
The cover image was created by the transcriber and is placed in the public domain.

The cover image was created by the transcriber and is placed in the public domain.
i
ii
iii
Mr. E. E. Austen, of the British Museum, has been good enough to undertake the translation of my book on “Venoms.” For the presentation of my work to the scientific public in an English dress I could not have hoped to find a more faithful interpreter. To him I express my liveliest gratitude for the trouble that he has so kindly taken, and I thank Messrs. John Bale, Sons and Danielsson for the care they have bestowed upon the preparation of this edition.
Institut Pasteur de Lille, A. Calmette, M.D. June 17, 1908. iv
v
In the month of October, 1891, during the rains, a village in the vicinity of Bac-Lieu, in Lower Cochin-China, was invaded by a swarm of poisonous snakes belonging to the species known as Naja tripudians, or Cobra-di-Capello. These creatures, which were forced by the deluge to enter the native huts, bit four persons, who succumbed in a few hours. An Annamese, a professional snake-charmer in the district, succeeded in catching nineteen of these cobras and shutting them up alive in a barrel. M. Séville, the administrator of the district, thereupon conceived the idea of forwarding the snakes to the newly established Pasteur Institute at Saigon, to which I had been appointed as director.
At this period our knowledge of the physiological action of venoms was extremely limited. A few of their properties alone had been brought to light by the works of Weir Mitchell and Reichard in America, of Wall and Armstrong in India and England, of A. Gautier and Kaufmann in France, and especially by Sir Joseph Fayrer’s splendidly illustrated volume (“The Thanatophidia of India”), published in London in 1872.
An excellent opportunity was thus afforded to me of vi taking up a study which appeared to possess considerable interest on the morrow of the discoveries of E. Roux and Behring, with reference to the toxins of diphtheria and tetanus, and I could not allow the chance to escape. For the last fifteen years I have been occupied continuously with this subject, and I have published, or caused to be published by my students, in French, English, or German scientific journals, a fairly large number of memoirs either on venoms and the divers venomous animals, or on antivenomous serum-therapeutics. The collation of these papers is now becoming a matter of some difficulty, and it appeared to me that the time had arrived for the production of a monograph, which may, I hope, be of some service to all who are engaged in biological research.
Antivenomous serum-therapy, which my studies, supplemented by those of Phisalix and Bertrand, Fraser, George Lamb, F. Tidswell, McFarland, and Vital Brazil, have enabled me to establish upon scientific bases, has now entered into current medical practice. In each of the countries in which venomous bites represent an important cause of mortality in the case of human beings and domestic animals, special laboratories have been officially organised for the preparation of antivenomous serum. All that remains to be done is to teach its use to those who are ignorant of it, especially to the indigenous inhabitants of tropical countries, where snakes are more especially formidable and deadly. This book will not reach such people as these, but the medical men, naturalists, travellers, and explorers to whom it is addressed will know how to popularise and apply the information that it will give them. vii
I firmly believe also that physiologists will read the book with profit. Its perusal will perhaps suggest to them the task of investigating a host of questions, which are still obscure, relating to toxins, their mode of action upon the different organisms, and their relations to the antitoxins. There is no doubt that in the study of venoms a multitude of workers will, for a long time to come, find material for the exercise of their powers of research.
At the moment of completing this work I would like to be allowed to cast a backward glance upon the stage that it marks in my scientific career, and to express my heartfelt gratitude to my very dear master and friend, Dr. Émile Roux, to whom I owe the extreme gratification of having been able to dedicate my life to the study of experimental science, and of having caused to germinate, grow, and ripen a few of the ever fertile seeds that he sows broadcast around him.
I am especially grateful to those of my pupils, C. Guérin, A. Deléarde, F. Noc, L. Massol, Bernard, and A. Briot, who have helped me in my work, while showering upon me the marks of their confidence, esteem, and attachment; to my former chiefs, colleagues, and friends of the Colonial Medical Staff, Drs. G. Treille, Kermorgant, Paul Gouzien, Pineau, Camail, Angier, Lépinay, Lecorre, Gries, Lhomme, and Mirville; and to my numerous foreign or French correspondents, George Lamb, Semple, C. J. Martin, Vital Brazil, Arnold, de Castro, Simon Flexner, Noguchi, P. Kyes, Morgenroth, J. Claine, Piotbey, and R. P. Travers, several of whom have come to work in my laboratory, or have obligingly procured for me venoms and venomous animals. viii
I have experienced at the hands of a large number of our ministers, consuls, or consular agents abroad the most cordial reception on repeatedly addressing myself to them in order to obtain the papers or information of which I was in need. It is only right for me to thank them for it, and to acknowledge the trouble that M. Masson has most kindly taken in publishing this book.
Institut Pasteur de Lille, A. Calmette. March 10, 1907. ix
| PAGE | ||
| Fig. 1.— | A. Skull of one of the non-poisonous Colubridæ (Ptyas mucosus) | 7 |
| B. Skull of one of the poisonous Colubridæ (Naja tripudians) | 7 | |
| C. Skull of one of the poisonous Colubridæ (Bungarus fasciatus) | 7 | |
| D. Skull of one of the Viperidæ (Vipera russellii) | 7 | |
| E. Skull of one of the Viperidæ Crotalinæ (Crotalus durissus) | 7 | |
| F. Skull of one of the Colubridæ Hydrophiinæ (Hydrophis pelamis) | 7 | |
| Fig. 2.— | A. Maxillary bone and fangs of one of the Viperidæ (Vipera russellii) | 8 |
| B. Maxillary bone and fangs of one of the Colubridæ (Naja tripudians) | 8 | |
| C. Maxillary bone and fangs of one of the Colubridæ (Bungarus fasciatus) | 8 | |
| D. Maxillary bone and teeth of one of the non-poisonous Colubridæ (Ptyas mucosus) | 8 | |
| Fig. 3.— | A. Fang of one of the Viperidæ (Vipera russellii) | 8 |
| D. Transverse section of the fang | 8 | |
| Fig. 4.— | B. Fang of one of the Colubridæ (Naja tripudians) | 9 |
| E. Transverse section | 9 | |
| Fig. 5.— | C. Fang of one of the Hydrophiinæ (Hydrophis pelamis) | 9 |
| F. Transverse section | 9 | |
| Fig. 6— | Three transverse sections of a poison-fang of one of the Colubridæ | 9 |
| Fig. 7— | Marks produced on the skin by the bites of different species of snakes | 10 |
| Fig. 8— | Poison-gland and fangs of a venomous snake (Naja tripudians) | 11 |
| Fig. 9— | Muscular apparatus and poison-gland of Vipera russellii | 12 |
| Fig. 10— | Muscular apparatus and poison-gland of Vipera russellii | 12 |
| Fig. 11— | Muscular apparatus and poison-gland of Naja tripudians | 13 |
| Fig. 12— | Muscular apparatus and poison-gland of Naja tripudians | 13 |
| Fig. 13— | Arrangement of the scales of the head in one of the non-poisonous Colubridæ (Ptyas mucosus) | 14 |
| Fig. 14— | Arrangement of the scales of the head in one of the poisonous Colubridæ (Naja tripudians) | 15 |
| Fig. 15— | Feeding a poisonous snake (first stage) | 18 |
| Fig. 16— | Feeding a poisonous snake (second stage) | 18 xiv |
| Fig. 17— | Capture of a Naja tripudians (first stage) | 19 |
| Fig. 18— | Capture of a Naja tripudians (second stage) | 20 |
| Fig. 19— | Hindu carrying two captured Cobras in “chatties” | 21 |
| Fig. 20— | Maxillary bone, mandible, and head of Cœlopeltis monspessulana | 23 |
| Fig. 21— | (1) Vipera berus; (2) Vipera aspis; (3) Vipera ammodytes; (4) Vipera ammodytes | 25 |
| Fig. 22— | Vipera aspis, from the Forest of Fontainebleau | 28 |
| Fig. 23— | Skull of Bungarus | 31 |
| Fig. 24— | Bungarus fasciatus (India) | 32 |
| Fig. 25— | Skull of Naja tripudians | 34 |
| Fig. 26— | Naja tripudians (Cobra-di-Capello) on the defensive, preparing to strike | 35 |
| Fig. 27— | Naja tripudians (Cobra-di-Capello) | 36 |
| Fig. 28— | Vipera russellii (Daboia) | 45 |
| Fig. 29— | Pseudocerastes persicus | 47 |
| Fig. 30— | Echis carinatus (India) | 48 |
| Fig. 31— | Ancistrodon hypnale (Carawalla, of Ceylon) | 50 |
| Fig. 32— | Lachesis okinavensis | 52 |
| Fig. 33— | Lachesis flavomaculatus | 55 |
| Fig. 34— | Skull of Dendraspis viridis | 65 |
| Fig. 35— | Skull of Causus rhombeatus | 68 |
| Fig. 36— | Skull of Bitis arietans (Puff Adder) | 70 |
| Fig. 37— | Bitis arietans (Puff Adder) | 71 |
| Fig. 38— | Bitis cornuta | 73 |
| Fig. 39— | Bitis rasicornis | 74 |
| Fig. 40— | Cerastes cornutus | 75 |
| Fig. 41— | Echis coloratus | 77 |
| Fig. 42— | Skull of Atractaspis aterrima | 79 |
| Fig. 43— | Skull of Glyphodon tristis (Australian Colubrine) | 83 |
| Fig. 44— | Pseudelaps krefftii | 85 |
| Fig. 45— | Pseudelaps harriettæ | 85 |
| Fig. 46— | Pseudelaps diadema | 85 |
| Fig. 47— | Diemenia psammophis | 86 |
| Fig. 48— | Diemenia olivacea | 86 |
| Fig. 49— | Diemenia textilis | 86 |
| Fig. 50— | Diemenia nuchalis | 87 |
| Fig. 51— | Pseudechis porphyriacus (Black Snake) | 87 |
| Fig. 52— | Denisonia superba (Copperhead) | 89 |
| Fig. 53— | Denisonia coronoides | 89 |
| Fig. 54— | Denisonia ramsayi | 90 |
| Fig. 55— | Denisonia signata | 90 |
| Fig. 56— | Denisonia maculata | 91 |
| Fig. 57— | Denisonia gouldii | 91 |
| Fig. 58— | Hoplocephalus bitorquatus | 94 |
| Fig. 59— | Notechis scutatus (Tiger Snake) | 95 |
| Fig. 60— | Skull of Acanthophis antarcticus (Death Adder) | 96xv |
| Fig. 61— | Acanthophis antarcticus | 97 |
| Fig. 62— | Rhynchelaps australis | 98 |
| Fig. 63— | Skull of Furina occipitalis | 99 |
| Fig. 64— | Furina occipitalis | 99 |
| Fig. 65— | Skull of Elaps marcgravii | 101 |
| Fig. 66— | Elaps fulvius (Harlequin Snake) | 105 |
| Fig. 67— | Head and Skull of Crotalus horridus (Horrid Rattle-snake) | 109 |
| Fig. 68— | Ancistrodon piscivorus (Water Viper) | 110 |
| Fig. 69— | Lachesis lanceolatus (Fer-de-Lance) | 112 |
| Fig. 70— | Lachesis neuwiedii (Urutù) | 116 |
| Fig. 71— | Sistrurus catenatus (Prairie Rattle-snake) | 121 |
| Fig. 72— | A. Horny appendage (rattle) of a Crotalus horridus | 122 |
| B. Horny appendage, longitudinal section | 122 | |
| C. Separated segments of the appendage | 122 | |
| Fig. 73— | Crotalus terrificus (Dog-faced Rattle-snake) | 123 |
| Fig. 74— | Crotalus scutulatus (Texas Rattle-snake) | 126 |
| Fig. 75— | Crotalus confluentus (Pacific Rattle-snake) | 128 |
| Fig. 76— | Crotalus cerastes (Horned Rattle-snake) | 130 |
| Fig. 77— | Skull of Hydrus platurus | 132 |
| Fig. 78— | Hydrus platurus | 133 |
| Fig. 79— | Hydrophis coronatus | 134 |
| Fig. 80— | Hydrophis elegans | 135 |
| Fig. 81— | Skull of Distira | 137 |
| Fig. 82— | Enhydrina valakadien (E. bengalensis) | 138 |
| Fig. 83— | Skull of Platurus colubrinus | 139 |
| Fig. 84— | Platurus laticaudatus (P. fischeri) | 140 |
| Fig. 85— | Collecting venom from a Lachesis at the Serotherapeutic Institute of São Paulo (Brazil) | 154 |
| Fig. 86— | Chloroforming a Cobra in order to collect venom at Pondicherry (first stage) | 155 |
| Fig. 87— | Chloroforming a Cobra in order to collect venom at Pondicherry (second stage) | 157 |
| Fig. 88— | Collecting Cobra-venom at Pondicherry (third stage) | 158 |
| Fig. 89— | Mongoose seized by a Cobra | 225 |
| Fig. 90— | Indian Snake-charmer at Colombo (Ceylon) | 230 |
| Fig. 91— | Indian Snake-charmer at Colombo (Ceylon) | 231 |
| Fig. 92— | Musical instrument used by Indian snake-charmers to charm Cobras | 232 |
| Fig. 93— | Vaccinating a horse against venom at the Pasteur Institute, Lille | 244 |
| Fig. 94— | Aseptically bleeding a horse, vaccinated against venom, in order to obtain antivenomous serum, at the Pasteur Institute, Lille | 245 |
| Fig. 95— | Technique of injecting antivenomous serum beneath the skin of the abdomen | 264 |
| Fig. 96— | Lactrodectus mactans | 275 |
| Fig. 97— | Scorpio occitanus | 277 |
| Fig. 98— | Scolopendra morsitans | 280xvi |
| Fig. 99— | Poison-apparatus of the bee | 281 |
| Fig. 100— | Interior of the gorget of the Bee | 282 |
| Fig. 101— | Synanceia brachio var. Verrucosa | 291 |
| Fig. 102— | Cottus scorpius (Sea Scorpion, or Father Lasher) | 292 |
| Fig. 103— | Scorpæna grandicornis | 293 |
| Fig. 104— | Scorpæna diabolus | 294 |
| Fig. 105— | Pterois artemata | 295 |
| Fig. 106— | Pelor filamentosum | 296 |
| Fig. 107— | Trachinus vipera (Lesser Weever) | 297 |
| Fig. 108— | Operculum and opercular spine of the Lesser Weever | 298 |
| Fig. 109— | Callionymus lyra (Dragonet) | 300 |
| Fig. 110— | Batrachus grunniens | 302 |
| Fig. 111— | Thalassophryne reticulata | 302 |
| Fig. 112— | Lophius setigerus | 303 |
| Fig. 113— | Serranus ouatabili | 304 |
| Fig. 114— | Holacanthus imperator | 305 |
| Fig. 115— | Tetrodon stellatus | 306 |
| Fig. 116— | Tetrodon rubripes | 306 |
| Fig. 117— | Chilomycterus orbicularis | 307 |
| Fig. 118— | Chilomycterus tigrinus | 307 |
| Fig. 119— | Silurus glanis | 308 |
| Fig. 120— | Muræna moringa | 310 |
| Fig. 121— | Salamandra maculosa (Spotted Salamander) | 314 |
| Fig. 122— | Triton marmoratus (Marbled Newt) | 314 |
| Fig. 123— | Cryptobranchus japonicus (Great Japanese Salamander) | 315 |
| Fig. 124— | Heloderma horridum | 322 |
| Fig. 125— | Ornithorhynchus paradoxus (Duck-billed Platypus) | 324 |
1
VENOMS.
A large number of animals possess special glandular organs capable of secreting toxic substances called venoms.
Sometimes these substances are simply discharged into the surrounding medium, and serve to keep off enemies (toad, salamander); sometimes they mingle with the fluids and digestive juices, and then play an important part in the nourishment of the animal that produces them (snakes); in other cases, again, they are capable of being inoculated by means of stings or teeth specially adapted for this purpose, and then they serve at once as a means of attack or defence, and as a digestive ferment (snakes, spiders, scorpions, bees).
An animal is said to be venomous when it possesses the power of inoculating its venom.
Venomous species are met with in almost all the lower zoological groups, in the Protozoa, Cœlenterates, Arthropods, Molluscs, and in a large number of Vertebrates (fishes, amphibians, and reptiles).
The reptiles are best endowed in this respect, and it is in this class of creatures that we meet with the species most dangerous 2 to man and to mammals in general. The study of their venom, too, is of considerable interest, since it should lead to the quest of means of protection sufficiently efficacious to preserve us from their attacks.
Venomous reptiles are not always easy to distinguish from those devoid of any inoculatory apparatus. For this reason both classes alike have at all times inspired mankind with a lively dread, which is displayed among the various races in legends and religious beliefs.
In ancient days the cult of the snake occupied a prominent place. In Genesis the serpent is the incarnation of the Evil One, tempting and deceitful. In Greece it was the symbol of wisdom and prudence. In Egypt it was associated with the Sacred Scarabæus and the flowers of the lotus to represent Immortality!
At Rome epidemics ceased when the snake sacred to Æsculapius was brought from Epidaurus.
According to Kraff, the Gallas of Central Africa consider the snake as the ancestor of the human race, and hold it in great respect.
In India the cult of the Seven-headed Naja, or serpent-god, was formerly almost as flourishing as that of Buddha. It is still regarded as a crime to kill a Cobra when it enters a hut; prayers are addressed and food is offered to it. Its presence is an omen of happiness and prosperity; it is believed that its death would bring down the most terrible calamities on whomsoever should have brought it about, and on his family.
Nevertheless, in the Indian Peninsula alone, the Cobra, the Krait, and a few other extremely poisonous species of snakes cause every year an average of 25,000 deaths. The number of fatalities from the same cause is likewise considerable in Burma, Indo-China, the Dutch Indies, Australia, Africa, the West Indies and Tropical America generally.
The temperate regions of the globe are less severely affected; 3 but in North America the Rattle-snake and the Moccasin are especially deadly.
In France the Common Viper abounds in Jura, Isère, Ardèche, Auvergne, Vendée, and the Forest of Fontainebleau. Three hundred thousand have been killed in twenty-seven years in the Department of Haute-Saône alone. Every year this snake causes the death of some sixty persons. Cow-herds, shepherds, and sportsmen fear it greatly, since it is very dangerous to cattle, sheep, and dogs.
Poisonous snakes are divided by naturalists into two great Families, the Colubridæ and Viperidæ, distinguished from each other by certain anatomical characters, and especially by the dentition.
The Colubridæ resemble harmless snakes, which renders them all the more dangerous.
They are divided into two groups: Opisthoglypha (ὄπισθεν, behind; γλυφὴ, a groove) and Proteroglypha (πρότερον, before; γλυφὴ, a groove).
The Opisthoglypha have the upper jaws furnished in front with smooth or non-grooved teeth, but behind with one or several rows of long, canaliculate teeth.
This group includes three Sub-families:—
A. The Homalopsinæ, having valved nostrils, placed above the snout.
B. The Dipsadomorphinæ, in which the nostrils are lateral in position, and the dentition is highly developed.
C. The Elachistodontinæ, which have but rudimentary teeth only on the posterior portion of the maxillary, on the palatine and on the pterygoid bones.
Almost all the snakes belonging to these three sub-families are 4 poisonous, but only slightly so. They are not dangerous to man. Their venom merely serves to paralyse their prey before deglutition takes place; it does not afford them an effective means of defence or attack.
All the Homalopsinæ are aquatic; they bring forth their young in the water, and are met with commonly in the Indian Ocean, starting from Bombay, and especially in the Bay of Bengal, on the shores of Indo-China and Southern China, from Singapore to Formosa, in the Dutch Indies, in Borneo, the Philippines, New Guinea and the Papuan Archipelago, and as far as the north of Australia.
The Dipsadomorphinæ comprise a large number of highly cosmopolitan genera and species, found in all the regions of the earth except the northerly portions of the Northern Hemisphere. None of these reptiles is capable of causing serious casualties among human beings, owing to the peculiarly defective arrangement of their poison-apparatus. I therefore do not think it worth while to linger here over their description.
The Elachistodontinæ are of even less importance; at the present time only two species are known, both of small size and confined to Bengal.
The Proteroglypha group of the Colubridæ is of much greater interest to us, since all the snakes belonging to it are armed with powerful fangs, in front of the upper maxillaries. These fangs, which are provided with a channel in the shape of a deep groove, communicate at the base with the efferent duct of poison glands, which are often of very large size.
The group is composed of two Sub-families:—
A. The Hydrophiinæ (sea-snakes), provided with a flattened oar-shaped tail. The body is more or less laterally compressed; the eyes are usually small, with circular pupils; the scales of the nose have two notches on the upper labial border.
The normal habitat of all the members of this sub-family is the 5 sea, near the shore, with the exception of the genus Distira, which is met with in the fresh water of a lake in the Island of Luzon, in the Philippines. They are frequently found in very large numbers in the Indian seas and throughout the tropical zone of the Pacific Ocean, from the Persian Gulf to the west coast of the American Continent, but they are entirely absent from the West Coast of Africa.
B. The Elapinæ (land-snakes), with a cylindrical tail, and covered with smooth or carinate scales. These serpents are frequently adorned with brilliant colours. Some of them (belonging to the genus Naja) have the faculty of expanding the neck in the shape of a parachute, by spreading out the first pairs of ribs when they are alarmed or excited: the breadth of the neck then greatly exceeds that of the head. They are distributed throughout Africa, Asia, and North and South America, and are also found in Australia, where almost all the snakes that are known belong to this sub-family.
The Family Viperidæ is characterised by a triangular head, which is widened posteriorly, and by the general aspect of the body, which is usually thick-set and terminated by a short tail. The bones of the face are movable. The præfrontal bone is not in contact with the nasal; the maxillary is greatly shortened and may be articulated perpendicularly to the ectopterygoid; it bears a pair of large poison-fangs, one on each side, and these are always accompanied by several teeth to replace them, folded back in the gum; these latter teeth come in succession to take the place of the principal tooth, when this is broken or falls out of itself when the snake sheds its skin.
The poison-fangs are not grooved, as in the Proteroglyphous Colubridæ; they are pierced by a perfectly formed canal, the upper end of which inosculates with the efferent duct of the corresponding poison-gland, while its lower extremity opens to the exterior a little above and in front of the tip. The latter is always very sharp. 6
The palate and lower jaw are furnished with small hooked teeth, which are solid and non-venomous.
With the exception of the species of Atractaspis, these snakes are all ovoviviparous. The majority are terrestrial; a few lead a semi-aquatic existence, while others are arboreal.
Their distribution includes Europe, Asia, Africa (with the exception of Madagascar), and North and South America. They do not exist in Australia.
They are divided into two Sub-families:—
A. The Viperinæ, in which the head, which is very broad and covered with little plates and scales, has no pit between the nose and the eyes;
B. The Crotalinæ (κρὁταλον, a rattle), in which the head is incompletely covered with scales, and exhibits a deep pit on each side, between the eye and the nostril.
Among snakes, the characters that serve as a basis for the determination of genera and species are the general shape of the body, especially that of the head, the arrangement of the cephalic scales, the cranial skeleton, and the dentition.
Cranial Skeleton.—The cranium is composed of a certain number of bones, the homologues of which are found in the mammalian skeleton; but the bones are complex, and subject to modifications according to the structure and habitat of each species.
The special arrangement of the bones of the face is above all characteristic of the poisonous snakes. Those forming the upper jaw, the palate and the mandibles or “inter-maxillaries” are movable upon each other and on the cranium. The upper and lower maxillaries are united by an extensile ligament and articulated with the tympanic bone, which permits the mouth to be opened very widely when the animal swallows its prey.
Dentition.—The non-poisonous snakes have two rows of teeth in the upper jaw—one external, the maxillary, usually composed of 7 8 from 35-40 small, backwardly curved teeth; the other internal, the palatine, which only numbers from 20-22 teeth, having the same curvature (fig. 1, A).

