

ONE OF A SERIES ON
UNDERSTANDING THE ATOM
Nuclear energy is playing a vital role in the life of every man, woman, and child in the United States today. In the years ahead it will affect increasingly all the peoples of the earth. It is essential that all Americans gain an understanding of this vital force if they are to discharge thoughtfully their responsibilities as citizens and if they are to realize fully the myriad benefits that nuclear energy offers them.
The United States Atomic Energy Commission provides this booklet to help you achieve such understanding.

Edward J. Brunenkant
Director
Division of Technical Information
This smiling youngster in the chute of a large whole body counter has just emerged from the opening (beyond her feet) of a hollow tank of scintillation liquid, where she lay while the radioactivity in her body was being “counted.” In a minute she will step into her slippers (on the ramp, right) and be ready for play. The sensitive, heavily shielded radiation-detecting equipment shown has many uses that are described in this booklet.

John H. Woodburn teaches chemistry at Walter Johnson High School in Rockville, Md. In the past he taught at Michigan State University, Illinois State Normal University, and Johns Hopkins University. He received his A.B. from Marietta College, his M.A. from Ohio State University, and his Ph. D. from Michigan State University. He is the author of the book Radioisotopes (J. P. Lippincott 1962), which is a student’s introduction to this subject.

Frederick W. Lengemann is associate professor of radiation biology at New York State Veterinary College, Cornell University. He received his B.S. and M.N.S. from Cornell and his Ph. D. from the University of Wisconsin. He has been research associate in radiation biology and assistant professor of chemistry at the University of Tennessee and formerly was a biochemist with the Atomic Energy Commission, Division of Biology and Medicine.
By JOHN H. WOODBURN
and FREDERICK W. LENGEMANN
Whole body counters are sensitive radiation detecting and measuring instruments that provide information not easily obtainable otherwise about that most important of all chemical systems, the human body. They can do this because, strange as it may seem, every person who ever lived is slightly radioactive.
Quickly, accurately, and painlessly, whole body counters reveal the kinds and amounts of radioactive substances that have accumulated in the body from natural sources, from man-made fallout, or from tracer isotopes given for medical purposes. They count emissions from these radioactive materials, as do other kinds of instruments known as “counters”.
In contrast to devices that disclose concentrations of radioactivity in a small area or a particular organ, whole body counters usually are used to total up the burden of radioactivity in all parts of a human body. They also are distinguished from many radiation detecting instruments with the same general purpose by their large size, their heavy shielding, and their sensitivity to low levels of radioactivity.
Whole body counters are useful in many studies of physiological activity in living persons and animals. They have proved valuable in calculating the radiation absorbed by victims of overexposure to radioactive materials. They can show a doctor how much of his patient’s body is fat and how much lean. Whole body counters also gave medical scientists 2 clues to the relation of potassium deficiency to muscular dystrophy and other diseases. And new medical and scientific uses are being found regularly.
The need for an instrument that would measure whole body radioactivity was first felt in the 1920s when the hazardous nature of radium was recognized. Other sorts of instruments had to be used to estimate the amount of radium that some factory workers inadvertently had absorbed while painting luminous watch dials with a radium-containing coating. (See pages 15 and 16.) But instruments then available were without adequate shielding to eliminate background radiation, and so the measurement efforts were of limited value.
It was not until the 1950s that new types of radiation-detecting instruments were designed, making use of the discovery that some crystals, liquids, and plastics give off light when struck by gamma rays (one form of radioactive emission). Two principal types of instruments have been developed to detect these emissions in human tissues.
The most common whole body counter employs a sodium iodide crystal as the radiation detector. The person being examined usually sits in a tilted chair in a room that has thick steel walls to absorb background radiation. During the counting period, the crystal is centered a few inches above the subject. This type is useful for examination involving low levels of radiation or emissions from more than one kind of radioactive atom.
In the other type the subject is surrounded by a tank of a liquid that detects gamma rays. This type is faster, but less sensitive, than the crystal type.
This booklet is intended to enable you to make imaginary visits to several whole body counters, to understand the scientific principles that are applied in their design, to learn the interesting ways they are used, and to appreciate the promise they hold for increasing our knowledge of ourselves and the world we live in.
In general, all whole body counters must have (1) a mechanism that reacts to the energy emitted by some kinds of disintegrating, or radioactive, atoms; (2) a device that displays or records these reactions; and (3) adequate shielding to exclude unwanted rays from other sources.
Figure 1 Types of whole body counters.
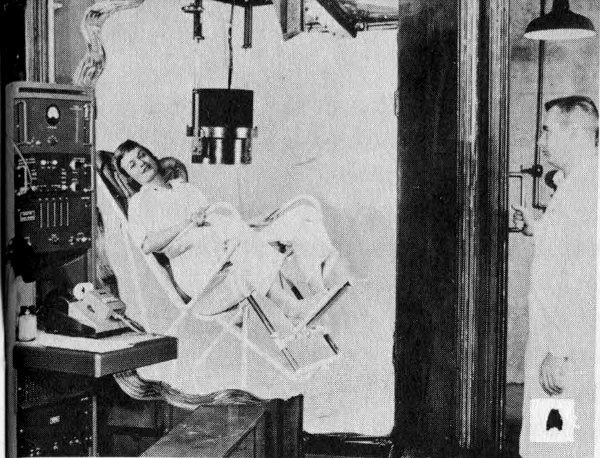
A The subject may be seated in a chair in an iron-shielded room and under a scintillation detecting crystal.
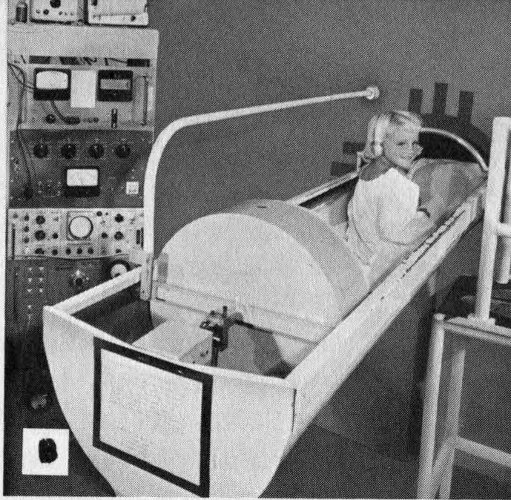
B The subject may lie in a bed that slides into the end of a hollow cylindrical tank filled with scintillation fluid.

