivers. Besides the silt transported in suspension, larger fragments are carried by rolling along the stream bottom. Measurements of the bottom load are lacking except in a solitary case—that of the Mississippi—in which it amounted to about 10% of the whole. It is difficult to define any precise difference between bottom load and suspended load, the former being only a limiting case of the latter. When the water is fully charged with rock debris the highest proportions are found near the bottom and sides, and at a point in mid-stream at about one-third the depth from the surface—the position of the stream lines of maximum velocity. In clear water the rolling power reaches its maximum value, for apart from fluid friction, energy is expended in no other way. Most rivers fall between these extremes. In making actual measurements, samples are taken from representative points in the river and used to give the average over the whole section. In the final estimate it seems probable that a large proportion of the bottom load is accounted for. [Pg 55] Even if a correction ought to be applied it would be pedantic to do so except for rivers which have been under observation for several consecutive years, since the variation from the mean annual load is very great from year to year. The Nile varies by 40% and the Potomac by as much as 100%. In the United States the mean variation is about 50%. With uncertain data of this kind a correction of less that 10% may safely be disregarded.
Of all the rivers of the world, the Mississippi has been most favoured by measurements of the kinds required, and many estimates of the rate of continental degradation, of the rate of deposition of sediments and of the age of the earth have been based upon them. There is no doubt, however, that the Mississippi is working more rapidly than any other river of importance in North America, except, perhaps, the Colorado River. The high declivity in the west, the Tertiary elevation of the plains to which the streams are not yet adjusted, and the abundance of easily eroded glacial drift are all factors which promote this activity. In the case of rivers other than those of North America for which data are available, the same high rate of denudation obtains; the Rhone and the Po, for example, being amongst the most energetic workers in the world. Generalising for the whole earth from these rivers alone, would obviously give misleading results. The work of Dole and Stabler again comes to our aid, and in the following table their aggregate measurements for the whole area of the United States are tabulated. [Pg 56] Similar evidence for four widely separated rivers is also given:
| Drainage basins. | Area in sq. miles. |
Millions of tons of Material removed per Year. |
Denudation ratio. |
|
|---|---|---|---|---|
| In Solution |
In Suspension |
|||
| United States | 3,088,500 | 241·5 | 468 | 0·34 |
| Mississippi | 1,265,000 | 122 | 304 | 0·29 |
| Nile | 1,100,000 | 21 | 52 | 0·29 |
| Uruguay | 150,000 | 7·5 | 15 | 0·33 |
| Rhone | 34,800 | 8·5 | 36 | 0·19 |
Leaving out the Mississippi because of its inclusion in the United States and weighing each result according to the area over which it holds, the mean denudation ratio is 0·31, a figure which agrees very well with our previous estimate. If now we use the denudation ratio to calculate the material removed by mechanical denudation over the whole land surface, we should not be far from the truth. It is clear that if from the 40 million square miles which drain into the oceans the quantity of material carried in solution represents 0·3 of the total material removed, then, as the former amounts to 2440 millions of tons annually, the quantity carried away mechanically must be 5700 million tons. That this figure is of the right order is favoured by another consideration. The mean elevation of North America is very nearly that of all the land areas of the earth. Moreover, according to Clarke’s figures the rate of denudation over North America is slightly higher [Pg 57] than the average for all the lands, but more closely approaches it than does that of any other continental area. We may therefore take the rate of denudation of North America as a fair average and apply it with some confidence to all the drainage areas of the globe. Doing this, the total amount of suspended material annually discharged into the oceans is computed to be 6000 million tons.
We may sum up the work of denudation in round figures as follows:
| Material annually removed in solution | 2500 | million | tons |
| Material annually removed in suspension | 6000 | ” | ” |
| Total | 8500 | million | tons |
These figures may also be expressed in terms of the rate at which the land areas are being worn down. By solvent denudation a degradation of one foot in 30,000 years is implied, and by mechanical denudation, one foot in 12,000 years. Taking both together the average rate of denudation is found to be one foot in 8600 years. It should be clearly understood that individual areas may be lowered at rates very different from this. The maximum rate is attained in the Irawadi basin, one foot of which is removed in 400 years. The Po is also an exceptional river, and lowers its basin by one foot in 850 years. On the other hand, in the Hudson Bay district of North America the drainage only carries away one foot in 47,000 years.
No minimum figure can be given, for wherever deposition of sediment [Pg 58] takes place on the land areas the temporary rate of denudation locally becomes negative. In making these calculations, the density of rock material is taken as 2·6; the weight of a cubic foot as 165 lbs; and the weight of a cubic mile as 10,800 million tons. Although the surface covering of loam or earth weighs only about 100 lbs per cubic foot, the denser and more closely packed underlying rock need alone be considered, for it is by its decay and expansion that the superficial blanket above is produced.
Our final problem is to determine the nature and quantity of the sediments which are ultimately formed on the continental shelves. This can only be done roughly, but the results will suffice to serve our purpose. First of all, two serious difficulties must be met before the way is open to take this step. So far, marine denudation has been left out of account. It is not yet possible to make a wholly satisfactory estimate of the relative magnitude of the supply of detritus captured directly by the sea. The unknown factor is the average encroachment of the sea upon the coasts. For the British Isles, Croll suggested an average of three feet per century, and the figure assumed by Sir A. Geikie about the same time was ten feet per century. A much later estimate by Prof. Watts places the average retreat of the English coast at a hundred feet per century. Along parts of our East coast marine erosion is still more rapid than this, the conditions being exceptionally favourable. On the other hand, Geikie considers that all the force of the Atlantic beating upon the N.W. coast of Scotland may [Pg 59] not wear it away at more than one foot per century. What the average between these extremes may be can only be guessed at. If for convenience we accept Geikie’s figure as affording a likely average for all the coast lines of the earth (125,000 miles), and if the average height of the cliffs be taken as 150 feet, then the mass of material annually removed will be about 700 million tons.
The other difficulty is concerned with the annual amount of material which remains in the oceans in solution, and also of that which is deposited on the ocean floor outside the limits of the continental shelves. For the former, a knowledge of the age of the oceans is necessary. Considering all the evidence, the amount retained at the present day seems to be about 200 million tons, but this is certainly too high as a figure representing the average increase throughout the history of the oceans.
For the deep-sea deposits little more than a guess is possible, although we can now approximate to the right order of magnitude by considering the circulation of radium. The radium in the material removed from the lands is redistributed between the sediments on the continental shelves, the deep-sea deposits and the water of the oceans. Applying our present knowledge of the distribution of radium (see p. 131) the annual mass of the deep-sea deposits is found to be about ¹/₃₀ of the whole, i.e. about 300 million tons.
The difficulties can scarcely be avoided by balancing them against each other. There still remain 200 million tons (700 - 300 - 200) to be added to the 8500 million tons already found as the total for [Pg 60] sub-aerial denudation. This gives us 8700, or as an extra safeguard, say 9000 million, tons as the mass of sediment annually deposited on the continental shelves. It is unfortunate that to arrive at this figure an element of doubt should be introduced by associating the results of careful experimental work with the vague conclusions just arrived at. The bugbear of the whole investigation is marine erosion; but if it is remembered that the figures given in that connection are meant to be suggestive rather than final, no erroneous impression need be carried away.
If the sediments ultimately formed are shales (20% quartz), sandstones (75% quartz) and limestones (75% calcium carbonate), their proportions will be as follows:
| Shales | 70% | or | 6300 | million | tons. |
| Sandstones | 16% | “ | 1440 | “ | “ |
| Limestones | 14% | “ | 1260 | “ | “ |
| Total | 100% | = | 9000 | million | tons. |
In the two following chapters the application of denudational statistics to the measurement of geological time will be considered.
[Pg 61]
Halley’s proposal in 1715—Joly’s application of modern data in 1899—Further studies by Sollas, Clarke, and Becker—Data of the problem—Corrections for disseminated sodium, wind-borne sodium, human agencies, and marine erosion—Uniformity not capable of proof—Importance of cyclic circulation of sodium—Origin of chlorine now fixed in salt—Hour-glass method applied—Land areas of the past and their elevation compared with those of to-day.
In 1715 the famous astronomer Edmund Halley published a paper which he entitled, A short Account of the Cause of the Saltness of the Ocean, and of the several Lakes that emit no rivers; with a Proposal by help thereof to discover the Age of the World. He showed that since the water removed from lakes by evaporation is perfectly fresh, “the saline particles brought in by the rivers remain behind, while the fresh evaporate; and hence it is evident that the salt in the lakes will be continually augmented and the water grow salter and salter.” Applying the same principle to the oceans, he wrote, “It is not improbable but that the ocean itself is become salt from the same cause, and we are thereby furnished with an argument for estimating the duration of all things.” Two hundred years ago it seemed hopeless to attempt to determine the annual increment of salt added to the oceans, but Halley [Pg 62] used his argument “to refute the ancient notion some have of late entertained of the eternity of the world.” The paper was completely forgotten, until Dr. G. F. Becker again drew attention to it a year or two ago.
As we saw in the last chapter, the analysis of river waters has now made possible a determination of the annual amount of material carried in solution by the rivers into the oceans. T. Mellard Reade was the first to contemplate the application of solvent denudation to the measurement of geological time, and the data he gathered together in support of his arguments came as a great surprise to those who had concentrated attention merely on the mechanical work of erosion. No independent advance, however, was possible until 1899, when Joly pointed out that of the many elements which enter into the composition of sea water, sodium alone tends to accumulate. All the others are sooner or later rejected, associating themselves with the detrital sediments, or forming chemical or organic sediments by their ultimate precipitation. Joly then proceeded to use sodium as the age-index of the oceans. He assumed that the annual increment Naᵣ of sodium added to the oceans by all the rivers of the world has remained practically constant throughout geological time. If Naₒ represents the total amount of sodium now accumulated, the ratio Naₒ/Naᵣ gives the time which has elapsed since the oceans first existed and denudation began to wear down the lands. Joly’s first estimate was from 80 to 90 million years, and shortly afterwards he increased this to 100 million years. Sollas [Pg 63] attacked the problemafresh in 1909, and from a most careful survey of all the data and a detailed enquiry into every phase of the subject, he concluded that the most probable estimate of the age of the oceans would appear to lie between 80 and 150 million years.
In 1910 a further study was made by Clarke and Becker. The latter departed from the uniformitarian basis on which all the other calculations had been founded. He inferred that sodium accumulation progressed more rapidly in the past than at present. All the original sodium must have been derived from igneous rocks, and Becker considers that at the time when the oceans were first possible the surface of the earth must have consisted exclusively of such rocks. At the present day three-quarters of the land areas are covered by sedimentary rocks which can supply no further important additions to the sodium content of the ocean. Assuming that the production of sodium has been always proportional to the area of igneous exposures, and that the total land area of the globe has averaged 80% of the present area, he finds that the age indicated is about 70 million years.
We may now proceed to examine the problem in detail. The fundamental data on which the method is based may be summarised as follows:
| Mean density of the ocean | (Murray) | 1·026 | |
| Volume of the ocean | (Murray) | 323,800,000 | cu. miles |
| ”” | (Joly) | 339,248,000 | ” |
| ”” | (Clarke) | 302,000,000 | ” |
| ”” | (Karsten) | 307,496,000 | ” |
[Pg 64] Clarke now accepts the latter as being the best estimate, and we therefore calculate that the
| Mass of the ocean | = | 1,178,270 × 10¹² tons. | |
| Total salinity of the ocean | (Dittmar) | 3·5% | |
| Sodium of the ocean | (Dittmar) | 1·08% | |
| Accumulated sodium, Naₒ | = | 12,600 × 10¹² tons. | |
| Annual increment, Naᵣ | = | 156,000,000 tons. |
From these values the quotient Naₒ/Naᵣ gives the age of the oceans to a first approximation as 80·8 million years. This figure, however, cannot be regarded as final. Two of the assumptions on which it is based are that all the sodium liberated from igneous rocks is contained in the ocean, and that all the sodium carried annually to the ocean has been liberated from such rocks for the first time. The obvious corrections to be applied will increase the numerator of the ratio and decrease the denominator, thereby increasing the age estimate. The degree to which the numerator must be augmented is, as far as we know, relatively insignificant. Saline deposits, such as those of the Stassfurt district, are only of trivial importance. The salt of the ocean, if spread regularly over the whole land areas, would form a layer about 120 feet thick, and all the beds of rock salt which have accumulated by evaporation become negligible beside this vast quantity. The amount of sodium in ground waters and disseminated through the sedimentary rocks is to be taken into consideration more carefully. Marine sediments at the time of their formation are saturated with sea water, and, when raised up to form land, they must, therefore, be strongly charged with [Pg 65] salt. The actual proportion of sodium abstracted from the ocean in this way cannot at one time be very great, probably not more than 1% of the whole. Throughout geological time, however, a certain amount of sodium has been in cyclic circulation between land and sea. A rough idea of the influence of this circulation on the age estimate may be gained from a knowledge of the total volume of the sedimentary rocks, and of their average pore space.
Neither of these factors is yet known with the desirable accuracy, but as far as present needs are concerned, the total bulk of the sediments, excluding deep-sea deposits, may be placed at 70 million cubic miles, and their pore space at 10%. The latter figure is probably too low, for fresh sediments have a pore space of 40% to 50% of their volume, and in consolidated sandstones 20% is common. Many of the sediments have been denuded and re-deposited time after time; on the average, probably three times. On the other hand, if the ocean has progressively increased in salinity, its average sodium content must have been about half that of the present. Basing our calculations on these considerations, the total volume of sediments which have ever existed is about 210 million cubic miles; the pore space to be filled by sea water would then be 21 million cubic miles. The total amount of sodium precipitated within the rocks by evaporation could not therefore exceed 420 million million tons, and at the present rate of denudation its removal would require nearly three million years. To what extent this result would be increased by absorption effects, whereby interstitial [Pg 66] salt solutions are concentrated in order that they may be in equilibrium with normal sea water, cannot be estimated. This corrective factor would probably not be large.
A still more important cyclic circulation is brought about by wind-borne sea salt. Fine particles of spray are swept from the foaming crests of waves by the wind and carried often many miles inland. Near the coast the salt blown over the land is naturally most abundant. It falls in sea fret and rain, and is ultimately returned to the sea in the drainage from the land. Fortunately, the amount of sodium supplied in this way can be estimated by analyses of rainwater and a measurement of the rainfall. As before, the cyclic sodium must be excluded from that entering into the denominator as primary sodium. Joly allows an additive time correction of 10%, Clarke of 7%, and Becker of 6%. The time estimate is therefore increased by a further five or six million years.
Clarke has suggested that the present rate of sodium accumulation has been accelerated by human agencies. Nearly six million tons of sodium are annually produced as common salt, and, as sewage and chemical refuse, much of this is again returned to the oceans. Subtracting this from the denominator, the age is increased by other three million years.
A number of corrections may be made to decrease the first rough estimate of the ocean’s age. Solvent denudation due to marine erosion must not be forgotten. Experiments by Joly have shown that sea water is several times (2½-14) more active as a solvent than fresh water. He attributes to its action over the tide-swept strand—covering a total [Pg 67] area of about 60,000 square miles—a supply of sodium amounting to 3% of that derived from the normal drainage areas. This is a very liberal estimate, and in applying a negative correction of three million years we are granting all that can reasonably be claimed.
Finally, there are other possibilities of uncertain magnitude which deserve mention. The ocean may conceivably have contained sodium before the fluviatile contributions commenced. By volcanic eruptions, sodium-bearing materials may be cast widespread over the sea. Juvenile waters expelled from igneous magmas may presumably afford a supply of sodium. The importance of saline waters derived from the heated interior of the earth has been particularly insisted upon by the great geologist Suess. As a source of sodium, such waters are probably of little moment, for the whole amount of sodium in the ocean is already more than explained by the erosion of igneous rocks. Solvent denudation involves a loss from average igneous rock of 1·57% due to sodium abstraction alone. According to Clarke, the average sodium content of igneous rocks is 2·52%, and the amount retained by the detrital material which goes to make up the sediments is 0·95% of the original rock. Now, if the total mass of sodium which has accumulated in geological time is 12·6 × 10¹⁵ tons and this represents 1·57% of the igneous rock in which it originally occurred, it is evident that the mass of the igneous rocks which have been denuded away amounts to 800 × 10¹⁵ tons. The mass of one cubic mile of rock may be taken as 10,800 [Pg 68] million tons, and hence the volume of igneous rock which has been removed is 74 million cubic miles. The sediments derived from the latter ought therefore to occupy a volume of the same order. A number of independent estimates of the volume of sediments existing on the land areas have been attempted, and while no great accuracy can be hoped for, it is significant that all agree in assigning a volume somewhat greater than 74 million cubic miles. The sediments hidden under the oceans would add still further to the total. If any reliance at all is to be placed on these estimates, it is clear that the sodium in the ocean ought to be more than it is; at any rate, there would appear to be little room for sodium derived from other sources, such as the interior of the earth.
We may sum up the results of this discussion as follows:
| Approximate age, Naₒ/Naᵣ | = | 81 | million | years. |
| Correction for | ||||
| (a) Disseminated Sodium | = | 3 | million | years. |
| (b) Wind-borne Sodium | = | 6 | ” | ” |
| (c) Human agencies | = | 3 | ” | ” |
| (d) Marine erosion | = | -3 | ” | ” |
| Age of the oceans | 90 | million | years. | |
Another assumption on which this computation is founded must now be examined; that is, the practical uniformity of the annual addition of sodium throughout the period involved. Little weight can be attached to the argument that the source of supply has been gradually impoverished [Pg 69] in alkalies, for the soils from limestones, which are among the poorest of the alkali-bearing rocks, may contain more sodium than do the soils derived from granites. The sedimentary rocks being less able to resist erosion are more rapidly disintegrated and removed than are the igneous rocks. The composition of streams considered in relation to the rocks which they drain is of interest here. Hanamann has shown that in the Elbe and its tributaries three times more sodium is carried away from a square mile of Cretaceous sediments than from an equal area of granite. In the same connection, it is well known that the waters flowing through stratified rocks carry more salt than those from igneous rocks. The greater exposure of the latter in former land areas, if such were the case, would certainly not contribute a greater sodium income to the ocean than if sedimentary rocks prevailed. Joly considers that an approximate uniformity has been preserved whatever the distribution of rock types may have been. It would not, however, be difficult to prove by actual analyses that a greater area of igneous rocks would result in a reduction of the sodium increment, and that the present rate may be too high because of the predominance of sedimentaries.
This unexpected conclusion raises the question of the validity of the method at its most critical point. Can we be sure that the cyclic circulation of sodium has been taken sufficiently into consideration? The only source to which the anomalous proportion of sodium from sedimentary rocks can be traced, apart from the factors already dealt [Pg 70] with, is constituted by the ground waters. These waters often contain minute quantities of salt of which the origin is very obscure. Whence came the chlorine? Igneous rocks themselves only contain about 2% of the chlorine necessary to convert the liberated sodium into salt. The emission of chlorine from volcanoes and solfataras probably affords the necessary supply, but most of this never reaches the ground waters directly. Becker, dealing with the same difficulty in the case of river water, calculates that 40% of the annual sodium increment is combined with chlorine of which the origin cannot be traced. He accordingly rejects this amount as not being of normal or primary origin. If it has all been derived from the oceans, which he regards as an extreme possibility, it must be subtracted from the apparent income, since it is simply a circulation of capital.