Fig. 1.—A, Cranial skeleton of one of the non-poisonous Colubridæ (Ptyas mucosus); B, cranial skeleton of one of the poisonous Colubridæ (Naja tripudians); C, cranial skeleton of one of the poisonous Colubridæ (Bungarus fasciatus); D, cranial skeleton of one of the Viperidæ (Vipera russellii); E, cranial skeleton of one of the Viperidæ Crotalinæ (Crotalus durissus); F, cranial skeleton of one of the Colubridæ Hydrophiinæ (Hydrophis pelamis).
In the poisonous snakes the maxillary bones are shorter, and the outer row is represented by a single long and tubular or grooved tooth (the fang), fused with the maxillary bone, which is itself movable (fig. 1, B, C, D, E, F).

Fig. 2.—A, Maxillary bone and fangs of one of the Viperidæ (Vipera russellii); B, maxillary bone and fangs of one of the Colubridæ (Naja tripudians); C, maxillary bone and fangs of one of the Colubridæ (Bungarus fasciatus); D, maxillary bone and teeth of one of the non-poisonous Colubridæ (Ptyas mucosus). (After Sir Joseph Fayrer.)
Certain species (Dipsas) have maxillary teeth which increase in size from front to rear; the longest teeth are grooved and serve for the better retention of prey, and also to impregnate it with saliva; but they are not in communication with the poison-glands.
The poison-fangs are normally covered with a fold or capsule of mucous membrane, in which they are sheathed. This fold conceals a whole series of reserve teeth in different degrees of development, which eventually become attached to the extremity of the maxillary when the principal tooth falls out or is broken (fig. 2).

Fig. 3.—A, Fang of one of the Viperidæ (Vipera russellii); D, transverse section of the fang.
Poison Apparatus.—The grooved or furrowed teeth in the Proteroglypha and the canaliculate teeth in the Solenoglypha are arranged, not for the purpose of seizing 9 prey, but in order to deal it a mortal blow by injecting the venom into its flesh.
In the normal position they lie almost horizontally, and exhibit no mobility of their own. But, when the animal prepares to bite, their erection is effected by the snake throwing its jaw back; and this movement, which is always very sudden, enables it at the same time to compress its poison-glands, by the aid of special constrictor muscles.
On examining the various species of poisonous snakes, we observe very sharply marked differences in the arrangement and dimensions of the teeth. Thus, in the Viperidæ they are long, extraordinarily sharp, and capable of producing deep wounds (fig. 2, A, and fig. 3). They are traversed by an almost completely closed canal, from the base, which communicates with the poison-duct, to the neighbourhood of the point, where it opens very obliquely on the convex surface (fig. 3, A and D).

Fig. 4.—B, Fang of one of the Colubridæ (Naja tripudians); E, transverse section.

Fig. 5.—C, Fang of one of the Hydrophiinæ (Hydrophis pelamis); F, transverse section.

Fig. 6.—Three transverse sections of a poison-fang of one of the Colubridæ, through A A, B B, C C; P P, pulp cavity; V V V, poison-canal (groove). (After C. J. Martin.)
In the Elapinæ sub-family of the Colubridæ, and especially in the Hydrophiinæ, the teeth are much shorter and simply grooved or canaliculate; that is to say, the canal communicates with the exterior throughout its extent by a narrow slit, which traverses the entire convex surface of the tooth (figs. 4, 5, and 6).
It does not follow from this that the bites of these reptiles are less dangerous; 10 the contrary, indeed, is the case, for their venom is infinitely more active.
These differences, as well as the particular mode of arrangement of the other little non-poisonous teeth in both jaws, enable us in many cases to recognise, by the mere appearance of the bite, the species of snake by which the bite has been inflicted.

Fig. 7.—Marks produced on the Skin by the Bites of different Species of Snakes.
I. Non-venomous Colubrine.—The bite is marked only by the imprint of from 35-40 small palatine or pterygoid teeth, and 20-22 upper maxillary teeth (on the outside of the foregoing) on each side.
II. Venomous Colubrine (Naja tripudians, Proteroglypha).—The bite exhibits 25 or 26 punctures from the pterygoid or palatine teeth, and, on each side, one or two, rarely three, circular wounds produced by the principal poison-fangs and by the reserve teeth.
III. Viperidæ (Solenoglypha).—The sole indication of the bite consists of 8 or 10 punctures from the palatine or pterygoid teeth, and one little round wound, on each side, produced by the poison-fangs.
(After Sir Joseph Fayrer.)
Fig. 7, above, shows how it is possible to distinguish the marks left by a non-venomous reptile, and by one of the Proteroglypha or Solenoglypha respectively.
Poison-glands.—The poison-glands occupy an extensive inter-muscular space behind the eyes, on each side of the upper jaw. 11 They are oval in shape, and may, in Naja tripudians for example, attain the size of a large almond (fig. 8).
Their structure is the same as that of the salivary glands of the larger animals. The poison that they secrete accumulates in their acini and in the efferent duct that opens at the base of the corresponding fang.
Each gland is surrounded by a capsule, to which are partly attached the fibres of the masseter muscle, which violently compresses it and drives the poison, just as the piston of a syringe would do, into the canaliculi or groove of the fang.

Fig. 8.—Poison-gland and Fangs of a Venomous Snake (Naja tripudians, Colubridæ). (Natural size.)
L, Lobe of the gland; D, poison-duct; F, fang attached to the maxillary bone; G G, gland; M, capsule of mucous membrane surrounding the fangs; R, reserve fangs; A A, muscular fascia covering the gland.
(After Sir Joseph Fayrer.)
In a few venomous snakes the gland is developed to such an extent that it extends as far as the first ribs.
The species, on the contrary, that have the poison-teeth placed in the hinder part of the mouth (Opisthoglypha) have glands but little developed.
Muscular Apparatus of the Head.—Figs. 9 and 10, 11 and 12 show the arrangement of the principal muscles that work the jaws and glandular organs in Vipera russellii and Naja tripudians, which respectively represent the most formidable types of Viperidæ and venomous Colubridæ.
It is not necessary to give a detailed description of each of these muscles. Let it suffice to point out that all contribute in giving the greatest elasticity to the jaws, and at the same time strength 12 13 14 sufficient to retain the prey and to cause it to pass from front to rear towards the œsophagus, by a series of alternate antero-posterior movements and analogous lateral ones. By means of these movements, which are participated in by the upper and lower maxillary bones, the palatines, mandibles or inter-maxillaries, and the pterygoids, the animal in a manner draws itself over its prey like a glove, since the arrangement of its dentition does not admit of mastication.

A A, Fascia covering the anterior and posterior temporal muscles; B, small gland; C, tendinous insertion of the fascia; D, poison-duct; E, poison-fang; F, reserve fangs; G, mandible; H, ectopterygoid muscle; J, poison-gland covered by the masseter; K, masseter inserted in the mandible; L, insertion of the temporal muscle; M, digastric muscle.

A A, Ectopterygoid muscle; B, præ-spheno-pterygoid muscle; C, intermandibular muscle; D, præ-spheno-palatine muscle; E præ-spheno-vomerine muscle; F, capsule of mucous membrane surrounding the fangs; G, long muscle of the neck.
Figs. 9 and 10.—Muscular Apparatus and Poison-gland of Vipera russellii
(Viperidæ). (After Sir Joseph Fayrer.)

A, Tracheo·mastoid muscle; B B, digastric muscle; C C, posterior temporal muscle; D D, anterior temporal muscle; E E, masseter; F, poison-gland, covered by the masseter and fascia; G, poison-duct; H, maxillary bone; I, neuro-mandibular muscle; J, costo-mandibular muscle.

A, Entopterygoid muscle; B, poison-gland; C, poison-duct; D, poison-fangs; E, præ-spheno-palatine muscle; F, præ-spheno-vomerine muscle; G, capsule of mucous membrane surrounding the fangs; H, præ-spheno-pterygoid muscle (which erects the fangs); J, inter-mandibular muscle; K, ectopterygoid muscle; L, long muscle of the neck (longus colli).
Figs. 11 and 12.—Muscular Apparatus and Poison-gland of Naja tripudians
(Colubridæ). (After Sir Joseph Fayrer.)

Fig. 13.—Arrangement of the Scales of the Head in one of the Non-poisonous Colubridæ (Ptyas mucosus). (After Sir Joseph Fayrer.)
A, Rostral scale; B, anterior frontals; B1, posterior frontals; C, vertical; D, occipitals; E, supra-ciliaries; F, temporals; L, M, nasals; N, loreals, or frenals; O, anterior oculars, or præ-orbitals; P, posterior oculars, or post-orbitals; Q, supralabials; G, median infralabial; H H, lateral infralabials; I K, mentals.
The enormous extensile power of the mouth and œsophagus thus enables snakes to swallow animals, the size of which is several times in excess of their own diameter.
Deglutition is slow and painful, but the gastric and intestinal juices are so speedy in action, that the digestion of the most 15 resistant substances rapidly takes place. The very bones are dissolved, and the fæces, which are voided some days later, contain only a few osseous remains and a felt-like material composed of hair or feathers.
Scales.—The skin of snakes, which is very elastic and extensile, is covered with scales, small on the back, and in great transverse plates on the entire ventral surface.

Fig. 14.—Arrangement of the Scales of the Head in one of the Poisonous Colubridæ (Naja tripudians, OR Cobra-di-Capello). (After Sir Joseph Fayrer.)
The shape and dimensions of the scales of the head are highly characteristic in each species. It is therefore necessary to know their names and the arrangement that they exhibit: these details are shown with sufficient clearness in figs. 13 and 14.
Coloration.—The colouring exhibited by the scales of snakes is governed generally by the biological laws of mimicry. It is therefore not a character of specific value, and may be modified several times in the course of the existence of the same reptile, according to the surroundings in which it is obliged to live. 16
“Nature,” write Dumeril and Bibron, “seems to have caused the tints and colours of snakes to vary in accordance with their habits and modes of life. Generally speaking, the colours are greyish or dull in species that are wont to live among sand, or which bury themselves in loose earth, as also in those that lie in wait on the trunks or large boughs of trees; while these hues are of a bluish-green, resembling the tint of the leaves and young shoots of plants, in snakes that climb among bushes or balance themselves at the end of branches. It would be difficult to describe all the modifications revealed by a general study of the colours of their skins. Let us imagine all the effects of the decomposition of light, commencing with white and the purest black, and passing on to blue, yellow, and red; associating and mixing them together, and toning them down so as to produce all shades, such as those of green, of violet, with dull or brilliant tints more or less pronounced, and of iridescent or metallic reflections modified by spots, streaks, and straight, oblique, undulating, or transverse lines. Such is the range of colours to be found in the skin of snakes.”
This skin is covered by a thick epidermis, which is periodically detached in its entirety, most frequently in a single piece. Before effecting its moult, the reptile remains in a state of complete repose for several weeks, as if asleep, and does not eat. Its scales grow darker and its skin becomes wrinkled. Then one day its epidermis tears at the angle of the lips. The animal thereupon wakes up, rubs itself among stones or branches, divests itself entirely of its covering as though it were emerging from a sheath, and proceeds forthwith in quest of food.
The moult is repeated in this way three or four times every year. 17
All poisonous snakes are carnivorous. They feed on small mammals (rats, mice), birds, batrachians, other reptiles or fish, which they kill by poisoning them by means of their fangs.
They almost always wait until their prey is dead before swallowing it.
Some of them are very fond of eggs, which they well know how to find in the nests of birds, and swallow whole.
When a poisonous snake wishes to seize its prey, or strike an enemy, it raises its head, and depresses the lower and elevates the upper jaw in such a way that the fangs are directed straight forward. Then, with the quickness of a spring when it is released, the reptile makes a sudden dart and strikes its victim. After inflicting the wound it draws back, doubles up its neck and head, and remains prepared to strike again.
So rapid is the action of the venom, that the wounded animal falls to the ground almost immediately; it is forthwith stricken with paralysis, and dies in a few moments. In most cases the snake holds it in its mouth until death ensues; the reptile then sets to work to swallow its victim, an operation which is always slow and painful.
In captivity poisonous snakes almost always refuse to take any food whatever. If it be desired to keep them for a long time, it is often necessary to resort to artificial feeding. For this purpose the snake is seized by the head by means of a strong pair of long forceps; it is then grasped by the neck with the left hand without 18 19 squeezing too hard (fig. 15), taking care at the same time not to give the body a chance of coiling itself round anything. Next, one or more lumps of beef or horse-flesh are introduced into the jaws, and gently forced down deep into the œsophagus by means of a glass rod, which is polished in order not to injure the mucous membrane. The œsophagus is then gently massaged in a downward direction, in order to cause the bolus of food to descend into the stomach (fig. 16). This operation is repeated every fortnight.

Fig. 15.—Feeding a Poisonous Snake, First Stage.

Fig. 16.—Feeding a Poisonous Snake, Second Stage.

Fig. 17.—Catching a Cobra-di-Capello (Naja tripudians), First Stage. (At the French Settlement of Pondicherry, in India.)
In this way, at my laboratory, I have been able to preserve, in perfect condition for more than two years, Indian Cobras and 20 Fers-de-lance from Martinique, taking care to keep them in a hothouse, at a temperature of about 82° to 86° F.
It is also very important to place inside the cases a vessel full of water, which should be frequently changed, for almost all snakes drink often and like to bathe for whole days at a time.

Fig. 18.—Catching a Cobra-di-Capello (Naja tripudians), Second Stage. (At the French Settlement of Pondicherry, in India.)
Within their reach should be placed in addition branches and rockwork, against which they rub at the moulting times, in order periodically to rid themselves of their scarf-skin.
While moulting, snakes must neither be touched nor fed, since to force them to take food at such a time would be fatal.
Snake-catching.—The capture of poisonous snakes, in order to keep them alive, can only be performed without danger by skilful persons, who are possessed of much coolness. 21
The best way of securing them is suddenly to pin the neck to the ground by means of a stick held horizontally, or a small two-pronged fork of wood or metal (fig. 17).
The stick is rolled along until close to the occiput (fig. 18). The animal can then be seized with the hand immediately behind the head, in such a way that it is impossible for it to turn and bite. It is then put into a wire cage, provided with a small movable trapdoor, with the fastening on the outside.
In this way poisonous snakes can be sent to a distance, and left without food for one or even two months, provided that they be kept in a place which is somewhat moist and sufficiently warm.
Fig. 19 shows how captured cobras are carried in India, in the environs of Pondicherry. They are enclosed in earthern chatties, or in baskets of plaited bamboo, which are provided with covers, and are very convenient for carrying snakes short distances.

Fig. 19.—Hindu carrying Two Captured Cobras in “Chatties.” 22
Poisonous snakes are especially common in the tropical zones of the Old and New Worlds. The species found in Europe are but of small size and not very formidable. In hot countries, on the other hand, they attain large dimensions, their venom is much more active, and, although they hardly ever attack man, and in most cases avoid him, they cause a considerable number of fatal accidents.
It is sometimes a rather difficult matter to recognise from the mere appearance of a snake whether it is poisonous or not. Naturalists themselves are occasionally deceived. It is therefore useful to learn to distinguish the most dangerous species by their external characters, and to know in what countries there is a risk of their being encountered.
Of the continents of the Old World, the poorest in poisonous snakes is Europe. The only species found there are a Cœlopeltis (belonging to the Sub-family Dipsadomorphinæ of the Opisthoglypha), and certain Viperinæ, which rarely exceed 75 centimetres in length.
Cœlopeltis, the cranial skeleton and head of which are represented in fig. 20, is characterised by a narrow, concave frontal shield, 23 projecting supraciliaries, short snout, large eyes, with round pupils, two poison-fangs at the back of the upper maxillaries, and a cylindrical body. The scales of the back are finely grooved, and in the adult slightly concave.