C The subject may stand in a semicylindrical double-walled tank filled with scintillation fluid. (See Figure 2.)
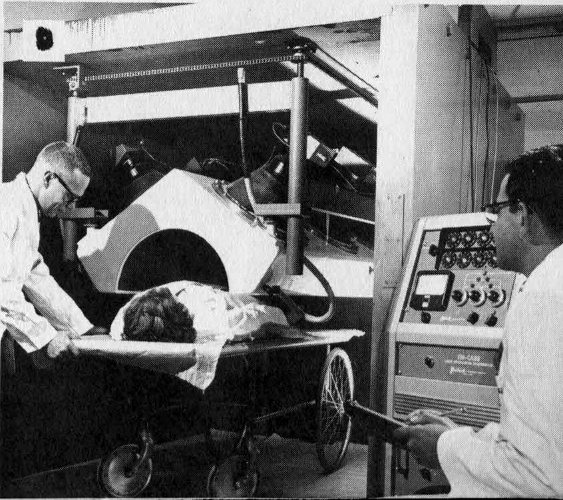
D The subject may lie on a wheeled cart and be wheeled beneath a shielded detecting crystal.
One of the first whole body counters was shown at an atomic science conference in Geneva, Switzerland, in 1955 (Figures 1D and 2). While it was on display, 4258 visitors to the meeting climbed a set of stairs to enter a 10-ton lead-walled chamber. Here they stood still for 40 seconds while the radioactive atoms in their bodies were being “counted”, or recorded. This device, because it was the first one persons could walk into, aroused great interest.

Figure 2 How a “walk-in” whole body counter, such as the one demonstrated at Geneva, works.
Shielding for the Geneva counter consisted of 3 inches of lead. Only the most energetic background gamma rays and 5 cosmic rays can penetrate this amount of shielding, and the number that do so remain almost constant during successive counting periods. This constant remaining “background” radiation level, once determined, could be subtracted from the recorded number of emissions to provide the correct radiation total from the body of each person examined.

Figure 3 Typical crystals and liquid materials used to produce scintillations for whole body counters and other radiation-detecting instruments. Scintillation counters provide much faster recording of radiation than Geiger counters, and are widely used in experiments with high-energy particle accelerators, as well as in whole body counters.
To detect the gamma rays emitted by radioactive atoms disintegrating within the body, whole body counters take advantage of a property of radiation that has been known since 1896. In that year the English physicist William Crookes discovered that X rays react with certain chemicals to produce fluorescence. A few years later a New Zealand-born physicist, Ernest Rutherford (later Lord Rutherford), found that this glow consisted of many tiny individual flashes or scintillations, each caused by the emission of a single alpha particle. He laboriously counted individual flashes by observing them through a magnifying glass. If you examine a luminous watch with a hand lens in 6 a dark room, you can see these fascinating scintillations, just as Rutherford saw them long ago.
Today, scientists have found several crystals, liquids, and plastics that are especially effective in showing scintillations caused by nuclear radiations. One of these substances, with the challenging name 2,2′-p-phenylene bis [5-phenyloxazole], often shortened to POPOP, was used in the scintillating liquid of the Geneva counter. How the flashes are detected can be appreciated by considering the infinitely small world of individual atoms and following a single atom as it disintegrates. (For a more complete explanation of radioactivity, see the companion booklet Our Atomic World in this series.)
Let us assume that we are looking at a single potassium-40 atom in the body of the person to be examined and that it is about to disintegrate. (Potassium-40 is naturally radioactive. It is the most abundant radioisotope in our bodies.) In any sizable portion of potassium-40, we know that half of the atoms will disintegrate over a period of 1.3 billion years, but, since this process is random, there is no way for us to know when any particular atom will do so. However, when it does, one of two alternative events will occur: either a beta particle (that is, an electron) will be ejected from the nucleus, creating an atom of nonradioactive calcium-40, or the nucleus will capture one of its own orbital electrons, resulting in creation of an atom of stable argon-40 and the emission of a gamma ray. (The beta emission process occurs in 89 out of every 100 disintegrations. See Figure 4.)
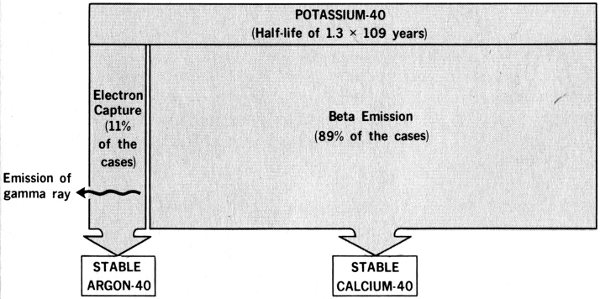
Figure 4 Comparison of potassium-40 disintegration methods.
Assume that the particular gamma ray is traveling in the direction of the scintillating liquid in the counter. Remember that the gamma ray is tiny in comparison with an atom, which is mostly empty space. Therefore, any one gamma ray probably will miss all the material part of the atoms in the body of the person being studied. Nor will it collide with anything as it passes through his clothes and the stainless steel tank. It also may fail to collide with any of the atoms in the molecules of scintillation liquid, of course. But let us assume that the one we are watching does make a hit there. Its total energy will be converted instantaneously to a flash of many bits or photons of light.
These photons radiate from the collision scene and strike a light-sensitive surface in one or more of the counter’s photomultiplier tubes, which have been placed where they can “see” the scintillation liquid. Energy transformations result, and a tiny pulse of electricity is originated. These photomultiplier devices are similar to the equipment in the familiar “electric eye” door openers. As their name suggests, photomultiplier tubes (see Figures 6 and 9) do more than merely respond to the light flashes produced in the scintillation liquid. They also amplify the weak electron disturbances into electrical pulses to operate meters that record each scintillation and count the total.
The Geneva counter recorded about 25% of the total gamma rays emitted by each subject. Since this sample was a constant proportion of the total body radiation, it could be converted to whole body measurements with about 97% reliability.
In addition to finding persons with actual body contamination among those counted at Geneva, the 1955 counter revealed some interesting sideline information. People who failed to remove radium-dial watches were soon spotted. And one small boy who had picked up a sample of uranium ore at a nearby exhibit “jammed” the instrument.
Each of the 25 persons who were found to have above-normal levels of radiation could recall having worked with 8 radium or some other radioactive substance at some time in the past.
A visit to this type of counter recalls the first glimmers of scientific insight that the radiation in the human body could be counted. In the early 1950s, Frederick Reines and Clyde L. Cowan, two scientists at the Los Alamos Scientific Laboratory, Los Alamos, New Mexico, built a large liquid scintillation counter hoping to prove or disprove that neutrinos really existed. Neutrinos are elusive, uncharged particles with essentially no mass. They had been predicted in theory nearly 20 years earlier to explain how beta particles of different energy levels can be emitted from atoms with apparently identical nuclei.
Figure 5

Dr. Frederick Reines (left) and Dr. Clyde L. Cowan (right), co-discoverers of the neutrino, lower a fellow worker into the first “whole body counter”, the scintillation assembly used in their experiment. Below, Dr. Wright Lanham, inside the counter, peers from the opening.