A maximum figure for the age of the oceans (still on the assumption of uniformity) ought theoretically to be attainable by accepting the whole of the sodium-chloride of the rivers as cyclic. We have the following figures:
| Chlorine | = | 138 | million | tons. |
| Sodium combined with chlorine | = | 87 | ” | ” |
| Sodium uncombined with chlorine | = | 69 | ” | ” |
| Total Sodium, Naᵣ | = | 156 | million | tons. |
The unchloridised sodium implies an age of 180 million years, an estimate which would in general be condemned as excessive. [Pg 71]
The difficulties presented by sediments and ground waters are almost insuperable, and it may be safer to adopt a means by which they can be entirely disregarded. The sodium lost from igneous rocks amounts by mass to 1·57% of the latter. Igneous and metamorphic rocks are at present exposed over one-fifth of the drainage areas, or 8,000,000 square miles. Many of the metamorphic rocks are partially or wholly of sedimentary origin, and ought not to enter into the computation. We have already seen that the average rate of denudation is one foot in 8600 years, and at this rate, one cubic mile of primary rock, or 10,800 million tons, would be denuded away in 4·54 years. In one year the supply of sodium would therefore be 37 million tons, and the time required for the accumulation of the existing store of sodium in the oceans would be 340 million years.
We meet with a curious discrepancy at this point. The actual amount of unchloridised sodium is 69 million tons, while that which is drawn from the igneous rocks cannot exceed 36 million tons. In each case sub-aerial denudation alone is being considered, so that the failure to take marine denudation into account does not affect the matter. The figures imply that a great deal of unchloridised sodium is drawn from the sedimentaries. Even if the extreme assumption is made for the moment, that the igneous rocks lose all their sodium, 2·52%, the annual supply could only reach 60 million tons. The discrepancy is evidently due to the facts that sediments freshly formed from primary rocks retain a considerable percentage of sodium, and that it is only after [Pg 72] the material has been re-assorted, perhaps several times, that the element is withdrawn to the extent indicated by Clarke’s average figures. At the present day, even when the igneous rocks have been deprived of 37 million tons of sodium, the sedimentary rocks, to bring up the total to 69 million tons, must lose more than half of their primary sodium. It is interesting to observe that if all the sodium now carried each year to the oceans—156 million tons—were derived directly from the rocks, igneous and sedimentary, the latter would be obliged to lose nearly twice as much sodium as they actually contain! This remarkable conclusion indicates either that our statistics of the annual production of sediments are hopelessly wrong, or else that the chloridised sodium is almost wholly cyclic. The latter inference, supported as it is by the impossibility of otherwise accounting for the chlorine, thus receives further support.
In the application of the above data to the age question, it has been tacitly assumed that the present exposures of primary rocks are neither greater nor less in area than the average of such exposures since the oceans began.
It is impossible to know what the average may have been, for while the original lands must have been entirely formed of igneous or pseudo-igneous planetesimal material, it is equally certain that their area was but a fraction of that of the present land areas. The evolution of the earth’s surface features has tended to intensify the difference between the ocean depths and the mountain heights. The ocean [Pg 73] basins have gradually deepened and become more stable, and their relative permanence is generally admitted. The lands are now more extensive than ever they have been; initially it is probable that only low and scattered islands emerged from the shallow primitive seas. We are faced here with many vague problems. The volume of the seas may have been less than it is to-day; the original rocks were probably not granitic in type, and were certainly poor in sodium; carbon-dioxide seems to have been the chief atmospheric companion of nitrogen, and solvent denudation would be correspondingly more vigorous. How far all these considerations affect the point at issue cannot be decided. To discuss them would lead us into a complex maze of speculation. The original basis of our calculation may afford as near an approach to the truth as could be arrived at by any other reasonable hypothesis of changing land areas, and of their composition.
On this view it is possible to calculate a minimum figure for the age of the oceans, but it must be remembered that the results based on an assumption, which is merely a uniformitarian guess, are themselves equally uncertain. If the igneous rocks had lost all their sodium, we have already found that the annual supply would be 60 million tons, and the sediments would be destitute of that element. The age would then be at least 210 million years. The contradiction between this minimum figure and the maximum deduced from the total unchloridised sodium, viz. 180 million years, is explicable on one or both of two further possibilities, that the average exposure of igneous [Pg 74] rocks in the past has been greater than that of the present day, or that the amount of primary sodium now being derived from the sedimentary rocks is considerably above the average for the past. Our interpretation of the data is made particularly difficult because of the widespread covering of glacial detritus, rich in felspathic constituents and easily eroded.
The contradictory results become still worse when it is remembered that the rate of denudation—one foot in 8600 years—is probably too high to be safely applied to areas of igneous rocks. Analysis of river waters draining such areas indicate a rate which is only about a half of the above. But even this correction is surpassed in importance by one arising out of the possibility that the present standard of measurement may be misleading in not being typical of geological time. The mean height of the drainage areas is subject to much fluctuation. By denudation it is steadily reduced; by earth movements, and particularly during the periods of intense diastrophism, it may become unusually great. It seems not unlikely that we are now near a period of extreme continental elevation, and that the geological processes which are thereby quickened up, cannot be accepted as affording a true standard. This suggestion will be dealt with further in the next chapter.
The high figures—210-340 million years—given above, must not be supposed to possess any serious value. The whole discussion merely serves to betray the uncertainty of the method and the doubtful [Pg 75] applicability of even the most accurate data. For the present we can only conclude that our knowledge of the part played by sodium and chlorine in the constant redistribution of the materials of the earth’s crust is still lamentably imperfect, and that quantitative deductions drawn from it must be regarded as being purely provisional.
[Pg 76]
The maximum thickness of the sedimentary rocks—Rate of deposit—Uniformitarian basis of the method as usually applied—Arguments against the validity of this assumption—Present a period of land extension and continental elevation—Present geological rates not true standards—Ideal sedimentation curve and rates of deposit—Difficulties in the application of the data—Summary of time estimates based on this method—The hour-glass method applied to the accumulation of sediments and of calcium carbonate.
The most familiar method of estimating geological time is based upon the total observed thickness of stratified rocks and the rate at which they may have been deposited. Our knowledge of the so-called maximum thickness of each of the stratigraphical systems has been carefully summarised by Sollas, to whom we owe the following table.
Maximum Thickness of the Geological Systems.
| FEET | ||
| Recent and Pleistocene | 4,000 | |
| Pliocene | 13,000 | |
| Miocene | 14,000 | |
| Oligocene | 12,000 | |
| Eocene | 20,000 | |
| 63,000 feet. [Pg 77] | ||
| Cretaceous | 44,000 | |
| Jurassic | 8,000 | |
| Triassic | 17,000 | |
| 69,000 ” | ||
| Permian | 12,000 | |
| Carboniferous | 29,000 | |
| Devonian | 22,000 | |
| 63,000 ” | ||
| Silurian | 15,000 | |
| Ordivician | 17,000 | |
| Cambrian | 26,000 | |
| 58,000 ” | ||
| Algonkian | 82,000 | 82,000 ” |
| Archean | ? | ? |
| Total | 335,000 feet. |
The rate of deposit of sediment on the ocean floor is a factor over which there has been much dispute. The rate varies between very wide limits, according to distance from the shore and from the mouths of great and active rivers. What is generally sought is to give an estimate which will correspond to the maximum thickness of accumulated material, i.e. an estimate of the average maximum rate of deposit. The same rate is then applied in turn to the whole of the geological column in the hope that no serious error will be introduced by the assumption of uniformity. This attitude was taken up in defence against the [Pg 78] attacks of Kelvin and his followers. The tendency to invoke more active geological agencies in the past, greater floods and tidal waves, a more stupendous upheaval of mountains and more violent volcanic eruptions, did not commend itself to most geologists. Geikie stated the case very clearly in his eloquent address of 1892, when he affirmed that “the geological record furnishes a mass of evidence which no arguments drawn from other departments of Nature can explain away, and which, it seems to me, cannot be satisfactorily interpreted save with an allowance of time much beyond the narrow limits which recent physical speculation would concede.” But while the conception of greater activity in the past met with little favour, the assertion of uniformity was as far as geologists dared to go. No one suggested that we might be living in an age of more than average activity. Yet there are many reasons which favour this hypothesis in preference to the alternative views.
That the average land area of the past was less than that of to-day has already been stated. According to the palæo-geographical researches of Mr. C. Schuchert, the mean area of North America since Cambrian times has been four-fifths of its present area. In the case of the other continents a smaller fraction would probably be more representative. Of very much greater importance is the fact that the average height of the land areas above sea-level has often been less than it is, so that the present average is excessive when viewed from the broader standpoint of geological time. The chief defect in the time estimates based on the [Pg 79] rate of sedimentation lies, according to Chamberlin, in the too full dependence on standards derived from the geological processes now in action. It is tacitly assumed that current rates are representative, or that the departure from the mean rate is not such as to involve any grave error. Joly, for example, in discussing the divergent evidence of geological processes and radioactive minerals, points out that to bring the different methods into agreement we must assume “that the rivers are now bearing to the sea about 14 times the average percentage of the past—not less than 9 times.” Then he says, “It seems quite impossible to find any explanation of such an increase.”
In the present high relief of the earth’s surface at least a partial explanation may be found. Prof. Chamberlin writes in a private communication to the author: “Because of the relatively high gradients, the wash of clastic material from the slopes and its deposition in the basins, as well as the transfer of salts to the sea, are to-day more rapid than in average times. We seem to be at, or near, one of the great extremes of intensification of the processes of solution and degradation. And so, whether conclusions are based upon degradation and clastic deposition, or upon solvent action and the accumulation of solutes in the sea, the present rates are high rates, and if these are made the basis of time estimates, the estimates are minimum ones. There are abundant evidences that periods of base-levelling have occupied a notable part of geological time. There is cogent evidence that the Archean and Proterozoic (Algonkian) terranes were reduced well towards [Pg 80] base-level in pre-Cambrian times, and that subsequently extensive base-levelling clearly seems to have intervened at repeated intervals. To me the evidence seems to support the existence of a dozen or a score of stages of peneplanation, some of which appear to have made a notable advance toward complete base-levelling.”
During those intervals, when the average continental height was low, denudation and deposition would proceed very slowly. How slowly, we have no adequate means for determining. A very careful study of drainage basins with reference to their mean elevation would be a step towards a sounder method than it has yet been possible to apply. That the departure from present rates, in past time, may have been very considerable, is indicated crudely by a very simple calculation. It has been found experimentally that the carrying power of water varies as the sixth power of its velocity. Roughly, we may say it varies as the sixth power of the mean square of the heights of the drainage basin from which it finds its way to the sea. If this were strictly true, then, if the present day contours of the land were reduced to half their value, the power of removing material would be reduced to less than one four-thousandth. Of course, other factors would begin to operate which would prevent the attainment of any variation as extreme as this. Nevertheless, our faith in the value of present standards as applied to geological time cannot but be seriously shaken. Chamberlin feels warranted in thinking “that the substitution of mean velocities of denudation, deposition, and saline accumulation, if it could be made [Pg 81] to approach the realities of the case, would have the effect of multiplying by a considerable figure the best estimates that have been made on the basis of current velocities.” Other factors tending to increase the age estimate are numerous, but comparatively insignificant beside those already briefly discussed.
In the last chapter we concluded with an estimate of the quantity of different types of sediment which are now annually deposited.
| Shales | 6300 | million | tons. |
| Sandstones | 1440 | ” | ” |
| Limestones | 1260 | ” | ” |
| 9000 | million | tons. |
If it be assumed that all the arenaceous sediments form littoral, deltaic and estuarine types, i.e. that they are concentrated over an area of say 100,000 square miles, the rate of accumulation would be one foot in 150 years, or dealing only with fresh unconsolidated sediment, of one foot in about 100 years. But these figures are obviously too high, for a great deal of sandstone comes under shallow water marine types. Fortunately, in the deltaic deposits of two important rivers human remains of recognised age are found buried; and from measurements of the thickness in each case it is known that the Nile has deposited loose sediment at one foot in 320 years, and the Po at one foot in 174 years.
We have here, as in all deltaic deposits, a mode of growth analogous to that of the glacial clays studied by De Geer. It by no means follows [Pg 82] that these rivers will continue to raise their beds at the same rates, for the main growth of deposit is not upwards, but seawards.
To get a rough idea of the rate of sedimentation, the ideal section opposite has been constructed. The continental shelves are assumed on the average to be representable as a band of 100 miles in width fringing a coastline of 100,000 miles. The average thickness of a deposit laid down according to this plan would be about 0·4 of the greatest thickness, the latter, on the scale of the diagram, having reached 400 feet. In the particular case illustrated the land is slowly sinking, and as the sea encroaches upon it the deposits gradually overlap.
If deposited over one square mile, as consolidated sediments of density 2·5, our 9000 million tons of material would form a rectangular mass 4570 feet in thickness. Being deposited over the ten million square miles of the continental shelf, the average thickness of the layer is 0·000457 feet, corresponding to a rate of deposit of one foot in 2200 years. The maximum rate is therefore one foot in 880 years. If this figure be applied to the total thickness of the geological column, then, disregarding its imperfection, the time implied would be about 300 million years.
The distribution of conglomerate, sandstone and shale is sufficiently indicated in the diagram. It is difficult to know how to distribute the argillaceous and calcareous types. They are mutually exclusive, for limestone cannot form by organic agencies in places where mechanical detritus is being deposited. They are therefore taken together, on the basis that where one is not being formed the other is. [Pg 83]

Fig. 11.
Section illustrating the formation of Sediments on the
Continental Shelf while the latter is being slowly depressed.
[Pg 84] The following table gives the rates of deposit at various distances from the shore, for the case illustrated in Fig. 11:
| Distance from Shore in Miles. |
Years for Deposit of One Foot. | |||||
|---|---|---|---|---|---|---|
| Fresh density = 1·8. |
Consolidated density = 2·5. |
|||||
| Sandstones | 0 | |||||
| 10 | 2,780 | 2,000 | ||||
| 20 | Shales | 1,670 | 1,200 | |||
| 30 | 1,120 | 880 | ||||
| 40 | 1,430 | 1,030 | ||||
| 50 | 2,280 | 1,660 | ||||
| 60 | 5,210 | 3,750[3] | ||||
| 70 | 10,420 | 7,500[4] | ||||
| 80 | 13,900 | 10,000[5] | ||||
| 90 | 20,850 | 15,000[6] | ||||
| 100 | 41,700 | 30,000 | ||||
These figures, however, have but little value, for there is no single law of deposition. The effects of ocean currents, of earth movement, and of the presence or absence of great rivers should all be considered, and they provide a problem so complex that as yet it is hopelessly beyond a general solution. Difficulties are encountered at every stage; not only are the estimated rates of doubtful value, but their application is discredited by our ignorance as to what constitutes the real maximum thickness of sediments. [Pg 85]
As already indicated, the latter difficulty is due to the tendency for successive beds to overlap while the cycle of deposition is running its course. As was admirably stated by Prof. Watts in his Presidential address to the Geological Society in 1911, deltaic deposits gradually extending seawards are more characteristic during periods when the land is being elevated relative to sea-level. On the contrary, during periods of depression estuarine conditions prevail and the beds grow landwards.
The same problem has been attacked by Sederholm from a rather different point of view. He says, “As the layers successively formed cover each other like scales or roof-tiles, no vertical section contains them all. If we mean by maximum thickness the sum of the maxima of the layers formed in successive years, it certainly measures millions of feet.” In this case the rates of deposit as ordinarily found would not, of course, be applicable.
Before proceeding farther, it may be well to review the various estimates of time which have been founded upon the geological method. The earlier geologists believed that the sediments were deposited widespread over the ocean floor and the rate of deposition was therefore taken as even less than that of denudation. Then came the Challenger expedition in 1872-5, and it became certain that the formation of all except the less important deep-sea deposits takes place almost entirely on the submarine continental shelves. A considerable modification of the estimated rates of deposition was then made necessary and the time periods were correspondingly shortened. [Pg 86]
| Date. | Author. | Maximum Thickness in feet. |
Rate of Deposit Years for one foot. |
Time in Millions of Years. |
|---|---|---|---|---|
| 1860 | Phillips | 72,000 | 1332 | 96 |
| 1869 | Huxley | 100,000 | 1000 | 100 |
| 1871 | Haughton | 177,200 | 8616 | 1526 |
| 1878 | Haughton | 177,200 | ? | 200 |
| 1883 | Winchell | — | — | 3 |
| 1889 | Croll | 12,000[7] | 6000[8] | 72 |
| 1890 | de Lapparent | 150,000 | 600 | 90 |
| 1892 | Wallace | 177,200 | 158 | 28 |
| 1892 | Geikie | 100,000 | 730-6800 | 73-680 |
| 1893 | McGee | 264,000 | 6000 | 1584 |
| 1893 | Upham | 264,000 | 316 | 100 |
| 1893 | Walcott | — | — | 45-70 |
| 1893 | Reade | 31,680[9] | 3000[10] | 95 |
| 1895 | Sollas | 164,000 | 100 | 17 |
| 1897 | Sederholm | — | — | 35-40 |
| 1899 | Geikie | — | — | 100 |
| 1900 | Sollas | 265,000 | 100 | 26·5 |
| 1908 | Joly | 265,000 | 300 | 80 |
| 1909 | Sollas | 335,800 | 100 | 80 |
Most of these estimates are little more than rough guesses. We do not know how much of the story is lost to us, or how much is hidden away. The time which has usually been regarded as expressing the geological requirements most adequately is 100 million years. The fanciful figures arrived at by Winchell, and McGee (who suggested a probable age of 6000 [Pg 87] million years) are merely illustrations of how the data could be twisted to produce impossibly extreme results. The latest estimate, due to Sollas, includes an allowance of 25·4 million years for the duration of pre-Cambrian time—the same period as that which has apparently elapsed since. A further allowance is made for unconformities, those gaps in the sequence which are unrepresented by sediment. Taking the great unconformities as probably numbering six, each being equivalent to 40,000 feet of sediment, 24 million years are added. For minor unconformities and interruptions in the record other 5 millions are granted and the total is thus brought up to 80 million years.
In an attempt to free ourselves from the difficulties with which this method is beset, we may adopt a mode of procedure similar to that followed in the last chapter. It was there assumed that the total volume of the sediments which have ever existed amounts to some 210 million cubic miles. The present annual supply of sediment when ultimately compressed and consolidated would occupy 0·83 of a cubic mile. If the present rate of accumulation were reliable, geological time would then be of the order 250 million years. A still nearer approach to the truth may be made by calculating on a uniformitarian basis how long the existing sediments, which we placed at 70 cubic miles, have taken to form. From the 8,000,000 square miles of igneous and metamorphic rocks, one cubic mile would be denuded away in 4·54 years, which implies that one cubic mile of consolidated sedimentaries [Pg 88] would be formed in about five years. The age then works out at 350 million years. Making a further correction for the slower rate of denudation of igneous rocks, this figure may perhaps be doubled. Finally, there is the correction for average rate in place of present rate, and to what extent this would increase the estimate it is impossible to say.