Fig. 20.—Maxillary, Mandible, and Head of Cœlopeltis monspessulana.
The coloration, olivaceous-brown, or deep red on the back, becomes on the ventral surface pale yellow with brown streaks, and from five to seven longitudinal series of small spots, which are blackish and edged with yellow on the sides.
The mean total length is 1,800 millimetres. The tail is somewhat tapering, and about 350 millimetres long.
The only European species is Cœlopeltis monspessulana, which is met with pretty commonly in France, in the neighbourhood of Montpellier, and Nice, near Valencia in Spain, and in Dalmatia. It is likewise found throughout North Africa, and in Asia Minor.
A second species, Cœlopeltis moilensis, occurs in Southern Tunis, Egypt, and Arabia.
The European Viperinæ belong exclusively to the Genus Vipera, the principal zoological characters of which are as follows:—
Head distinct from the neck, covered with small scales, with or without frontal and parietal shields; eyes small, with vertically 24 elongate pupils, separated from the labials by scales; nostrils lateral. Body cylindrical. Scales keel-shaped, with an apical pit, in from 19-31 rows; ventral scales rounded. Tail short; subcaudal scales in two rows.
The Genus Vipera is represented in Europe by several species, which are likewise found in Western Asia and North Africa.
These species are:—
V. ursinii, V. berus, V. aspis, V. latastii, and V. ammodytes. 1
Snout obtuse, soft on its upper surface, with the frontal and parietal shields distinct, the former about one and a half times as long as broad, and almost always longer than the parietals. A single series of scales between the eyes and the free margin of the lips.
Temporal shields smooth. Body scales in from 19 to 21 rows, strongly keeled on the back, less strongly on the sides.
Colour yellowish or pale brown above, grey or dark brown on the sides, sometimes uniform brown; spots more or less regular on the vertebral column, oval, elliptic or rhomboidal, dark brown or flecked with white, sometimes forming an undulous or zigzag band; two or three longitudinal series of dark brown or black spots on the sides; small dark dots running obliquely from the eye to the angle of the mouth; nose and lips white, and one or two dark angular streaks on the head; chin and throat yellowish; belly black, with transverse series of white or grey dots. No sexual differences in coloration.
Total length from 420-500 millimetres; tail 50-55. 25
Habitat: South-east France (Basses-Alpes); Italy (Abruzzi); Istria; Mountains of Bosnia; Plains of Lower Austria; Hungary (environs of Buda-Pesth).
Snout rounded, short and truncate; pupil vertically elongate; vertical diameter of the eyes equal to or greater than the distance separating them from the mouth; frontal and parietal shields distinct, the former as long as broad, usually shorter than the space separating it from the rostral shield; 6-13 scales round the eyes; one or rarely two series of scales between the eyes and the lips; nasal shield single, separated from the rostral by a naso-rostral shield; temporal scales smooth. Body scales in 21 rows (exceptionally 19 or 23), strongly keeled; 132-150 ventral shields; 38-36 subcaudals.
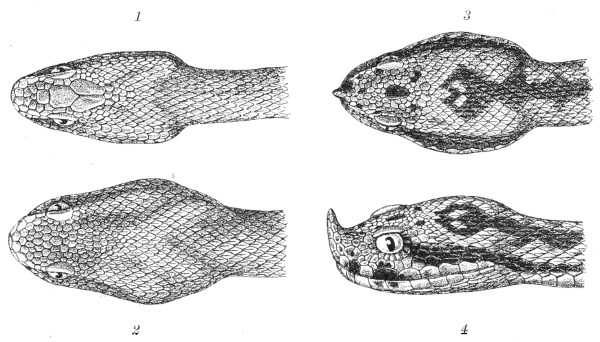
Fig. 21.—(1) Vipera berus; (2) Vipera aspis; (3, 4) Vipera ammodytes.
(Natural size.)
Colour very variable, grey, yellowish, olive, brown, or red above, 26 generally with an undulating or zigzag band along the vertebral column, and a series of lateral spots. A black spot shaped like a V, an X, or a circumflex accent, on the head. The tip of the tail is yellow or reddish. Some specimens are entirely black.
Total length from 350 to 700 millimetres; tail 75 to 90.
Habitat: Northern Europe, and especially the mountains of Central Europe; irregularly distributed in Southern Europe; Northern Spain and Portugal, Northern Italy, Bosnia, Caucasus.
This viper, which is very common in France, ranges as far as the Scandinavian Peninsula to about the 65th parallel of North Latitude. It is sometimes met with among the mountains at an altitude of about 6,500 feet (2,000 metres). It is found on heaths, in grass-lands, vineyards, and forests. Certain parts of the sandy moors of North Germany are literally infested with it. It abounds in the Jura, Isère, Ardèche, Auvergne, Brittany, Vendée, and the Forest of Fontainebleau.
It seeks its prey by night, and feeds on voles, small birds, frogs, lizards, and small fish. During the summer it shows a preference for moist places, often even remaining in the water, in which it swims with ease.
Light and fire attract it. It does not climb trees, but is frequently found coiled up on boughs of dead wood scattered on the ground.
When on the defensive, and preparing to bite, it throws its head back, and makes a sudden dart of from a foot to sixteen inches. If irritated it makes a sort of hissing noise.
To pass the winter it retires into the crevices of rocks or into old tree-trunks, where it entwines itself closely with a number of its congeners. In this way ten or fifteen vipers are frequently found together in the same hole.
In April, the whole company awakes, and copulation then takes place. The eggs are laid in August and September, and the young immediately crawl out of the shell, already prepared to bite, and 27 capable of finding their own food. Their length at birth amounts to 230 millimetres.
The two glands of an adult adder contain about 10 centigrammes of poison. This small quantity is sometimes sufficient to cause death; out of 610 persons bitten, Rollinger returns 59 deaths, or about 10 per cent.
In the departments of Vendée and Loire-Inférieure alone, Viaud Grand Marais has noted during a period of six years 321 cases of bites from adders, 62 of which were followed by death. In Auvergne, Dr. Fredet2 (of Royat) returns 14 cases, which caused 6 deaths.
Snout slightly turned up, soft and squarely truncate; vertical diameter of the eyes equal to the space separating them from the mouth; upper surface of the head usually covered with small, imbricate, smooth or feebly keeled scales, in 4-7 series, between the supraocular shields, which are prominent. The frontal and parietal shields are usually wanting; sometimes they are distinct, but small and irregular; the former are separated from the supraoculars by two series of scales; 8-13 scales round the eyes; two (rarely three) series of scales between the eyes and the labials; nasal shield single, separated from the rostral by a naso-rostral shield. Body scales in 21-23 rows, strongly keeled; 134-158 ventrals; 32-49 subcaudals.
Coloration very variable, grey, yellowish, brown, or red above, with a zigzag band as in V. berus. Usually a black U-shaped mark on the hinder part of the head, with a longitudinal black streak behind the eyes; upper lip white, or yellowish. Ventral surface yellow, white, grey, or black, with lighter or darker markings.
Total length, 620-675 millimetres; tail 75-95. 28
Habitat: France (especially Vendée, the Forest of Fontainebleau, and the South), Pyrenees, Alsace-Lorraine, the Black Forest, Switzerland, Italy and Sicily, and the Tyrol.
This viper especially frequents dry, rocky, and arid hillsides, which are exposed to the sun. Like the adder, it hibernates in tree-trunks and old walls. It lays from 6 to 15 eggs, from which the living young immediately issue, provided with poison. It feeds upon small rodents, worms, insects, and young birds. Raptorial birds, storks, and hedgehogs pursue it and devour it in large numbers.

Fig. 22.—Vipera aspis. (Natural size.) (From the Forest of Fontainebleau.)
Intermediate between V. aspis and V. ammodytes. Snout less turned up into a corneous appendage than in the latter. Head covered with small, smooth, or feebly keeled, subimbricate scales, among which an enlarged frontal shield may sometimes be distinguished; 5-7 longitudinal series of scales between the supraocular shields; 9-13 scales round the eyes; 2 or 3 series between the eyes and the labials; nasal shield entire, separated from the rostral by a naso-rostral. Body scales in 21 rows, strongly keeled; 125-147 ventrals; 32-43 subcaudals.
Coloration grey or brown above, with a longitudinal zigzag band, usually spotted with white; head with or without spots on 29 the vertex; black streak behind the eyes; ventral surface grey, spotted with black and white; tip of the tail usually yellow or with yellow spots.
Total length, 550-610 millimetres; tail 80-85.
Habitat: Spain and Portugal.
Snout terminated in front by a horny appendage covered with 10-20 small scales; vertical diameter of the eyes less than the distance separating them from the mouth; upper surface of the head covered with small, smooth, or feebly keeled scales, among which an enlarged frontal and a pair of parietal shields are sometimes distinguishable; 5-7 longitudinal series of scales between the supraoculars; 10-13 scales round the eyes; two series between the eyes and the labials; nasal shield entire, separated from the rostral by a naso-rostral. Body scales in 21-23 rows, strongly keeled; 133-162 ventrals; 24-38 subcaudals.
Coloration grey, brown, or reddish above, with a zigzag dorsal band, usually spotted with white; black streak behind the eyes; belly grey or violaceous; end of the tail yellow, orange, or coral-red.
Total length, 550-640 millimetres; tail 70-80.
Habitat: Southern Tyrol, Carinthia, Styria, Hungary, Danubian principalities and kingdoms, Turkey. Does not pass beyond the 48th parallel of North Latitude.
This viper loves very sunny places, and hillsides planted with vines. It rarely hibernates.
In districts in which it is plentiful, it is only necessary to light a fire at night in order to attract this species in swarms; this is the best method of taking it.
Its food consists of small rodents, lizards, and birds. 30
The species of snakes most dangerous to man are found in the warmer regions of Asia. India especially is infested by the famous Cobra-di-Capello (Naja tripudians), which possesses the highly remarkable faculty of dilating its neck in the form of a hood when irritated, and whose sculptured image appears on almost all the Hindu monuments.
We shall describe in a separate section (see below, F.) the Hydrophiinæ, or Sea-snakes, a large number of species of which frequent the shores of the Indian Ocean, the Strait of Malacca, the China Sea, the Moluccas, Celebes, and North Australia. In the case of certain species the area of distribution includes the whole of the tropical and sub-tropical zones of the Pacific Ocean, as far as the West Coast of America. It is therefore preferable to group them together for the purpose of comprehensive study.
Besides the above, the continent of Asia harbours a multitude of poisonous snakes belonging to the two Families Colubridæ and Viperidæ.
The genera and species belonging to these are so diverse, that we must confine ourselves to mentioning the essential characters of those that present most interest.
| I.—Family Colubridæ. | |
|---|---|
| Subfamily Elapinæ: Genera | |
| { (a) Bungarus. | |
| { (b) Naja. | |
| { (c) Hemibungarus. | |
| { (d) Callophis. | |
| { (e) Doliophis. | |
(a) Bungarus.
Head hardly distinct from the neck; eyes small, with round or vertically elliptic pupils; nostril between two nasal shields. Two large poison-fangs followed by one or two small, slightly grooved 31 teeth (fig. 23). Scales smooth, oblique, in 13-17 rows, enlarged and hexagonal in shape on the vertebral column; ventral scales round. Tail relatively short; subcaudal scales in one or two rows.
Two very dangerous snakes found in India and Indo-China belong to this genus, B. fasciatus and B. candidus (var. cæruleus). Both are fairly common. In Ceylon B. ceylonicus is met with, and in South China B. candidus (var. multicinctus). The length of these snakes is from 1,000-1,500 millimetres. The back is compressed in the shape of a keel. The neck is not dilatable.

Fig. 23.—Skull of Bungarus. (After G. A. Boulenger, op. cit.)
Colour bright yellow, ringed with black, with a black band commencing between the eyes, and broadening behind upon the nape and neck (fig. 24). 32
Especially abundant on the Coromandel Coast, in Bengal, and in Burma. In the North-west Provinces of India it is known as the Koclia-Krait. Its bite is very serious, but does not cause nearly so many fatalities as that of the Cobra, since its fangs are smaller.
Dogs bitten by B. fasciatus die in from four to five hours.

Fig. 24.—Bungarus fasciatus (India). (After Sir Joseph Fayrer.)
Blackish-brown or bluish, with narrow transverse white streaks, or small white spots, or alternate rings of yellow and dark brown; belly white. Smaller than the foregoing, scarcely exceeding 1,000 millimetres in length. It is known as the “Krait” in India, where, 33 after the Cobra, the variety cæruleus causes most deaths among human beings. It is found in jungles and rice-fields, and commonly secretes itself in old trees and old walls. It frequently penetrates into houses, verandahs, bathrooms, and even beds. Sir Joseph Fayrer relates the story of a lady, who, when travelling in a palanquin, found on arriving at her destination a “Krait” coiled up in her luggage, the snake having thus made the journey with her throughout a whole night.
The Krait may easily be confused with Lycodon aulicus, a harmless snake which closely resembles it, though it can at once be distinguished by examining its mouth.
Head scarcely distinct from the neck; eyes with round pupils; nostril between two nasal shields and an internasal. A pair of solid grooved poison-fangs. Body elongate, cylindrical, terminated by a conical and pointed tail. Scales smooth, disposed obliquely, in 15-25 rows. Ventral scales round.
Head small, covered with large shields, a frontal as long as broad, a supraocular, a præocular, 3 postoculars, 2 + 3 or 3 + 3 temporals, 7 upper labials, 4 lower labials. Neck dilatable by the separation of the first cervical ribs; 21-35 scales round the neck, 17-25 round the middle of the body; 163-205 ventrals; 42-75 subcaudals.
Total length, 1,500-1,900 millimetres; tail 230.
Coloration very variable, usually cinereous grey or almost black with a bluish sheen; belly lighter, sometimes tinted with red. The head is frequently tinged with golden-yellow; it is spotted with yellowish-white above, and is pure white underneath. 34
This species is distributed throughout the whole of Southern Asia, from the south of the Caspian Sea to South China and the Malay Archipelago.

Fig. 25.—Skull of Naja tripudians. (After G. A. Boulenger, op. cit.)
Several varieties occur, and of these the principal are:—
(1) Var. Typica (fig. 27), with a black-and-white spectacle-shaped mark on the middle of the dorsal surface of the most dilatable portion of the neck, and one or more dark transverse bands on the ventral surface, behind the head.
Habitat: India, Ceylon. 35
(2) Var. Cæca.—Colour, pale brown or uniform dark grey, without mark on the neck, and with one or more dark transverse bands on the anterior part of the belly.
Habitat: Transcaspian region, India, Java.

Fig. 26.—Naja tripudians (Cobra-di-Capello) on the Defensive, preparing to Strike.
(3) Var. Fasciata.—Colour, brown, olive, or black, with more or less distinct light transverse bands. White spot edged with black in the shape of a ring or of a U on the neck, behind; a black spot on each side in front.
Habitat: India, Indo-China and South China, Hainan, Cambodia, Siam, Malay Peninsula.
(4) Var. Sputatrix.—Black or dark brown, with yellow or orange-coloured spots on the sides of the head and neck. The young have a pale spot in the shape of a U or an O on the middle of the dorsal surface of the neck, and the throat is whitish.
Habitat: Chusan Islands and South China, Burma, Malay Peninsula, Sumatra, Java.
(5) Var. Leucodira.—Brown or black, without mark on the neck. Throat yellowish-white, followed by a black transverse band.
Habitat: Sumatra, Malay Peninsula. 36
(6) Var. Miolepis.—Brown or black; sides of the head and throat yellowish, no mark on the neck. Young with white rings completely encircling the body and tail.
Habitat: Sarawak, Labuan, Borneo.