According to theory, neutrinos are created whenever negative beta-emitting atoms are produced. On this basis, Drs. Reines and Cowan were convinced that the fission of the nuclear fuel in the reactors of the Hanford atomic plant at Richland, Washington, should create high densities of neutrinos. They went to Hanford and set up an elegant neutrino-catching experiment that hinged on detecting and counting 9 gamma rays of definite energy. To accomplish this, they built a large liquid scintillation detector and shielded it from stray gamma radiation. As their work progressed, someone realized that the equipment was large enough to allow a person to crawl inside. After further research at the Atomic Energy Commission’s Savannah River Plant in South Carolina, they were successful in finding their long-sought neutrino. In doing so, they also developed the sort of instrument that can study the human body.
Figure 6 shows a person about to enter one version of a Los Alamos counter, the instrument’s 140-gallon tank of scintillation fluid, and 45 of its 108 photomultiplier tubes. When the instrument is in use, the tank slides into, and is shielded by, a 20-ton barrier of 5-inch lead.

Figure 6 A liquid scintillation whole body counter at Los Alamos National Laboratory, showing (left) a subject in the chute before it is slid into the shielded detector chamber. Below, the same instrument’s detecting assembly, showing the photomultiplier tubes, removed from the shielding.
The loading chute will hold a person 6 feet 4 inches tall and weighing up to 260 pounds. The subject lies in the chute as it slides into the counter. A lead plug behind his head closes the end of the cylinder to add shielding. Counters of this type have “panic buttons” with which subjects may signal if they become uneasy on being confined. Most counts are completed in less than 5 minutes, however, so the buttons are rarely used.
Since the detector fluid almost completely surrounds the subject when the chute is in place, this type of counter captures twice as large a fraction of the emitted gamma rays as does the Geneva type.
Each radioactive substance emits gamma rays with an energy level characteristic of that substance. Whole body counters are able to measure this specific energy spectrum, or “fingerprint”, and so identify the kind of atom producing the radiation.
The number of light photons produced in the scintillation fluid is proportional to the energy transferred by the incoming gamma rays. For example, gamma rays emitted by potassium-40 have 1.46 million electron volts (Mev) energy; those of cesium-137 have 0.660 Mev energy. When both these radionuclides are producing flashes of light in the scintillation fluid at once, the photomultiplier tubes produce two different strengths of electrical pulses. Electronic devices called multichannel pulse-height analyzers sort and record the number of each.
Data from whole body counters indicate that potassium-40 is the most abundant radionuclide in the human body. Our bodies also contain other naturally radioactive substances but the numbers of atoms usually present are so low (as with radium for instance) that they cannot be detected with whole body counters. Several man-made radionuclides also have found their way into body tissues and organs in quantities that sometimes are large enough to be detected and counted.
Measurement of disintegrating potassium-40 atoms in the tissues of a human body can be used to determine the total amount of potassium (both radioactive and stable) in the body. It is known that potassium-40 makes up 0.0119% of all potassium and that 11% of all disintegrating potassium-40 atoms emit high-energy gamma rays that are measurable by the counter.
The method of determining the amount of potassium in a human subject is to compare the number of gamma rays from a known amount of potassium placed in a phantom, or dummy body, with the number counted from the human subject (see photo on next page). Phantoms are artificial bodies, approximately the size, shape, and density of a human body, used for calibrating counters. They are designed 11 so the radioisotopes they contain have similar distribution to the distribution of the isotopes expected in the real body. This is how a test might work:
| Counts per minute from 140 grams of potassium in the phantom | 16,800 |
| Counts per minute with nothing in counter (background) | 12,000 |
| Net counts per minute from 140 grams of potassium | 4,800 |
| Counts per minute with a 77-pound boy in counter | 14,400 |
| Background cunts per minute | 12,000 |
| Net count per minute from boy | 2,400 |
| Calculated amount of potassium in boy | 70 grams |

Figure 7 Phantom used for iodine-131 studies. The radiation spectrum from the thyroid area in the neck is being obtained with a sodium iodide crystal, left.
We can appreciate the sensitivity of whole body counters by comparing the number of gamma rays recorded by the instruments with the total number emitted from the body being counted. The following data, also simplified, illustrate this comparison:
| Total atoms in 70 grams of potassium | 1.08 × 10²⁴[1] |
| Number of atoms of potassium-40 in 70 grams of potassium | 1.3 × 10²⁰ |
| Half-life of potassium-40 in minutes | 6.4 × 10¹⁴ |
| Number of potassium-40 atoms disintegrating | |
| per minute in 70 grams of potassium | 141,000 |
| Number of potassium-40 atoms disintegrating per minute with emission of measurable gamma rays | 15,510 |
| Number of counts recorded | 2,400 |
| Detection efficiency: 2,400 ÷ 15,510 = 15.5% |
Fatty tissues are known to have a low potassium concentration and muscle tissue a higher level. It is therefore apparent that potassium-40 determinations provide a way to indicate the amount of lean muscle in any individual, and indirectly the amount of fat. Estimates of the amount of fat based on the measurement of the specific gravity of the subject, often used in the past, have never been satisfactory. Not only do variable and unmeasurable air spaces change body density, but the process of submerging a person in a tank—to determine his specific gravity by the amount of water he displaces—is a clumsy and uncomfortable one.
Significant variations in potassium content have been found in persons suffering from muscle diseases or malfunctions. For example, a sharp drop in potassium content accompanies the profound muscle weakness that follows diabetic coma. Administration of potassium produces striking improvement in the condition known as familial periodic paralysis.
Whole body counter data from a study of muscular dystrophy and myotonia atrophica patients showed there is a gradual and progressive decrease of body potassium during the unrelenting courses of these diseases. Otherwise healthy children of muscular dystrophy patients, or their brothers and sisters, also may be deficient in potassium. By assisting in muscle research, whole body counters help doctors learn more of how potassium relates to muscle function and muscle health.
Whole body counting is an improvement over potassium determination based on chemical analysis of body fluids. If counters are not used, one way to measure body potassium is to inject a known quantity of potassium-42 (another radioactive form of potassium), wait until this has been uniformly mixed with the potassium already in the body, and then record the radioactivity of a volume of blood serum. From the degree of dilution of the injected potassium-42, the total body potassium can be calculated. This widely used method is uncomfortable for the patient since it involves use of syringes to inject and withdraw fluids. Because about 95% of the body potassium is inside the cells, rather than in fluids between the cells, this method may also be inexact if the mixing process does not continue long 13 enough. (See Radioisotopes in Medicine, another booklet in this series, for a full discussion of medical treatment with radioactive materials.)