We may revert to the maximum thickness of the sedimentary rocks to support the estimate of the total volume of sediments which has ever existed. On the basis of the sedimentation curve, the sediments have been deposited on an area of 10 million square miles, and if laid down everywhere at their average maximum thickness, 60 miles, they would cover about 0·4 of that area. The total volume which can ever have existed, leaving unconformities out of the question, is therefore, 60 × 10,000,000 × 0·4 cubic miles, or 240 million cubic miles.
Finally, a crude estimate may be based on the amount of calcium carbonate which has accumulated in geological time. Several estimates of the volume of limestones in existence have been made, e.g.:
| Dana | 18·40 | million | cubic | miles. |
| Reade | 10·00 | ” | ” | ” |
| Van Hise | 6·25 | ” | ” | ” |
The limestones now forming make up 14% of the total sediments which collect on the continental shelves. On the land the proportion must be lower than this, because limestone is denuded at a rate well above the average. If we take limestone formations at 10%, the volume would be [Pg 89] about 7 million cubic miles, a figure not far from that of Van Hise. The calcium carbonate may now be estimated. Limestones contain on an average 75% and shales and sandstones together about 7%. The total volume, calculated at density 2·6, would therefore be in round figures 10 million cubic miles.[11] Igneous rock contains 3·43% of calcium, and if in the process of denudation all of this is dissolved and removed, the present rate of production of calcium would be equivalent to 1 cubic mile of calcium carbonate in 32 years, on the same basis as in previous calculations. The time estimate at this rate would be 320 million years.
We may now sum up our various results as follows:
| 1. Accumulation of Sodium. | MILLION YEARS. |
| (a) Uncorrected quotient Naₒ/Naᵣ | 80·8 |
| (b) Partially corrected Naₒ/Naᵣ | 90 |
| (c) Unchloridised sodium alone | 180 |
| (d) Primary sodium alone | 210-340 |
| 2. Accumulation of Sediments. | |
| (a) Maximum thickness | 300 |
| (b) Total volume which has ever existed | 250 |
| (c) Total volume now existing | 350 |
| 3. Accumulation of CaCo₃. | |
| (a) Total volume now existing | 320 |
[Pg 90] Not one of these estimates is to be regarded as final; the uncertainties are too many and too great. The whole trend of this chapter has been to show that whatever may be the true reading of the hour-glass of denudation and deposition, it ought probably to be very much higher than has been generally assumed.
[Pg 91]
Geological application—Röntgen rays—Becquerel rays—Mme. Curie and the discovery of Radium—The gold-leaf electroscope—The nature of α-, β- and γ-rays—Helium and its production from radium—The heat output of radium—Transformation of the radium atom—Disintegration theory—The half-life period of radium and its generation from uranium—The uranium, thorium and actinium families—Direct measurement of the heat output—Radioactivity independent of external influences—Pleochroic haloes—Measurement of radium in rocks and minerals.
In the limited space at our disposal, only a brief outline of the salient facts of the new science of radioactivity can be given. It is necessary to give some account of these recent discoveries because of their immediate application to the study of the earth itself. In a science like geology, the function of which is to study the consequences of energy transformations in the earth’s crust during the long ages of her gyrations through space, the recognition of sources of energy previously undreamed of is a stimulant to research which must profoundly affect the interpretations of the phenomena we observe. For our present purpose it will be necessary to review only those facts which have a bearing on the question of the earth’s age, and of its thermal history.
[Pg 92]
In 1895 Röntgen gave his first account of A New Kind of Radiation. This extraordinary radiation, now familiar to us as the X, or Röntgen rays, revealed itself by its remarkable property of passing through objects which are entirely opaque to ordinary light. It was found, for example, that a photographic plate was not protected from its influence by being wrapped in black paper, but was affected just as though it had been exposed.
This discovery awakened the deepest interest amongst all scientific workers, and the idea arose that phosphorescence and the Röntgen rays might in some way be vitally connected. Certain substances, after being exposed to sunlight, are able to shine in the dark with a phosphorescent light, and it was this radiation which was examined from the new point of view. Working on these lines, with uranium salts, Becquerel found, almost by accident, that they gave out rays which were capable of penetrating black paper and making themselves evident by their effect upon a photographic plate wrapped within it. This observation in 1896 marks the commencement of the harvest of wonderful discoveries which have revolutionised our conception of the structure of matter. [Pg 93]
Fig. 12.
Taken in the dark by its own rays.

Fig. 13.
Taken in ordinary daylight.
PHOTOGRAPHS OF PITCHBLENDE.
The emission of rays was found to be an inherent property of uranium and entirely independent of the phosphorescence exhibited by certain of its compounds. Physical and chemical changes made no difference; the radioactive properties evidently belonged to the atom itself, for they could be detected under whatever conditions uranium happened to be present. Mme. Curie at once began an exhaustive examination of all the other known elements, and came to the conclusion that thorium alone possessed radioactive properties similar to those of uranium. In the course of her researches, she found that uranium-bearing minerals such as pitchblende (uraninite) were far more active in their emission of rays than could be accounted for by the uranium present. Systematically following up so significant a fact, she traced the activity to the bismuth and barium separated from these minerals. In 1898 two new elements were announced. One, associated with bismuth, was called Polonium; the other, found in minute quantities with the barium, was appropriately named Radium.
The spontaneous radiations of these elements, collectively known as the Becquerel rays, were soon distinguished from the Röntgen rays by their greater complexity. Their analysis is largely due to the scientific genius of Rutherford, who has classed them into three well-marked types, the Alpha or α-, Beta or β-, and Gamma or γ-rays. The instrument chiefly employed in their detection is the gold-leaf electroscope, which not only is simple in construction, but is capable of extreme sensitiveness. Under ordinary conditions the air is a good insulator, and a charged electroscope loses its charge only very slowly. However, if radium is brought near the instrument, the discharge proceeds much more rapidly, the effect of the Becquerel rays being to ionise the air and so render it conductive to electricity. In the electroscope this effect can be readily observed and accurately measured, for the leaf falls at a regular rate, which depends on the intensity of the [Pg 94] radiation. The sensitiveness of this method so far surpasses that of the most delicately adjusted balance that by its means a quantity of radium can be detected which would need to be multiplied thousands of millions of times before the balance would turn to its weight.
It is easy—by interposing between the electroscope and the radium a gradually increasing thickness of suitable material such as thin sheet aluminium—to establish the existence of three kinds of rays which differ greatly in their power of penetrating matter. The γ-rays are, on the average, about a hundred times more penetrating than the β-rays, and these in turn are equally the superior of the α-rays. In each case the penetration in different materials depends approximately on their density. The three types can also be distinguished by their behaviour in a strong magnetic field. Giesel showed that the β-rays were deflected in such a way as to indicate that they consisted of particles carrying a negative charge of electricity. The α-rays were much more difficult to deflect, but Rutherford successfully turned them from their path by applying a very intense field, and from the direction of their deflection he concluded that they consisted of positively charged particles. The γ-rays have, however, resisted all attempts made to alter their course. Their real nature is not yet beyond dispute, and from the great similarity between their properties and those of the Röntgen rays they are generally considered to be, like the latter, either electro-magnetic pulses in the ether, or uncharged corpuscles of a kind not yet understood. [Pg 95]
When an electric discharge is passed through a high-vacuum tube, rays are emitted from the cathode which, on reaching the walls of the tube, produce a yellowish-green phosphorescence. The cathode rays are identical with β-rays in every essential respect, and both are proved to be tiny, negatively charged particles, called electrons. The velocities of both cathode and β-particles are inconceivably high; in the latter case even approaching that of light. Now Röntgen rays, whatever may be their true nature, are set up by the sudden stoppage of electrons when they impinge upon the anti-cathode of a vacuum tube. In the same way it is supposed that the β-rays, by their sudden expulsion, give rise at the same moment to γ-rays. It is a significant fact in this connection that the β- and γ-rays are always associated together, whereas the α-ray may be quite independent.
The story of the brilliant researches which culminated in the measurement of the mass of an electron, and of the charge it carries, is one of the most fascinating in the annals of science. But here we must be content with the bare statement that the electron has only ¹/₁₇₀₀ of the mass of a hydrogen atom. The application of similar methods to the α-particle has demonstrated that it is of atomic dimensions, and consists either of a hydrogen atom with a single ionic charge, or of a helium atom with twice that charge. Other evidence decides conclusively in favour of the second alternative.
Of all the elements, helium has probably the most interesting history. It was first discovered in the sun during the eclipse of 1868, but it [Pg 96] was not until 1895 that it was detected in terrestrial materials. In that year Sir W. Ramsay identified it in uranium- and thorium-bearing minerals by means of its highly characteristic spectrum. Even then, before radioactivity had been recognised, the association of helium with these elements had forced itself upon his notice. In 1902 the genetic relationship between helium and certain of the radioactive elements was predicted by Rutherford and Soddy. During the following year Ramsay and Soddy working in conjunction sought for, and actually observed, its generation from radium. Using a radium preparation from which helium was at first entirely absent they were able to prove, with the aid of the spectroscope, that in the course of a few months sufficient of the gas had been generated to be identified. This important discovery has received abundant confirmation during the last decade, and to-day we even know the number of helium atoms which a gram of radium emits in a given time. Moreover, the production of helium has been demonstrated from uranium, thorium and actinium, and from most of the other radioactive elements which are characterised by the emission of α-rays. The rate of generation of helium will be considered in more detail in Chapter X, where it finds an important application.
An observation of fundamental importance was made by Curie and Laborde in 1903 when they discovered that radium is able to maintain a temperature above that of its environment. The source of this emission of heat lies in the kinetic energy of the Becquerel rays. Projected [Pg 97] from radium, they are sooner or later completely absorbed by the matter through which they penetrate. Simultaneously, their energy is given up to the molecules with which they collide, and it therefore reappears as heat. By far the greater proportion—over 85%—of the energy is carried by the relatively heavy α-particles which move with velocities nearly one-tenth that of light. Knowing their masses and velocities, and the number liberated per hour from a gram of radium, it is a simple dynamical problem to calculate their total kinetic energy. Expressing this quantity in heat units, it is found to be equivalent to an hourly production of 113 calories. (Ap. A, p. 177.) As we shall see later, this figure is in close agreement with the results of direct measurements.
The most remarkable feature about these phenomena is that they appear to continue unceasingly. Year after year the spontaneous production of helium goes on, accompanied by a steady evolution of heat. But what of the atoms that remain? They evidently cannot continue to be the same element after having given up part of their energy and expelled from themselves the material particles of the α- and β-rays. A close examination reveals the genesis of a new element—a gaseous substance known as radium emanation. Its atom represents the residual fraction of the radium atom after the separation of a single α-particle. The emanation is highly unstable; so rapidly does it give up energy and liberate helium that its original intense activity decreases by half every four days. In its place arises another product, radium A, of [Pg 98] still more transitory existence. A long succession of similar transmutations has been traced, each accompanied by a setting free of energy.
In 1902 Rutherford and Soddy advanced the theory of the disintegration of the radioactive atoms. The radium atom is essentially unstable and ultimately breaks up, explosively emitting α- and β-particles. Each atom has a certain expectation of life which distinguishes it from the atoms of other radioactive substances; but such is the distribution of instability that a constant proportion of the total number of atoms present is always breaking up. Out of a million atoms of radium a definite number disintegrates each second, and the proportion has been proved to be invariable in every case it has been possible to test. From element to element it is this proportion, and therefore the average promise of life, which varies. An equilibrium between formation and transformation is gradually attained, and once it is established a given quantity of radium produces as many atoms of emanation as the emanation produces of radium A, and so on. When in equilibrium the longer-lived radio-elements are present in greater quantity than those of shorter lives in order to compensate for their comparatively slow rate of decay, and so to enable them to keep pace with their more rapidly changing associates.
We may now calculate the average life period of radium. The volume of emanation in equilibrium with one gram of radium has been measured and found to be O·58 cubic millimetres—less than the volume of a pin’s head. The rate of change into radium A also lends itself to direct [Pg 99] measurement, and from these two quantities it can be shown (see Ap. A, p. 179) that in one year ¹/₂₆₀₀ of the radium originally in existence must undergo transformation. The same result can be arrived at more directly. We know the number of atoms in a gram of radium and also the number of helium atoms expelled in the course of a year. The proportion which has suffered disruption follows at once, and is found to be ¹/₂₅₀₀.
From evidence of this kind it is concluded that after about 1850 years only half of any given quantity of radium then maintains its identity as that element. Now it is clear that radium must have a progenitor by which it is formed as fast as it decays, or otherwise there could now, after the lapse of millions of years, be none in existence.
That progenitor is undoubtedly uranium—an element which disintegrates so slowly that its half-life period is three million times as long as that of radium. This association is revealed by the study of radioactive minerals. For every gram of uranium there exists in equilibrium with it, 3·4 × 10⁻⁷ grams of radium. The constancy of this proportion points to a genetic connection and admits of no other interpretation. The generation of radium has been experimentally verified by Soddy, who observed it after a number of years in a uranium preparation originally quite free from it. In practice this work is complicated, and the desired effects are retarded, by the intermediate production of another radio-element, ionium, which has an average life many times longer than that of radium. The time taken to establish equilibrium is therefore very long. The primary minerals from which the [Pg 100] equilibrium ratio of radium to uranium is deduced, have existed for hundreds of millions of years and so far as they have remained unaltered by percolating waters they afford a safe guide. But in secondary minerals, like autunite, the age of which is often to be reckoned only in thousands of years, the equilibrium stage has not always been reached, and the ratio between the two elements is consequently somewhat lower than that found in the older minerals.
In the accompanying diagram the complete family of uranium is summarised together with the half-life period of each member and the rays it emits. Another series of elements related in the same way is derived from thorium which is a parent of even longer life than uranium. As to the final products of transformation, which, being stable, ought to survive and in the course of time to accumulate, helium is the only one which has been directly observed. That lead is the end product of the uranium series is most probable, as we shall see in a later chapter. Of the stable element to which the thorium series ultimately leads we are still in ignorance. Neither lead nor bismuth, which suggest themselves by their appropriate atomic weights, satisfy the conditions, for their presence in a mineral bears no relation to the quantity of thorium (see p. 190).
The actinium group of radio-elements calls for little notice here. Boltwood finds a fixed ratio between actinium and uranium in minerals, and the former would therefore appear to be a descendant of uranium just as radium is. Actinium, however, does not fall in the main line of [Pg 101] descent. There is some probability that it marks the beginning of a collateral series, also springing from uranium. The proportion of uranium which disintegrates in this direction rather than towards radium can only be very small.[12] It is possible that uranium atoms, or those of one of its products, may be of two different types, according to their internal structure, and that on disruption each may give rise to an independent chain of elements.
The heat output of radium and its relation to the energy of the α-particle has already been mentioned. The first direct measurement of the heat generated was made by Curie and Laborde. Using suitably constructed calorimeters, they were able to demonstrate a regular evolution of about 100 calories in an hour from a gram of radium—heat enough to raise the temperature of an equal mass of water from freezing to boiling-point. The radium in these experiments was in radioactive equilibrium with the emanation and radium A, B and C, so that altogether four α-particles were disengaged for each atom of radium which transformed. The approximate correctness of this first estimate will be seen from the following table, in which the best results up to date are quoted:
| 1903. | Curie and Laborde | 100 | |
| 1904. | Rutherford and Barnes | 110 | gram calories emitted per hour per gram of radium |
| 1904. | Runge and Precht | 105 | |
| 1908. | v. Schweidler and Hess | 118 | |
| 1909. | Duane | 120 | |
| 1911. | Duane | 117 | |
| 1912. | St. Meyer and Hess | 132 |
[Pg 102] The correspondence of these results with the theoretical figure, 113 calories, which represents about 85% of the energy,[13] is eminently satisfactory (see Ap. A, p. 177).
The disintegration theory affords a simple explanation of the origin of this energy. From the new point of view the atom is no longer indivisible, no longer the ultimate foundation of all matter; rather is it regarded as a tiny universe of electrons, a whirling assemblage of charged particles. These minute entities move with very high velocities within the atom itself. In general the energy is locked up, but in the radioactive elements the atomic structure is unstable and from time to time some of the energy can escape. The intrinsic energy of the atom itself is the real source of the heat liberated by radium, and the apparent permanence of its output is simply an expression of the immense supplies of energy which an atom can store within itself.
The essential features of radioactivity are two: the spontaneous emission of Becquerel rays, and the atomic character of the change. The radiation from a radioactive substance is proportional to the quantity of the substance and to nothing else. Temperature, being a function of molecular movement, is without influence upon atomic transformation. Experiments made at temperatures ranging between that of liquid air and 1600° C. show that the same law of disintegration holds throughout. Similarly, the effect of very high pressures indicates that these inter-atomic processes are quite independent of any external influence. [Pg 103] Whether in solution or in chemical combination, whether taking part in energetic chemical reactions or suffering bombardment by its own radiations, the atom continues to obey the law which determines its life period, unmoved by any experiences through which we can oblige it to pass. The radioactive properties are simply superimposed upon the chemical and other properties of the substance, and as far as laboratory conditions are concerned, they are entirely spontaneous and can in no way be affected.
Whether or not radioactivity is a general property of atomic matter cannot yet be announced. Potassium and rubidium are feebly radioactive—a thousand times less than uranium—and emit β-rays. It has been thought that some other metals may share in a minor degree the same attributes, but up to the present no reliable results have been forthcoming.
In its geological applications radioactivity possesses a great interest. The most important bearing of all lies in the part it plays as a source of terrestrial heat. Radium is widespread amongst the surface materials of the earth’s crust. Rocks and soils, and natural waters all contain radium as a constituent, widely diffused but always present. In the following two chapters the influence of radio-thermal action in the earth and sun, and on speculations as to their ages, will be dealt with. Before passing on to these cosmic aspects of the subject, a brief account may be given of the way in which minute quantities of radium can be detected and accurately measured—quantities which in rocks average little more than a billionth of a gram in each gram of the rock. [Pg 104]
Radium itself is not directly dealt with; it is the emanation in equilibrium with it which is actually measured. The emanation is separated from a suitable amount of rock, mixed with air, and introduced into an electroscope. After about three hours, the emanation has generated radium A, B and C in their equilibrium amounts, and the emission of three sets of α-particles then steadily proceeds. In the electroscope, the rate of fall of the changed leaf depends directly on the conductivity of the air or on the number of ions which are utilised as carriers of electricity. Now each α-particle, in virtue of its enormous energy, is capable of producing in air about 100,000 ions, and the total number present is therefore determined almost wholly by the number of α-particles. The β- and γ-rays certainly have an ionising effect, but so small that in this connection it may be ignored. To be quite sure that all the ions are taking an active part in the discharge and collapse of the leaf, it is only necessary to observe the latter before its divergence from the central support falls to less than, say, 10°. The rate of collapse can easily be gauged by observing the leaf through a microscope with a divided scale attached to the eyepiece, the time during which the leaf passes over a given number of divisions being taken with a stop watch. The rate of fall is proportional to the conductivity, which depends in turn on the number of α-particles expelled, and therefore on the quantity of emanation present in the electroscope; this quantity, provided that it was initially in equilibrium with the radium in the original rock, determines the amount [Pg 105] of that element which was present. A constant can be determined once for all by experiment with known materials, which directly connects the rate of fall in scale divisions per hour with the equivalent amount of radium or uranium in grams. Some idea of the delicacy of the method may be gained from a particular example. In the apparatus devised by Prof. Strutt and used both in his own investigations and those of the present writer, a leakage of one scale division per hour corresponds to 9 × 10⁻¹³ grams of radium.