Fig. 27.—Naja tripudians (Cobra-di-Capello). (After Sir Joseph Fayrer.)
Internasal shields shorter than the præfrontals, and in contact with the præoculars; 1-3 large occipital shields behind the 37 parietals; 1 præocular and 3 postoculars; 2 + 2 or 2 + 3 temporals; 7 supralabials, 4 infralabials; 21-23 scales across the neck, 17-19 across the middle of the body; 159-175 ventrals; 45-50 subcaudals.
Coloration black or sometimes yellowish above; pale brown or yellowish on the belly; neck black.
Total length, 1,000 millimetres; tail 160.
Habitat: Philippine Islands.
A pair of large occipital shields; 1 præocular; 3 postoculars; 2 + 2 temporals; 7 supralabials, 4 infralabials; 19-21 scales across the neck, 15 across the middle of the body; 215-262 ventral scales, 80-117 subcaudals. Neck dilatable.
Coloration very variable, yellowish, brown, olive, or black, with or without dark transverse bands.
Total length, 3,900 millimetres; tail 630.
Habitat: India, Burma, Indo-China, Siam, Southern China, Malay Peninsula and Archipelago.
The species of Naja are oviparous, and usually lay some twenty eggs, elliptical in shape and as large as those of a pigeon, with a soft shell.
These snakes do not fear the proximity of man, and feed upon rats, mice, and birds; they seek their prey chiefly in the evening, after sunset.
They swim extremely well, and frequent the neighbourhood of water-courses.
Indian legends relate that Brahma, having descended on earth and fallen asleep one day at high noon, a Naja placed itself in front of him and, dilating its broad neck, procured for him kindly shade. In order to repay it for the service rendered, Brahma gave 38 Naja the marks that it bears on its neck, intended to frighten the kites and other birds of prey, which are implacable enemies of this snake.
When a native of the Malabar Coast finds a Naja in his dwelling, he begs it in a friendly way to depart; if the request be without avail, he offers it food in order to attract it outside; if the snake still does not move, the Hindu goes in search of the pious servitors of one of his divinities, who, procuring an offering, address the most touching supplications to it (Brehm).
The mortality due to the bite of this snake, which is by far the most common in India, is considerable. In the course of a period of eight years, from 1880 to 1887, it amounted on the average to 19,880 human beings and 2,100 head of cattle every year.
In 1889, 22,480 persons and 3,793 head of cattle perished from snake-bite. Since then, the annual tale of fatalities always fluctuates between 16,000 and 22,000, in spite of the rewards for the destruction of snakes which the Indian Government has been obliged to institute, which represent an expenditure of about £10,000 per annum.
For every 100 persons bitten, it is estimated that on an average from 25 to 30 die, and in most cases death supervenes in from two to twelve hours after the bite.
Naja bungarus, or the Hamadryad, is the largest and most formidable of poisonous snakes. It is very vigorous and very aggressive, but is more rarely met with than Naja tripudians. It loves the vicinity of rivers and streams, lives in forests and jungles, and climbs trees with facility. It feeds upon other snakes (whence its name Ophiophagus), and also on birds, fish, and small mammals.
Hindu snake-charmers assert that it is very difficult to capture, and dangerous owing to its strength; they handle it only after having extracted its poison-fangs.
A very intelligent Hindu told Torrens how he had seen the way in which the Hamadryad procures the snakes that form its favourite 39 food. The Hindu in question happened to be on the flat roof of his house, when a young Hamadryad appeared quite close to him. The snake raised its head, expanded its neck, and emitted a shrill hissing noise. Thereupon a dozen snakes came crawling up from all directions and assembled round the Hamadryad, when the latter made a dart at one of them and hastened to devour it (Fayrer).
The Hamadryad is dreaded with good reason, for not only is it aggressive, and hurls itself boldly upon its adversary, but it also pursues him, a trait exhibited by no other poisonous snake.
Cantor relates that in Assam an officer met with several young Hamadryads which were being watched over by their mother. The latter turned towards its enemy, who took to his heels with all speed, pursued by the terrible reptile. The course taken led to a river, which the fugitive did not hesitate to swim in order to gain the opposite bank, hoping thus to make good his escape; all, however, to no purpose. The snake still pursued him, and the officer saved himself only by a stratagem. He dashed his turban on the ground; the snake threw itself upon it and savagely bit it several times, thus giving the officer time to reach a place of safety.
Cantor’s experiments show that the venom of the Hamadryad is extremely rapid in its action. A dog usually dies a quarter of an hour after being bitten, and Nicholson states that he has seen an elephant bitten by a snake of this species die in three hours.
This genus includes several species of snakes of somewhat small size, rarely exceeding 700 millimetres in length, with an elongate, cylindrical body; the head is scarcely distinct from the neck, the pupil round, and the tail short, while the nostril is situate between two nasal shields. The temporal shields are arranged in a single row. The poison-glands sometimes extend into the abdominal 40 cavity. Scales in 13 or 15 rows; 190-260 ventrals, 12-44 subcaudals in 2 rows.
Four species belonging to this genus are known:—
(1) H. calligaster.—2 + 3 temporal scales, 6 supralabials.
Colour purple, with black transverse bands separated by narrow white bars; belly and end of tail red; snout yellow, with a black band on the upper lip below the eyes.
Total length, 520 millimetres; tail 30.
Habitat: Philippine Islands.
(2) H. collaris.—No anterior temporal scales.
Colour black on the back, with black and red bands on the belly; a yellow collar on the occiput.
Total length, 430 millimetres; tail 15.
Habitat: Philippine Islands.
(3) H. nigrescens.—Scales in 13 rows. A single temporal scale; 218-251 ventrals; 33-44 subcaudals.
Belly uniformly red; upper lip yellow in front of and behind the eyes.
Total length, 1,100 millimetres; tail 115.
Habitat: Hills of Western India, from Bombay to Travancore.
(4) H. japonicus.—Scales in 13 rows; 190-216 ventrals; 28-29 subcaudals; temporals 1 + 1.
Colour red on the back, with 1-5 black bands crossed by other black bands edged with yellow. Snout and sides of head black. Belly yellow, with large black spots alternating with black transverse bands.
Total length, 520 millimetres; tail 40.
Habitat: Loo Choo Islands.
This genus is characterised by the maxillary bones extending forwards beyond the palatines, with a pair of large poison-fangs, but without other teeth. Head and eyes small, pupils round; 41 nostril between two nasal shields. Body cylindrical, greatly elongate. Scales smooth, in 13 rows; ventrals rounded; subcaudals in 2 rows.
Five species are known:—
(1) C. gracilis.—Red or pale brown, with three longitudinal black lines passing through brown, or black spots; the lateral spots alternating with the vertebræ. Black and yellow bands under the tail and on the belly.
Total length, 740 millimetres; tail 35.
Habitat: Malay Peninsula, Sumatra.
(2) C. trimaculatus.—Head and nape black, with a yellow spot on each side of the occiput; belly uniform red; tail with two black rings.
Total length, 335 millimetres; tail 21.
Habitat: India and Burma.
(3) C. maculiceps.—Head and nape black, with one or two yellow bands on each side. Belly red, two black rings on the tail. Diameter of the eyes equal to two-thirds of the space separating them from the mouth.
Total length, 485 millimetres; tail 30.
Habitat: Burma, Indo-China, Malay Peninsula.
(4) C. macclellandii.—Head and neck black, with a yellow transverse band behind the eyes. The space separating the eyes equal to that separating them from the mouth. Colour reddish-brown on the back, with regular and equi-distant black streaks; belly yellow, with black bands or quadrangular spots. The head exhibits two black transverse bands separated by a yellow band.
Total length, 620 millimetres; tail 55.
Habitat: Nepal, Sikkim, Assam, Burma, Southern China.
(5) C. bibronii.—Met with by Beddome in the forests of Malabar, at an altitude of 3,280 feet. Back purplish-brown, with a pearly lustre, and about forty irregular black transverse bands, extending to the tip of the tail. Head black in front, cherry-red on the occiput. 42
Total length, 640 millimetres; tail 50.
Habitat: Malabar.
All the snakes belonging to the genus Callophis are remarkable for their bright and varied colours, whence the generic name, which signifies “beautiful snakes.”
They feed exclusively on other snakes belonging to the Family Calamaridæ; consequently they are not found in regions where Calamaridæ do not occur, as, for instance, in Ceylon.
They are essentially terrestrial, and live in old tree-trunks, or clefts in rocks. They are sluggish, slow-moving, and chiefly nocturnal.
As a rule they do not seek either to defend themselves or to bite; consequently fatal accidents caused by them are scarcely known in the case of human beings. Their venom, however, is very toxic to animals.
This genus exhibits the same characters as Callophis, except that the poison-glands, instead of being confined to the temporal region, extend a very long way on each side of the body, to about one-third of its length, gradually growing thicker and terminating at the base of the heart.
It includes four species:—
(1) D. bivirgatus.—Colour reddish-purple or black on the back, red on the head, tail, and belly.
Total length, 1,610 millimetres; tail 190.
Habitat: Burma, Indo-China, Malay Peninsula, Sumatra, Java, and Borneo.
(2) D. intestinalis.—Brown or black on the back, with darker or lighter longitudinal streaks; tail red beneath; belly red, crossed with black streaks.
Total length, 580 millimetres; tail 45. 43
Habitat: Burma, Malay Peninsula, Sumatra, Java, Borneo, Celebes.
(3) D. bilineatus.—Black on the back, with two white streaks along the whole length of the body. Snout white; belly striped with black and white bands. Tail orange, with two or three black rings or spots.
Total length, 710 millimetres; tail 45.
Habitat: Philippine Islands.
(4) D. philippinus.—Back with dark brown cross-bands, merging into black transverse ventral streaks, which are separated by yellow or red interspaces. Head brown, with small yellow spots.
Total length, 430 millimetres; tail 35.
Habitat: Philippine Islands.
The Family Viperidæ is represented in Asia by a considerable number of snakes belonging to the two Subfamilies Viperinæ and Crotalinæ.
The Asiatic Viperinæ belong to the genera:—
(a) Vipera. (b) Pseudocerastes. (c) Cerastes. (d) Echis.
The Crotalinæ consist of only two genera:—
(e) Ancistrodon. (f) Lachesis.
We shall not recapitulate here the characters of the Genus Vipera, which we described in dealing with the vipers of Europe. The genus is represented by several species, the geographical range of which is chiefly confined to Eastern and Central Asia. 44
(1) Vipera renardi.—Resembles V. berus, but the snout is pointed and soft, with a turned-up tip; a single series of scales between the eyes and the lips; nostril pierced in the lower half of a single nasal shield; 8-9 supralabial shields; 4 infralabials. Body scales in 21 rows; 130-150 ventrals; 24-37 subcaudals.
Coloration the same as in the European V. ursinii, but the snout and lips are spotted with black or brown.
Total length, 395-620 millimetres; tail 40-75.
Habitat: Central Asia, Turkestan.
(2) V. raddii.—Snout rounded; supraocular shields erectile; eyes surrounded by a complete circle of 14-17 scales; 9-10 supralabials; body scales in 23 rows; 150-180 ventrals; 23-32 subcaudals.
Coloration pale brown or grey on the back, with a dorsal series of small reddish spots arranged in alternating pairs. A black mark like a circumflex accent on the occiput, and a black band behind the eyes. Belly yellow, speckled with black and white.
Total length, 740 millimetres; tail 50.
Habitat: Armenia.
(3) V. lebetina.—Snout rounded and obtuse, with a well-marked prominence; 7-12 longitudinal series of scales between the eyes; supraocular shields well developed or narrow, or broken up into several small portions; 12-18 scales round the eyes; 9-12 supralabials; 4-5 infralabials; body scales in 23-27 rows; 147-180 ventrals; 29-51 subcaudals.
Coloration variable, grey or pale brown on the back, with a series of large dark spots. Large brown mark like a circumflex accent on the crown of the head and another on the occiput. Belly whitish, speckled with grey-brown; end of tail yellow.
Total length, 960 millimetres; tail 120. The female may attain the length of 1,350 millimetres.
Habitat: Cyprus, Galilee, Syria, Asia Minor, Transcaspia, Persia, Mesopotamia, Afghanistan, Baluchistan, Cashmir. 45
(4) V. russellii (Daboia, or Russell’s Viper). (fig. 28.)—This viper, which may attain a length of as much as 2,000 millimetres, is magnificently coloured. Its dorsal surface is brownish-yellow, marked with large oval spots of blackish-brown, edged with yellow or white. The belly is covered with transverse bands, with beautiful triangular black spots, bordered with white. The head, which is long, ends in front in a thick, rounded snout; it is covered above 46 with small keeled scales. The nostril, which is large and laterally placed, is surrounded by three shields and soft smooth skin.

Fig. 28.—Vipera russellii (Syn. Vipera elegans. Daboia, or Russell’s Viper). India.
(After Sir Joseph Fayrer.)
The species is found throughout India, from Bombay to Bengal, in Ceylon, Burma, and Siam. It is particularly common in Burma, around Rangoon. For walking in the jungle and rice-fields, the natives of this region encase their feet and legs in a special kind of jack-boots made of coarse jute-cloth, in order to protect themselves from the bites of this snake, which cause a large number of fatal accidents.
The Daboia ascends the Himalayas to an altitude of 5,250 feet. It lives in thickets, under stones, and in the clefts of rocks. When disturbed it makes a terrible hissing, but bites only when attacked or irritated.
It feeds upon small vertebrates, such as mice, rats, birds, and frogs, and often enters houses in pursuit of rats.
“Schrott had the opportunity of observing a Daboia on the defensive. A lady carrying a child on her arm was returning home towards evening; she had almost reached her house when a bulldog accompanying her began to bark furiously. Although the lady saw nothing, she was, nevertheless, frightened and called for help. Schrott, who was not far away, ran to the spot, and saw a Daboia lying across the path by which the lady had to proceed. The reptile had its neck thrown back and its head in a horizontal position; its bright eyes followed all the movements of the dog, to whose barks it replied by shrill hisses. It was only waiting for an opportunity to strike. Schrott called off the dog, and the snake at once disappeared among the high grass close by. Next day it was killed at the same spot” (Brehm).
The venom of this viper is terribly potent. According to Russell, a large dog exhibited symptoms of poisoning five minutes after being bitten. At the end of a quarter of an hour it lay down, uttering heartrending cries, began to breathe with difficulty and noisily, was seized with spasms of the jaws and cramps, and died in frightful agony less than half an hour after the wound was inflicted. Fowls 47 in most cases die in less than two minutes. A horse succumbed in half an hour, and another in eleven hours.
It appears that in India many cattle are killed by Daboias while grazing (Fayrer).
This genus is represented by a single species (Pseudocerastes persicus), which appears to be exclusively confined to Persia.
The head is very distinct from the neck, and covered with small imbricate scales; the eyes, which are small, have vertical pupils; they are separated from the lips by small scales. The nostrils are directed upwards and outwards. The snout is very short and rounded. The cylindrical body has 23-25 rows of scales; 151-156 ventrals; 43-49 subcaudals.

Fig. 29.—Pseudocerastes persicus.
(After Duméril and Bibron.)
The coloration is gray or brown, with four series of large black spots, and the head exhibits two longitudinal black streaks behind the eyes. The belly is whitish, dotted with black.
Total length, 890 millimetres; tail 110.
The vipers belonging to this genus are much more common in North Africa, and we shall therefore study them in conjunction with the African snakes. Cerastes cornutus alone, the special habitat of which is Egypt, is sometimes met with in Arabia and on the eastern bank of the Suez Canal. 48
Echis carinatus (the Phoorsa). (Fig. 30).—This viper is characterised by the subcaudal shields being arranged in a single row. It is savage and very aggressive, being always ready to attack. Its length does not exceed 600 millimetres at the most. The colour of the body is grey, more or less dark and adorned with streaks, spots, and dots of blackish-brown. The back displays yellowish-white wavy lines, forming X-shaped markings. The upper side of the head exhibits a yellow spot surrounded by brown, and other small black spots, the whole arrangement forming a fairly good representation of a cross.
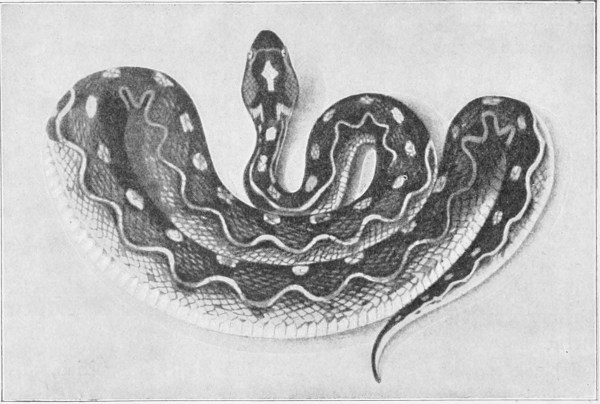
Fig. 30.—Echis carinatus. India. (After Sir Joseph Fayrer.)
This species is found in India, Persia, Baluchistan, Arabia, and Palestine; while, as we shall see, it also occurs in Africa. It is fairly common in the environs of Aden. 49
In moving over the ground a peculiar sound is produced by the friction of its scales. It is capable of springing with great agility a fairly long distance on to its prey. When it believes itself in danger it coils up, doubling its body twice in the shape of a crescent, in the middle of which it places its head, ready to strike. Its venom is very rapid in taking effect.
The snakes belonging to this genus of Crotalinæ are found in Central and Eastern Asia, but three important species occur in the New World, in the United States and Central America. The head is covered with nine large symmetrical shields, but the internasals and præfrontals are sometimes broken up into scales. The body is cylindrical; the tail rather short; the subcaudal scales are arranged in one or two rows.
A. acutus.—The snout of this snake is prolonged into an appendage directed forwards. The head-shields are finely granulate. Body scales arranged in 21 rows; 162-166 ventrals; 58-60 subcaudals.
Coloration brown on the back, with blackish-brown X-shaped spots; head dark brown, yellow on the cheeks, with a black band running from the eye to the angle of the jaw; belly yellowish, spotted with brown, with a series of large black transverse blotches.
Total length, 1,500 millimetres; tail 200.
Habitat: Upper Yang-tse, China.
A. halys.—Snout prolonged into an upturned appendage, blunt at the tip; 7-8 supralabial scales, the third of which forms part of the margin of the eye; body scales in 23 rows; 149-174 ventrals; 31-44 subcaudals.
Coloration yellowish, grey, red, or pale brown above, with darker spots or cross-bars. A black spot on the snout; two black spots 50 on the vertex; a dark, light-edged band on the temple; belly whitish, more or less speckled with grey or brown.
Total length, 490 millimetres; tail 65.
Habitat: From the borders of the Caspian Sea and the Ural River to the Upper Yenisei; Turkestan.
A. intermedius.—Resembles the foregoing very closely, but the snout is not turned up at the end.
Total length, 750 millimetres; tail 85.
Habitat: Central Asia, Eastern Siberia, Mongolia, and Japan.
A. blomhoffii.—Similar to A. halys, but the snout is not turned up at the end, and the body scales are in 21 rows; 137-166 ventral scales; 29-55 subcaudals.
Coloration very variable; grey, brown, or red above, with large black-edged spots arranged in pairs; black, light-edged band on the temple; upper lip uniformly yellow or red; belly yellow or reddish, more or less spotted with black, or almost entirely black.
Total length, 720 millimetres; tail 100.
Habitat: Eastern Siberia, Mongolia, China, Japan, Siam.
A. himalayanus.—Snout scarcely turned up, with a hard tip; 5-7 supralabial shields. Body scales in 21 (rarely 23) rows; 144-166 ventrals; subcaudals in 35-51 pairs.

Fig. 31.—Ancistrodon hypnale.
(The Carawalla of Ceylon.)
(After Sir Joseph Fayrer.)
51
Coloration brown, with black spots or transverse bands; black, light-edged band from the eye to the angle of the mouth; belly dark brown, or more or less whitish.
Total length, 590 millimetres; tail 90.
Habitat: Himalayas (5,000 to 10,000 feet), especially in the North-west; Khasi Hills.
This snake feeds chiefly on mice.
A. rhodostoma.—Snout pointed, somewhat turned up at the tip: 7-9 supralabial shields; body scales in 21 rows; 138-157 ventrals; 34-54 subcaudal pairs.
Coloration reddish, grey, or pale brown above, with large angular, dark brown, black-edged spots arranged in pairs or alternating. Vertebral line almost black; lips yellow, speckled with brown; brown, black-edged band, running from the eye to the angle of the mouth. Belly yellowish, spotted with greyish-brown.
Total length, 810 millimetres; tail 90.
Habitat: Java.
A. hypnale (fig. 31).—Snout more or less turned up, with a hard, pointed end; 7-8 supralabial shields; body scales in 17 rows; 125-155 ventrals; 28-45 subcaudal pairs.
Coloration very variable; brown, yellowish, or greyish above, sometimes with dark brown spots or transverse bands. Cheeks brown, with a longitudinal, white, black-edged streak on each side of the neck. Belly more or less speckled with dark brown.
Total length, 480 millimetres; tail 65.
Habitat: Ceylon, and Western Ghats of India as far north as Bombay.
In Ceylon this snake is known as the Carawalla. It is much dreaded, but its bite is not rapidly fatal.
This genus has many representatives in Asia and the New World. The American species are for the most part of larger size and more formidable. 52
They are characterised by a triangular head, covered with small scales or small shields, and by a cylindrical or compressed body. The Asiatic species have the subcaudal scales in two rows and the tail short, and often prehensile, which enables them to climb trees or bushes, where they lie in wait for their prey.
Their classification is based upon the following characters:—
A. First infralabial scale in contact with its fellow.
I. Scales in 21-25 (rarely 27) rows; 129-158 ventrals; 21-57 subcaudals; 5-9 series of scales between the supraocular shields; tail non-prehensile.

Fig. 32.—Lachesis okinavensis. (After G. A. Boulenger, op. cit.)
(1) L. monticola.—Supraocular shields large, separated by 5-8 scales; snout obtuse.
Colour brown or yellowish above, brown or pale yellow on the sides, with a brown temporal streak. Belly white, spotted with brown.
Total length, 740 millimetres; tail 115.
Habitat: Tibet, Himalayas (2,000 to 8,000 feet), Hills of Assam, Burma, Malay Peninsula, Singapore, Sumatra.
(2) L. okinavensis (fig. 32).—Supraocular shields large, separated 53 by a transverse series of 6-9 scales; end of snout pointed and turned up.
Colour brown above, with dark transverse bands and a light temporal streak. Belly brown, spotted with black, especially on the sides.
Total length, 350 millimetres; tail 60.
Habitat: Okinawa, Loo Choo Islands.
(3) L. strigatus.—Supraocular shields small, sometimes broken up, separated by 8-11 convex scales in juxtaposition.
Colour brown above, with black spots often forming a median zigzag band; temporal band black; belly whitish, spotted with grey or black; end of tail yellow or reddish.
Total length, 480 millimetres; tail 55.
Habitat: Hills near Bombay, Deccan, Anamallays and Nilgherries.
II. Scales in 27-37 rows; 174-231 ventrals; 54-90 subcaudals; tail non-prehensile.
(4) L. flavoviridis.—Scales in 33-37 rows; 222-231 ventrals; 75-90 subcaudals; 8-9 supralabials.
Coloration pale brown or greenish-yellow above, marbled with black; longitudinal black streaks symmetrically disposed on the head; belly yellow or greenish, with darker spots.
Total length, 1,215 millimetres; tail 220.
Habitat: Loo Choo Islands.
This snake is not infrequently termed by naturalists Trimeresurus riukianus.
(5) L. cantoris.—Scales in 27-31 rows; 174-184 ventrals; 55-76 subcaudals; 13 supralabials.
Coloration pale brown or dull green, with small black spots; a whitish streak along the sides of the body; belly white or greenish.
Total length, 1,020 millimetres; tail 140.
Habitat: Andaman and Nicobar Islands. 54
III. Scales in 21-27 rows; 160-218 ventrals; 54-92 subcaudals; tail not or scarcely prehensile.
(6) L. jerdonii.—7-9 scales in a line between the supraocular shields; scales in 21 or 23 rows; 164-188 ventrals; 54-67 subcaudals.
Coloration greenish-yellow or olive above, mixed with black, a dorsal series of reddish-brown transverse rhomboidal spots; upper lip yellow, with one or two black spots; belly yellow, more or less marbled with black.
Total length, 930 millimetres; tail 145.
Habitat: Assam, Tibet, Upper Yang-tse.
(7) L. mucrosquamatus.—10-15 scales in a line between the supraocular shields; scales in 25-27 rows; 183-218 ventrals; 72-92 subcaudals. Colour brownish-grey above, with a series of large black median spots and smaller ones on the sides; a black streak from the eye to the angle of the mouth; belly brownish, spotted with white.
Total length, 1,050 millimetres; tail 210.
Habitat: Formosa, Assam.
(8) L. luteus.—12 or 13 scales in a line between the supraocular shields; scales in 23-25 rows; 182-186 ventrals; 72-74 subcaudals; supraocular shields large.
Colour yellow above, with a series of dark rhomboidal spots, and a dorsal, black-spotted zigzag band; a black streak on each side of the head behind the eyes; belly yellowish, spotted with grey.
Total length, 945 millimetres; tail 164.
Habitat: Loo Choo Islands.
(9) L. purpureomaculatus.—12-15 scales in a line between the supraocular shields; scales in 25-27 rows; ventrals 160-182; subcaudals 55-76; supraocular shield very narrow, sometimes broken up.
Coloration purplish-black above, sometimes variegated with pale green; flanks pale green; belly olive or greenish-white, uniform or spotted with black. Some specimens are entirely green. 55
Total length, 980 millimetres; tail 150.
Habitat: Himalayas, Bengal, Assam, Burma, Malay Peninsula, Andaman and Nicobar Islands, Pulo-Pinang, Sumatra.
IV. Scales in 21 rows (rarely 19 or 23); 7-13 series of scales between the supraoculars; tail more or less prehensile.
(10) L. gramineus (Syn. Trimeresurus gramineus. The Green Pit Viper).—145-175 ventral scales; 53-75 subcaudals; snout slightly projecting; supraocular shield narrow.
Colour bright green, rarely olive or yellowish, with or without darker transverse bands; end of tail yellow or red; belly green, yellow, or white.
Total length, 870 millimetres; tail 150.
Habitat: South-eastern Asia, Darjeeling, Himalayas, Ganges Delta, Siam, Southern China, Hong Kong, Formosa, Java, Sumatra, Timor.