Figure 8 A crystal whole body counter “iron room” under construction above, and in use.
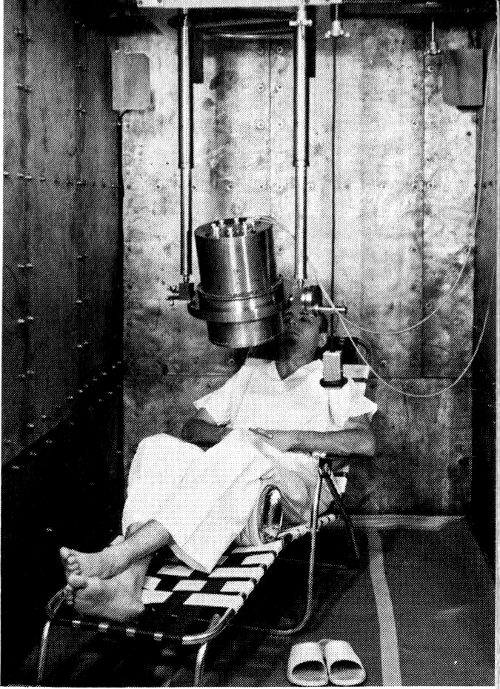
When we visit a crystal counter, shown under construction in Figure 8, walls of battleship steel 6 to 8 inches thick are the first things we see. Rather than using shielding only around the detecting instrument, as was done in the Geneva counter and early versions of the Los Alamos counter, crystal counters have shielding around the entire counting room. With this arrangement the instruments are available for adjustment and servicing.
This type of counter also uses a different detection device: a solid, rather than a liquid scintillator. A large crystal, usually of sodium iodide sensitized with thallium, is used to convert gamma rays to light photons.
Let us return to the shielding problem for a moment. Tanks of water, bricks, stone, and lead have been tried by scientists seeking effective, cheap, and convenient shielding. Some early counters were built deep underground in the hope of avoiding cosmic radiation. Radioactive elements are so widely distributed in the rocks, soil, water, and air, however, that there is no place where background radiation does 14 not exist. Not even the crystals, glass, or metals used in the detection system are free of radioactivity.
Pre-World War II surplus armor plate came to be the preferred shielding material. Thick slabs of battleship steel were available after the war at low cost. Furthermore, steel produced since the war may contain unwanted radioactivity originating in fallout from nuclear tests and make it undesirable for shielding. Sometimes cobalt-60 used as a tracer to measure deterioration of blast furnace walls causes problems in postwar steel, too, so old warship armor is used when possible.
Some whole body counters have additional shielding. In the counter at the Brookhaven National Laboratory, Upton, New York, the steel room is lined with ¼-inch lead sheets, covered by thin layers of cadmium and copper. The lead is intended to absorb the secondary X rays produced in the iron by the interaction of high-energy gamma and cosmic rays. The cadmium and copper absorb the secondary radiation that is similarly produced in the lead.
The doors of these rooms often weigh 6 tons or more. A special escape hatch was built into the counter room at the University of California at Los Angeles, to be used if the main door should be jammed by an earthquake. In newer whole body counting laboratories, such as the one at the National Institutes of Health, Bethesda, Md., the steel rooms are concealed in the interior design and are so pleasantly furnished that the patient scarcely is aware of the thick walls around him.

Figure 9 A sodium iodide crystal, right, and a cluster of 7 photomultiplier tubes that fit under it to record its scintillations.
Figure 9 shows a sodium iodide crystal used to react with the gamma rays that traverse it. To the left of the crystal is a cluster of seven photomultiplier tubes that “watch” for the scintillations, convert them to electrical pulses, and amplify them so they can be sorted, counted, and recorded. A trace of thallium added to the sodium iodide improves its scintillation properties.
In addition to being of convenient size and easy to maintain, crystal detectors have another advantage over liquid systems. The energy of the incident gamma rays from crystals is more accurately indicated by the quality of the flashes of light impinging on the photomultiplier tubes. If two or more radionuclides are emitting gamma rays, a crystal detector distinguishes between their energy levels with much more precision and sensitivity than does a liquid system. Crystal instruments separate gamma rays differing by no more than 0.05 Mev.
The energies of the gamma rays emitted by nuclides have all been determined and are listed in handbooks. A scientist can thus identify the data delivered by a multichannel pulse-height analyzer as coming from potassium-40, zinc-65, or any other nuclide.
Counters using sodium iodide crystals intercept, and therefore count, a much smaller fraction of the gammas emitted by the subject’s body than liquid systems, but they also pick up a smaller amount of background. When speed is important, the liquid counter is more effective, but the crystal counter is preferred when radionuclides emitting gammas of nearly the same energies are to be separated and counted.
Radium-226 in the human body poses unique problems for whole body counters. People who have accumulated this nuclide only because of the minute amounts occurring naturally in food and water have counts of only two or three disintegrating atoms per second, and this amount cannot be distinguished from background radiation. Whole body counters are useful, however, in diagnosing effects in persons who have been overexposed to radium. These include 16 persons who formerly were employed to paint watch dials with a luminous paint containing radium. (See table below.)

Figure 10 A group of radium dial painters at work in a watch factory in 1922. Almost all these employees have been identified and the living ones recently have participated in a study at Argonne National Laboratory to determine the extent of radium accumulation in their bodies. Whole body counters aided in their examinations.
Excerpts from case records of one research center show the high counts found in several patients and the source of the radium or thorium (a closely related element) that their bodies had taken up:
| Case | Body burden in disintegrating atoms per second |
|---|---|
| Born 1900, drank 210 bottles of “Radithor” in 1927[2] | 63,640 |
| Born 1897, drank approximately 78 ounces of “Radium Water” in 1932[2] | 32,780 |
| Born 1925, worked as radium chemist since 1946 | 14,800 |
| Born 1922, radium chemist for 7 years | 7,000 |
| Born 1898, two injections of “Thorotrast” for X-ray diagnosis | 3,300 |
| Born 1898, radium dial painter in watch factory, 1918 to 1921 | 72,500 |
| Born 1902, radium dial painter for 4½ months in 1924 | 1,924 |
Scientists at the Argonne National Laboratory, Argonne, Illinois, have attempted to improve crystal whole body counters so that they will be more useful in determining the amount of radium-226 in humans. Rolf Sievert at the Swedish Atomic Energy Commission also has studied the radium-226 detection. He devised a highly accurate whole body counter with 10 ion chambers arranged around a curved aluminum bed on which the subject rested. The instrument was installed below ground to reduce the interference of background radiation.
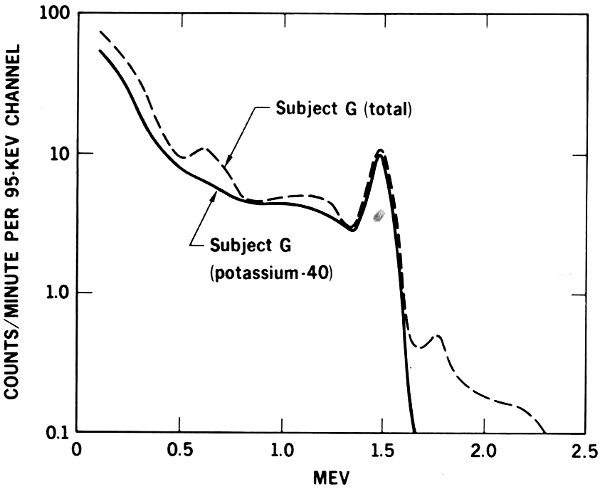
Figure 11 The spectrum of gamma radiation emitted by the body of a man three years after he accidentally inhaled radium-226. The solid line shows the normal radiation due to potassium-40, the dotted line the total from potassium-40 and radium-226.
In 1955, Charles E. Miller and L. D. Marinelli were measuring human potassium levels with the whole body counter at the Argonne laboratory. They were puzzled by finding several people who emitted 0.660-Mev gamma rays. Gamma rays of this energy, which are emitted by cesium-137, had not previously been detected in humans. To add to the perplexity, when the same persons were examined a few months later, the count of these gamma rays had increased. The Argonne findings indicated strongly that radiocesium, which is known to occur in fallout from nuclear explosions, was finding its way into people’s bodies. (See Fallout from Nuclear Tests, another booklet in this series.)