To use this apparatus it is necessary to bring the rock into solution before the emanation can be extracted. The solution is stored up for a few weeks until the equilibrium amount has been generated and the flask A containing it is then attached to the water condenser B (Fig. 14). The emanation can be expelled by vigorous boiling, and greatly diluted with air, it passes out into the gas-holder C. The stream condenses in B and drops back. At the end of an hour the cooling water is run out of B and the steam then drives all the air charged with emanation into C. In order that none should be allowed to escape back, the connection at D is closed. Meanwhile the electroscope F has been exhausted and the emanation is made to pass into it through the tap at E, the gas-holder being slowly filled with water. After three hours the measurements may then be made. [Pg 106]
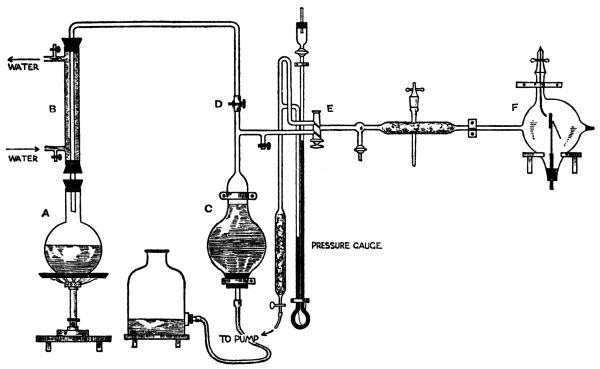
Fig. 14.
Apparatus for estimating Radium by its Emanation.
[Pg 107] Joly has recently varied this method in order to save the labour attending the process of getting a rock into clear solution. The mixture of finely-powdered rock and fusion mixture is heated in an electric tube-furnace, and the expelled gases containing the emanation are drawn straight from the furnace. Carbon-dioxide is absorbed by soda-lime, and the remaining gas is collected and finally passed into the electroscope as before. The absence or presence of thorium makes no appreciable difference in these measurements, for the life of its emanation is many thousands of times less than that of radium emanation. Joly has utilised this fact in his solution-method for determining minute quantities of thorium in rocks and minerals—a method similar in principle to that described above for radium.
The presence of radioactive elements in rocks sometimes reveals itself in a most beautiful way. In mica, cordierite, hornblende, chlorite, tourmaline, and a few other minerals, small circular spots known as pleochroic haloes are sometimes seen. A tiny crystal can generally be detected in the centre, usually of zircon, but sometimes of apatite, epidote, rutile, or sphene. Until quite lately the nature and origin of these intensely pleochroic spots was entirely unknown. Joly showed in 1907 that they are due to the radioactivity of tiny inclusions around which they spread spherically outwards. The α-rays discharged from the minute central crystals are able to ionise the biotite (or other mineral) and this effect, to which the colouring action is due, spreads just as far as the α-particle can penetrate. The range of the different α-particles from the uranium family is given overleaf, both for air and for biotite. [Pg 108]
| Source of α-Particle. | Range in Millimetres. | |
|---|---|---|
| In Air. | In Biotite. | |
| Radium C | 70·6 | 0·033 |
| Radium A | 48·3 | 0·023 |
| Ra. Emanation | 42·3 | 0·020 |
| Radium F | 38·6 | 0·018 |
| Radium | 35·4 | 0·017 |
| Ionium | 28·0 | 0·013 |
| Uranium 1 and 2 | 27·0 | 0·013 |
Now Bragg has shown that the ionising effect of an α-particle is greatest just before it comes to rest. We should therefore expect to find a number of intensely coloured spherical shells existing around the central crystal and corresponding in each case to one or other of the range limits here given. In section, and magnified 700 diameters, the appearance of the shells would be as in the following diagram.
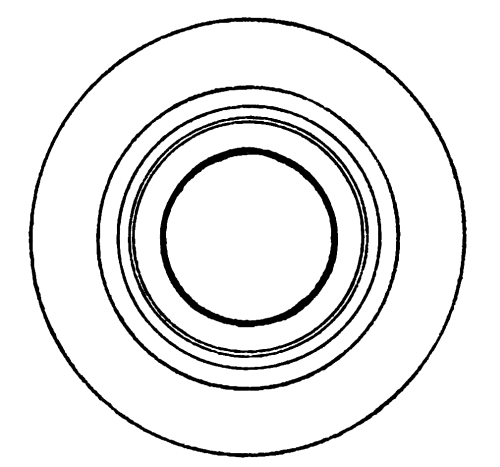
Fig. 15.
Pleochroic Haloes in Biotite due to Uranium and its transformation products.
Magnified 700 diameters.
[Pg 109] The dimensions of the actual haloes are exactly in agreement with the distances to which the different α-particles can travel, and occasionally the successive spheres of ionisation and colouring are beautifully developed. In the biotite of Co. Carlow granite, Joly has found several very perfect examples, and by his courtesy four of his photographs are reproduced in the Frontispiece. Not only is the proof that the haloes are due to α-rays conclusive beyond question, but the correctness of Bragg’s laws from which the range in biotite is calculated are established in an unexpected way. Thorium haloes are also found (as in Fig. 2, upper left-hand part of the field), and these again have diameters in perfect accord with the demands of theory.
A point of interest in the history of igneous rocks is that the haloes disappear when the rock is heated. The presence of well-formed haloes, therefore, implies the maintenance for very long periods of fairly uniform conditions. So slowly do these haloes form that it may become possible, when they have been further investigated, to give a rough minimum estimate of the age of the minerals in which they occur. The radioactivity of a zircon is much more intense than that of the rock enclosing it, but the smallness of the quantities involved is such that two or three weeks may elapse between the expulsion of successive α-particles or helium atoms. By the accumulative effects of millions of these atoms a spherical halo, faint in youth, but assuming a deeper tint with age, is gradually produced.
[Pg 110]
The sun’s heat and the conservation of energy—Mayer’s meteoric hypothesis—Helmholtz and contraction under gravity—The earth’s dependency upon solar radiation—The work of Kelvin and Ritter—The insufficient contributions of atomic disintegration—Arrhenius’ view of the importance of molecular energy—Cyclic development of the universe.
Before the doctrine of the conservation of energy was established, the steady radiation of solar light and heat was not, in its quantitative aspect, a phenomenon to be wondered at. Regarding the sun merely as a gigantic fire, philosophers such as Leibnitz and Kant were satisfied that the intense emission of energy was sustained simply by combustion. As soon as the chemistry of combustibles came to be studied, it was at once evident that the energy derived from burning alone would be hopelessly insufficient. If the sun had been originally a colossal mass of the most powerful explosives known to us, then, under the most favourable conditions for maintaining the present output of heat, all would have been at an end within a few thousand years. The total amount of available energy would not have sufficed even for the historical period—a period which is merely a ripple in the vast ocean of geological time.
[Pg 111] What, then, can be the source of the energy which for millions of years has enabled the sun to bathe the earth in a welcome glow of light and heat? How is the loss to be accounted for? For how long can the sun continue to radiate its energy without becoming perceptibly colder? These critical questions must have appealed to Mayer with some force when he recognised the truth that energy could neither be created nor destroyed. For the first time a sound explanation became an imperative demand. Mayer realised that in the collision and friction of bodies, heat energy is acquired in strict equivalence to the energy of motion which has apparently disappeared. He saw that the mechanical generation of heat would be of vastly greater importance in cosmic evolution than the limited possibilities of combustion. A piece of coal falling into the sun from infinite space would yield, by the stoppage of its motion, six thousand times as much heat as it could provide by burning.
Applying these principles, Mayer thought that the sun’s heat might be traced to the kinetic energy of swarms of meteorites. These bodies falling into the sun with enormous velocities, would be competent, if only the supply were ample, to generate the heat annually required. Kelvin at first also advocated this view, but he was soon compelled to relinquish it in favour of another explanation less at variance with known facts. The observations of astronomers were all against there being a circulation and influx of meteorites to the extent required by the sun if income and expenditure of heat were to balance. Comets would [Pg 112] suffer resistance in their passage round the sun. The rotational velocity of the sun would be constantly impeded, and probably, far back in the past, it would have been brought to a standstill. The sun’s mass would be appreciably increased every year, and an immediate effect of this would be to hurry up the earth in its orbit, so that each year would be notably shorter than the preceding one. Happily for the stability of the solar system, there is no evidence for an infalling of meteorites on the scale first contemplated.
In 1856, Helmholtz, another early worker in the domain of energy, found a more satisfactory escape from the dilemma. Instead of looking outside the sun for the origin of the heat supply, he sought for an internal source, and found one—certainly a more efficient substitute—in the contraction of the sun’s diameter under its own strong gravitation. Knowing the amount of heat annually radiated, it is easy to calculate that a shrinkage of 1000 feet would make up the loss for five years. The decrease in the apparent diameter would, at this rate, never become detectable in human experience. Helmholtz imagined a time when the sun existed as a nebula spreading far out into space. As it slowly cooled and contracted, the mechanical work of shrinkage would reappear as heat. Assuming the present sun to be a globe of uniform density, Helmholtz calculated that its past history must have been restricted to about 20 million years.
The annual output of heat had been determined by Pouillet, and his result, which was too low, was used by Helmholtz in this estimate. The [Pg 113] solar constant of radiation is measured by the heat in calories, which would be absorbed in one minute by a surface of one square centimetre placed outside the earth’s atmosphere at right angles to the sun’s rays. Allowance is made in this way for the absorptive effects of gases and of the load of dust held by the lower strata of the atmosphere. Pouillet’s value in these units was 1·76 and the results of subsequent experiments, made up to 1905, varied between his figure and 4·1. This was unsatisfactory, and, under the auspices of the Smithsonian Institution, work has recently been done to clear up the discrepancy. The constant is now known with some certainty to be 1·95. Using this figure the duration of the sun’s heat would, according to Helmholtz, be limited to 18 million years.
The surface temperature of the earth can owe but little to its internal energy. Taking the temperature gradient at 1° C. in 32 metres, and the average conductivity of rock as 0·004, the temperature maintained by this flow of heat alone would reach only 34° Absolute (239° C. below zero). It is evident then that the genial warmth of the greater part of the earth’s surface is maintained almost wholly by the absorption of solar radiation. For this reason the active life of our planet is intimately bound up with that of the sun, and any age limit assigned to the latter becomes a still more embarrassing restriction in its application to the earth.
We may now return to our discussion of the sun’s vast expenditure of energy, armed with data worthy of confident acceptance, and with the [Pg 114] knowledge that, for at least as long as the earth has been a habitable globe, so long has the sun emitted its life-giving rays at a rate not very different from that of the present. The evidence of geology is clear on this point. The geographical distribution of plants and coral reefs in past ages betrays no sign of a steadily cooling sun. In some of the oldest sedimentary rocks which are known, the imprints of raindrops have been found, and the size and force of the latter were evidently not very different from those which fall to-day by the shores of seas and lakes. The intensity of climatic forces has remained, on the average, unchanged.
Kelvin somewhat mitigated the consequences of Helmholtz’s extreme view in his later treatment of the problem. Helmholtz had assumed a sun of uniform density; but Kelvin pointed out that, as the density probably increases enormously towards the centre, the amount of heat which has been already available may have been very much greater than that previously calculated. Kelvin’s cautious spirit was not shared by his contemporaries, who readily accepted the smaller estimates. The more daring investigations of Ritter, and of other physicists who followed his lead, did not support any period which exceeded 12 million years. Ritter showed that as the sun contracted from the nebulous state its temperature would at first begin to rise. Not only would contraction supply the energy necessary to sustain radiation, but an even greater quantity of energy would be available for heating purposes. An [Pg 115] interesting summary of Ritter’s work will be found in the second of the fascinating little volumes by Arrhenius on The Life of the Universe.
Geologists found no consolation in these speculative studies, and even from Kelvin’s more liberal allowance of time, an element of embarrassment was not absent. While an annual shrinkage of the sun’s diameter by 200 feet would suffice for the present, yet, unless at some time the sun’s temperature begins to fall, it is not clear why shrinkage should continue. Cooling is none the less certain because it is temporarily delayed, nor because its rate is for a time diminished. Increasing density would gradually put an end to effective contraction, and the sun would then cool as a white-hot ball would do—its capacity for replenishing its losses having been exhausted for ever. If this were all, then, in the course of a few more million years, an icy death would overtake the earth. In the last gleams of the fading solar twilight our planet would disappear—a barren and frozen world.
More recent views lead to a less pessimistic outlook, and the twilight of the sun, though ultimately inevitable, is removed to an indeterminately remote future. Gravitation is manifestly an insufficient cause to maintain the sun’s heat for the periods required. We need a supply not for less than a dozen million years, nor even for the 100 million years which would have satisfied geologists a decade ago. Some source a hundred times as fruitful as contraction under gravity is required. Happily there is no longer any need to regard the sun as a serious difficulty, for there are locked within its atoms and [Pg 116] molecules stores of potential energy capable of fulfilling every terrestrial requirement.
The importance of radio-thermal action in the sun was pointed out by Rutherford and Soddy in 1903. A month or two later, W. E. Wilson made a calculation of the amount of radium which, if distributed throughout the sun, would entirely explain the evolution of its radiant energy. He found that 2·5 parts of radium in every million of the sun’s mass would be necessary. In uranium the equilibrium amount of radium is only 0·34 part in a million, so that, even if the sun consisted entirely of uranium and its disintegration products, the heat generated would account for only one-seventh of the total expenditure. Wilson used in his calculation a solar constant of three calories per minute, which is certainly too high. His result must also be modified to allow for the heating effects of the other radioactive bodies. According to the most recent determinations, a million grams of uranium would give out 77 calories per hour, and in the same time a million grams of thorium would give out one-third as much, each element being in radioactive equilibrium. The energy from the sun when divided throughout its mass averages 300 calories per hour per cubic metre. To sustain this steady evolution of heat four million grams of uranium would be required. The average weight of a cubic metre of solar matter is only 1·44 million grams, so that by no possibility could more than one-third of the sun’s heat be accounted for by radioactivity.
That radioactive bodies do exist in the sun can admit of little doubt. [Pg 117] Helium was first known as a solar element and its abundance suggested the presence of those radioactive elements from which it could have been generated. Direct spectroscopic evidence has not yet revealed any traces of radium, and, indeed, so minute are the quantities involved that until recently there was little hope of detecting it in this way. Uranium lines have now been found in the sun’s spectrum, and unless the laws of radioactivity are totally different at the temperature of the sun, we may safely assume that uranium exists in equilibrium with its associated elements. The sun’s radiation is destitute of the Becquerel rays, but this in no way denies their emission from solar matter. Before reaching the earth, the rays would be obliged to pass through both solar and terrestrial atmospheres, and the latter alone would be more than sufficient to absorb them completely.
In the materials of the earth’s crust, uranium only averages about one part in 150,000, and thorium one part in 50,000. If the proportions of these elements which enter into the constitution of the sun are of the same order, their contributions to the sun’s energy can only be very small. The importance of radio-thermal phenomena is not felt until cooling has progressed to a more advanced stage, as exemplified by the earth, when the heat lost is balanced against that set free by atomic disintegration. We must therefore find some other means of escape from the embarrassment of a rapidly cooling sun.
Arrhenius has made a bold attempt in this direction. He calls to his aid a universal law, first enunciated by Le Chatelier, which may be [Pg 118] stated as follows: If a system in equilibrium is subjected to external influences which disturb its equilibrium, then the internal reaction within the system will be such as to oppose the external influences, i.e. the normal effects of the latter will be partially overcome. This general statement may be illustrated by the particular example to which the chief appeal is made. In the chemical changes which constitute combustion, heat is evolved, and the reaction is said to be exothermic. But the heat so liberated tends all the while to prevent the reaction from proceeding, and were an external source of heat of sufficient intensity applied so as to raise the temperature, the compounds previously formed would be again separated into their constituent elements. The liberated elements possess a greater quantity of intrinsic energy than when they are united to form a compound. That is to say, not all the heat supplied is able to exercise its normal effect of raising temperature; the internal reaction is responsible for the withdrawal and absorption of part of the energy. If now still more heat is applied the same opposing tendency continues, and the elements will again combine, this time forming endothermic compounds characterised by further absorption of heat, and consequently by higher intrinsic energy than that which the free elements possessed. The system shows a conservative disinclination to be made hotter, and as more and more heat is supplied, enormous quantities of energy are accumulated in the recesses of the molecule itself. In the case of water, the general tendency is well illustrated. Ice at the freezing [Pg 119] point absorbs 80 calories and becomes water at the same temperature. Water at boiling-point absorbs 540 calories and becomes steam. This in turn, when raised to about 3000° C., dissociates into hydrogen and oxygen, the absorption of energy being 3800 calories. Although laboratory conditions do not allow us to experiment further, there are no grounds on which to suggest that this is the end of the process.
While matter thus opposes an increasing temperature by its internal reactions, it nevertheless resists the reverse change quite as actively, and for the same reason. If an intensely energetic exothermic compound be allowed to cool, it will strenuously refuse to do so at numerous stages. The energy previously gained is emitted steadily or explosively according to the thermal environment.
In discussing the constitution of the sun and the source of its powerful radiation, this Law of Reaction, as it is called, finds a pertinent application. With its help Arrhenius has pointed out the path which appears to lead us safely out of the difficulty in which we were left by Helmholtz. The chromosphere which, disregarding the mysterious corona, constitutes the outermost strata of the solar atmosphere, is largely composed of free elements at a temperature of 6000°-7000° C. Lower down in the photosphere, 9000° C. is probably attained. Temperature and pressure both increase enormously with depth, and indeed, the average solar temperature has been estimated at a thousand times that of the chromosphere. Under these conditions it [Pg 120] seems reasonable to suppose that the sun’s interior is characterised by compounds charged with a high concentration of energy. Brought by convection currents towards the surface such highly explosive compounds would dissociate with expansion and an immense evolution of heat. It may be to explosions of this sort that the prominences are due. These violently projected gaseous tongues are shot out with velocities which sometimes reach a thousand times that of the swiftest rifle bullet. Since energy is proportional to the square of the velocity, it would appear from this that solar energies are at least a million times greater than those of our most powerful explosives. It has already been stated that if the life of the sun depended solely on the latter it would endure for only a few thousand years. As it is, the energy seems amply sufficient to last a million times as long. Here then, furnishing a regular and sufficient income from within, we have found an almost inexhaustible source of heat, which is competent to maintain the sun’s present expenditure for inconceivably long ages, as most probably it already has done in the past.