Fig. 33.—Lachesis flavomaculatus.
(After G. A. Boulenger, op. cit.)
(11) L. flavomaculatus (fig. 33).—170-187 ventral scales; 53-73 subcaudals; snout projecting, obliquely truncate; supraocular shield narrow.
Colour bright green or olive, sometimes barred with reddish-brown; belly green, olive, or greenish-yellow; end of tail sometimes red.
Total length, 1,060 millimetres; tail 160.
Habitat: Philippine Islands. 56
(12) L. sumatranus.—180-191 ventral scales; 58-82 subcaudals; supraocular shield large.
Coloration bright green, with or without black transverse bands; yellowish band on each side; belly yellow or green, with or without black speckles; end of tail red.
Total length, 1,100 millimetres; tail 180.
Habitat: Singapore, Sumatra, Borneo, Palawan.
(13) L. anamallensis.—138-158 ventral scales; 44-58 subcaudals.
Colour green, olive, yellowish, or reddish-brown; a black temporal band; belly pale green, olive, or yellow; tail usually black and yellow.
Total length, 730 millimetres; tail 110.
Habitat: Anamallay and Nilgherry Hills, South India.
(14) L. trigonocephalus.—Scales in 17-19 rows, 145-170 ventrals; 53-67 subcaudals.
Coloration green, with or without black transverse bands or spots; a black temporal streak; belly green or yellow; end of tail usually black.
Total length, 790 millimetres; tail 130.
Habitat: Ceylon.
(15) L. macrolepis.—Scales in 13-15 rows; 134-143 ventrals; 48-56 subcaudals.
Colour bright green or olive; belly pale green.
Total length, 680 millimetres; tail 120.
Habitat: Southern India.
B. First lower labial shield divided; the portion separated off forms a pair of small supplementary dental shields; 144-176 ventral scales; 38-57 subcaudals; tail prehensile.
(16) L. puniceus.—Scales in 21-23 rows; 12-14 series of scales between the supraocular shields.
Colour grey, brown, or red; belly spotted with brown; end of tail red.
Total length, 640 millimetres; tail 90. 57
Habitat: Sumatra, Java, Borneo, Natuna Islands.
(17) L. borneensis.—Scales in 19-21 rows; 10-11 series of scales between the supraocular shields; 152-168 ventrals; 43-65 subcaudals.
Total length, 770 millimetres; tail 105.
Habitat: Borneo, Sumatra.
C. (18) L. wagleri.—Scales in 19-27 rows, 127-154 ventrals; 45-55 subcaudals; tail prehensile.
Colour green, with darker or lighter markings, black and yellow, or nearly entirely black.
Total length, 980 millimetres; tail 150.
Habitat: Malay Peninsula and Archipelago.
In Africa, poisonous snakes abound throughout the whole of the tropical and sub-tropical zones. The fatal accidents to human beings caused by them in this continent are fewer than in India, since the population of Africa is less dense, but several species occur, the venom of which is especially dangerous to life.
These species belong in nearly equal numbers to the Families Colubridæ and Viperidæ.
The poisonous Colubridæ of Africa are all included in the Subfamily Elapinæ, of which the following are the eight principal genera:—
(a) Boulengerina. (b) Elapechis. (c) Naja. (d) Sepedon. (e) Aspidelaps. (f) Walterinnesia. (g) Homorelaps. (h) Dendraspis.
58
A small snake 240 millimetres in length, the principal characters of which are as follows:—
Maxillary bones equal to the palatines in length, with a pair of relatively large poison-fangs, followed by three or four small teeth.
Head hardly distinct from the neck. Eye small, with round pupil; nostril between two nasal shields. Body cylindrical; scales smooth, in 21 rows; ventrals rounded. Tail moderate; subcaudal scales in 2 rows.
The only known species, B. stormsi, is brown, with black transverse bars on the neck; tail black; belly white in front, brown behind, with a black border to the scales.
It is found in the region of Lake Tanganyika.
This genus is characterised by the size of the maxillary bones, which is equal to that of the palatines; a pair of large poison-fangs followed by two to four small teeth; head not distinct from the neck; eye small with round pupil; nostril between two nasal shields. Body cylindrical; scales oblique, smooth, in 13 or 15 rows; ventrals rounded. Tail very short, subcaudal shields in 2 rows.
Six species are known:—
(1) E. guentheri.—Scales in 13 rows. Snout short, rounded; first lower labial shield in contact with its neighbour behind the symphysis. Internasal shorter than the præfrontals; length of frontal three-fourths of that of the parietals.
Coloration whitish or grey above, with black, white-edged cross-bands. Belly dirty white, brownish, or grey.
Total length, 520 millimetres; tail 50.
Habitat: Gaboon, Congo, Angola, Central Africa.
(2) E. niger.—Snout and arrangement of scales as before. 59 Internasal shields three-fourths of the length of the præfrontals; frontal two-thirds of the length of the parietals.
Colour, black all over.
Total length, 420 millimetres; tail 30.
Habitat: Zanzibar.
(3) E. hessii.—Snout as in foregoing. Symphysial shield of the lower lip in contact with the anterior chin-shields.
Colour grey, with black cross-bars; a series of black spots on the sides, between the bars; large black blotch on nape; belly white.
Total length, 160 millimetres; tail 12.
Habitat: Congo.
(4) E. decosteri.—Snout obtusely pointed.
Colour, dark grey, each scale edged with black; belly white.
Total length, 380 millimetres; tail 38.
Habitat: Delagoa Bay.
(5) E. sundevallii.—Snout obtusely pointed.
Colour reddish-brown, with yellow transverse bands, the scales in which are edged with reddish-brown; upper lip and belly yellow.
Total length, 510 millimetres; tail 43.
Habitat: Caffraria.
(6) E. boulengeri.—Scales in 15 rows.
Colour black on the back, with narrow white transverse bands; head white, with a black streak along the parietal suture; belly blackish-grey.
Total length, 170 millimetres; tail 14.
Habitat: Zambesi.
Six species of Naja are found in Africa:—
(1) N. haje or haie.—Scales in 21-23 rows on the neck, which is dilatable, although slightly less so than that of N. tripudians, the Cobra-di-Capello of India. 60
Six or seven large supralabial shields, sixth or seventh in contact with the lower postocular. Eyes separated from the labial shields by the suboculars. 191-214 ventral scales; 53-64 subcaudals.
Colour yellowish, olive, or uniform black; belly yellowish; black or brown band on the neck; head sometimes blackish.
Total length, 1,180 millimetres; tail 290.
Habitat: Borders of the Sahara, Egypt, Southern Palestine, East Africa as far south as Mozambique.
Naja haje (the Egyptian Cobra) is common throughout the Nile Basin, the Sudan and Central Africa. Livingstone mentions it several times. In Egypt it is met with in the vicinity of ruined monuments, under the large blocks of stone or among brushwood.
The Egyptians are greatly in dread of it, and hunt it down as often as possible. When pursued, N. haje turns bravely and faces its adversary, raising itself upon its tail, puffing out its neck and hissing fiercely. If too hard pressed it strikes at its enemy.
“A friend of mine,” writes Anderson, “had great difficulty in escaping from one of these snakes. While botanising one day a Naja passed quite close to him. My friend darted backwards with all speed, but the Naja pursued and was about to attack him, when he stumbled against an ant-hill and fell flat on his back. This, no doubt, frightened the snake, which disappeared in a twinkling.”
Another case of the same kind is narrated by Waller, who writes: “A young girl met with her death in a truly dramatic fashion. She was walking behind some porters on a narrow path, when suddenly an Aspic came out of a thick bush, attacked and bit her in the thigh; in spite of all efforts to save her the unfortunate girl died in less than ten minutes.” This instance, which is absolutely authentic, proves the truth of the statements made by various travellers. Natives assert that a full-grown Naja invariably pursues either a man or an animal, however large, when either passes within its range (Brehm). 61
Snake-charmers, of whom there are many in Egypt, always employ Naja haje for their performances. They know how to capture it, and tear out its fangs by making it bite a bundle of rags.
This species rarely lives more than six or eight months in captivity, and is quite untamable. It is fond of bathing, and remains in the water for hours together.
“The Ancient Egyptians,” write Duméril and Bibron,3 “are known to have worshipped the Naja, which they regarded as the protector of their crops. They allowed it to live and multiply among the cultivated lands, which they apparently entrusted to the care of their tutelary guardian, recognising that this snake freed them from the ravages of the rats, the immense number of which otherwise caused terrible destruction and even actual famine. It was, therefore, from motives of gratitude that the Egyptian Cobra was venerated in this way; that its image was hung up in the temples; that its skin was embalmed; and that its effigy, so easy to recognise and to reproduce roughly, was graven or sculptured on the stones of their monuments. This is the explanation of the fact that paintings representing N. haje are frequently reproduced in hieroglyphics and on Egyptian sarcophagi.”
The Naja was the tutelary deity of the temples, whose duty was to prevent the profane from entering. Thus, in one of the crypts of Denderah we find represented Serpent-genie, figures with a head like that of a Naja supported by the body of a man, with the hands armed with enormous cutlasses (Mariette, Dendérah, p. 91, 1875).
(2) N. flava.—Same arrangement of scales. Neck dilatable. 200-227 ventrals; 50-67 subcaudals.
Colour very variable, yellowish, reddish, brown, or black, uniform or with light spots; sometimes a black transverse band on the neck.
Total length, 1,470 millimetres; tail 230.
Habitat: South Africa. 62
(3) N. melanoleuca.—Coloration very variable. Sides of the head yellow or whitish; labial shields tinged with black on the posterior edge.
Total length, 2,400 millimetres; tail 400.
Habitat: Tropical Africa.
(4) N. nigricollis.—Third supralabial shield the deepest, sixth and seventh not in contact with the postoculars; 183-228 ventral scales; 55-68 subcaudals.
Coloration very variable; lower surface of the neck with a black transverse bar.
Total length, 2,000 millimetres; tail 300.
Habitat: From Senegambia and Upper Egypt to Angola and the Transvaal.
(5) N. anchietæ.—Scales in 17 rows on the neck and on the body; 181-192 ventrals; 52-62 subcaudals.
Colour blackish or brown above; end of snout and cheeks yellow; belly yellow or pale brown, with or without black cross-bar under the neck.
Total length, 1,800 millimetres; tail 340.
Habitat: Angola and Ovamboland.
(6) N. goldii.—Eyes large, two-thirds the length of the snout in adults. Scales in 15 rows on the neck and on the body; 194—195 ventrals; 88 subcaudals.
Colour black, uniform, or with transverse series of small whitish spots; cheeks and end of snout white, with a black border at the margin of the shields; belly white in front, black behind; subcaudal scales black.
Total length, 1,750 millimetres.
Habitat: Lower Niger.
Maxillary bones projecting considerably beyond the palatines, with a pair of large poison-fangs; no other maxillary teeth. Head not distinct from neck; eyes of moderate size, with round pupils; 63 nostril between two nasal shields and an internasal. Body cylindrical; scales oblique, keeled, in 19 rows; ventrals rounded. Tail moderate; subcaudal scales in 2 rows.
S. hæmachates (The Spitting Snake, or Ring Hals Slang).—This snake, which is about 700 millimetres in length, is of a bluish-brown colour, with numerous narrow, undulating and denticulate cross-bands of yellow or yellowish-white. The throat is black or dark red; the belly grey.
It is met with throughout West, East, and South Africa, as far as the Cape of Good Hope, where it is very common. It lives among bushes in sandy places, where the ground is full of the holes of rats, moles, and small rodents, upon which it feeds. It is very active and exhibits great ferocity.
Natives at the Cape declare that this snake is able to project its venom to a distance of more than a yard, especially if the wind is blowing in the same direction, and that, if the poison happens to enter the eye, the inflammation that results therefrom often leads to loss of sight (Smith).
As to this, a very definite statement is given by Bavay,4 on the authority of Le Naour:—
“While hunting in Dahomey,” wrote Le Naour to Bavay, “I thrice met with the snake called the Spitter. On two occasions my dog was struck in both eyes by the liquid projected by the reptile. Immediately (less than two minutes afterwards), symptoms of conjunctivitis manifested themselves, with considerable swelling of the pupils; the conjunctivitis seemed as though it were going to be very serious, and yielded only after twelve 64 days’ treatment with boracic lotion, aided by a few cauterisations with sulphate of copper.
“During my sojourn at Porto-Novo, a store-keeper at the Dogba trading station was a victim of the Spitter. While working at his store he received in his eye a jet of liquid, which produced violent conjunctivitis.”
Maxillary bones extending forwards beyond the palatines, as in Sepedon, with a pair of large poison-fangs; no other maxillary teeth. Head slightly distinct from the neck. Eyes of moderate size, with round or vertically elliptic pupils. Rostral shield very large, detached on the sides. Body cylindrical; scales oblique, smooth or keeled, in 19-23 rows; ventrals rounded. Tail short, obtuse; subcaudal scales in 2 rows.
(1) A. lubricus.—146-167 ventral scales; 20-28 subcaudals.
Colour orange or red, with black rings; a black bar below the eyes; upper surface of head sometimes entirely black.
Total length, 590 millimetres; tail 55.
Habitat: Cape Colony and Namaqualand.
(2) A. scutatus (Fula-fula of Mozambique).—115-135 ventrals; 24-38 subcaudals.
Colour pale grey, with black spots or cross-bars, and a black mark shaped like a circumflex accent on the head. A black collar encircling the neck, and a black vertical streak below the eye. Belly whitish.
The total length may be as much as 520 millimetres.
Habitat: Natal, Delagoa Bay, Mozambique.
Maxillary bones prolonged forwards beyond the palatines, with a pair of large poison-fangs; no other maxillary teeth. Head distinct from the neck; eyes rather small, with round pupils; 65 nostril between two or three nasal shields and the internasal. Body cylindrical; scales smooth or feebly keeled, in 23 rows; ventrals rounded. Tail rather short; subcaudal scales for the most part in 2 rows.
W. ægyptia.—Colour blackish-brown on the back, paler on the belly.
Total length, 1,170 millimetres; tail 170.
Habitat: Egypt.

Fig. 34.—Skull of Dendraspis viridis (Poisonous West African Colubrine). (After G. A. Boulenger, op. cit.)
Maxillary bone curved upwards, bearing a pair of powerful poison-fangs, not fissured, and not followed by other teeth; a 66 long tooth at the end of each ramus of the mandible. Head narrow, elongate; eye moderate, with round pupil; nostril between two shields. Body slightly compressed; scales smooth, narrow, very oblique, in 13-23 rows; ventrals rounded. Tail long; subcaudals in 2 rows.
(1) D. viridis.—211-225 ventral scales; 107-119 subcaudals.
Colour uniform olive-green. Shields on the head edged with black; lips yellow; belly and tail yellow, with scales and shields bordered with black.
Total length, 1,830 millimetres; tail 460.
Habitat: West Africa, from the Senegal to the Niger; St. Thome Island.
(2) D. jamesonii.—Coloration as before. Scales in 15-19 rows (19-21 on the neck); 210-235 ventrals; 99-121 subcaudals. Tail sometimes black.
Total length, 2,100 millimetres; tail 560.
Habitat: West Africa, from the Niger to Angola; Central Africa.
(3) D. angusticeps (The Mamba).—202-270 ventral scales; 99-121 subcaudals.
Colour green, olive, or blackish, uniform; belly yellowish or pale green; caudal scales and shields not bordered with black.
Total length, 2,000 millimetres; tail 430.
Habitat: West Africa, south of the Congo; Central Africa, East Africa, Transvaal, Natal.
(4) D. antinorii.—Scales in 21-23 rows; 248 ventrals; 117 subcaudals. Colour olive on the back, yellowish on the belly.
Total length, 2,690 millimetres; tail 545.
Habitat: Abyssinia.
The African Viperidæ all belong to the Subfamily Viperinæ, of which the following are the seven principal genera:— 67
(a) Causus. (b) Vipera. (c) Bitis. (d) Cerastes. (e) Echis. (f) Atheris. (g) Atractaspis.
Head distinct from the neck, covered with symmetrical shields; nostril between two nasal shields, and the internasal; eyes moderate, with round pupils, separated from the lips by subocular shields. Body cylindrical; scales smooth or keeled, oblique on the sides, in 15-22 rows; ventral scales rounded. Tail short; subcaudals in 2 rows or single.
Four species:—
(1) C. rhombeatus.—Snout obtuse, moderately prominent. Scales in 17-21 rows; 120-155 ventrals; 15-29 subcaudals.
Colour olive or pale brown, usually with a series of V-shaped brown spots bordered with white, and a large spot in the form of a circumflex accent at the back of the head; lips bordered with black; belly yellowish or grey.
Total length, 700 millimetres; tail 75.
Habitat: Tropical and South Africa, from the Gambia to the Cape.
(2) C. resimus.—Snout prominent, more or less turned up; scales in 19-22 rows; 134-152 ventrals; 17-25 subcaudals.
Colour greyish-olive on the back; uniform white on the belly.
Total length, 470 millimetres; tail 40.
Habitat: Central and East Africa, Angola.
(3) C. defilipii.—Snout prominent, more or less turned up. Scales in 17 rows; 113-125 ventrals; 10-18 subcaudals. 68
Colour grey or pale brown above, with a series of large rhomboidal or V-shaped blackish-brown spots; a large A-shaped dark brown mark on the occiput; an oblique dark streak behind the eye; supralabial shields edged with black; belly yellowish.
Total length, 400 millimetres; tail 22.
Habitat: Central and East Africa, Transvaal.

Fig. 35.—Skull of Causus rhombeatus. (After G. A. Boulenger, op. cit.)
(4) C. lichtensteinii.—Snout obtuse; scales in 15 rows; 142-144 ventrals; subcaudals 15-21, single.
Colour greyish, with rather indistinct darker chevron-shaped cross-bands.
Total length, 413 millimetres; tail 35.
Habitat: West Africa (Gold Coast), Congo. 69
In North Africa are found Vipera latastii, V. ammodytes, and especially V. lebetina, the range of which extends from Morocco to Northern India. Vipera superciliaris, which occurs on the coast of Mozambique, has the snout rounded, and the head covered with small, imbricate, keeled scales, with a large supraocular shield; nostril very large, between two nasal shields; scales on the body strongly keeled, in 27 rows; 142 ventrals; 40 subcaudals.
The colour is pale reddish-brown or orange, with blackish transverse bars broken by a longitudinal yellow band on each side; the belly is white, spotted with black.
Total length, 570 millimetres; tail 77.
The Viperidæ belonging to this genus have the head very distinct from the neck, covered with small imbricate scales; the eyes rather small, with vertical pupils, separated from the lips by small scales; the nostrils directed upwards and outwards, usually pierced in a single nasal shield, with a rather deep pit above, closed by a valvular supranasal. The postfrontal bones are very large, in contact with the ectopterygoids. Scales keeled, with apical pits, in 22-41 rows; ventrals rounded. Tail very short; subcaudal scales in 2 rows.
(1) B. arietans (the Puff Adder; fig. 37).—This viper has the nostrils on the upper surface of the snout, and two series of scales between the supranasal shields; 12-16 supralabials; 3-5 lower labials. The scales on the body are in 29-41 rows, and strongly keeled; ventrals 131-145; subcaudals 16-34.
The body is thick, the head large and triangular, and the tail very short. The colour is dirty yellow or orange, with large, 70 transverse or oblique, chevron-shaped black bands; an oblique black band extends behind the eye. The belly is dirty yellow, uniform, or marked with small black spots.
Total length, 1,350 millimetres, sometimes more; tail 160.