Long-lived radionuclides, particularly cesium-137, accumulate on plants.

Reindeer herds graze on lichens and mosses.

A substantial portion of the Laplanders’ diet is reindeer meat.

Here personal data and eating habits are recorded for each person to be counted.

A Laplander in a whole body counter.

The University of Helsinki (Finland) study revealed a close correlation between the consumption of reindeer meal and the Lapps’ body burdens of cesium-137. However, these levels did not exceed acceptable limits.

Figure 12 Using a whole body counter to determine radioactivity in milk.
The idea that this nuclide had entered the body with food later was tested by placing various foods in the whole body counter. All foods tested were found to contain some cesium-137, but beef and dairy products had highest levels. The radiation spectra of persons of the same age but different diet habits were compared, and correlation was found between their cesium-137 content and the amount of dairy products they ate.
In October 1960, Kurt Liden at the University of Lund in Sweden encountered evidence of the source of cesium-137 in humans. While he was using the whole body counter at the University’s Radiation Physics Department, Liden found several Norwegians whose bodies contained quantities of cesium-137 several times higher than previously recorded. He substantiated these data by counting 15 additional Norwegians from Oslo and 6 from Bergen. The Oslo residents averaged 21 nanocuries (abbreviated nc) of cesium-137 and the Bergen group 60 nc. Swedes averaged only 8 nc. (A nanocurie is one billionth of a curie, the standard unit of radioactivity.)
Curiosity regarding these high values in the Norwegians led him to investigate goat cheese, which Norwegians consume in larger quantities than Swedes. Goat cheese at that time showed a high cesium-137 content of 41 nc per kilogram. In northern Norway, near Bergen, another main food is reindeer meat, which was found to contain 28 nc of cesium-137 per kilogram, compared to 0.1 nc per kilogram in beef. These factors indicated that goat cheese and reindeer meat were responsible for the high cesium-137 count in the Norwegians.
Investigation of the soil in northern Norway revealed that it averaged 20 nc of cesium-137 per square meter. Only 21 thin covers of lichens grow in this region, and reindeer must graze over large areas to obtain sufficient food. It was obvious that cesium-137 from fallout was collecting on lichens and then was being concentrated in the bodies of the reindeer before they were killed for meat.
Similar studies have been conducted by scientists of the Pacific Northwest Laboratory at Richland, Washington, who since 1959 have been measuring radioactivity of plants and animals in Alaska. When they found high levels of cesium-137 in lichens and caribou, they became interested in the body burdens of fallout isotopes in the Eskimos.
A portable crystal type of whole body counter was used to measure the cesium-137 content of Eskimos at five villages. The Eskimos cooperated willingly; in some communities nearly everyone accepted the invitation to be counted. This table presents the results:
| Cesium-137 (in nanocuries) | ||||
| Village | Number of subjects | Minimum | Maximum | Average |
| Diomede | 12 | 8 | 35 | 22 |
| Barrow | 259 | 8 | 166 | 51 |
| Point Hope | 107 | 3 | 119 | 17 |
| Kotzebue | 132 | 17 | 518 | 138 |
| Anaktuvuk | 52 | 83 | 719 | 421 |
(For comparison, the average body burden of cesium-137 of residents of Richland, Washington, during this period was between 5 and 7 nanocuries.)
These data show that the Eskimos who lived inland at Anaktuvuk and ate heavily of caribou meat carried cesium-137 burdens up to 20 times greater than Eskimos who lived in the four villages along the coast and had more variety in their food. (See pages 18 and 19 for photo story of a similar project in Finland.)
For chemists or others working with arsenic, cyanide, or other chemical poisons, safety depends on recognizing the materials and keeping them where they belong. When accidents do happen and poisons are swallowed or breathed in, successful treatment requires that someone find out 22 exactly what and how much of the poisonous material was involved.
In the event of accidents in nuclear laboratories or reactors, it would be equally essential to identify accurately and quickly the quantity and kind of unstable nuclides the victim has absorbed. A reactor accident conceivably could add a unique hazard since neutron radiation might change the normally stable, nonradioactive atoms in the bodies of nearby workers into radioactive ones. Even gold or silver fillings in their teeth might become radioactive.
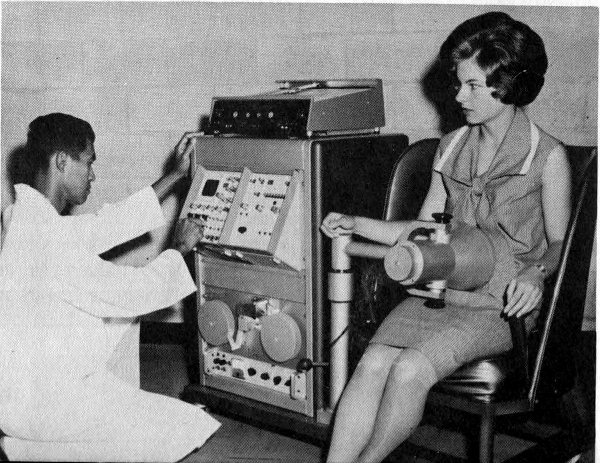
Figure 13 A portable whole body counter used for research as well as routine monitoring of personnel in an atomic reactor installation. Usually gamma-emitting radionuclides can be detected in 10 minutes. This counter is in almost constant use at the AEC’s National Reactor Testing Station in Idaho.
When human tissue (for example, hair) is bombarded by fast neutrons, sulfur atoms in molecules of proteins are converted to the radionuclide phosphorus-32. In atomic shorthand, this reaction is:
³²₁₆S + ¹₀n → ³²₁₅P + ¹₁p
or, in still more abbreviated form, ³²Sn,p³²P. Phosphorus-32 atoms do not emit gamma rays upon disintegration and so are not detected in standard whole body counters. Attempts are being made, however, to adapt some whole body counters to pick up the secondary radiations, called bremsstrahlung, that occur when the high-energy beta particles that are emitted by phosphorus-32 collide with other atoms.
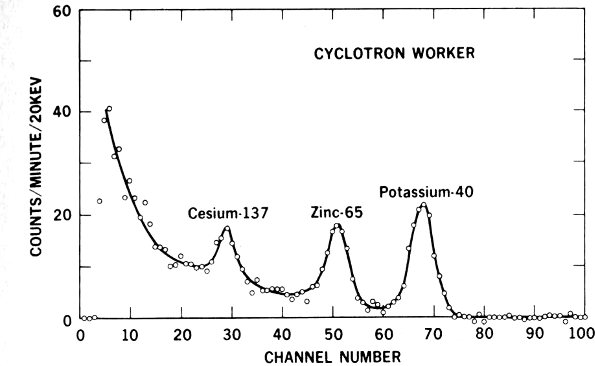
Figure 14 Whole body counter record of evidence that a cyclotron worker has picked up higher-than-average quantities of radioactive zinc-65. Note how the graph shows peaks at specific energy levels identifying the radionuclides.
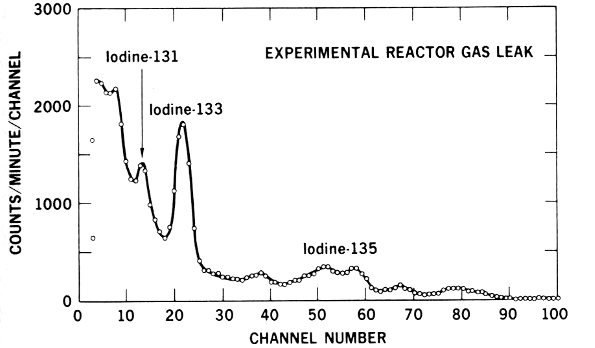
Figure 15 Graphic evidence that a radioactive gas accidentally inhaled by a worker at an experimental reactor contained 3 radioactive isotopes of iodine.
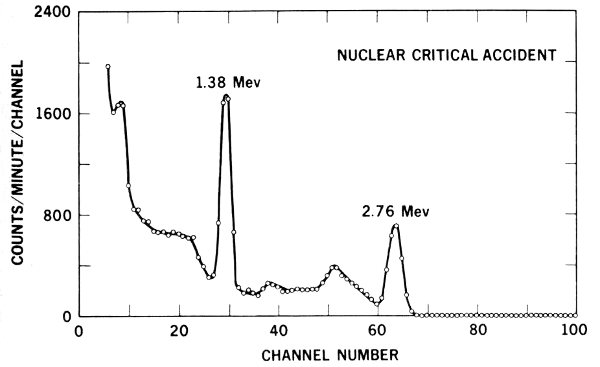
Figure 16 The number of 1.38-Mev gamma rays emitted by sodium-24 in the body of a reactor accident victim indicates he received about 900 rads of neutron irradiation.
Figures 14 through 16 illustrate the kind of data that whole body counters provide to help physicians care for people involved in accidents.
Similarly, neutron bombardment of natural sodium (²³Na) atoms in the body produces sodium-24. This reaction is written: ²³Na(n, gamma ray)²⁴Na. The 1.38-Mev gamma rays emitted by sodium-24 are detected effectively by whole body counters. Since a given neutron dose converts a known proportion of ²³Na atoms to ²⁴Na, it is possible to determine how much neutron exposure a worker has received by obtaining his body count of radioactive sodium.
Crystal type counters also were used in an interesting special case of excessive radiation exposure. Seven natives of the Marshall Islands were examined by the whole body counter at the Argonne National Laboratory in 1957. Another whole body counter, mounted in a Navy amphibious landing ship, was taken to Rongelap Atoll in the Marshalls several times to check on the health of all the residents of the atoll. These people had accumulated zinc-65 in their bodies as a result of contamination of crabs and other food items by fallout from the March 1954 atomic bomb tests at the Pacific Proving Ground. Although normal radioactive decay progressively reduced the total amount of radioactivity in the area, the Marshallese still were carrying this nuclide in their bodies after several years. (See Atoms, Nature, and Man, another booklet in this series, for a more complete report of this study.)
Study of the transfer of nutrients and other substances from an expectant mother’s body to that of her unborn child is one of the most challenging areas of biological research. A team of scientists headed by N. S. MacDonald has used a whole body counter at the University of California at Los Angeles to study one aspect of this problem by comparing the concentrations of radioactive materials in newborn infants, in babies who are born dead, and in tissues of the mothers-to-be.
In these studies the scintillation crystal was placed directly beneath a plastic bassinet holding the babies. Twenty-eight infants, 6 to 24 hours old, were counted for 45 minutes each. The only radionuclide found was the ever-present potassium-40.
The bodies of seven stillborn babies were counted for at least 10 hours each. More kinds of radionuclides were found than in the living babies, although the large counting time may have affected the results.
The same counting techniques were used with placental tissues from mothers of three of the stillborns. The placenta is the organ that nourishes an unborn child and through which substances from the mother’s bloodstream are exchanged with those in the baby’s blood. The graphs in Figure 17 show data from this experiment and illustrate the method of interpreting whole body counter data. When the counts per minute at each band of gamma-ray energy recorded from the placental tissues (b) were subtracted from corresponding values from the stillborns (a), it was found (c) that the placentas contained more of the isotopes ruthenium-103, ruthenium-106, and zirconium-95 than did the babies that had been nurtured by these placentas. The babies’ bodies contained more niobium-95 and potassium-40 than the placentas. Niobium-95 is produced by the radioactive disintegration of zirconium-95. This suggested that zirconium-95 atoms do not pass readily through the placenta, but, after they have decayed to niobium-95, they pass into the baby’s bloodstream easily.
Actually, the gamma-ray energies of zirconium-95 and niobium-95 are so similar that the counter cannot distinguish between them. The two isotopes, however, were separated chemically, and whole body spectra were prepared from samples of the pure elements. The spectrum (d) of pure zirconium-95 subtracted from that of pure niobium-95 was strikingly similar to the spectrum of “stillborn baby minus placenta” on the graphs. Cesium-137 was added to the synthetic spectrum to provide a reference mark at the 0.660-Mev point. This revealed that the ratio of cesium-137 to potassium-40 is lower in babies than in adults.