True, we are in the face of a new difficulty. Whence arose this absorption and concentration of energy in the first place? It is evident that once extinct, our sun could not be re-awakened to the warmth of its former activity merely by collision. Gravitational energy alone affords no escape from the ultimate Wärmetod, the thermal extinction towards which the universe would appear to be tending. If the development of the universe be everywhere toward the equalisation of [Pg 121] temperature implied by the laws of thermo-dynamics, the question arises—Why, in the abundance of past time, has this melancholy state not already overtaken us? Either we must believe in a definite beginning, in the creation of a universe furiously ablaze with energy, or else we must assume that the phenomena which we have studied simply reflect our limited experience. Toward the latter alternative we readily incline, the more so because of the hint it affords of cyclic processes in the scheme of Nature. Not only is energy being diffused; somewhere, our hazy conception tells us, energy is being elevated and stored up. With profound insight, Spencer pointed out in 1864 that it is to the attenuated nebulæ that we should look for the absorption and concentration of energy. In the universe nothing is lost, and perhaps its perfect mechanism is the solitary and only possible example of perpetual motion. In its cyclic development we may find the secret of its eternity and discover that the dismal theory of thermal extinction is, after all, but a limited truth.
[Pg 122]
Temperature gradients, conductivity, and the rate at which the earth loses energy—Kelvin’s attempt to estimate the age of the earth—King’s treatment of the problem—Estimates by Becker and Suzuki—The distribution of radium in rocks—The thermal equilibrium of the earth—Concentration of radium towards the surface—Constitution of the earth.
As the earth’s crust is penetrated by bore-holes, tunnels and mines, a steady increase of temperature with depth is encountered. The rate of increase varies greatly from place to place as the following records show:
| Locality. | Depth. | Temp. Gradient. | ||||
|---|---|---|---|---|---|---|
| Anzin, France | — | 1° C. | in | 15·3 | metres | |
| Wigan | 750 | metres | 1° C. | “ | 30·0 | “ |
| Sperenberg | 1700 | ” | 1° C. | “ | 36·5 | “ |
| Mt. Cenis Tunnel | 1600 | ” | 1° C. | “ | 43·0 | “ |
| Minas Geraes, Brazil | — | 1° C. | “ | 86·0 | “ | |
| Calumet, Michigan | 1430 | ” | 1° C. | “ | 12·20 | “ |
Such a variation would naturally be expected when the chemical reactions of weathering, cementation and metamorphism are remembered, and the unequal distribution of radioactive elements in the earth’s crust. The solution of silicates by ground waters takes place with liberation of heat, and it has been estimated that 120 calories are [Pg 123] released when one gram of rock is decomposed by weathering. The processes of metamorphism take place at the expense of the earth’s heat, and the net result of the complete cycle in which (a) an igneous rock is eroded, (b) the resulting sediments buried and transformed into schists and (c) the latter brought again to the surface by earth movement and denudation, is a running down of energy involving a permanent loss. Moreover, the earth loses heat from the interior, not only by conduction, but also by convection. The circulation of ground waters and the activities of vulcanism—which include the upward movement of molten magmas, heated waters and gases—all result in the transference of heat from the interior to the outer zones of the crust. It is generally accepted, that when cooling by convection is left out of account, the temperature gradient due to conduction alone is of the order of 1° C. in 32 metres. If this estimate should be in error, it is more likely to be too high than too low.
To calculate the rate at which heat escapes from the earth by conduction from the interior, it is necessary to know the average conductivity of rocks. This factor, in turn, varies greatly in different materials, but in the case of the most predominant rocks the conductivity is accurately known, and the value k = 0·004 may be accepted as very close to the true average value. The variation of conductivity with increasing temperature and pressure scarcely affects the problem. The former tends to diminish the conductivity, the latter to augment it. As far as our present knowledge goes, these two effects [Pg 124] almost exactly balance each other, and the assumption that conductivity remains fairly constant with depth is therefore justified. If r represent the earth’s radius, and dθ/dr the temperature gradient, θ being the temperature, then the quantity of heat which passes from the surface per second is given by
| 4πr² × k × | dθ | and can be readily calculated. |
| dr |
According to the Laplacian hypothesis, the earth’s original store of heat was derived from the nebula from which it separated. Kelvin made the assumption that as the molten globe cooled down it was preserved by convection currents at a temperature nearly uniform from centre to surface. On attaining the point of solidification, it would gradually become solid throughout, most probably starting from the centre, and only when solidification was complete could the surface continue to cool further. The problem which Kelvin set himself to solve was this: Given a solid globe originally at a uniform temperature of 7000° F. (3871° C.), and subsequently cooling down, to calculate the time which would be required for the establishment of the present surface gradient.
Taking the most probable values for conductivity k, density of rock ρ, specific heat of rock σ, and temperature gradient dθ/dr, he applied his data to Fourier’s differential equation for the linear conduction of heat—
| dθ | = | k | · | d²θ |
| dt | ρσ | dr² |
Solving this equation for the unknown factor t, the required time period was found. The high initial temperature was chosen from [Pg 125] very meagre data, as representing a maximum figure for the melting-point of rock. Kelvin was particularly anxious that his treatment should provide an over- rather than an under-estimate of time. The experiments of Dr. C. Barus have shown that diabase, a good typical rock, becomes thoroughly liquid at 1200° C. If Kelvin had used this temperature instead of his arbitrary 3871° C., his periods of cooling would have been reduced to less than one-tenth of those actually arrived at.
For enormous periods of time, the development of a temperature gradient would be restricted to the earth’s outer zones. The interior, in complete thermal isolation, would remain unaffected, its loss of heat being quite insensible. The limited thickness of the outer shell, in which cooling would make itself felt, is made clear by the following figures:
| 160 | miles | in | 100 | million | years. |
| 240 | ” | ” | 240 | ” | ” |
| 320 | ” | ” | 600 | ” | ” |
| 570 | ” | ” | 1000 | ” | ” |
The different periods which Kelvin favoured in his famous contributions to this problem have already been mentioned, and are tabulated below.
| 1862. | 96 | million | years | (limits 20-400). |
| 1876. | 50-90 | ” | ” | |
| 1897. | 20-40 | ” | ” |
Clarence King, in 1893, applied a new criterion to the subject, taking into consideration the effect of pressure in raising the melting-point of rocks, and the necessity for an earth which should be stable under [Pg 126] the influence of tidal stresses. Barus had measured the melting-point of diabase at various pressures, and a law of variation of melting-point with depth was formulated on his experimental results. If this law were to hold as far as the centre, diabase would there be able to exist in the solid state at any temperature below 76,000° C. Thus the hypothesis arose that solidification would begin at the centre owing to the high pressure obtaining there. Under these conditions, however, a temperature gradient was already developed. If the gradient were to exceed the rate by which the fusion point of rocks is raised by pressure, the former would catch up, and at a certain depth the temperature would reach the fusion point and a zone of fluid rock would be inevitable.
King accepted diabase as a representative rock, and rejected any distribution of heat which would demand a liquid zone in that part of the earth’s crust where diabase or similar rocks would be expected to prevail. This procedure is justified by the consideration that were such a zone to exist, the earth would be incapable of maintaining tidal stability, and the crust would break down. King found that in the admissible cases, the initial temperature of crustal solidification would not exceed 2000° C., and that the period of cooling, which would reduce the gradient to that of the present was limited by 24 million years. Higher initial temperature would involve fluidity, and superior age necessitate a lower surface gradient. The following curves represent the gradients of Kelvin’s earth of 100 million years, and [Pg 127] King’s earth of 20 million years, in relation to the diabase fusion point curve. It will be seen that according to King’s argument, Kelvin’s earth implies the impossible condition of a liquid zone from A to B.
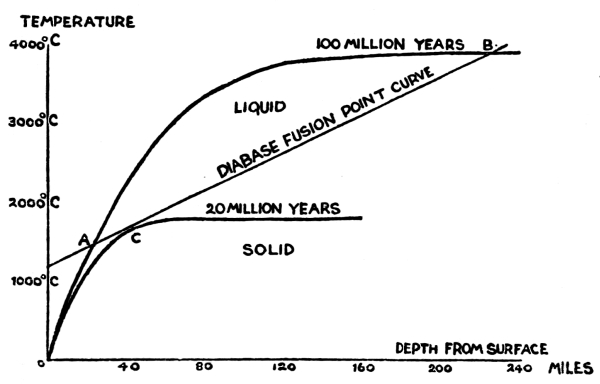
Fig. 16.
Temperature Gradients in relation to the Fusion point of Diabase.
In 1910, Becker attempted to deal with the same problem without relying on the temperature gradient, it being considered that owing to the presence of radium in rocks the gradient could not be trusted. In its place he assumes that the crustal strains associated with upheaval and subsidence are completely relieved at the surface of easiest fusion—and that according to the calculations of Hayford on isostatic compensation, the present depth of that surface is 71 miles. At that depth, therefore, the temperature curve and the diabase curve approach most closely, so that the additional temperature required to produce [Pg 128] fusion and relief of strain there becomes a minimum. In the above diagram, C would represent the point of easiest fusion. Becker justifies his choice of diabase by showing that on the Laplacian law of density, rocks of this type become predominant at depths greater than 40 miles, the more acid rocks, lying above, being more refractory. Tidal stability is provided for by rejecting any temperature curve which crosses the diabase line in the zone of basic rocks. The most probable earth, according to Becker, is one with an initial temperature of 1300° C., which would attain a surface gradient of 1° C. in 42 metres in 60 million years. He concludes that only “a tenth of the heat emitted by the earth can be ascribed to radioactivity plus all other exothermic chemical transformations.”
A Japanese estimate of the time elapsed since the molten surface of the earth began to solidify appeared in 1912. Suzuki makes the assumption that a thin solid crust has gradually increased in thickness, so that the latent heat of fusion liberated at the junction of solid and liquid rock is equal to the heat lost at the surface. The present thickness of the crust is assumed, on the authority of Milne and Arrhenius to lie between 30 and 40 miles. Granting these postulates, the thermal constants for basalt and granite lead to an age of 20 to 60 million years according to the thickness of the crust and the material of which it is composed.
It is surprising that Becker and Suzuki should have treated the problem in this restricted way. The heat evolution attending atomic disintegration was established in 1903, and in dealing with the earth [Pg 129] this phenomenon must be regarded as one of fundamental importance. To ignore the significance of radio-thermal energy is to reduce the problem to a mathematical exercise, interesting, no doubt, but with little value in its geological application. Let us make a simple calculation of the quantity of radium, which, if uniformly distributed throughout the earth, would make good the loss of heat. If Q is the heat generated per second by the radium in each cubic centimetre, then we have
| 4πr² × k × | dθ | = ⁴/₃ × πr³Q, |
| dr |
Now 1 gram of radium in complete radioactive equilibrium emits 216 calories per hour and consequently all the heat would be supplied by 10⁻¹³ grams per cubic centimetre, or 1·8 × 10⁻¹⁴ grams per gram of earth material. [Pg 130]
Radium per Gram of Igneous Rock
in Billionths (10⁻¹²) of a Gram.
| Observer. | Acid. | Intermediate. | Basic. | Ultrabasic. | ||||
|---|---|---|---|---|---|---|---|---|
| Strutt | 11[14] | 2·59 | 4 | 2·25 | 9 | 0·52 | 4 | 0.46 |
| Farr and Florence | 3 | 1·83 | 4 | 1·68 | 6 | 0·54 | ||
| Buchner | 8 | 2·61 | 15 | 1·64 | 4 | 0·73 | ||
| Fletcher | 4 | 0·85 | 20 | 0·85 | 5 | 0·71 | ||
| Holmes | 8 | 2·80 | 4 | 0·85 | 10 | 0·51 | ||
| Mean | 34 | 2·63 | 43 | 1·28 | 28 | 0·66 | 14 | 0·50 |
| Joly | 86 | 3·01 | 48 | 2·57 | 31 | 1·28 | ||
[Pg 131] Turning to the rocks themselves, the actual amount of radium is found to be a hundred times more than we want. Strutt was the first to discover this embarrassing richness, and his results, with those of later investigators, are summarised in the adjoining table. Joly’s results are given apart from those of other observers, for they were arrived at by the fusion method, and, moreover, separate rocks were not examined. A composite mixture of typical rock specimens was made up and a single analysis then sufficed to determine the average radium content. It will be noticed that Joly’s results are consistently higher than those found by the solution-method. Joly claims that his own procedure is more reliable than that followed in the solution method. Up to 1909, Joly had himself employed the latter method in the examination of 126 igneous rocks. In striking disagreement with the results of other workers, he found an average radium content of 7 × 10⁻¹² grams per gram of rock. An explanation of the discrepancy is not yet forthcoming, but in the light of his most recent work, which gives an average of 2·5 × 10⁻¹², he has now suggested that his earlier results be set aside. Measurements of thorium in rocks are not yet so plentiful as those of radium, and most of our present knowledge of the distribution of this element is due to Joly and Fletcher. The most probable averages of the data now available may be summarised as follows:
| Type of Rock. | Average per Gram of Rock. | |||
|---|---|---|---|---|
| Radium. | Thorium. | |||
| Igneous | 2·5 × 10⁻¹² | grs. | 2·0 × 10⁻⁵ | grs. |
| Sedimentary | 1·5 × 10⁻¹² | ” | 1·0 × 10⁻⁵ | ” |
| Metamorphic | 2·0 × 10⁻¹² | ” | 1·5 × 10⁻⁵ | ” |
| Deep-sea deposits | 5·0 × 10⁻¹² | ” | ||
Accepting these figures for igneous rocks provisionally and combining them with the respective heat emission of radium and thorium in complete radioactive equilibrium, viz.,
| Radium[15] | per | gram | 6 × 10⁻² | calories | per | second. |
| Thorium | ” | ” | 7·5 × 10⁻⁹ | ” | ” | ” |
[Pg 132] it is clear that each gram of the earth’s crust is a source of heat supplying on an average 15 × 10⁻¹⁴ calories per second on account of its radium content, and 15 × 10⁻¹⁴ calories on account of its thorium content. The total heat emission is therefore of the same order in each case, and amounts altogether to 30 × 10⁻¹⁴ calories per second. The whole mass of the earth is 6 × 10²⁷ grams, and if this were the source of as much radio-thermal energy throughout, the supply of heat in 1000 million years would have been sufficient to raise its temperature to about 40,000° C., and the present gradient should be many times greater than it is. This conclusion cannot be reconciled with the evidence afforded by the crustal rocks, both their structure and temperature gradients being decisively against any such possibility.
There are three cases which may be considered. The earth may be in thermal equilibrium, gaining as much heat as it loses and cooling only as the slow decay of the radio-elements permits; or it may be growing hotter, or, which is very unlikely, it may be cooling more rapidly than it would do if in radio-thermal equilibrium. The first case is the one now regarded with most favour. If the earth has cooled at all, and there seems to be no sound reason why we should altogether abandon that venerable conception, it must at some time have attained a condition of equilibrium. With the slow march of atomic disintegration its own rate of cooling would then keep time. The temperature gradient would be maintained solely by radioactivity for an immeasurably long period. [Pg 133]
The superabundance of radium which seemed to be implied by Strutt’s original work is certainly, as he then suggested, restricted to the surface rocks. The interior of the earth must be relatively free from radium and thorium. It is easy to calculate the thickness of the outer zone of the earth’s crust, which would suffice to supply the stream of heat passing to the surface. The temperature θᵣ at any distance r from the surface is given by the following equation, where k is conductivity, h the heat production of radium and thorium per second, in each gram of rock, ρ the density of the rock, and D the total depth of the radioactive layer:
| θᵣ = | hrρ | ( | D - | r | ) |
| κ | 2 |
At the base r becomes equal to D and the temperature θᴰ is therefore given by
| θᴰ = | hρD² |
| 2κ |
Using the figures given above, the thickness of the radioactive layer would be restricted to about ten miles and the basal temperature would reach only 250° C. This result cannot be held to express the facts, for there can be no doubt that the radium and thorium content decreases with depth for the same reason that the type of rock varies with depth. A glance at the table on p. 130 will show that there is a rough proportionality between the acidity or percentage of silica of a rock [Pg 134] and its radium content. The more basic rocks are much poorer in radium, and, as would be expected, in thorium also. Now we have good reason to suppose that the more deep-seated rocks of the earth’s crust are of basic and ultra-basic composition, and that below the 30-mile crustal zone they are exclusively ultra-basic, perhaps similar in composition to the material of stony meteorites, with which they agree almost exactly in density (3·4). This information, which it might be thought would be for ever withheld from us, is derived from the study of earthquake waves. The latter in passing through the earth’s interior carry with them a record of the type of material they have penetrated. Within the stony zone, which extends downwards for several hundred miles, and separated from it somewhat sharply, lies the heavy core of the earth (density about 7·8), probably of metallic composition, like the iron meteorites. If we may judge from the latter, this nucleus is entirely free from radium, and that there is safety in this analogy is indicated by the very low radium content of such native iron as has found its way in basaltic magmas up to the surface.
We have already seen (p. 30) that a first differentiation of the original heterogeneous material from which the earth was built would result in the formation of a metallic core surrounded by a stony zone. The further differentiation of the latter, whereby the crust with its abundant variety of acid and basic rocks was developed, is of too complex a nature to be considered here. It is very probable that as the more siliceous constituents separated and became concentrated towards [Pg 135] the surface, they carried with them their store of radio-elements. In this way, basing our ideas on evidence quite independent of the temperature gradient, we are led to the remarkable conclusion that the radium and thorium of the earth are to be found almost exclusively in the earth’s crust. The most probable depth of the radioactive layer may therefore be placed at 30 miles and the basal temperature in this case would be about 750° C., which would be more in accordance with the requirements of volcanic phenomena. Moreover, it must not be forgotten that the heat lost by the upward movement and convection currents of rock magmas, heated waters and gases, has also to be accounted for. The radio-thermal equivalent must be substantially increased to include this phase of the subject. The basal temperature of 750° C. is only a minimum, and the higher temperatures demanded by geology are not therefore inconsistent with the facts. However, until more data are accumulated, it would be rash to attempt to deduce the exact distribution of the radio-elements in the crust, but already we may assert with confidence that the crustal average is somewhat lower than that of the surface rocks in which granitic types form so large a proportion. What the average actually may be cannot yet be decided.
Kelvin’s problem must now be reversed. It is impossible to deduce the earth’s age from its thermal condition. We can only say that the age must be very much greater than Kelvin calculated. The new problem which presents itself is to determine the thermal history of the earth, [Pg 136] accepting its antiquity as a known or partially known factor. The science of radioactivity is a welcome addition to the tools which the geologist employs in his difficult task of elucidating the earth’s history, and it is peculiarly valuable in helping him just where he has hitherto had most cause for despair.
[Pg 137]
The rate of helium production from uranium and thorium—Lead the final product of the uranium family—Its accumulation in geological time—Lead and helium ratios as a measure of geological time—Assumptions to be granted—Value of analyses in deciding quality of material for estimating ratios—Necessity for fresh, stable, primary rock minerals—Strutt’s work on the helium ratio—The lead ratio—Boltwood’s collection of analyses—Examples, with geological data—The author’s work on the Devonian minerals of Norway.
As we saw in Chapter VII the α-particles which are emitted at certain points in the line of descent of the radioactive elements have been identified with helium. Fortunately the evidence is conclusive, for upon this identification depends the latest and most elegant method yet devised of measuring geological time.