Fig. 36.—Skull of Bitis arietans (the Puff Adder). (After G. A. Boulenger, op. cit.)
Habitat: This snake is met with throughout Africa, from Southern Morocco, Kordofan, and Somaliland, to the Cape of Good Hope, and also in Southern Arabia. It is especially common near the Niger and on the Congo.
When irritated, it puffs itself out to such an extent that its 71 body becomes twice the ordinary size. It then doubles back its head and neck in the shape of an S, and emits a loud and prolonged hiss. Before biting, it first strikes a blow with its head as with a battering-ram, thus justifying its French name, vipère heurtante (Striking Viper).

Fig. 37.—Bitis arietans (the Puff Adder). (After Duméril and Bibron.)
The natives of South Africa assert that this Viper is able to 72 spring high enough to strike a rider on horseback. It feeds upon rats and mice, in search of which it often approaches habitations.
The Hottentots hunt it in order to obtain its venom; they pound its head between stones, and mix the pulp with the juice of certain plants for the purpose of poisoning their arrows.
It lives for a fairly long time in captivity. At the Pasteur Institute at Lille I have succeeded in keeping one of these snakes for two years, feeding it by forcing milk and eggs down its throat.
(2) B. peringueyi.—Nostrils opening upwards and outwards. Head covered with small, strongly keeled scales, which are smallest on the vertex; 11 scales round the eye; 3 series of scales between the eye and the lip; 11-14 supralabials. Scales on the body in 25-27 rows, strongly keeled; 130-132 ventrals; 19-28 subcaudals.
Colour greyish-olive, with 3 longitudinal series of grey or blackish spots; head sometimes with a trident-shaped dark mark, followed by a cross; under surface whitish, with small dark spots.
Total length, 325 millimetres; tail 26.
Habitat: Angola and Damaraland.
(3) B. atropos.—Nostrils opening upwards and outwards, 13-16 scales round the eye; 2-5 series of scales between the supranasals; 10-12 supralabials; 3-4 infralabials. Scales on the body in 29-31 rows, all strongly keeled; 124-145 ventrals; 18-29 subcaudals.
Colour brown or grey-brown, with 4 longitudinal series of dark spots, edged with black and white; two large black marks on the head; belly grey or brown, with darker spots.
Total length, 350 millimetres; tail 25.
Habitat: Cape of Good Hope.
(4) B. inornata.—Eyes smaller than in B. atropos, and separated from the lips by 4 series of scales; supraorbital region raised, but without erect horn-like scales; 15-17 scales across the head; 13-14 supralabials; 3 lower labials. Scales on the body in 27-29 rows, all keeled; 126-140 ventrals; 19-26 subcaudals. 73
Total length, 350 millimetres; tail 30.
Habitat: Cape of Good Hope.
(5) B. cornuta (fig. 38).—Nostrils opening upwards and outwards. Head covered with small, imbricate, strongly keeled scales; 2-5 raised scales, like horns, above each eye; 12-14 scales round the eye; 12-15 supralabials; 2-3 infralabials. Scales on the body keeled, in 25-29 rows; 120-152 ventrals; 18-36 subcaudals.
Colour grey or reddish-brown, with black spots, edged with white and arranged in 3 or 4 longitudinal series; a dark, oblique streak from the eye to the mouth; belly yellow or brown, uniform or spotted.
Total length, 510 millimetres; tail 35.

Fig. 38.—Bitis cornuta.
(After Duméril and Bibron.)
Habitat: Cape Colony, Namaqualand, Damaraland.
(6) B. caudalis.—Nostrils opening upwards and outwards. 12-16 scales from one eye to the other across the head; above each eye a single, erect, horn-like scale; 10-16 scales round the eye; 10-13 supralabials; 2-3 infralabials. Scales on the body in 22-29 rows, strongly keeled; 112-153 ventrals; 18-33 subcaudals.
Colour reddish or sandy-grey, with 2 series of brown spots with light centres, and frequently a vertebral series of narrow spots; belly dull yellow, uniform, or with small black spots on the sides.
Total length, 360 millimetres; tail 25.
Habitat: South-west Africa, from Angola to Namaqualand.
(7) B. gabonica (Gaboon Viper, or River Jack Viper).—Nostrils directed upwards and outwards. Head covered with small, moderately keeled scales, smallest on the vertex, 13-16 from eye to eye; 15-19 scales round the eye; a pair of erectile, triangular, nasal “horns,” consisting of sometimes tricuspid shields, between the supranasals; 13-16 supralabials; 4-5 infralabials. Scales 74 on the body in 33-41 rows, strongly keeled; lateral scales slightly oblique; 125-140 ventrals; 17-33 subcaudals.
This viper, which often attains a length of 1,200 millimetres, is brown, with a vertebral series of quadrangular, yellowish, or light brown spots connected by black markings; the belly is dull yellow, with small brown or blackish spots.
Habitat: Tropical Africa (West Africa, from Liberia to Damaraland; Zanzibar, Mozambique).
This species, which is nocturnal, is often met with on the Gaboon, and in the forests near the banks of the Ogowai. Its head is enormous, triangular in shape, and wider above; it has a bulky body, and a very short tail, terminating abruptly in a point.
The Gaboon Viper is a savage snake, with very active venom, and its poison-glands are of the size of large almonds. It lives in virgin forests, among dead wood and rocks. I have several times met with it in manioc plantations on the edge of the woods. In broad daylight it is sluggish, moves somewhat slowly, and never attacks man. It bites only when surprised.

Fig. 39.—Bitis nasicornis.
(After Duméril and Bibron.)
(8) B. nasicornis (fig. 39).—Nostrils opening upwards and outwards. Head covered with small strongly keeled scales, smaller on the vertex, 14-16 from one eye to the other; 2 or 3 pairs of compressed, erectile, horn-like shields between the supranasals, usually separated in the middle by 1 or 2 series of small scales; 15-18 supralabials; 4-6 infralabials. Scales on the body in 35-41 rows, strongly keeled; 124-140 ventrals; 16-32 subcaudals.
Colour purple or reddish-brown above, with pale olive or dark 75 brown spots; a vertebral series of brown, black-edged spots, which assume a rhomboidal form; sides of head dark brown, with a triangular light mark in front of the eye, and an oblique light streak from behind the eye to the mouth; belly pale olive, spotted with black or yellow.
Total length, 1,250 millimetres; tail 125.
Habitat: West Africa, from Liberia to the Gaboon.
Head very distinct from the neck, covered with small juxtaposed or slightly imbricate scales; eyes small, with vertical pupils, separated from the lips by small scales; nostrils opening upwards and outwards. Body cylindrical; scales keeled, with apical pits, in 23-35 rows. Tail short; subcaudals in 2 rows.
(1) C. cornutus (fig. 40).—Snout very short and broad; two erectile horns above the eyes, which are separated by 15-21 scales and surrounded by 14-18; 4-5 series of scales between the eyes and the lips; 12-15 supralabials; 3 infralabials; scales on the body in 27-35 rows; 130-165 ventrals; 25-42 subcaudals.

Fig. 40.—Cerastes cornutus.
(After Duméril and Bibron.)
Colour yellowish-brown or grey, with or without brown spots, forming 4-6 regular series, the two middle ones sometimes forming cross-bars; an oblique dark streak behind the eye; belly white; end of tail sometimes black.
Total length, 720 millimetres; tail 90.
Habitat: Northern border of the Sahara, Egypt, Nubia, Arabia, and Southern Palestine.
(2) C. vipera.—Snout very short and broad; head covered with small, tubercularly keeled scales, to the number of 9-13 from eye to eye; no “horns”; 9-14 scales round the eye; nostril between 76 two small shields, separated from their neighbours by 5-6 series of scales; 10-12 supralabials; 3 infralabials. Scales on the body in 23-27 rows; 102-122 ventrals, rather strongly keeled at the sides; 18-26 subcaudals. Colour dull yellow, pale brown or reddish, with or without black spots; end of tail often black above; ventral surface white.
Total length, 340 millimetres; tail 30.
Habitat: Northern border of the Sahara, from Algeria to Egypt.
The snakes belonging to this genus live constantly hidden in the sand, lying in wait for small birds, which alight beside them without suspicion, mistaking their horns for insects or larvæ; they also feed upon mice. Their poison-fangs are of relatively large size.
These small and exceedingly active vipers, whose colour harmonises marvellously with their surroundings, are very dangerous to the Arabs and blacks, who walk barefooted; they frequently cause fatal accidents.
They are able to exist for a very long time without drinking. They are attracted by the fires which are lighted at night round caravan encampments.
(1) E. carinatus (Efa, Viper of the Pyramids).—The same as met with in Persia, Arabia, and India. Very common in the environs of Cairo, and throughout Egypt and Abyssinia. It often makes its way into towns and villages. Brehm records that he more than once found an Efa in his house at Khartoum, and that on one occasion he discovered one of these vipers coiled up beneath the covering of his bed. At another time, getting up in the night, he put his foot on one of these animals and was not bitten, the reptile being very fortunately just at that moment in the act of devouring a tame bird which it had seized.
It hardly ever happens that a native of Egypt can bring 77 himself to destroy an Efa, of which he has the greatest dread. If, as often occurs, he finds one of these reptiles in his house, he addresses himself to the Hani or juggler, in order that, by his magic art, he may expel the dangerous visitor. From this custom the juggler evidently derives no small advantage, for, as is only right, he does not ply his craft for nothing. In many cases, indeed, the juggler releases a snake in a house, and then goes and informs the owner that he knows that a reptile is concealed in his dwelling, and that, in consideration of a stipulated reward, he will rid him of it (Brehm).

Fig. 41.—Echis coloratus. (After G. A. Boulenger, op. cit.)
(2) E. coloratus (fig. 41).—Scales on the snout and vertex convex, smooth or bluntly keeled, 13-15 from eye to eye; no supraocular shield; 17-22 scales round the eye; 12-15 supralabials; scales on the body in 31-35 rows; 174-205 ventrals; 42-52 subcaudals. No cruciform mark on the head.
Total length, 750 millimetres; tail 80.
Habitat: Palestine, Arabia, Socotra. 78
Head very distinct from neck, covered with imbricate scales; eyes large, with vertical pupils, usually separated from the labial shields by small scales; nostrils lateral. Body slightly compressed; scales keeled, with apical pits. Tail moderate, prehensile; subcaudal scales in a single row.
(1) A. chlorechis.—No supraciliary horn-like scales; 9-11 scales from eye to eye; 25-36 rows of scales in the middle of the body, strongly keeled; 154-165 ventrals; 53-62 subcaudals.
Colour green, uniform or with small yellow spots; end of tail yellowish or blackish.
Total length, 520 millimetres; tail 85.
Habitat: West Africa, from Liberia to the Ogowai.
(2) A. squamiger.—No supraciliary horn-like scales; 7-8 scales from eye to eye; 15-25 rows of scales in the middle of the body, strongly keeled; 153-173 ventrals; 51-95 subcaudals.
Colour olive, uniform or with more or less regular, narrow yellow cross-bands, or yellow with green spots; belly pale olive, marbled with black or yellow, or uniform yellow.
Total length, 550 millimetres; tail 100.
Habitat: West Africa, from the Cameroons to Angola.
(3) A. ceratophorus.—Several erect, supraciliary horn-like scales; 9-10 scales from eye to eye; 25 rows of scales in the middle of the body, strongly keeled; 142 ventrals; 55 subcaudals.
Colour dark olive, with black spots forming cross-bands; belly pale olive, speckled with black.
Total length, 210 millimetres; tail 65.
Habitat: East Africa.
This genus is characterised by enormous poison-fangs, a few teeth on the palatines, and none on the pterygoids. The mandible, which is edentulous in front, has only two or three small teeth in 79 the middle of the dentary bone. Head small, not distinct from the neck, covered with large symmetrical shields; nostril between two nasal shields; eyes minute, with round pupils; postfrontal bone absent. Body cylindrical; scales smooth, in 17-37 rows; ventrals rounded. Tail short; subcaudals in 1 or 2 rows.
(1) A. hildebrandtii.—Six supralabials; no præocular shields; frontal shorter than the parietals; scales on the body in 17 rows; ventrals 167-174.
Colour uniform dark brown.
Total length, 450 millimetres; tail 53.
Habitat: East Africa.
(2) A. congica.—Five supralabials, of which the fourth is the larger; postocular in contact with a large temporal; one præocular; frontal as long as or slightly shorter than the parietals. Scales on the body in 19-21 rows; 209-230 ventrals; 19-23 subcaudals.

Fig. 42.—Skull of Atractaspis aterrima (African Viperine). (After G. A. Boulenger, op. cit.)
Colour uniform dark brown or black.
Total length, 450 millimetres; tail 35.
Habitat: Congo, Angola.
(3) A. irregularis.—Characters as before, but scales on the body in 25-27 rows; 220-257 ventrals, subcaudals 22-28 pairs.
Colour uniform black or dark brown.
Total length, 560 millimetres; tail 35. 80
Habitat: West Africa, from the Gold Coast to the Congo; Central Africa.
(4) A. corpulenta.—Postocular shield in contact with a large temporal; second lower labial very large, fused with the chin-shields. Scales on the body in 23-27 rows; 178-193 ventrals; 23-27 subcaudals.
Colour uniform blackish-brown; tail sometimes white.
Total length, 345 millimetres; tail 33.
Habitat: West Africa, from Liberia to the Gaboon.
(5) A. rostrata.—Snout very prominent, cuneiform. Third lower labial very large; first lower labial in contact with its fellow, behind the symphysial. Scales on the body in 19-23 rows; ventrals 227-248.
Colour uniform dark brown, or blackish.
Total length, 600 millimetres; tail 37.
Habitat: East and Central Africa.
(6) A. bibronii.—Characters as before. Snout prominent, subcuneiform. Ventral scales, 221-260.
Colour dark purplish-brown above, dull yellow or pale brown on the belly.
Total length, 600 millimetres; tail 25.
Habitat: Eastern districts of Cape Colony, Natal, Namaqualand, Angola.
(7) A. aterrima.—Characters as before. Snout rounded; 251-300 ventral scales.
Colour uniform dark brown or black.
Total length, 650 millimetres; tail 30.
Habitat: West and Central Africa.
(8) A. dahomeyensis.—Characters as before. Symphysial shield in contact with the chin-shields. Scales on the body in 31 rows; 240 ventrals; 24 subcaudals.
Colour black above, brown on the belly.
Total length, 490 millimetres; tail 32.
Habitat: Dahomey. 81
(9) A. micropholis.—Temporal shields small, 2 + 3 or 4; fourth or fifth infralabial larger; scales on the body in 25 rows; 210-215 ventrals; 29-30 subcaudals. Frontal shield slightly longer than broad, much longer than the parietals.
Colour uniform dark brown.
Total length, 330 millimetres; tail 28.
Habitat: Cape Verd.
(10) A. leucomelas.—Characters as before. Frontal one and two-fifths as long as broad, as long as the parietals.
Colour black, with a vertebral white line, occupying one row and two half rows of scales; ventrals and subcaudals white; neck black, head white, with a black spot covering the nasals and upper head-shields.
Total length, 575 millimetres; tail 40.
Habitat: Somaliland.
(11) A. microlepidota.—Characters as before. Scales on the body in 29-37 rows; 212-245 ventrals; 26-37 subcaudals.
Colour uniform dark brown.
Total length, 540 millimetres; tail 45.
Habitat: Central and East Africa.
The Sunda Islands and the whole of Malaysia are rich in poisonous snakes. Those that are found there belong for the most part to species that we have already met with in India or the Malay Peninsula. We shall therefore not describe them again here.
All those that inhabit Australia are included in the great Family Colubridæ and the Subfamily Elapinæ. There are no Viperidæ; but certain genera of poisonous Colubridæ are peculiar to this continent.
These reptiles have been particularly well studied by Gérard 82 Krefft, formerly Director of the Australian Museum at Sydney, from whose work5 we shall borrow a considerable portion of the following notes, and the figures accompanying them.
The genera represented in Australia are:—
| (a) | Ogmodon. |
| (b) | Glyphodon. |
| (c) | Pseudelaps. |
| (d) | Diemenia. |
| (e) | Pseudechis. |
| (f) | Denisonia. |
| (g) | Micropechis. |
| (h) | Hoplocephalus. |
| (i) | Tropidechis. |
| (j) | Notechis. |
| (k) | Rhinhoplocephalus. |
| (l) | Brachyaspis. |
| (m) | Acanthophis. |
| (n) | Elapognathus. |
| (o) | Rhynchelaps. |
| (p) | Furina. |
This genus is characterised by the maxillary bones extending forwards as far as the palatines, and bearing, in addition to the poison-fangs, 6-7 grooved teeth. The head is not distinct from the neck; the eyes are very small. The body is cylindrical, and covered with smooth scales in 17 rows. Tail short; subcaudal scales in 2 rows.
O. vitianus.—Snout elongate, pointed; 139-152 ventral scales; 27-38 subcaudals. 83
Colour dark brown, lighter on the sides; belly brown or white, more or less spotted with black; tail black.
Total length, 360 millimetres; tail 45.
Habitat: Fiji Islands.

Fig. 43.—Skull of Glyphodon tristis (Australian Colubrine).
(After G. A. Boulenger, op. cit.)
General characters the same; snout rounded; poison-fangs followed, after a wide interspace, by 6 small grooved teeth; anterior mandibular teeth strongly developed (fig. 43). Head and 84 eyes small; pupils round or vertically subelliptic; nostrils pierced between 2 nasal shields. Body cylindrical; scales smooth, in 17 rows; tail short; subcaudals in 2 rows.
G. tristis.—Ventral scales 165-179; subcaudals 38-52.
Colour dark brown; occiput often yellowish, or pale reddish-brown; belly yellow.
Total length, 900 millimetres; tail 125.
Habitat: North-eastern Australia and South-eastern New Guinea.
Maxillaries extending forwards as far as the palatines, with a pair of large grooved poison-fangs, and, after a wide interval, 8-12 small grooved teeth. The anterior mandibular teeth are of large size, almost like fangs. Head hardly distinct from the neck; eyes small, with vertically elliptic pupils. Body cylindrical; scales smooth, in 15-17 rows. Tail moderate or short; subcaudal scales in 2 rows.
(1) P. muelleri.—Scales in 15 rows. Nasal shield divided, in contact with the præocular; 2 + 2 temporals; 139-176 ventral scales; 21-35 subcaudals.
Colour brown, with a light vertebral line; a more or less distinct dark, light-edged streak on each side of the head, passing through the eye; belly yellowish or coral-red; uniform or spotted with black.
Total length, 500 millimetres; tail 70.
Habitat: Moluccas, New Guinea, New Britain.
(2) P. squamulosus.—Scales in 15 rows. Nasal shield divided, in contact with the præocular; 1 + 2 temporals; 170-183 ventrals; 34-52 subcaudals.
Colour brown, with a yellowish streak round the snout and through the eyes to the nape; belly whitish, with confluent black spots forming lines on each side. 85
Total length, 375 millimetres; tail 55.
Habitat: New South Wales.
(3) P. krefftii (fig. 44).—Nasal shield entire, in contact with the præocular; 1 + 2 temporals; 146-156 ventrals; 26-38 subcaudals.
Colour dark brown, with a light longitudinal line on each scale; a yellowish cross-band on the occiput, connected with another yellow band which encircles the snout.
Belly yellowish in front, black behind; subcaudals white, with a longitudinal black band running between them.

Fig. 44.—Pseudelaps krefftii.

Fig. 45.—Pseudelaps harriettæ.

Fig. 46.—Pseudelaps diadema.
Total length, 255 millimetres; tail 33.
Habitat: Queensland.
(4) P. harriettæ (fig. 45).—Nasal shield entire, in contact with or narrowly separated from the præocular; 1 + 2 temporals; 176-193 ventrals; 29-35 subcaudals.
Colour dark brown, with a light longitudinal line on each scale; a long yellow blotch on the nape, and, connected with this, a yellow ring round the snout; ventrals and subcaudals brown or black, edged with white.
Total length, 415 millimetres; tail 45.
Habitat: Queensland.
(5) P. diadema (fig. 46).—Nasal shield entire, widely separated from the præocular; 2 + 2 temporals; third and fourth upper labials entering the eye; 164-203 ventrals; 40-62 subcaudals. Colour pale brown or reddish, with a brown edging to each scale forming a reticulate pattern; a yellow cross-band on the occiput; belly uniformly white.
Total length, 600 millimetres; tail 80. 86
Habitat: Eastern, Northern, and Western Australia.
(6) P. warro.—Characters the same as in the previous species; 143 ventrals. Colour brown; a broad lunate black collar on the nape; head black above, but paler than the collar.
Habitat: Port Curtis, Queensland.
(7) P. sutherlandi.—Characters as before. Scales on the body in 17 rows; 160 ventrals; 40 subcaudals. Colour red-brown on the back, yellow on the belly; a broad lunate black collar on the nape, with a lighter edging; light bars across the head, body, and tail.
Habitat: Norman River, Queensland.