Figure 17 Results of experiment studying transfer of nutrients from an expectant mother to her unborn child.
Radioactive tracer atoms, either natural or purposefully built into molecules of vital materials like proteins, are revealing how these substances function in the body to produce 27 energy or to form new tissues. When we know accurately the normal totals and kinds of radioactive substances in the body, we can undertake new kinds of tracer studies without using large amounts of additional radiation. Small instruments called scanners (see Figure 18) usually are used to track tracer isotopes, but whole body counters are useful in special circumstances.
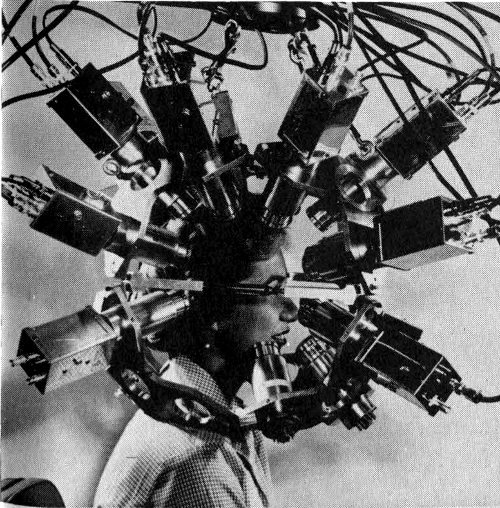
Figure 18 A multidetector positron scanner to record radiations with opposed pairs of detection crystals. Scanning devices are commonly used for noting the fate of tracer isotopes in medical diagnosis.
Two types of adaptations enable whole body counters to locate accumulations of radioactive materials in specific organs or small portions of the body. At the National Institutes of Health, Bethesda, Md., one counter is fitted with three rows of six 12-by-12-inch plastic blocks. Each block has four photomultiplier tubes to collect the scintillations from the crystal. The rows are curved so as to be equally distant from the patient’s body.
The current pulses from each of the 18 blocks can be fed individually into the pulse sorter, counter, and recorder. Thus the kinds and numbers of gamma rays from the sector of the patient adjacent to any block can be studied individually. Similarly, the path and speed of the administered materials can be followed by taking recordings from the blocks sequentially.
At the U. S. Naval Hospital in Bethesda, Md., a whole body counter is fitted with a crystal that can be moved at 28 controlled speed past the body of the person being studied (Figure 19). To increase its scanning efficiency, the crystal is fitted with a slit and a focusing device. Instruments record the body radioactivity visibly at timed intervals as the crystal moves along the patient’s body. A television screen enables the operator to observe the patient during the counting.

Figure 19 The U. S. Naval Hospital whole body counter, showing the moving crystal, left, and instruments, including the television screen used by the operator. The moving crystal makes it possible to use this whole body counter for scanning.
The role of iron in preventing one form of anemia has been clarified by using iron-59 as a tracer. Persons suffering from chronic infections or such blood diseases as leukemia and polycythemia vera have been checked for the amount of iron carried by their red blood cells. Cobalt-60 atoms have been substituted for stable cobalt in molecules of vitamin B-12 so that the way the body makes use of this vitamin can be studied. Similarly, the body’s use of sodium can be studied by labeling sodium chloride with sodium-22 29 and then administering solutions of the tagged salt orally or by injection.
Whole body counters used in tracer studies cause a minimum of inconvenience for the patient. Their sensitivity permits use of smaller quantities of radioactive material than is required with small scanning instruments.
Those are unusual jobs for whole body counters, however. Scanners or other types of instruments are used more typically in following tracer isotopes.

Figure 20 Dogs about to be examined in a whole body counter.
How do dogs accumulate fallout isotopes in their bodies? This question was answered effectively by placing dogs in whole body counters and comparing the count from radioactive strontium-emitted gamma rays originating in their bodies with the count from a masonite phantom dog containing a known amount of radioactive strontium-85. It was found that female dogs increase their strontium retention while they are nursing newborn puppies. Strontium is much like calcium, which is a major component of milk. One dog measured had broken a leg in a fight. The counter showed above-average strontium accumulation for this dog, and it was conjectured that strontium, a “bone-seeking” element, had followed calcium to the point of bone repair and new bone growth.

Figure 21 A wild deer under a whole body counter.
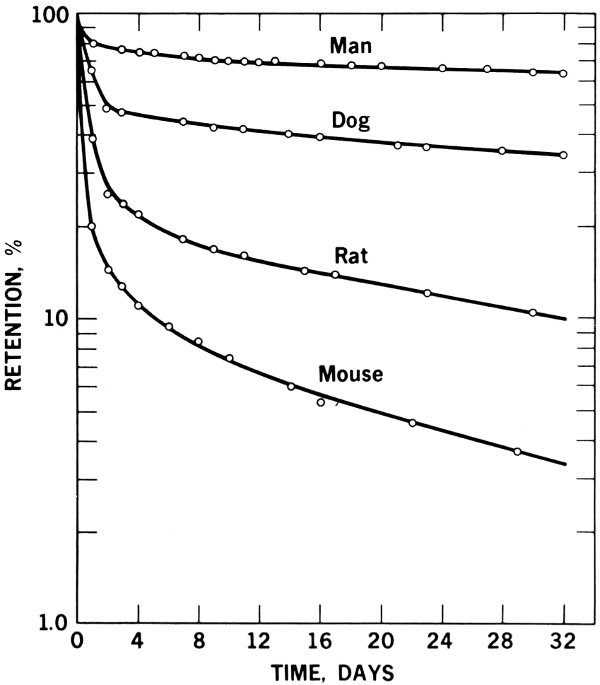
Figure 22 Graph of whole body counter survey of four different animal species, showing differences in their retention of orally administered radioactive zinc-65.
Figure 22 shows how animals differ in their retention of orally administered zinc-65, as revealed by a whole body counter. It is apparent that counters can be used to determine the differences in the metabolism of different animal species used for research. Standard data developed in this way can serve to reduce error that may occur if results from one species are used for interpretation of data for another species, such as man.
A University of Illinois project to breed meat animals with a high lean-to-fat ratio has been aided by whole body counters. The tendency to deposit fat seems to be inherited, and breeding stock with low fat content can be selected, using “muscle-seeking” potassium-40 to show the proportion of muscle in each potential parent. The Illinois counter is unique in being large enough to examine an adult steer (Figure 23). A similar counter at Cornell University has been used to study animals infested with internal parasites, comparing them with parasite-free animals. The counter revealed that a positive relationship exists between the level of parasite infestation and loss of iron-59-labeled blood from the digestive tract. The possibility of using this method to evaluate parasite-killing drugs is being considered. The Cornell counter is kept clean by covering the animals with plastic sheeting. The same counter also can serve human patients, who are positioned in a wheeled hospital stretcher. (See Figure 1D.)
This booklet has presented a sample of the ways whole body counters add to man’s knowledge and increase his ability to manage conditions and processes important to his health and well-being. Radioactive substances occur naturally within our own bodies and all other materials. Whole body counters measure this radioactivity, or any which may have been added from artificial sources.
We have seen how these sensitive instruments help to prove hypotheses difficult to verify otherwise and how they thereby may stimulate new and fruitful scientific experimentation. We have learned how whole body counters add to our knowledge of normal processes in healthy bodies and detect disease or abnormalities resulting from dangerous conditions.