When all the members of a genetically related series of radio-elements are in equilibrium, the transformation proceeds in such a way that an equal number of atoms of each element disintegrates in the same time. When an atom of uranium disintegrates it does so with the production of two atoms of helium. At the same time, as indicated in the diagram, page 190, each of six other members of the family also emit a single [Pg 138] atom of helium. Consequently, the total number of helium atoms which are liberated in the course of the complete transformation of a single atom of uranium is eight. From this result and the remarkable measurement made by Rutherford and Geiger—that 3·4 × 10¹⁰ α-particles or atoms of helium are expelled per second from a gram of radium—it is possible to calculate that the annual production of helium from a gram of uranium in equilibrium with all the other products of the series is 10·7 × 10⁻⁸ cubic centimetres. Measurements of the ionising power of thorium and its chain of dependent elements may be utilised to calculate the same rate in the case of the thorium family. It is found that one gram of thorium is equivalent in helium generation to 0·26 gram of uranium. The details of these calculations will be found in Appendix A.
While there is little uncertainty in these indirect results, it is gratifying to know that Prof. Strutt, during 1909-10, verified them both by a direct appeal to experiment. In certain minerals which contain radioactive constituents, the evolution and accumulation of helium must have been steadily proceeding for very long periods. Before a direct determination of the rate of helium production can be made, it is evidently necessary to use material entirely free from that element. Strutt worked with richly radioactive minerals, from large quantities of which he expelled completely the accumulated store of helium. This was done by preparing solutions with every precaution to avoid the presence of undissolved particles of mineral, and afterwards boiling them till the helium was removed. The solutions were then put aside [Pg 139] until a fresh supply of helium had been generated in sufficient quantity to be detected and measured. To isolate the gas from the solutions and accurately to determine its volume was obviously a matter of great experimental difficulty, so minute were the volumes dealt with. However, as the result of the exquisite delicacy of his methods, Strutt brought his experiments to a successful issue.
The minerals used were pitchblende and thorianite, the former containing uranium alone, and the latter both uranium and thorium. The annual production of helium per gram of the parent element (in radioactive equilibrium with its respective family) was found to be:
The remarkable concordance of these results with the theoretical requirements is an eloquent tribute to the refined methods and experimental skill with which the measurements were carried out.
Experiments carried out by Boltwood and Rutherford in 1911 afford an equally striking confirmation of the conclusions on which the [Pg 140] theoretical results were based. They measured the rate of production of helium from radium, the latter being in equilibrium with its early disintegration products, three of which also emit α-particles. Radio-lead and polonium were completely removed. The annual evolution of helium to be expected was 158 cubic millimetres. The first direct determinations were made by Sir James Dewar in 1908, and his best results corresponded to 169 cubic millimetres. Boltwood and Rutherford arrived at a much closer agreement, their figure being 156 cubic millimetres.
These results cannot fail to inspire the conviction that our atomic theory of matter is essentially correct. We are in possession of two experimental facts. The number of helium atoms expelled per second from a gram of radium has been directly counted, and the volume of helium accumulated in a year has been directly measured. The number of atoms in a given volume of helium (at N.P.T.) can be deduced at once, and the calculation is independent of any underlying theory. Calculation gives 2·69 × 10¹⁹ atoms per cubic centimetre; the atomic theory demands 2·72 × 10¹⁹.
Although there can now be no doubt that helium is one of the stable disintegration products, yet there is no direct evidence as to the identity of the ultimate products in the direct line of descent. In the uranium series indirect evidence points to lead with a considerable degree of certainty, but the end product of the thorium family is still unrecognised. In every uranium-bearing mineral the parent element slowly breaks down, while the final product of the transformation [Pg 141] accumulates at its expense. Hence, if lead is the favoured element it ought to be found in association with uranium in all minerals which contain the latter. Moreover, in minerals which can be proved to be of the same antiquity, the amount of lead per gram of uranium should be constant; further, in minerals of various geological ages the proportion of lead should vary according to the latter. A mineral which began its accumulation of lead in pre-Cambrian times should certainly contain more at the present time than one in which lead has been collecting only since, say, the Tertiary outburst of igneous activity. The same statements apply equally well to the case of helium.
In so far as these principles may be used conversely to test the identity of lead with the ultimate product, they lend every support to that important conclusion. Dr. Hillebrand, the leading authority on the analysis of uranium-bearing minerals, has never in the course of a long experience found uranium unaccompanied by lead. It was this constant association which led Boltwood, in 1905, to suggest the probability of a genetic relationship existing between these two elements. In 1907, Boltwood went farther and showed that for minerals of the same age the amount of lead for each gram of uranium, or the ratio Pb/U, was, in general, nearly constant. He collected all the best analyses of primary uranium minerals, but unfortunately he omitted to give the geological details of their occurrence. As will be seen in the present chapter, when the relative ages of the minerals are compared with their lead ratios, a striking proportionality discloses itself. [Pg 142]
The evidence of atomic weights is also favourable to lead. The complete disintegration of an original atom of uranium may be expressed as follows:
| U | ➜ | 3He | + | Ra | ➜ | 8He | + | Pb |
| 238·5 | 3·994 | 226·36 | 3·994 | 207·08 | ||||
| 238·5 | 238·34 | 239·03 | ||||||
Atomic weights are appended to each symbol, and the totals, which should be equal, are placed underneath. The agreement is close, but not as convincing as one could desire. Two alternative explanations of the discrepancies are suggested. Either lead is not the final product, or the atomic weights of both radium and uranium are too low by about 0·5. Neither alternative can readily be granted, but it may be pointed out that since uranium is the heaviest known element and radium follows not far behind, any impurities whatever, with the exception of thorium, would have the effect of lowering the observed atomic weights.
Accepting the above equation as substantially correct, the mass of lead generated in one year from a gram of uranium can now easily be calculated. For eight atoms of helium, one of lead is produced, or, mass for mass, six and a half times as much.
Consequently, as a gram of uranium involves the annual production of 1·88 × 10⁻¹¹ grams of helium, the associated lead which remains must amount to 1·22 = 10⁻¹⁰ grams. If this rate were constant we could find how long it would take for any mass of uranium to become completely [Pg 143] converted into helium and lead. However, the rate is not constant, but is proportional at every moment to the quantity of uranium remaining unchanged. As the parent element becomes exhausted, it disintegrates more and more slowly.
Now in very considerable periods amounting to hundreds of millions of years only a very small fraction of the uranium originally in existence is decayed. For this reason, if only a small proportion of lead or helium has collected in a mineral since it began its life-history, then no serious error will be made in assuming their rates of evolution to have been constant. In minerals which have been in existence for 400 million years the slowing down is only about 5%. If an appreciable error should arise in ignoring this decline, then in place of the present day percentage of uranium the time-average must be substituted (see Ap. A, p. 179); that is to say, the amount of uranium which, if it did break up at a regular rate, would evolve the same quantities of the ultimate products.
Having calculated this uranium average, Uₘ, with the necessary approximation, the total quantity of lead or helium accumulated in a mineral would then provide a direct measure of its age.
Using lead as the age-index, and knowing its percentage, Pbₜ, in the mineral, then the time it has taken to collect, i.e. the age of the mineral, Pbₜ, is given by Pbₜ/Uₘ × 8200 million years.
Using helium as the age-index, both thorium and uranium must be estimated. The amount of thorium may be conveniently expressed in terms [Pg 144] of uranium, since the latter is four times as active in its helium production as thorium. The total equivalent quantity of uranium, Uₑ, is thus known, and thorium then need play no further part in the calculations. It is found most convenient to measure the amount of helium, Heₜ, as the volume in cubic centimetres per gram of mineral. In this notation the age is given by Heₜ/Uₑ × 9·6 million years.
The validity of this procedure evidently demands the granting of certain obvious assumptions. Our choice of suitable minerals will not only be limited by these considerations, but the reason for a particular choice will be justified. The assumptions fall under the following headings:
The first and second suppositions bring up the whole problem of the origin of minerals. It is possible, by means of a physical examination, to decide whether a mineral is of the same age as the rock in which it occurs, or whether it is older or younger. In igneous rocks, such as a granite, the majority of the minerals are of the same antiquity as the rock, that is, they date from the period of consolidation of the rock magma. The component mineral particles of most sedimentary and detrital [Pg 145] rocks existed long before the strata were laid down, whereas the cementing materials by which they are consolidated are partly furnished by percolating solutions and therefore may be subsequent to the period of deposition. In the same category come those ore deposits which occupy the fissures and crevices of pre-existing formations.
In whatever way a mineral may occur, its history can always be traced back either directly or by conjecture to an igneous rock, and it is rarely that it is possible to go beyond the magma from which such a rock must have consolidated. And even if this can be done in exceptional cases, there lies behind still another magma to which the material can be referred. Consequently, the minerals of igneous rocks are regarded as primary or original, and here we may briefly consider a few facts relative to their crystallisation from a molten condition.
A molten rock is regarded as a solution in which the numerous constituents are dissolved one in another. Now certain of the constituents can only remain in solution provided the latter is, with regard to them, very dilute. That is to say, they are only slightly soluble in a solvent composed of the rest of the rock material. Consequently, substances of which these constituents form an essential part will, as a general rule, be the first to crystallise; as examples, zircon, sphene, and apatite may be cited. For the same reason it happens that the magma does not remain homogeneous, but rejects certain [Pg 146] of the rarer elements which collect together in a subsidiary magma of peculiar composition. To this concentrate of exceptional constituents, the gases and water vapour expelled during solidification are also added, and serve to maintain it in a state of æquo-igneous fusion, even when the bulk of the rock has already crystallised. The residual liquors yield the minerals of pegmatites and of drusy cavities. Certain minerals which are conspicuously rare in the body of the normal rock, are often developed on a large scale in pegmatite dykes, and it is from these that the most perfect and beautiful crystal forms are generally obtained.
Amongst the elements of limited solubility in a rock magma, uranium and thorium must be placed. The accessory minerals of ordinary igneous rocks, such as those already mentioned—zircon, sphene, and apatite—are rich in the radioactive elements when compared with commoner minerals like felspar and hornblende. In general, the richness of a mineral in uranium seems to depend on its position in the order of consolidation. The minerals first to be formed claim the greater part of the available store. The original surplus, unable to dissolve in the magma, is held over, and, should it be rich in radioactive ingredients, uranium- and thorium-bearing minerals may be formed during the later stage of pegmatitic intrusions. It is a striking fact that, as primary constituents, these minerals invariably occur in pegmatites associated with granite or syenite. As examples, pitchblende or uraninite, thorite, thorianite, and monazite may be mentioned.
We must now consider the part played by lead and helium during the [Pg 147] genesis of minerals. Before the consolidation of the magma, both these elements must, of course, have been generated within it for an unknown period. As to the effect of physical conditions upon radioactive transformations it has already been shown (p. 102) that all the evidence points to the conclusion that these atomic changes are independent of the temperatures and pressures under which a molten magma exists. The helium already present at the time of crystallisation appears to behave physically in no way different from the other gases. There is no evidence that it tends to congregate in any particular mineral. A small proportion may be distributed through the resulting rock, but probably the larger share is expelled.
The lead which may be originally present follows a similar course. The metal is rejected, not only by the primary magma but, with rare exceptions, by the residual magma also. It finds no definite place in igneous rocks. Doubtless a certain amount of lead is retained in the molecular network of crystals, but that amount is not high. In the rocks of Leadville, Colorado, Hillebrand found an average of less than 0·002 per cent of lead. In the nepheline syenite of Southern Norway, using specimens free from minerals which one would expect to be comparatively rich in accumulated lead, the present writer was able to determine a percentage of only 0·0004. If, then, there should be initially a greater quantity of original lead, where are we to look for it? Probably the most of it goes to form lead-ores, such as galena. [Pg 148] Separated from the pegmatites it appears in the later phases of ore deposition which follow on the heels of igneous activity. In company with hot gases, sulphide solutions, and a number of metallic companions, our lead is carried away and deposited in the fissures encountered by the mineralised waters. If the agency of magmatic gases appears to have been an important factor in the production of ore bodies, the origin of the latter is said to be pneumatolytic. Brögger has shown that in Southern Norway galena was one of the minerals to be formed in this way.
In the last phase of this complex series of operations, magmatic waters contribute their share to the filling of mineral veins, and it is amongst these hydatogenetic ores that galena is most usually found. The important point is that lead, for the most part, is drawn from the primary magma at, or perhaps before, the time of crystallisation, and it is not until the igneous activities have declined that it again appears in an active rôle.
Let us now consider the effect of the original distribution of lead and helium in a newly-formed rock. It will be clear that an analysis of the rock as a whole would give values of Pb/U and of He/U much higher than those corresponding to the period since consolidation. Of the total amounts of lead and helium, part would be originally segregated in the rock and part would be due to subsequent genesis. In most rocks, the former part is of sufficient magnitude altogether to invalidate the use of the ratios as age indices. This difficulty can be avoided by confining attention to particular minerals—indeed, to just those [Pg 149] minerals which concentrate within themselves the radioactive parent elements. Within them lead and helium may accumulate to such a degree that the amount initially present becomes negligible. Zircon from the Devonian syenites of Southern Norway contains more than twenty times as much lead as the rock in which it occurs. Roughly, we may say that since the zircon came into being its content of lead has multiplied twenty times. Thorite may accumulate a hundred or a thousand times as much lead as it possessed at first. For the same reason, minerals like zircon and sphene often contain hundreds of times as much helium as the rock from which they are taken, and there is little possibility of error in assuming that they have themselves generated the whole of their supply.
Another difficulty, and a more serious one, must now be faced. Can we be sure that for periods of hundreds of thousands years a mineral has remained comparatively unaltered by external agencies? With regard to helium there is undoubtedly a tendency to escape. Strutt has demonstrated that when a radioactive mineral has been powdered, helium begins to leak, rapidly at first, then at a diminishing rate. Even crystals washed out of their original matrix showed a considerable leakage of helium. The observed rate of escape always exceeds the rate of generation, and it therefore follows that during the life-history of a mineral the conditions must be specially favourable to the retention of helium, for otherwise the latter could not have accumulated. Nevertheless, these experiments prove conclusively that the majority of [Pg 150] minerals do not contain their full store of helium, and it is a matter for surprise that they contain so much. Consequently, ages deduced from the helium content of minerals can be regarded only as a fraction of the true age.
Dealing with lead and uranium, we must consider the tendency to alteration of the minerals in which they occur. From the surface down to the permanent level of the ground waters, rock material is subject to weathering. The more soluble constituents are leached out and complex silicate minerals are decomposed by the combined action of water, oxygen, and carbon-dioxide. It is in this belt of weathering that igneous rocks suffer most change. Many of the primary minerals are broken down and alteration products take their place. All the reactions involve considerable increase in volume, and not only are minerals altered in place, but material is carried away and deposited elsewhere as secondary minerals. Can we be sure that lead and uranium have remained untouched during this redistribution? In some cases we cannot, but fortunately for our purpose many of the most valuable minerals, like zircon, are dense and exceptionally stable. A mineral is only stable over a limited range of conditions. It adapts itself more or less readily to its physical environment. Certain minerals, however, are much more capable than others of withstanding great changes without undergoing metamorphism or alteration, and amongst these are many of the uranium-bearing minerals.
When there has been a migration of lead or uranium, an appeal to analysis will rarely fail to dispel the difficulty by disclosing the [Pg 151] fact. It is inconsistent with the chemical properties of these elements that both should have been affected in the same proportion, and hence the ratio of lead to uranium obtained from different minerals of the same geological age affords an immediate test of the extent to which they have suffered from alteration in the course of their history. If the analyses give consistent results, it can be safely assumed that the effects of alteration have been inconsiderable; if there are marked discrepancies the results must be rejected as valueless from a chronological point of view. A microscopical examination of the minerals before analysis is a useful safeguard, for in this way altered material can often be detected. It is clear that reliable conclusions can only be drawn from minerals which are undoubtedly fresh.
Becker has criticised the method by directing attention to a suite of minerals from Llano Co., Texas. Their geological age is well defined. The Burnet granites with which they are associated are intrusive into a series of schists and quartzites, metamorphosed sediments of late Algonkian time. The Cambrian rocks lie upon this complex and the period of intrusion is therefore between two limits which are not very far apart. The lead ratios of these minerals are far from being constant, as the following examples show:
| Yttrialite | 1·15 |
| Yttrialite | 0·51 |
| Mackintoshite | 0·39 |
| Uraninite | 0·17 |
| Fergusonite | 1·04 |
| Fergusonite | 0·30 |
[Pg 152] Boltwood found a satisfactory agreement in four cases, the ratio being 0·17, but, as he pointed out himself, most of the minerals from this locality are unsuitable, because of incipient or advanced alteration. The quartzose pegmatites in which the minerals occur are riddled with alteration products and secondary minerals, and the whole series is altogether unfavourable to accurate age determination. It is doubtful whether the apparent agreement of the ratios quoted by Boltwood ought to be accepted without further verification; for the present they cannot be regarded without suspicion.
This example shows how the actual results indicate the vicissitudes, varying from mineral to mineral, which the lead and uranium contents may have undergone. The method confirms or denies the validity of its application in every case. Judging from the relative solubilities of the constituents in question, uranium is likely to be abstracted from a mineral during the process of weathering more readily than lead, and consequently the age deduced from a weathered or altered specimen should in general be too high. A differential effect of this kind would account for the high ratios given by the Llano Co. minerals.
Strict attention must be paid to the question of origin, and secondary minerals avoided as carefully as altered primary minerals. Pitchblende is often secondary, e.g. when it occurs in veins with metalliferous sulphides. Other examples, of a rather different type, are autunite and carnotite. Secondary minerals are necessarily more recent than the [Pg 153] rocks in which they occur, and many of them date back to no very remote period. Autunite is sometimes formed quite near the surface, within a few inches in fact. Its antiquity cannot therefore be more than a few thousand years, and in this time a detectable quantity of lead could not be generated. The traces actually found were probably in the original possession of the mineral. In an analysis made by the writer, only 0·06% of lead was found in specimens of autunite from Mozambique, where it occurs in bright green flakes attached to the large biotite crystals of pegmatitic dykes. In keeping with the age of the pegmatites and the high proportion of uranium—45%—it should have contained a hundred times as much had it been a primary mineral. The paucity of lead in autunite has even been put forward as an argument against the contention that lead is the ultimate product of disintegration of the uranium family. We now see how baseless is this argument when the origin of the mineral is remembered. It is not surprising that autunite should contain so little lead; on the contrary, it contains much more than the uranium can account for in the time at its disposal.
From these considerations it will be obvious that the only minerals to be chosen as material from which to determine the lead-ratio are fresh, stable primary rock-minerals. Having decided this, there remains a third possibility which might cast doubt upon the method. It can be objected that lead may originate as a product of some element other than uranium. Analytical results show clearly that thorium cannot give rise to lead, or a more proportionate relationship between these two [Pg 154] elements would have announced the fact. There is also a possibility that certain of the longer-lived members of the uranium family may themselves be segregated in a mineral, independently of uranium. If so, they would gradually disintegrate, leaving no trace of themselves other than the residual helium and lead. In a magma containing 10% uranium, the radium would amount only to 0·0000034%. In actual magmas, even of pegmatites, the quantity present is always much less than this, and even if it be allowed that such tiny quantities may saturate the magmatic solution, the precipitation and concentration in any particular mineral would not be sufficient to leave an appreciable residue of lead.