Fig. 47.—Diemenia psammophis.

Fig. 48.—Diemenia olivacea. (Northern
Australia and New Guinea).
(After Krefft.)

Fig. 49.—Diemenia textilis.
Maxillary bones extending forwards as far as the palatines, with a pair of large grooved poison-fangs, followed, after an interspace, by 7-15 small grooved teeth; anterior mandibular teeth much elongated, resembling poison-fangs. Head scarcely distinct from the neck; eyes rather large, with round pupils; nasal shield entire or divided; frontal elongate.
Body cylindrical; scales smooth, in 15-19 rows (more on the neck). Tail moderate or long; subcaudals all or for the most part in 2 rows.
Coloration very variable, orange-yellow, olive, red-brown, or pale brown.
Average length, 1,000-1,700 millimetres.
Habitat: South-eastern New Guinea, and Australia. 87
Seven species of this genus are known, divided into two groups as follows:—
(1) Scales on the body in 15 rows.
D. psammophis. (fig. 47).—Internasal shields at least half as long as the præfrontals.
D. torquata.—Internasals more than half as long as the præfrontals.
D. olivacea. (fig. 48).—Internasals not more than half as long as the præfrontals; snout broad.

Fig. 50.—Diemenia nuchalis.
(2) Scales in 17 or 19 rows.
D. modesta.—154-165 ventrals.
D. textilis (Brown Snake, fig. 49).—190-232 ventrals.
D. nuchalis (fig. 50).—184-224 ventrals.
Maxillary bones extending forwards as far as the palatines, with a pair of large grooved poison-fangs followed by 2-5 small solid teeth; anterior mandibular teeth long. Head distinct from the neck; eyes rather small, with round pupils; nostril between two nasal shields. Body cylindrical. Scales smooth, in 17-23 rows (more on the neck). Tail moderate; subcaudals in 2 rows, or partly single, partly in 2 rows.

Fig. 51.—Pseudechis porphyriacus.
Total length, 1,500-2,000 millimetres, sometimes more.
Habitat: Australia and New Guinea.
This genus includes eight species. 88
(1) P. porphyriacus (Black Snake; fig. 51).—Frontal shield longer than broad; 180-200 ventrals; 50-60 subcaudals.
Colour black on the back; outer row of scales red at the base; ventrals red, edged with black.
(2) P. cupreus.—199-210 ventrals; 57-72 subcaudals.
Colour coppery above, brown or orange below, all the scales and shields edged with brown.
(3) P. australis.—199-220 ventrals; 57-70 subcaudals; frontal shield once and two-thirds to twice as long as broad.
Colour pale brown on the back, yellowish on the belly.
(4) P. darwiniensis.—Frontal as broad as long; 212 ventrals; 54-64 subcaudals.
Colour reddish-brown; head pale brown; belly yellowish-white.
(5) P. papuanus.—Scales in 19-21 rows (26 or 27 on the neck); 221-224 ventrals; 49-55 subcaudals.
Colour uniform black; chin white.
(6) P. scutellatus.—Subcaudal shields in 2 rows; scales on the body in 23 rows (25-30 on the neck); 230-233 ventrals; 61-78 subcaudals.
Colour pale brown or dark brown; snout and cheeks pale brown or yellowish; belly yellow.
(7) P. microlepidotus.—30-36 scales across the neck, 23 across the middle of the body; 232-237 ventrals; 61-66 pairs of subcaudals.
Colour dark brown on the back, yellowish-grey on the belly; head sometimes blackish.
(8) P. ferox.—Snout very broadly rounded. Scales on the body in 23 rows; 235 ventrals; 60 pairs of subcaudals.
Colour black above, yellowish beneath.
Maxillary bones prolonged as far as the palatines, with a pair of large grooved poison-fangs, followed by 3-5 small solid teeth; 89 anterior mandibular teeth greatly developed. Head fairly distinct from the body; eyes small, with round or vertically elliptic pupils; nasal shield entire or divided. Body cylindrical; scales smooth, in 15-19 rows; tail moderate or short; subcaudals in a single row, except in one species.
According to the British Museum Catalogue, the genus Denisonia includes twenty-one species, the principal characters of which are as follows:—

Fig. 52.—Denisonia superba (The Copperhead).
(1) D. superba (The Copperhead. Fig. 52).—Scales in 15-17 rows; nasal shield entire, in contact with the præocular; ventral scales 145-160; subcaudals 41-50.
Colour brownish to dark olive on the back, often yellow or salmon-red on the sides; belly yellowish or greyish-olive.
Total length, 1,010 millimetres; tail 160.
Habitat: New South Wales, Southern Australia, Tasmania.
(2) D. coronata.—Scales in 15 rows; 138-151 ventrals; 38-51 subcaudals.

Fig. 53.—Denisonia coronoides.
Colour olive, with a black streak on each side of the head; belly yellowish or pale olive.
Total length, 480 millimetres; tail 95.
Habitat: Western Australia and New South Wales.
(3) D. coronoides (fig. 53).—Scales in 15 rows; 136-151 ventrals; 39-57 subcaudals.
Colour brown, lips yellow; belly salmon-red to dark olive-grey; end of tail salmon-red.
Total length, 440 millimetres; tail 80. 90
Habitat: Southern Australia and Tasmania.
(4) D. muelleri.—Scales in 17 rows; 118 ventrals; 38 subcaudals.
Colour grey-brown; lips and chin with yellow spots; belly grey.
Total length, 292 millimetres; tail 52.
Habitat: Queensland.
(5) D. frenata.—Scales in 19 rows; 167 ventrals; 35 subcaudals.
Colour olive-brown; upper lip yellow; belly white.
Total length, 390 millimetres; tail 54.
Habitat: Lake Elphinstone, Queensland.
(6) D. ramsayi (fig. 54).—Scales in 15 rows; 164 ventrals; 51 subcaudals.

Fig. 54.—Denisonia ramsayi.
Colour dark olive-green above, yellow beneath; subcaudals nearly black.
Total length, 265 millimetres; tail 50.
Habitat: New South Wales.
(7) D. signata (fig. 55).—Scales in 17 rows; 153-170 ventrals; 41-56 subcaudals.

Fig. 55.—Denisonia signata.
Colour dark olive or black, head brown; belly dark grey or black.
Total length, 640 millimetres; tail 120.
Habitat: Queensland, New South Wales.
(8) D. daemelii.—Scales in 17 rows; 147-168 ventrals; 33-45 subcaudals.
Colour olive, head darker; belly yellowish-white.
Total length, 380 millimetres; tail 60.
Habitat: Queensland.
(9) D. suta.—Scales in 19 rows; 157-164 ventrals; 25-30 subcaudals.
Colour pale olive-brown, head dark brown, nape black; upper lip and belly yellow. 91
Total length, 200 millimetres; tail 23.
Habitat: Southern Australia.
(10) D. frontalis.—Scales in 19 rows; 154 ventrals; 30 subcaudals.
Colour light brown, with a vertebral black line; belly pearly-white, with a median bronze-coloured band.
Total length, 400 millimetres; tail 50.
Habitat: New South Wales.
(11) D. flagellum.—Scales in 17 rows; 132-138 ventrals; 25-27 subcaudals.
Colour pale brown; vertex, occiput, and nape black; upper lip and belly white.
Total length, 380 millimetres; tail 40.
Habitat: Victoria.
(12) D. maculata. (fig. 56).—Scales in 17 rows; 121-136 ventrals; 20-30 subcaudals.

Fig. 56.—Denisonia maculata.
Colour dark grey-brown, or brown; a large dark olive-green or brown blotch on the head, with two or three unequal light grey spots; belly white.
Total length, 400 millimetres; tail 55.
Habitat: Queensland.
(13) D. punctata.—Scales in 15 rows; 160 ventrals; 25 subcaudals.
Colour pale brown; head and nape orange; upper lip and belly yellow.
Total length, 350 millimetres; tail 36.
Habitat: North-Western Australia.
(14) D. gouldii (fig. 57).—Scales in 15 rows; 140-170 ventrals; 22-23 subcaudals.

Fig. 57.—Denisonia gouldii.
Colour brownish-yellow; nape black; head covered with a broad greenish-blue blotch, extending from the nose to the neck; upper lip and belly yellow. 92
Total length, 435 millimetres; tail 50.
Habitat: Western and Southern Australia.
(15) D. nigrescens.—Scales in 15 rows; 170-200 ventrals; 30-46 subcaudals.
Colour dark olive; head black; belly yellow.
Total length, 545 millimetres; tail 75.
Habitat: New South Wales and Queensland.
(16) D. nigrostriata.—Scales in 15 rows; 180-184 ventrals; 50-64 subcaudals.
Colour yellow, streaked with black; head dark brown; upper lip and belly yellowish-white.
Total length, 380 millimetres; tail 52.
Habitat: Queensland.
(17) D. carpentariæ.—Scales in 15 rows; 166-183 ventrals; 31-43 subcaudals.
Colour brown; upper lip and belly yellowish-white.
Total length, 285 millimetres; tail 47.
Habitat: Northern Queensland.
(18) D. pallidiceps.—Scales in 15 rows; 170-178 ventrals; 37-38 subcaudals.
Colour dark olive-brown; head somewhat paler; belly yellowish. Total length, 590 millimetres; tail 80.
Habitat: Northern Australia.
(19) D. melanura.—Scales in 15 or 17 rows; 165-171 ventrals; 38-48 subcaudals.
Colour dark brown; head and sides usually reddish; belly yellow; tail black.
Total length, 1,000 millimetres; tail 140.
Habitat: Solomon Islands.
(20) D. par.—Scales in 16 rows; 164-166 ventrals; 40-49 subcaudals.
Colour reddish-brown, in broad bands with white intervals; head blackish-brown; belly white; tail with red rings.
Total length, 750 millimetres; tail 110. 93
Habitat: Faro and Howla Islands, Bougainville Straits, Solomon Islands.
(21) D. woodfordii.—Scales in 17 rows; 166-172 ventrals; 41-45 subcaudal pairs.
Colour brownish-white, with a reticulate pattern; head dark brown; belly white.
Total length, 670 millimetres; tail 100.
Habitat: New Georgia, Solomon Islands.
Maxillary bones extending forward as far as the palatines, with a pair of large grooved poison-fangs, followed by three small solid teeth; mandibular teeth longer in front. Head distinct from the neck; eyes very small, with round pupils; nostril between two nasal shields. Body cylindrical; scales smooth, in 15 or 17 rows. Tail short; subcaudals in 2 rows.
(1) M. ikaheka.—Scales in 15 rows; 180-223 ventrals; 39-55 subcaudals.
Colour yellow and black, in irregular cross-bars; head and tail black above; belly yellow.
Total length, 1,550 millimetres; tail 180.
Habitat: New Guinea.
(2) M. elapoides.—Scales in 17 rows; 208 ventrals; 35 subcaudals.
Colour cream, with 22 black bands, broader than the interspaces between them; snout and ocular region black.
Total length, 750 millimetres; tail 75.
Habitat: Florida Island, Solomon Group.
Characters the same as in Micropechis. Scales in 21 rows; ventrals angulate and notched laterally. Tail moderate; subcaudals in a single row. 94
(1) H. bungaroides (Syn. H. variegatus. The Broad-headed Snake).—204-221 ventrals; 40-56 subcaudals.
Colour black on the back, with yellow spots forming more or less regular cross-bands on the body; upper lip yellow, margined with black; belly blackish, yellow on the sides.
Total length, 1,620 millimetres; tail 210.
Habitat: New South Wales.
(2) H. bitorquatus (fig. 58).—Ventral scales strongly angulate laterally, 191-227; subcaudals 44-59.

Fig. 58.—Hoplocephalus bitorquatus.
Colour olive-green; head pale olive, with a bright yellow occipital blotch, and a large black blotch on each side of the nape; a pair of small spots in front of and between the eyes; three black transverse blotches on the vertex; belly greyish-olive or brown.
Total length, 510 millimetres; tail 95.
Habitat: Queensland, New South Wales.
(3) H. stephensii.—239 ventrals; 60 subcaudals.
Body barred alternately with black and white; the black bars are twice as broad as the white ones; head dark, spotted with yellow; a W-shaped yellow mark on the back of the head.
Total length, 760 millimetres.
Habitat: Port Macquarie, New South Wales.
Same general characters; nasal shield entire; scales on the body strongly keeled, in 23 rows. Tail moderate; subcaudals in a single row.
T. carinatus.—Colour dark olive, with darker cross-bands; belly yellow, more or less tinged with olive-green.
Total length, 730 millimetres; tail 120.
Habitat: New South Wales, Queensland. 95
Same general characters; pupil round; nasal shield entire. Body cylindrical; scales smooth, disposed obliquely, in 15-19 rows, the lateral scales shorter than the dorsals. Tail moderate; subcaudals in a single row.
N. scutatus (Syn. Hoplocephalus curtus. The Tiger Snake. Fig. 59).—Colour dark olive; belly yellow or olive; the shields often dark-edged.

Fig. 59.—Notechis scutatus (The Tiger Snake.)
Total length, 1,280 millimetres; tail 170.
Habitat: Australia and Tasmania.
Dentition as in Hoplocephalus. Head but little distinct from the neck; eyes small, with round pupils; no internasal shields. Body cylindrical, rigid. Scales smooth, in 15 rows. Tail short; subcaudals in a single row.
R. bicolor.—Colour greyish-olive on the back, yellowish-white on the belly; tongue white.
Total length, 395 millimetres; tail 55.
Habitat: Australia.
Characters the same, but head distinct from the neck; eyes small, with vertically elliptic pupils; nostril between two nasal shields. Body stout, cylindrical; scales smooth, slightly oblique, in 19 rows. Tail short; subcaudals in a single row.
B. curta.—Colour uniform olive-brown; belly yellowish. 96
Total length, 490 millimetres; tail 70.
Habitat: Western Australia.
Maxillary bones extending forwards as far as the palatines, with a pair of large grooved poison-fangs, followed by two or three small teeth; anterior mandibular teeth elongate, fang-like. Head distinct from the neck; eyes small, with vertically elliptic pupils; nostril in the upper part of a single nasal shield. Body stout, cylindrical. Scales more or less distinctly keeled, in 21-23 rows. Tail short, compressed at the end and terminating in a sort of long spine, turned upwards; anterior subcaudals in a single row, posterior in 2 rows.

Fig. 60.—Skull of Acanthophis antarcticus. (After G. A. Boulenger, op. cit.)
A. antarcticus (The Death Adder; fig. 61).—Supraocular shields often raised and angular, assuming the shape of little horns; scales on the crown of the head rugose and striated; 113-130 ventrals; 41-51 subcaudals (the last 5-27 are divided). 97
Colour yellow-brown or reddish, with more or less distinct dark cross-bands; black spots or small dark bars on the lips; belly yellowish-white; end of tail yellow or black, covered with spiny scales.

Fig. 61.—Acanthophis antarcticus (The Death Adder).
Total length, 850 millimetres; tail 150.
Habitat: Moluccas, New Guinea, Australia.
Maxillary bones extending forwards as far as the palatines, with a pair of moderately large grooved poison-fangs; no other maxillary teeth; mandibular teeth subequal. Eyes moderate, with round pupils; nasal shield entire. Body cylindrical; scales smooth, in 15 rows; ventrals rounded; tail moderate; subcaudals in a single row.
E. minor.—Colour dark olive, with a black occipital blotch in the young; belly yellow or greenish-grey.
Total length, 460 millimetres; tail 95.
Habitat: South-west Australia.
Maxillary bones extending forwards as far as the palatines, with a pair of moderately large grooved poison-fangs, and two small teeth near the posterior extremity of the bone; anterior mandibular teeth the longest. Head small, not distinct from the neck; eyes small, with vertically elliptic pupils; nostril in a single nasal shield. Body short, cylindrical; scales smooth, in 15-17 rows. Tail very short; subcaudals in 2 rows.
(1) R. bertholdi.—Scales in 15 rows; 112-126 ventrals. 98
Colour yellow, with 19-40 black annuli, usually narrower than the interspaces; head brown, with a large black blotch on the nape, and another on the vertex and temples.
Total length, 270 millimetres; tail 22.
Habitat: Southern and Western Australia.
(2) R. australis (fig. 62).—Scales in 17 rows; 152-163 ventrals; 18-20 subcaudals.

Fig. 62.—Rhynchelaps australis.
Colour red on the body, with ill-defined cross-bars formed of yellowish black-edged scales; a black blotch on the head, covering the vertex and temples, and surrounding the eyes; another large blotch on the nape; snout and occiput yellow; belly white.
Total length, 290 millimetres; tail 25.
Habitat: Queensland.
(3) R. semifasciatus.—Scales in 17 rows; 143-170 ventrals; 17-25 subcaudals.
Colour yellow, with brown cross-bands; large brown blotches on the head and nape; belly white.
Total length, 300 millimetres; tail 30.
Habitat: West Australia.
(4) R. fasciolatus.—Scales in 17 rows; 145-161 ventrals; 22-27 subcaudals.
Colour red, with numerous blackish-brown cross-bands; large blackish-brown blotches on the head and nape; belly white.
Total length, 335 millimetres; tail 30.
Habitat: West Australia.
Maxillary bones extending forwards beyond the palatines, bearing a pair of moderately large grooved poison-fangs, and one or 99 two small teeth near their posterior extremities; mandibular teeth subequal. Head small, not distinct from neck; eyes very small, with round pupils; nostril in a single nasal shield. Body cylindrical; scales smooth, in 15 rows. Tail very short, obtuse; subcaudals in 2 rows.
(1) F. calonota.—Six supralabial shields; 126-131 ventrals; 29-30 subcaudals.
Colour yellow, with a black vertebral stripe; a black bar across the end of the snout; a large black blotch covering the vertex and the parietals; belly white.
Total length, 215 millimetres; tail 33.
Habitat: West Australia.
(2) F. bimaculata.—Scales in 15 rows; 181-200 ventrals; 21-25 subcaudals.

Fig. 63.—Skull of Furina occipitalis. (After G. A. Boulenger, op. cit.)

Fig. 64.—Furina occipitalis.
Colour yellow, with large black blotches on the snout, middle of head, and occiput; belly white.
Total length, 330 millimetres; tail 25.
Habitat: West Australia.
(3) F. occipitalis (fig. 64).—Scales in 15 rows; 180-234 ventrals; 14-25 subcaudals.
Entire body ringed with black and white, annuli narrower on 100 belly; head black, with a broad white band across the occiput, and another narrower and irregular one across the snout; nose black.
Total length, 590 millimetres; tail 36.
Habitat: Australia.
There are no poisonous snakes in New Zealand. In New Caledonia no terrestrial poisonous snakes are known, but Hydrophiidæ abound on its shores, as on those of the majority of the islands of the Pacific.
In Australia, especially in New South Wales and farther to the north, fatalities due to the bites of poisonous snakes are not rare. The most dangerous species are: Acanthophis antarcticus (the Death Adder), Diemenia textilis (the Brown Snake), Pseudechis porphyriacus (the Black Snake), and Notechis scutatus or Hoplocephalus curtus (the Tiger Snake).
The health authorities of this country have accordingly taken the wise precaution of circulating very widely among the public coloured placards bearing illustrations of these four species, with a description of the essential anatomical details by which they may be recognised. Similar placards are exhibited in all the schools, and a generous distribution is made of instructions, printed on handkerchiefs, indicating the most effective method of treating poisonous bites.
In Queensland, according to information furnished to me by Mr. C. W. De Vis, late Director of the Queensland Museum, Brisbane, the number of deaths resulting from the bites of poisonous snakes has been only twenty-seven in ten years.
The fauna of the New World includes only a very small number of poisonous snakes belonging to the family Colubridæ. The Genus Elaps alone is represented there by twenty-eight species, scattered over Mexico, Central America, Bolivia, Ecuador, Peru, Colombia, and Brazil. 101
Viperidæ, on the other hand, are extremely numerous, and belong exclusively to the subfamily Crotalinæ; there are no Viperinæ.