Figure 23 A University of Illinois counter large enough to examine an adult steer, above. A Cornell University counter used for animal-parasite studies, below.


Figure 24 A trailer-mounted whole body counter used for research. School children were examined in a project to determine possible pathways by which radioactivity may enter the human body.
Each fact uncovered by whole body counters seems to trigger more penetrating research. This is how science advances. And with the advance of science comes knowledge on which we may build intelligent behavior and find solutions to problems affecting our lives.
Environmental Radioactivity, Merril Eisenbud, McGraw-Hill Book Company, Inc., New York. 1963, 430 pp., $13.50.
Radioactivity in Man: Whole Body Counting and the Effects of Internal Gamma Ray-Emitting Radioisotopes, George R. Meneely (Ed.). Charles C Thomas, Springfield, Illinois. 1965, 672 pp., $24.50.
Whole Body Counting, Proceedings of the Symposium on Whole Body Counting Held by the International Atomic Energy Agency at the Neue Hofburg, Vienna (June 12-16, 1961), National Agency for International Publications, 317 East 34th Street, New York 10016, 1962, 535 pp., $10.00.
How to Detect and Measure Radiation, Harold S. Renne, The Bobbs-Merrill Company, Inc., New York, 1963, 160 pp., $3.95.
An Introduction to Radiation Counters and Detectors, C. C. H. Washtell, George Newnes Ltd., London, 1960, 115 pp., $7.50.
Liquid Scintillation Counting, Proceedings of a Conference Held at Northwestern University (August 20-22, 1957), Carlos G. Bell, Jr., and F. Newton Hayes (Eds.), Pergamon Press, Inc., New York, 1958, 292 pp., $10.00.
Fundamental Nuclear Energy Research, A Special Report of the United Stales Atomic Energy Commission (December 1963), Superintendent of Documents, U. S. Government Printing Office, Washington, D. C. 20402, 407 pp., $2.50. Whole Body Counters as Medical Aids, pp. 11-13; Acute Whole Body Irradiation Effects, pp. 35-41; Chronic Whole Body Irradiation Effects, pp. 42-48.
Radioactive Contamination of Materials Used in Scientific Research, James R. DeVoe, Nuclear Science Series Report No. 34, National Academy of Sciences—National Research Council, Washington, D. C. 20418, 1961, $2.00. Appendix VIII, The Negotiations and Developmental Work on Low Activity Glasses for Use in Whole Body Counters, pp. 109-115.
Liquid Scintillation Counting of C¹⁴ and H³ Labeled Amino Acids and Proteins, M. Vaughan and others, Science, 126: 446 (Sept. 6, 1957).
Phosphorescence in Liquid Scintillation Counting of Proteins, R. J. Herberg, Science, 128: 199 (July 25, 1958).
Suspension Counting of Carbon-14 in Scintillation Gels, B. L. Funt and A. Hetherington, Science, 125: 986 (May 17, 1957).
Liquid Scintillation Counting of Aqueous Solutions of Carbon-14 and Tritium, J. Shapira and W. H. Perkins, Science, 131: 414 (Feb. 12, 1960).
35Alone in the Dark with a Panic Button: Purdue’s Whole Body Counter, Martin Mann, Popular Science, 181: 90 (October 1962).
Counter as a Test Instrument, W. H. Bucksbaum, Electronics World, 68: 48 (November 1962).
Spiral Capillary Plastic Scintillation Flow Counter for Beta Assay, B. L. Funt and A. Hetherington, Science, 129: 1429 (May 22, 1959).
Available for loan without charge from the AEC Headquarters Film Library, Division of Public Information, U. S. Atomic Energy Commission, Washington, D. C. 20545 and from other AEC film libraries.
Understanding the Atom: Radiation Detection by Scintillation, 30 minutes, black and white, sound, 1962. Produced by the Educational Broadcasting Corporation under the direction of the AEC’s Division of Isotopes Development. This semitechnical film describes the scintillation process. Solid and liquid scintillators are shown, a description of a photomultiplier is given, and the pulse-height analyzer principle is illustrated.
Human Radioactivity Measurements, 9 minutes, color and sound, 1958. Produced by AEC’s Los Alamos Scientific Laboratory. This film shows a method developed at LASL to monitor possible intake of radiation by personnel. The liquid scintillation counter is large enough to contain a man and sensitive enough to detect even the minute amounts of his natural gamma radioactivity.
Ionizing Radiation in Humans, 15 minutes, color and sound, 1958. Produced by AEC’s Argonne National Laboratory. Describes the design and operation of ANL’s whole body counter for determining identification, quantity, and location of internally deposited radioelements. Various techniques in accumulation of data are shown.
Liquid Scintillation Counting, 14 minutes, color and sound, 1958. Produced by the Jam Handy Organization for the U. S. Atomic Energy Commission. Describes the use of a liquid scintillator for counting low-energy beta emitters commonly used in biological and medical tracer experiments. Also describes counting techniques, how the counters work, and how a sample is prepared.
| Cover | courtesy Los Alamos Scientific Laboratory (LASL) |
| Figure 1 | A, Argonne National Laboratory (ANL); B and C; LASL; D, Cornell University. |
| Figure 2 | LASL |
| Figure 3 | Brookhaven National Laboratory (BNL) |
| Figure 5 | LASL |
| Figure 6 | LASL |
| Figure 7 | BNL |
| Figure 8 | LASL |
| Figure 9 | LASL |
| Figure 10 | Dr. Charles E. Miller, ANL |
| Figure 12 | LASL |
| Figure 13 | National Reactor Testing Station |
| Figure 18 | BNL |
| Figure 19 | National Naval Medical Center |
| Figure 20 | LASL |
| Figure 21 | Colorado State University |
| Figure 23 | Cornell University |
| Figure 24 | Pacific Northwest Laboratory |
| Center-spread | Professor J. K. Miettinen, University of Helsinki |
This booklet is one of the “Understanding the Atom” Series. Comments are invited on this booklet and others in the series; please send them to the Division of Technical Information, U. S. Atomic Energy Commission, Washington, D. C. 20545.
Published as part of the AEC’s educational assistance program, the series includes these titles:
A single copy of any one booklet, or of no more than three different booklets, may be obtained free by writing to:
USAEC, P. O. BOX 62, OAK RIDGE, TENNESSEE 37830
Complete sets of the series are available to school and public librarians, and to teachers who can make them available for reference or for use by groups. Requests should be made on school or library letterheads and indicate the proposed use.
Students and teachers who need other material on specific aspects of nuclear science, or references to other reading material, may also write to the Oak Ridge address. Requests should state the topic of interest exactly, and the use intended.
In all requests, include “Zip Code” in return address.
Printed in the United States of America
USAEC Division of Technical Information Extension, Oak Ridge, Tennessee
February 1967