The application of the accumulation of helium in minerals to the measurement of geological time, was first suggested by Rutherford in 1905, when he wrote: “I think that, when the constants required for these calculations are more definitely fixed, this method will probably give fairly trustworthy information as to the probable age of some of the radioactive minerals of the earth’s crust, and indirectly as to the age of the rocks in which they are found.”
During the years 1908-10, Strutt examined a great number of minerals, and determined the helium ratio whenever practicable. His first set of experiments dealt with phosphatic nodules and phosphatised bones. These may sometimes contain fifty times as much uranium as average rock material, and they have a further advantage in that they can be found in strata of nearly every age. As they frequently consist of fossils [Pg 155] characteristic of the formations in which they occur, their age is well defined. However, the power of retaining helium is both poor and variable in the case of these phosphates, and the time relation is therefore obscured. Such materials never retain more than a small fraction of the helium which has been generated within them.
More suitable in their power of retention are certain iron ores, from which significant results were obtained. The helium ratio, and therefore the numerical age derived from it, showed a marked dependency upon the geological age of the mineral, as the following examples illustrate:
| Mineral. | Geological Age. | Millions of Years. |
|---|---|---|
| Siderite | Upper Oligocene | 8·4 |
| Hæmatite | Eocene | 30·8 |
| Hæmatite | Upper Carboniferous | 141·9 |
| Hæmatite | Devonian | 145·2 |
Strutt next investigated the more compact minerals of igneous rocks, notably zircon and sphene. Zircon can be obtained from rocks belonging to several periods of igneous activity, and being a durable and stable mineral it is peculiarly fitted to retain the helium generated within it. Even allowing that the helium found does not represent the whole amount generated, it is unlikely that the fraction lost will vary as conspicuously as in the case of phosphates. In so far as that fraction [Pg 156] depends on the structure of the mineral it is probably more uniform for zircon than for most other minerals. The helium ratio ought therefore to stand in a close relation to the geological age of the specimen. That it does so is clearly demonstrated by Strutt’s results, which are given below. The geological ages have been taken from the most recent literature, and are given in greater detail than those published in the original paper. Where two periods are bracketed together they are to be understood as referring to the limits between which the age of the igneous rock may fall.
| Locality. | Geological Age. | Millions of Years. |
|---|---|---|
| Mt. Somma, Vesuvius | Recent | 0·1 |
| Pleistocene | ||
| Mayen, Eifel | Pleistocene | 0·1 |
| Campbell I., N.Z. | Pliocene | 2·5 |
| Expailly, Auvergne | Miocene | 6·3 |
| Brevig, Norway | Devonian | 54 |
| Cheyenne Canon, Colorado | Upper Cambrian | 141 |
| Archean | ||
| Green River, N. Carolina | Carboniferous | 147 |
| Archean | ||
| Ural Mts. | Pre-Devonian | 209 |
| Ceylon | Archean | 286 |
| Blue Ground, Kimberley | Archean | 321 |
| Sebastopol, Ontario | Archean | 622 |
The results for sphene add but little to the above table, for most of the rocks from which workable quantities of sphene can be obtained are of pre-Cambrian age. The most notable helium ratio was from a specimen [Pg 157] occurring in the Archean rocks of Ontario, and corresponded to an age of 715 million years. Summarising all the data afforded by Strutt’s work, we may graduate the geological column with a time-scale. It must be clearly understood, however, that the ages as expressed in years are, in the case of the helium ratio, minimum values only. How much greater the time represented by the geological periods actually is will appear from the ages as deduced from the lead-ratio. A few of these are placed in the table below for comparison.
| The Geological Systems. | Time-Scale in Millions of Years. | |
|---|---|---|
| Helium Ratio. | Lead Ratio. | |
| Pleistocene | 1 | — |
| Pliocene | 2·5 | — |
| Miocene | 6·3 | — |
| Oligocene | 8·4 | — |
| Eocene | 30·8 | — |
| Cretaceous | — | — |
| Jurassic | — | — |
| Triassic | — | — |
| Permian | — | — |
| Carboniferous | 146 | 340 |
| Devonian | 145 | 370 |
| Silurian | 209 | 430 |
| Ordivician | ||
| Cambrian | — | |
| Algonkian | 1000-1200 | |
| Archean | 710 | 1400-1600 |
The application of the lead-ratio to the measurement of the antiquity [Pg 158] of minerals was first due to Boltwood. He tested its reliability by an appeal to the best analyses published up to 1907. Some of these, with geological details, will now be given.
In Glastonbury, and also in Portland, Connecticut, primary uraninite is found in the felspar quarries. The pegmatite in which the mineral occurs is associated with a granite which intrudes Lower Carboniferous strata. It is probably to be referred to the close of the Carboniferous period, and is certainly pre-Triassic. Five different specimens gave lead ratios in striking agreement, corresponding to an age of 340 million years.
It should be observed in all the following tables that in calculating the lead ratios the time-average of uranium has been used, and not the amount actually present. The statement of analyses is, of course, in percentage.
| Uranium. | Lead. | Ratio. |
|---|---|---|
| 70 | 2·9 | 0·041 |
| 70 | 3·0 | 0·042 |
| 70 | 2·8 | 0·039 |
| 72 | 3·0 | 0·041 |
| 72 | 2·9 | 0·040 |
Similar crystals of uraninite have been furnished by the pegmatites of Branchville, Connecticut. The intruded strata are of either Silurian or Ordivician age, and the evidence suggests that the period of intrusion is possibly coincident with that of the earth movements which commenced [Pg 159] at the close of the Ordivician. Here again the lead ratios closely agree, the age being 430 million years:
| Uranium. | Lead. | Ratio. |
|---|---|---|
| 74 | 4·0 | 0·052 |
| 75 | 4·0 | 0·051 |
| 74 | 4·0 | 0·052 |
| 66 | 3·5 | 0·051 |
In North Carolina uraninite occurs in coarse pegmatites which are mined for their large flakes of mica. In this instance a good agreement is scarcely to be expected, as secondary products abound, and the three specimens from Spruce Pine which were examined by Hillebrand showed signs of incipient alteration. The fourth specimen was from South Carolina and this again lacked the freshness which is so essential. Zircon is also found in the North Carolina pegmatites, and the writer has examined two sets of specimens with the results given below. The material in this case appeared to be quite fresh.
The geological period is difficult to establish with certainty. The relations of the Appalachian rocks of Carolina are so obscure that the required age may be anywhere from pre-Cambrian to Carboniferous. Judging from the lead ratios, one is tempted to suggest that the age is not far from the Silurian, but until our time-scale is better determined one must be chary in this converse application of the method. It may confidently be hoped that in this way the geologist will [Pg 160] be greatly helped in his attempt to unravel the history of igneous activity in the earth’s crust. But the time is not yet, for the accumulation of facts and data has only just commenced.
| Mineral. | Uranium. | Lead. | Ratio. Pb/U. |
|---|---|---|---|
| Uraninite | 77 | 3·9 | 0·049 |
| Uraninite | 77 | 4·2 | 0·052 |
| Uraninite | 67 | 3·3 | 0·047 |
| Uraninite | 71 | 3·3 | 0·045 |
| Zircon | 0·076 | 0·0036 | 0·046 |
| Zircon | 0·130 | 0·0055 | 0·041 |
Other series of minerals could be given from the pre-Cambrian rocks of North America, Ceylon, and Mozambique, but it is unnecessary to multiply details further. In all cases the Archean seems to date from 1200-1600 million years.
We now turn to the pre-Cambrian rocks of Scandinavia and Finland, and for comparison with those of N. America the following classifications and correlations may be given. The chief unconformities are indicated by wavy lines. It should be pointed out that the correlation of the pre-Cambrian rocks over wide areas is, perhaps, the most difficult task the geologist has to attempt, and the scheme given opposite is based more on analogy than direct evidence.
| Fennoscandia. | North America. | |
|---|---|---|
| Jotnian | Keweenawan | Algonkian |
| Jatulian | Upper Huronian | |
| Upper Kalevian | Mid. Huronian | |
| Lower Kalevian | Lower Huronian | |
| Bottnian | ||
| Ladogian | Laurentian | Archean |
| Katarchean | Keewatin |
[Pg 161] It is not yet possible to support this correlation by a concordant system of chronology. Igneous rocks occur at most of the horizons, but it is extremely difficult to get samples of suitable mineral species for analysis. However, there is no doubt that in the future a definite time estimate will be attached to each of the above periods. When we are in full possession of this knowledge, and only then, will a reliable correlation of these rocks be possible.
The analyses collected by Boltwood include two groups which are of minerals taken from the pegmatites of Southern Norway, rocks famous for the occurrence of rare minerals. The first group, from the igneous complex of the Moss district, of which the average age is 1000 million years, is as follows:
| Mineral. | Uranium. | Lead. | Ratio. |
|---|---|---|---|
| Uraninite | 66 | 8·4 | 0·12 |
| Uraninite | 68 | 7·8 | 0·11 |
| Annerdödite | 15 | 2·2 | 0·135 |
| Uraninite | 66 | 9·3 | 0·13 |
| Uraninite | 57 | 8·0 | 0·13 |
| Uraninite | 65 | 8·8 | 0·135 |
| Uraninite | 68 | 8·8 | 0·12 |
| Uraninite | 76 | 9·0 | 0·11 |
| Thorite | 8·2 | 1·2 | 0·13 |
[Pg 162] The second group, from the complex of Arendal, demands an age of 1200 million years. The Scandinavian geologists believe that both groups of rocks are younger than the quartzites and other metamorphic sediments with which they are always associated, and that the latter rocks are of late Archean age. Sederholm, however, thinks they may be equivalent to his Kalevian division. The geological evidence, as far as it goes, does not point to any difference in the ages of these two sets of Norwegian minerals, but, on the other hand, there is no positive evidence that the ages are the same. An analogy made in ignorance cannot be held to constitute a proof. Field work in this case does not disqualify the testimony of the radioactive minerals; it rather invites their co-operation in the perplexing task of disentangling the intricate structural relations of the rocks.
| Mineral. | Uranium. | Lead. | Ratio. Pb/U. |
|---|---|---|---|
| Uraninite | 56 | 9·8 | 0·14 |
| Uraninite | 61 | 10·2 | 0·15 |
| Uraninite | 56 | 9·4 | 0·15 |
| Thorite | 9 | 1·5 | 0·16 |
| Orangite | 7·5 | 1·2 | 0·15 |
| Xenotime | 2·9 | 0·62 | 0·19 |
Amongst these rocks correlation is exceedingly difficult, and even their relative ages are hidden in obscurity. Högbom holds that the massifs of Moss and Arendal are contemporaneous with the Ser-archean [Pg 163] granites of Sweden. Similar hyperites and quartzites are found in nearly every locality and the granites always appear to be younger than these. After their intrusion, an enormous thickness of rock was denuded away before the Jatulian sediments were laid down. The physical break here indicated is one of the greatest in the history of the earth, and undoubtedly represents an immense lapse of time. The granites and pegmatites must therefore be considerably older than the Jatulian rocks.
With a view to testing the constancy of the lead-ratio in a series of minerals from a single igneous complex, the author, in 1911, made a number of experiments on carefully chosen material. There occurs in the Christiania district of Norway, a geologically depressed area of nearly 4000 square miles, which is separated by faults from the surrounding pre-Cambrian rocks on every side. Within this area there is a nearly complete sequence of early Palæozoic rocks, surmounted by a few beds of red sandstone of Lower Devonian age. Over these beds and intercalated with them are lava flows; and finally, penetrating the whole mass, and representing a later phase of the same period of igneous activity, are great intrusions of plutonic rocks. Amongst the earliest of the intrusions is a series of thorite-bearing nepheline-syenites. Brögger believes them to be of Middle or Lower Devonian age, most probably the latter. The minerals occurring in them are, in many instances, notably radioactive, and thus they afford an admirable series in which to investigate the consanguinity of lead and uranium. A suite of minerals [Pg 164] was obtained from Brevig, and estimations of these elements made, with the following results:
| Mineral. | Uranium. Grs. per 100 grs. Mineral |
Lead. Grs. per 100 grs. Mineral |
Pb/U. |
|---|---|---|---|
| Thorite | 10·1040 | 0·4279 | 0·042 |
| Orangite | 1·2437 | 0·0570 | 0·046 |
| Orangite | 1·1825 | 0·0542 | 0·046 |
| Thorite | 0·4072 | 0·0196 | 0·048 |
| Homelite | 0·2442 | 0·0121 | 0·049 |
| Zircon | 0·1941 | 0·0085 | 0·044 |
| Pyrochlore | 0·1923 | 0·0120 | 0·062 |
| Pyrochlore | 0·1855 | 0·0093 | 0·050 |
| Biotite | 0·1602 | 0·0069 | 0·043 |
| Tritomite | 0·0631 | 0·0026 | 0·041 |
| Freyalite | 0·0526 | 0·0028 | 0·053 |
| Mosandrite | 0·0432 | 0·0024 | 0·056 |
| Aegerine | 0·0253 | 0·0015 | 0·060 |
| Astrophyllite | 0·0140 | 0·0007 | 0·050 |
| Catapleite | 0·0132 | 0·0009 | 0·068 |
| Nepheline | 0·0010 | 0·0004 | 0·400 |
| Felspar | 0·0006 | 0·0003 | 0·500 |
It will be noticed, that with a few exceptions, the value of the ratio increases as the percentage of uranium diminishes. This is probably due to the relative importance of lead originally entangled in the minerals at the period of their crystallisation. Thus it would seem in the case of nepheline and felspar that almost the whole of the lead found was originally present, while that which has since been generated is very small in comparison. Minerals with so little uranium contain too much occluded lead to be reliable, and are, of course, valueless in age-estimations. When sufficient uranium is held by a mineral, the lead [Pg 165] generated becomes increasingly important, until the original amount is of negligible consequence. There is always the possibility that in some of the richer minerals larger quantities of lead were occluded than in felspar, but the agreement among the ratios renders this improbable. Rejecting all the results after that of biotite because of the low percentage of uranium, and omitting that of the first specimen of pyrochlore because the estimation could not be verified owing to lack of material, the mean ratio is 0·046. Replacing the uranium percentage by its time-average value, the ratio becomes 0·045 and the corresponding age 370 million years. It may be thought somewhat arbitrary to select certain results preferentially, but in view of the interpretation placed upon them the choice is not unfair, nor without justification.
Most of the available evidence drawn from radioactive minerals has now been passed in review. As yet it is a meagre record, but, nevertheless, a record brimful of promise. Radioactive minerals, for the geologist, are clocks wound up at the time of their origin. After a few years’ preliminary work, we are now confident that the means of reading these time-keepers is in our possession. Not only can we read them, but if they have been tampered with and are recording time incorrectly, we can, in most cases, detect the error and so safeguard ourselves against false conclusions.
[Pg 166]
The discrepancy between the geological and radioactive methods of estimating time—Uniformity of the rate of decay of uranium—Joly’s criticism—Comparison of the two time-scales—Doubtful assumptions made in the geological arguments—Possibility of reconciliation no longer hopeless.
Of the various methods which have been devised to solve the problem of the earth’s age, only two, the geological and the radioactive, have successfully withstood the force of destructive criticism. The other arguments may be dismissed without further discussion, as in every case their cogency has been vitiated by the detection of a fundamental error. From the mists of controversy which for half a century have hung over the subject, the two hour-glass methods alone emerge, and the final issue must be fought out between them. In the one the world itself is the hour-glass, and the accumulating materials are salt, the sedimentary rocks and calcium carbonate. Three concordant sets of results may be drawn from this triple scheme of measurement, but it must not be supposed that they are altogether independent. Each set of data is intimately related to the others and all stand or fall together. In the other case the accumulating materials are helium and lead, and the hour-glass is constituted by the minerals in which they [Pg 167] collect. Provided that the field-evidence is clear and convincing and that the relative geological age of a mineral specimen can be determined, the construction of an exact and precise time-scale is a task which can be dealt with successfully in the laboratory. The problem has advanced from the qualitative to the quantitative stage, and for the first time in historical geology accurate measurement founded on delicate experimental work has become possible.
It is a matter for regret that confidence in this pioneer work has been shaken by the advocates of the geological methods of attack. The surprises which radioactivity had in store for us have not always been received as hospitably as they deserved. With the advent of radium geologists were put under a great obligation, for the old controversy was settled overwhelmingly in their favour. But the pendulum has swung too far, and many geologists feel it impossible to accept what they consider the excessive periods of time which seem to be inferred. That there exists a serious discrepancy obviously points to a flaw in the underlying assumptions of one or the other or both of the methods. Evidently we are at the parting of the ways. The fundamental assumptions on which the arguments are based cannot both be right. One of them must be rejected. Which is it to be? Let us consider each in turn, and discuss the consequences of the two possible forms of reconciliation.
The only assumption which can reasonably be called into question is that of uniformity, and it is involved equally in both calculations. It is here, at the root of the problem, that the discrepancy really lies. [Pg 168] If we favour the uniformity of geological processes—a well-worn doctrine which has done good service—then we must reject uniformity of radioactive disintegration. Joly has drawn attention to the latter possibility. He asks: Is it assured that the parent substance, uranium, has always in the past disintegrated at the rate determined by its present average life period? As far as we know, the rate of decay for substances of rapid transformation is constant, and independent of temperature and pressure changes. On the grounds that a large number of radioactive bodies decay at a constant rate, it is believed that this constancy is a definite attribute of all the radioactive elements. In the case of uranium this assumption cannot be proved for periods commensurate with its half-life period. On analogy with the behaviour of the shorter lived elements, it is probable that had we lived in Cambrian times and experimented with Archean uranium-bearing minerals just as has been done during the last decade, the half-life period would then have been exactly the same as we now find it—about 5400 million years. In the case of radium emanation there can be no doubt that experiments in Cambrian times would have given results concordant with ours. It would be as unphilosophic to doubt this as to believe that the laws of physics and chemistry vary with time. The difference between uranium and its daughter elements, the difference which suggests to Joly a possible distinction, is simply one of origin. We are in complete ignorance of the genesis of uranium. It is not impossible that, owing its origin to some process other than atomic [Pg 169] transformation, the particular distribution of intrinsic energy among its atoms may not be such as to maintain a constant rate of decay. At the moment of its birth every radioactive atom has a definite expectancy of life, and when a sufficiently large number of atoms is under observation a definite fraction disintegrates every second. There is this difference in the case of parent elements. As they become aged with reference to the time of their origin, they are not reinforced by the addition of fresh, newly-born atoms, as are the other members of each series. Joly’s supposition seems to be that in the absence of this reinforcement the uranium in its early stages may possibly disintegrate more rapidly than it does now. However, it is not found that the younger atoms of the short-lived elements are, on an average, more prone to rapid decay than are their older companions. Whether an element is in equilibrium with the higher members of its family, or whether it is separated from them, its transformation proceeds with unaffected regularity.
It is very improbable that reconciliation will be found in the supposition of a progressive retardation of the rate of decay of uranium. There are three possibilities. Uranium may have disintegrated in the past exactly as it now does; or it may have decayed more slowly or more rapidly. The latter two alternatives do not favourably commend themselves. There is no evidence which can be cited in their support. On the other hand, the hypothesis of constant change is deduced from a well-established series of experimental facts, and is remarkably [Pg 170] in accordance with the general phenomena of radioactivity. Uranium, in other respects, does not present any anomaly, and with regard to the mechanism of its decay physicists are not likely to regard it as an exceptional case without very definite reasons for doing so. The discordance between the time estimates drawn from the rates of geological and radioactive changes cannot be held to constitute a sufficient reason for rejecting current opinions unless it is conclusively demonstrated that the geological estimates are beyond question. In the future the case for uranium may be established more securely, when the dynamics of atomic disintegration, and the conditions upon which the distribution of unstable atoms depends, becomes more intimately understood. At present there is only one means of testing the constancy of uranium decay, but unfortunately it affords only negative evidence. The range of α-particles from a radioactive element is connected in some way with its rate of decay. If then, uranium in the past disintegrated more rapidly, the radius of its particular pleochroic halo ought to record the difference. It is improbable that any variation—assuming for the moment that there were a variation—could be detected even in the most favourable cases.
We now turn with a double interest to the geological estimates. If it can be shown that they ought to be largely increased, as Chamberlin and a few other geologists believe, then not only is a reconciliation at once made possible, but, in turn, the constancy of uranium decay is [Pg 171] placed beyond doubt. Little need be added to the discussion in Chapter VI. It was there indicated that one factor previously over-looked—the average height of the continents in geological time—very largely controls the rate of denudation and therefore of sedimentation. Let us make an attempt to discover how past rates must be related to those of the present to make possible a complete reconciliation. In Fig. 17 the discrepancy is illustrated graphically by comparing the respective time scales from the close of the Archean (gneiss and granite phase) to the present day. All the sediments of which relics have remained to us are in this way taken into consideration. Lying buried in the Archean, the base of the record is obscured beyond recognition by the prevalence of metamorphic and plutonic igneous rocks. The extent to which the earliest sediments have been lost in the evolution of the earth’s crust, and the part they have played in the genesis of granites and gneisses are questions which betray our ignorance and offer food merely for wild speculations. These possibilities, however, do not touch the immediate point at issue, for in their time relations they lie outside the limits to which this discussion is restricted.
The curve A is plotted strictly against the maximum observed thickness of sediments, and corresponding to it is the sedimentation line A′ to which is granted 300 million years. According to these two graphs the greatest error lies beyond the Cambrian. The average rate of denudation and of sediment accumulation must now be nine times that of the pre-Cambrian periods, but if post-Cambrian is compared with present the ratio is reduced to two-and-a-half. In the B series, the lead ratios are plotted in a straight line and the stratigraphical column is extended in accordance with the palæontological evidence that pre-Cambrian time is at least as long as that which has elapsed since the beginning of the Cambrian. On this basis present rates are four times the average for post-Archean time. [Pg 172]
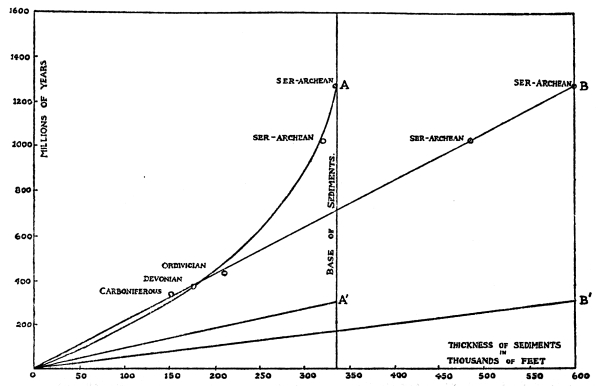
Fig. 17.
Geological Time Scales.
[Pg 173] Assuming that the true time-scale lies somewhere between the extremes of A and B, we are led to two conclusions which, if accepted, greatly lessen the severity of the discordance between A and A′ and B and B′. From A and A′ it appears that pre-Cambrian denudation took place much more slowly than has since been typical. This proposition is in complete accordance with the view that the pre-Cambrian continents, when viewed in the light of the reconstructed geographies of the later periods, were of limited area and restricted elevation. From B and B′ the broad time conception of palæontology gains further support, and the existence of great gaps in the pre-Cambrian succession is suggested. It is well known that the most important unconformities of the whole geological record are to be found in the imperfect succession of the earliest formations. It is impossible to do more than guess at the duration of time periods which are without their sedimentary equivalents. On the most extreme assumption, the Algonkian sediments should be represented not by 82,000 feet, but by more than 300,000 feet. Unwilling though we may be to consider the record imperfect to this incredible degree, it is of importance to point out that had such [Pg 174] an immense thickness existed in successive periods of time, the sodium content of the ocean would not necessarily be in any way different from what it is. The same amount of primary rock material may have been broken up, but instead of a reassorting of the materials to form three successive sets of sediments (on a rough average) we would be obliged to postulate six or more repetitions of the sorting process. The question need not be pursued farther. The suggestion here put forward is, that in the limitations of pre-Cambrian geography and in the imperfection of the sedimentary relics of those remote times, the discrepancy which is peculiar to the pre-Cambrian finds an adequate explanation.
If this be allowed, all that remains is to decide whether it is inconsistent with geological principles to assert that the modern hour-glass is running at two-and-a-half to four times its average rate. The decision depends largely on the broad point of view from which geological interpretation proceeds. From the standpoint of Catastrophism little progress was made. Uniformity proved a great advance, but in detail it is apt to lead us astray if applied too dogmatically. Modern interpretation is based on the more philosophic conception of Evolution, and in place of the earlier idea which was insisted upon by the older physicists—that changes have been such as would accompany a gradual running down of the earth’s internal kinetic energy—the form of development now favoured is that of cycles of phenomena, recurring in their broad features again and again and not necessarily hampered in their activity by any progressive diminution in [Pg 175] the store of available energy. Igneous action, deposition of sediments, marine transgression and recession, are all rhymic phenomena and the factor common to each one, whether as cause or effect, is earth-movement.
The conditions of the present day cannot then be accepted as representing average conditions, unless it were by a happy accident. Amidst all the details of earthquakes and volcanic eruptions some great cycle is now running its course, and only in relation to the particular phase of the cycle under which we happen to pursue our investigations will our conclusions be strictly tenable. Although we cannot hope to judge the exact place which the present takes in the larger scheme of terrestrial activity, yet in comparison with the past, the present epoch would seem to approach just those extremes most favourable to a high rate of denudation, and to a rapid accumulation of sediments. Marine recession, brought about by deepening of the ocean basins, and raising of the land areas have together brought about continental expansion and elevation. The vulcanism of the present day, whether regarded as a closing phase of a period of igneous action, or as the initiation of a new cycle, probably affords an example of more than average intensity and violence. The weathering capacity of rain must be enhanced in proportion to its content of dissolved acid gases, and this in turn is conditioned by the prevalence of vulcanism. Still another factor leading to higher rates, though of a different category, is due to recent glaciation. Over wide areas easily eroded deposits are exposed, the areas being generally those which would resist denudation [Pg 176] most successfully. In Fennoscandia, for example, four-fifths of the pre-Cambrian shield is buried beneath a thin covering of moraine.
It is not suggested that present rates have never before been reached, but only that they are characteristic of the more intense phases of denudation rather than of average conditions. If this be granted, reconciliation of the rival time estimates is no longer hopeless. There can be no doubt that agreement will never be brought about by the more convincing testimony of experimental demonstration. It must be almost entirely a question of interpretation. An attempt has been made to show that in the geological evidence there is nothing impossibly at variance with the dictates of the radioactive minerals. With the acceptance of a reliable time-scale, geology will have gained an invaluable key to further discovery. In every branch of the science its mission will be to unify and correlate, and with its help a fresh light will be thrown on the more fascinating problems of the Earth and its Past.
[Pg 177]
1 gram of radium in equilibrium with emanation, Ra. A B and C generates heat at the rate of 132 calories per hour (85% due to α-particles).
| e = charge on α-particle | = 9·3 × 10⁻¹⁰ E.S. units. |
| = 3·1 x 10⁻²⁰ E.M. ” | |
| m = mass of α-particle | see Table below. |
| v = velocity of α-particle | |
| N = number of α-particles liberated from | |
| 1 gram of radium | |
| = 3·4 × 10¹⁰ per second. | |
Energy E transformed per second is given by—
| E = ½ ∑ | Nmv² | × e |
| e |
| = | Ne | ∑ | mv² |
| 2 | e |
| Element. | v Cms. per sec. |
mv²/e E.M. Units. |
|---|---|---|
| Radium | 1·56 × 10⁹ | 4·78 × 10¹⁴ |
| Ra. emanation | 1·70 × 10⁹ | 5·65 × 10¹⁴ |
| Ra. A | 1·77 × 10⁹ | 6·12 × 10¹⁴ |
| Ra. C | 2·06 × 10⁹ | 8·37 × 10¹⁴ |
Substituting these values, we have—
Ne = 3·4 × 10¹⁰ × 3·1 × 10⁻²⁰ = 10·5 × 10⁻¹⁰ E.M. units.
| ∑ | mv² | = 10¹⁴(4·78 + 5·65 + 6·12 + 8·37) |
| e | ||
| = 24·9 × 10¹⁴ E.M. units; | ||
| whence E | = 4·73 × 10⁹ ergs per hour. [Pg 178] | |
| Now 4·19 × 10⁷ ergs = 1 gram-calorie. |
||
| ∴ E | = 113 calories per hour. | |
Uranium—
N = number of helium atoms liberated from 1 gram of radium alone = 3·4 × 10¹⁰ per second.(Rutherford and Geiger, 1908).
The equilibrium ratio of radium to uranium is 3·4 × 10⁻⁷. Hence for each gram of uranium in equilibrium the number of atoms produced amounts to—
3·4 × 10¹⁰ × 3·4 × 10⁻⁷ × 8 per second
= 29·1 × 10¹¹ per year.
Now the number of helium molecules, and therefore of atoms, in 1 cc. of the gas at N.P.T. is 2·72 × 10¹⁹.
The annual production of helium must consequently be
| 29·1 × 10¹¹ |
| 2·72 × 10¹⁹ |
i.e. 10·7 × 10⁻⁸ ccs., or 1·88 × 10⁻¹¹ grs. per gram of uranium.
An experimental determination gave 10·6 × 10⁻⁸ ccs. (Strutt, 1910).
Thorium—
The ionising power, or the energy of the α-particles from 1 gram of thorium, is 0·325 of that from 1 gram of uranium, each element being in complete equilibrium.
Average range of α-particles from thorium and its products = 5·4 cms.
Average range of α-particles from uranium and its products = 4·3 cms.
The average thorium α-particle is therefore 1·25 times as energetic as the average uranium α-particle.
Hence the actual production of α-particles or helium atoms from thorium is only 0·325/1·25 = 0·26 of that of uranium. Experimental determinations 0·23 (Strutt, 1910), 0·27 (Rutherford and Geiger, 1910). [Pg 179]
(1) The number of α-particles emitted from 1 gram of radium per second (n = 3·4 × 10¹⁰) is equal to the number of atoms disintegrating per second.
If N is the number of atoms in 1 gr. radium, then λ, the fraction which transforms per second, is given by—
| λ = | n |
| N |
The number of atoms in 1 gr. hydrogen is
6·24 × 10²³,
and as the atomic weight of radium is 226 times that of hydrogen,
| N = | 2·76 × 10²¹. |
| ∴ λ = | 1·25 × 10⁻¹¹ gr. per sec. |
| = | 3·94 × 10⁻⁴ gr. per year. |
| Half-Life period = | 0·69315 | = 1760 years. |
| λ |
(2) 1 gr. of radium is in radioactive equilibrium with 0·58 cubic millimetre, or 5·7 × 10⁻⁶ grs. of emanation (atomic wt. = 222).
If λ₁ = 2·085 × 10⁻⁶ is the fraction of emanation transforming per second, we have—
| λ = | 5·7 × 10⁻⁶ × λ₁ |
| = | 1·19 × 10¹¹ gr. per sec.; |
| whence—Half-Life period = 1850 years. | |
The earlier values given for the half-life period were 1760 and 2000 years, but the lower figure seems most accurate, with 1850 years as a probable value.
The half-life of uranium would then be—
| 1850 | = 5400 million years. |
| 3·4 × 10⁻⁷ |
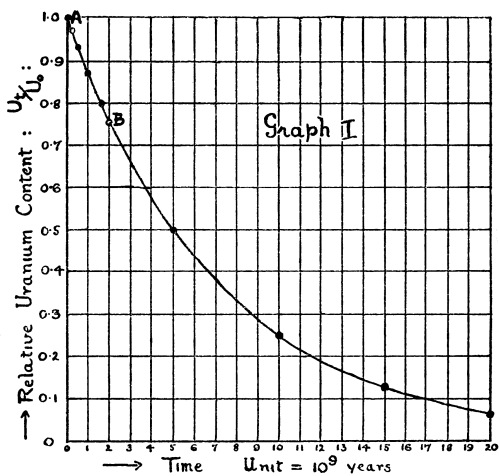
Graph I represents the rate of decay of uranium—according to the exponential law—
Uₜ = Uₒ e-λₜ.
There is one rate of decay which, if it remained constant throughout the time t, would have a total effect equivalent to that produced by [Pg 181] the actual slowly decreasing rate of decay. This average rate is represented by some point on the curve, and the corresponding quantity of uranium, Uₘ, is the time-average. Equating the amount of uranium transformed in each case, we have—
Uₒ - Uₗ = λUₘt
| whence Uₘ = | Uₒ - Uₜ | (a). |
| λt |
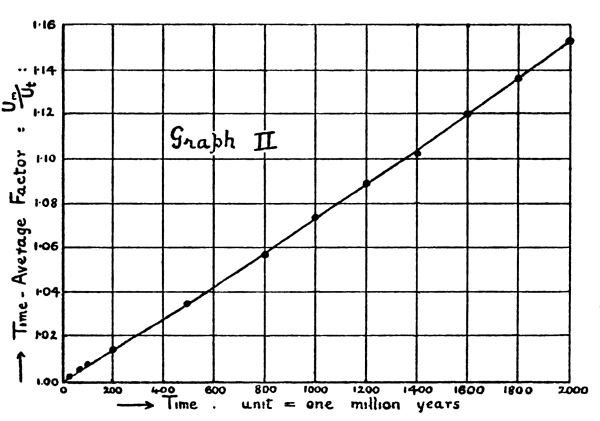
For periods less than 2000 million years, the time-average is nearly equal to the arithmetic mean of Uₒ and Uₗ.
| Uₘ = | Uₒ + Uₗ | (b). |
| 2 |
The value of Uₘ for 2000 million years is,
| according | to | equation | (a), | equal | to | 0·874 Uₒ |
| and ” | ” | ” | (b) | ” | ” | 0·879 Uₒ. |
[Pg 182] The ages of minerals rarely exceed 1500 million years, and therefore the error involved by using equation (a) in preference to (b) is quite negligible.
In Graph II the ratio Uₘ/Uₜ, i.e. the factor by which the present uranium content of a mineral must be multiplied in order to obtain the true time-average, is plotted against time.
An approximation to the age, t, of a mineral is afforded by the ratio Pbₜ/Uₜ. From the graph the factor corresponding to this time can be obtained, and thence the time-average. This in turn can be utilised to give the more correct age represented by Pbₜ/Uₘ.
A more straightforward method of correction is as follows:
Uₒ can be determined from known quantities according to the following equation:
| Uₘ = | Uₜ + Pbₜ + Heₜ |
| = | Uₜ + 1·15 Pbₜ; |
| whence, Uₘ = | Uₗ + 0·575 Pbₜ. |
The age of the mineral is then given by the ratio Pbₜ/Uₘ, or directly from Graph II.
(cited on p. 130)
| Acid Rocks— | Ra. per gram of rock | ||
| Granite, | Mozambique | 5·84 | × 10⁻¹² grs. |
| ” | ” | 2·61 | ” |
| ” | ” | 1·77 | ” |
| ” | N. Nigeria | 3·09 | ” |
| ” | Rhodesia | 2·43 | ” |
| ” | Transvaal | 2·12 | ” |
| ” | South Africa | 1·81 | ” |
| ” | ”” | 2·73 | ” [Pg 183] |
| Basic Rocks— | |||
| Basalt, | Mozambique | 0·94 | × 10⁻¹² grs. |
| Dolerite | ” | 0·85 | ” |
| Gabbro | ” | 1·07 | ” |
| Norite | ” | 0·54 | ” |
| Ultrabasic Rocks— | |||
| Composite analysis of 10 specimens from Scotland, | |||
| New Zealand, Africa, and Canada | 0·51 × 10⁻¹² grs. | ||
[Pg 184]
[Pg 190]
THE RADIOACTIVE ELEMENTS
URANIUM FAMILY
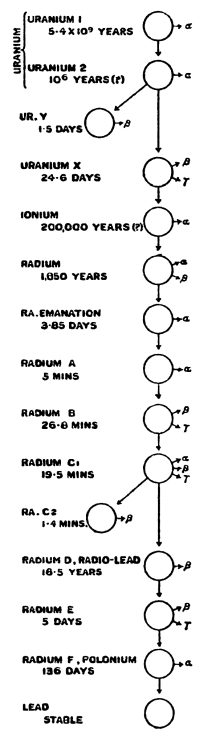
ACTINIUM FAMILY
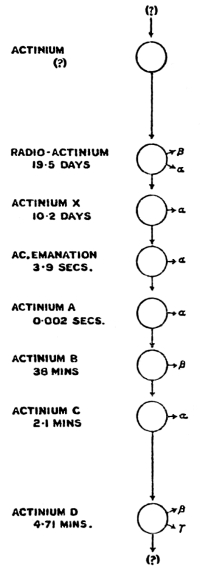
THORIUM FAMILY
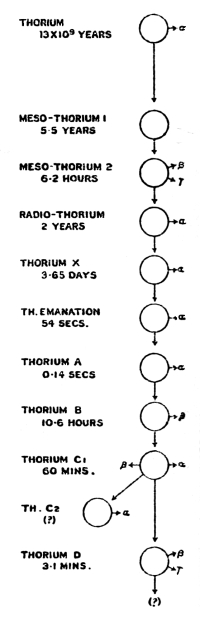
[Pg 191]
WILLIAM BRENDON AND SON, LTD.
PRINTERS, PLYMOUTH
Footnotes:
[1] A myriad = 10,000.
[2] The post-glacial layers number about 7000.
[3] Cf. Rates of deposit of the Cretaceous sediments of Colorado, p. 47.
[4] Cf. Rates of deposit of the Cretaceous sediments of Colorado, p. 47.
[5] Cf. Rates of deposit of the Cretaceous sediments of Colorado, p. 47.
[6] Cf. Rates of deposit of the Cretaceous sediments of Colorado, p. 47.
[7] Spread evenly over the land areas.
[8] Rate of denudation.
[9] Spread evenly over the land areas.
[10] Rate of denudation.
[11] Leith finds 12·5-22 million cubic miles.
[12] About 8%.
[13] Atomic recoil 6%, β-rays 4%, γ-rays 5%, approximately.
[14] The number of rocks examined in each case is given in the first of each pair of columns.
[15] Equivalent to 3,000,000 grs. of uranium.
Transcriber‘s Notes:
Deprecated spellings or ancient words were not corrected.
The illustrations have been moved so that they do not break up paragraphs and so that they are next to the text they illustrate.
Typographical and punctuation errors have been silently corrected.