Fig. 65.—Skull of Elaps marcgravii. (After G. A. Boulenger, op. cit.)
The characters of this genus are: Maxillary bones very short, extending beyond the palatines, and bearing a pair of large poison-fangs; pterygoid teeth few or absent; mandibular teeth all of equal length. No postfrontal bones; præfrontals meeting, or narrowly separated on the median line. Head small, not distinct 102 from neck; eyes small, with vertically elliptic or sub-elliptic pupils; nostril between two nasal shields. Body cylindrical; scales smooth, in 15 rows. Tail short; subcaudal scales in 2 rows, or partly single, partly in 2 rows.
(1) E. surinamensis.—Seven supralabials, of which the fourth enters the eye; frontal shield very narrow; 167-182 ventrals.
Colour red, with black annuli disposed in threes (the middle one broader), separated by narrow yellow interspaces; 7 or 8 sets of annuli on the body; the red scales dotted with black; head red above, with the shields black-edged, followed by a black cross-band behind the parietals.
Total length, 740 millimetres; tail 95. Grows to 1,900 millimetres.
Habitat: Venezuela, Guianas, Northern Brazil, North-east Peru.
(2) E. heterochilus.—Six supralabials, second and third entering the eye; snout narrow; 209 ventrals.
Colour red, with black annuli, as in E. marcgravii.
Total length, 553 millimetres; tail 43.
Habitat: Brazil.
(3) E. euryxanthus (Sonoran Coral Snake).—Seven supralabials, third and fourth entering the eye; internasals nearly as long as the præfrontals; 215-241 ventrals; 21-29 subcaudals.
Colour red, with 11 black annuli edged with yellow; head black.
Total length, 400 millimetres; tail 33.
Habitat: Arizona, Colorado, North-western Mexico. It is found in Arizona up to an altitude of nearly 6,000 feet (1,800 metres).
(4) E. gravenhorstii.—First lower labial in contact with its fellow; posterior nasal not reaching the præocular: 191 ventrals; 23 subcaudals.
Colour: Seven sets of black annuli disposed in threes, median annulus the broadest; head black, with a yellow transverse band behind the eyes.
Total length, 550 millimetres; tail 50.
Habitat: Brazil. 103
(5) E. langsdorffii.—Ventrals 204-225; subcaudals 37-54; 1 + 1 temporals.
Colour dark brown, with 63 transverse series of cream-coloured spots, each occupying one scale; belly yellow, with red cross-bands.
Total length, 300 millimetres.
Habitat: Upper Amazons.
(6) E. buckleyi.—203-211 ventrals; 39-43 subcaudals; 1 + 2 temporals.
Colour orange, with 48-60 black annuli edged with small yellow spots; head black; temples yellow.
Total length, 505 millimetres; tail 70.
Habitat: Northern Brazil, Eastern Ecuador.
(7) E. anomalus.—227 ventrals; 29 subcaudals; 1 + 1 temporals.
Colour: Body with 55 black annuli separated by narrow brownish-white bands; belly yellowish; anterior half of head black, posterior half yellow; tail yellow or red, with 4 black rings.
Total length, 280 millimetres; tail 23.
Habitat: Colombia.
(8) E. heterozonus.—Scales in 15 rows; 210-219 ventrals; 16-23 subcaudals; 1 + 1 temporals.
Colour red or brown, with 17 to 23 black rings, mostly narrower than the interspaces; a black band on the head across the eyes.
Total length, 900 millimetres; tail 40.
Habitat: Eastern Ecuador, Eastern Peru, Bolivia.
(9) E. elegans.—Scales in 15 rows; 189-221 ventrals; 29-47 subcaudals; 1 + 1 temporals.
Colour: Black annuli in sets of three, separated by reddish-brown interspaces; 12-17 sets; head black, with yellow blotches.
Total length, 730 millimetres; tail 70.
Habitat: Mexico and Guatemala.
(10) E. annellatus.—Scales in 15 rows; 200-211 ventrals; 30-45 subcaudals; 1 + 1 temporals.
Colour black, with 41-49 narrow white rings on the body, 4-7 on the tail; a white ring on the head. 104
Total length, 490 millimetres; tail 70.
Habitat: Eastern Peru.
(11) E. decoratus.—Scales in 15 rows; 196-213 ventrals: 29-37 subcaudals.
Colour red, with 15-16 sets of black annuli disposed in threes; head yellow, with end of snout black, and a black band across the eyes.
Total length, 625 millimetres.
Habitat: Brazil.
(12) E. dumerilii.—Scales in 15 rows; 197-204 ventrals; 50-53 subcaudals.
Colour: 8-9 sets of three black annuli on red and yellow; head black, with a yellow band on the occiput.
Total length, 410 millimetres; tail 65.
Habitat: Colombia.
(13) E. corallinus (The Coral Snake).—Scales in 15 rows; 179-231 ventrals; 30-47 subcaudals; 1 + 1 temporals.
Colour: Body with black annuli, separated by red interspaces edged with yellow; head bluish-black; temples yellow; a blue line from behind the eye to the lower jaw; tail white.
Total length, 790 millimetres; tail 70.
Habitat: Tropical South America and Lesser Antilles (St. Thomas, St. Vincent, Martinique).
(14) E. hemprichii.—Scales in 15 rows; 168-181 ventrals; 22-29 subcaudals; 1 + 1 temporals.
Colour: Black, with red or yellow annuli, a broad annulus between the narrow ones; occiput, upper lip, and temples yellow.
Total length, 720 millimetres; tail 65.
Habitat: Guianas, Colombia, Peru.
(15) E. tschudii.—Scales in 15 rows; 207-221 ventrals; 21-28 subcaudals.
Colour: Body with black annuli broader than the interspaces, disposed in sets; interspaces red and yellow; snout and occiput black. 105

Fig. 66.—Elaps fulvius (The Harlequin Snake, or Coral Snake). (After L. Stejneger.) 106
Total length, 430 millimetres; tail 35.
Habitat: Peru.
(16) E. dissoleucus.—Scales in 15 rows; 200 ventrals; 19 subcaudals.
Coloration as in foregoing species.
Total length, 1,070 millimetres; tail 35.
Habitat: Venezuela.
(17) E. fulvius (Harlequin, or Coral Snake, fig. 66).—Scales in 15 rows; 180-237 ventrals; 30-59 subcaudals.
Colour: Body with black, red, and yellow annuli; tail with black and yellow annuli; snout black.
Total length, 990 millimetres; tail 85.
Habitat: Eastern North America, from Southern Virginia, the Ohio River, and the Missouri to the Rio Grande, Mexico. Central America.
(18) E. psyches.—Scales in 15 rows; 188-214 ventrals; 32-47 subcaudals.
Colour: Body with alternate black and brown annuli, and 48-52 narrow yellow rings; head black, blotched with yellow.
Total length, 495 millimetres; tail 80.
Habitat: Guianas.
(19) E. spixii.—Scales in 15 rows; 201-219 ventrals; 22-29 subcaudals.
Colour: red, with 20-38 black rings disposed in threes; a black occipital collar, followed by a wide red space.
Total length, 1,400 millimetres; tail 70.
Habitat: Venezuela and Northern Brazil.
(20) E. frontalis.—Scales in 15 rows; 197-230 ventrals; 15-26 subcaudals. Tail ending very obtusely.
Colour: Body with black annuli disposed in threes, with red and yellow interspaces; head black, spotted with yellow or red.
Total length, 1,350 millimetres; tail 70.
Habitat: Southern Brazil, Uruguay, Paraguay, Argentina.
(21) E. marcgravii.—Scales in 15 rows; 210-240 ventrals; 23-42 subcaudals. 107
Colour: Body with black annuli in 6-10 sets of three, separated by broad red interspaces, the middle annulus usually wider; snout yellow, the end usually black; back of head red.
Total length, 1,120 millimetres; tail 100.
Habitat: Tropical South America.
(22) E. lemniscatus.—Scales in 15 rows; 241-262 ventrals; 30-39 subcaudals.
Colour: Body with 11-14 sets of black annuli disposed in threes, separated by red interspaces; head yellow; end of snout and a band across the middle of the head black.
Total length, 1,000 millimetres; tail 80.
Habitat: Guianas, Brazil.
(23) E. filiformis.—Scales in 15 rows; 290-308 ventrals; 35-45 subcaudals.
Colour: Body with black annuli disposed in threes, with red interspaces; head yellow; end of snout black; a black band across the eyes.
Total length, 575 millimetres; tail 40.
Habitat: Amazons, Colombia.
(24) E. mipartitus.—Scales in 15 rows; 210-278 ventrals; 24-34 subcaudals.
Colour: black, with 40-68 narrow white annuli; head black to between the eyes, elsewhere yellow.
Total length, 610 millimetres; tail 50.
Habitat: Central America and Tropical South America.
(25) E. fraseri.—Scales in 15 rows; 303 ventrals; 25 subcaudals.
Colour: black, with 75 narrow whitish rings with broken outlines; head black in front, yellow behind.
Total length, 780 millimetres; tail 40.
Habitat: Ecuador.
(26) E. mentalis.—Scales in 15 rows; 255-268 ventrals; 30-31 subcaudals.
Colour: Body black, with 58-70 narrow white annuli, which become 108 wider on the belly; snout black, occiput yellow; tail annulate, black and orange.
Total length, 490 millimetres; tail 30.
Habitat.—Colombia and Ecuador.
(27) E. ancoralis.—Scales in 15 rows; 258 ventrals; 31 subcaudals.
Colour: Body with 16 sets of black annuli disposed in threes, the middle one of each set a little wider; head light in front, spotted with black; an anchor-shaped black mark on the occiput and nape.
Total length, 780 millimetres; tail 57.
Habitat: Ecuador.
(28) E. narduccii.—Scales in 15 rows; 240-315 ventrals; 15-33 subcaudals.
Colour: black, beneath with yellow or red cross-bands or transversely oval spots, sometimes extending as triangular blotches up the sides; head with a yellow cross-band.
Total length, 720 millimetres; tail 50.
Habitat: Eastern Ecuador, North-eastern Peru, Bolivia.
The species of the genus Elaps, which are remarkable for the brightness and beauty of their colours, are generally found in forests.
“The traveller,” says Neuwied, “who ventures into the great Brazilian forests, where the ground is covered with leafy plants, is astonished to see shining through the verdure the black and red rings of the beautiful Coral Snake. Uncertainty as to whether the creature is dangerous alone prevents him from seizing it. The body of the snake is not lithe enough to enable it to climb trees. Its food consists of small animals.”
Dr. Lacerda relates that the Austrian naturalist Wertheimer, when in the Brazilian settlement of Philadelphia, was bitten by a Coral Snake in the back of the hand. The usual symptoms of poisoning manifested themselves immediately, and the unfortunate man died twelve hours later. Nevertheless, the small size and 109 slenderness of the fangs, the narrowness of their canals, and the considerable distance between the fangs and the anterior opening of the mouth, must necessarily render the bites of these snakes less serious and of rarer occurrence.
The Solenoglypha are infinitely more formidable in the two divisions of the New World. They are represented by a large number of species, some of which are feared in consequence of their size and ferocity, even more than on account of the deadliness of their venom (fig. 67).

Fig. 67.—Head and Skull of Crotalus horridus.
The American Crotalinæ are divided into two groups:—
The first of these comprises snakes not provided with the caudal appendage, which is characteristic of the Rattle-Snakes. It consists of two genera:—
(a) Ancistrodon.
(b) Lachesis.
The second group includes only those snakes the tails of which are terminated by the scaly appendage known as the “rattle.” 110
These are likewise divided into two genera:—
(c) Sistrurus.
(d) Crotalus.
Usually with 9 shields on the head, or internasals and præfrontals broken up into scales. Body cylindrical: scales smooth or keeled, with apical pits. Tail moderate or short; subcaudals single or in 2 rows.

Fig. 68.—Ancistrodon piscivorus (Water Viper). (After Stejneger.)
(1) A. piscivorus (Water Viper, or Cotton-mouth; fig. 68).—Snout rounded, soft above. Scales on the body strongly keeled, in 25 (rarely 27) rows; 130-147 ventrals; 33-51 subcaudals, all single or the posterior divided.
Colour pale reddish-brown to dark brown above, with more or less distinct dark brown cross-bands, or with alternating C-shaped dark markings each enclosing a central spot. Belly dull yellow spotted with black, or almost black.
Total length, 1,170 millimetres; tail 200.
Habitat: Eastern North America, from North Carolina and Indiana to Florida and Texas. The Water Viper lives for the most part in the vicinity of 111 rivers, and feeds upon fish, but also devours small warm-blooded animals. The Creoles call it the Congo Snake. It is fairly common in the environs of New Orleans. In summer time it conceals itself in the branches of trees at the edge of the water. It frequents inundated rice-fields, and is a terror to the blacks. It attacks readily, and opens its jaws some seconds before it bites.
(2) A. bilineatus.—Snout obtusely pointed. Scales more or less strongly keeled, in 23 (rarely 25) rows; 135-141 ventrals; 52-64 subcaudals, anterior single, posterior divided.
Colour dull yellow or reddish-brown, with more or less distinct darker cross-bands, or alternating transverse blotches, with yellow edges; a vertical yellow line on the rostral shield, and a fine yellow line round the snout; belly brownish or blackish, with white spots.
Total length, 1,100 millimetres; tail 200.
Habitat: Mexico, Guatemala, Honduras.
(3) A. contortrix (The Copperhead).—Snout rounded or truncate. Scales strongly keeled, in 23 (rarely 25) rows; 145-155 ventrals; 31-52 subcaudals, anterior single, posterior divided.
Colour dull yellow or reddish-brown, with dark brown or brick-red cross-bars; these bars are sometimes interrupted on the vertebral line, and form alternating triangles; belly yellow or reddish, more or less spotted with grey or brown.
Total length, 990 millimetres; tail 110.
Habitat: North America, from Massachusetts and Kansas to Northern Florida and Texas.
This snake is often more dreaded than a Rattle-Snake.
In Lachesis the caudal rattle is represented by a series of 10 or 12 rows of spiny scales, which are slightly hooked at the tips. The head is covered with small shields or smooth or keeled scales, with or without apical pits. The maxillary is much reduced; the transverse or pterygoid bone, on the contrary, is greatly developed. 112
The name is derived from one of the Parcæ, daughters of Night, who placed the thread on the spindle, and upon whom depended the fate of men.
In addition to the 19 Asiatic species, of which we have already given descriptions, the genus Lachesis includes 21 American species.
(1) L. mutus (Bushmaster, or Surucucu).—Two or three scales separating the internasals in front; 10 to 15 scales on a line between the supraoculars; 9 or 10 supralabials. Scales tubercularly keeled, feebly imbricate, in 35 or 37 rows; 200-230 ventrals; 32-50 subcaudals.

Fig. 69.—Lachesis lanceolatus (Fer-de-lance of Martinique). (After Stejneger.)
Colour yellow or pinkish on the back, with a series of large rhomboidal brown or black spots enclosing smaller light spots; a black streak from the eye to the angle of the mouth.
Total length, 1,995 millimetres; tail 170.
Habitat: Central and Tropical South America.
(2) L. lanceolatus (known as the Fer-de-lance in Martinique, and Jararacussu in Brazil; fig. 69).—Snout obtuse, slightly turned up; upper head-scales small, imbricate, more or less strongly keeled, in 5-10 longitudinal series between the supraoculars, which are large; 7 or 8 supralabials. Scales in 23-33 rows, 113 sharply keeled; 180-240 ventrals; 46-70 subcaudals, all or the greater part in two rows.
Coloration very variable, grey, brown, yellow, olive, or reddish; uniform or with more or less distinct dark spots, or cross-bands, or with dark triangles on the sides enclosing pale rhombs; a dark streak from the eye to the angle of the mouth; belly yellowish, uniform, or spotted with brown.
Total length, 1,600 millimetres; tail 190.
Habitat: Tropical America; Mexico, Martinique, St. Lucia, Bequia Island near St. Vincent, Venezuela, Guianas, Rio de Janeiro.
This snake does not exist in Guadeloupe. “In the islands in which it is found,” says Dr. Rufz de Lavison, “its presence makes itself felt even where man has built his dwelling and cultivates the soil. Because of it no one can carelessly lie down to rest in the shade of a tree; no one can walk in the woods or enter unconcernedly into the pleasures of the chase.” It is especially abundant in coffee and sugar plantations, and is met with from the sea-level up to the summits of the highest mountains in Martinique and St. Lucia. It not infrequently makes its way into human habitations, and is not uncommon in gardens, even entering those of the town of Fort-de-France.
It does not seek its prey by day, but remains on the alert, always ready to bite. With open mouth, and fangs projecting forwards, it strikes with the rapidity of lightning. It swims in the rivers and moves over the ground with great speed. Oviposition takes place in July, and the young are hatched forthwith, the usual number being from about 50-60.
It feeds upon lizards and rats, but also destroys a certain number of fowls and turkeys. All the large animals are afraid of it. Its bite is extremely dangerous, and causes about a hundred deaths in Martinique every year.
In striking at its prey or at a man, the Fer-de-lance throws back its head and opens its jaws widely, with the fangs directed forwards. It drives in its poison-teeth as with the blow of a 114 hammer, and quickly draws back again. When very excited, it resumes its position and strikes afresh. It never becomes tame, but is capable of living a fairly long time in captivity. I have kept a number of specimens of it for more than two years, caged in my laboratory.
(3) L. atrox (Labaria).—Closely resembling L. lanceolatus, but bulkier; the enormous head is armed with powerful fangs, which are often more than a centimetre in length; 7 supralabial shields; scales in 25-29 rows, strongly keeled; 161-216 ventrals; 47-73 pairs of subcaudals.
Colour brown, with dark cross-bands or triangular blotches, with the apices adjacent on the vertebral line; a dark streak from the eye to the angle of the mouth; belly yellowish-white, speckled with brown, or brown spotted with yellowish-white.
Total length, 1,110 millimetres; tail 180.
Habitat: from Central America to Peru and Northern Brazil.
(4) L. pulcher.—Two postocular shields and a subocular, separated from the labials by one series of scales; 7 supralabials; scales in 21 or 23 rows, strongly and tubercularly keeled; 156-172 ventrals; 58-64 pairs of subcaudals.
Colour olive-grey, with brown, light-edged cross-bands, which are continuous or broken on the vertebral line; a light streak from the eye to the angle of the mouth; belly covered with minute confluent brown markings, with darker and lighter spots on the sides.
Total length, 685 millimetres; tail 115.
Habitat: Andes of Ecuador.
(5) L. microphthalmus.—Snout short, rounded; eyes very small; 7 supralabials, of which the third and the sixth or seventh are the largest; scales in 23 rows, dorsals tubercularly keeled; 159-161 ventrals; 52-55 subcaudal pairs.
Colour yellowish-brown or pale olive on the back, with dark brown triangles on the sides; posteriorly, the united triangles form cross-bands; a yellowish band from the eye to the side of the neck; belly dark brown with yellowish spots. 115
Total length, 630 millimetres; tail 100.
Habitat: Peru, Ecuador.
(6) L. pictus.—Snout obliquely truncate; two series of scales between the eye and the labials; scales in 21-23 rows, strongly keeled; 157-172 ventrals; 40-74 pairs of subcaudals.
Colour pale brown, with a dorsal series of large black-edged brown spots, which may form a vertebral zigzag band; a black streak behind the eye, and a vertical bar below the eye; belly yellowish, spotted with brown.
Total length, 310 millimetres; tail 43.
Habitat: Peru.
(7) L. alternatus.—Head narrow, elongate; 8-9 supralabials; scales in 29-35 rows, very strongly keeled; 167-181 ventrals; 34-51 pairs of subcaudals.
Colour brown, very elegantly marked with opposite or alternating pairs of large C-shaped markings, which are dark brown, edged with black and yellow, and separated by narrow interspaces of the ground colour; head dark·brown above, with a ⅄̅-shaped light marking, the transverse bar between the eyes; belly whitish, spotted with brown or black.
Total length, 1,190 millimetres; tail 110.
Habitat: Southern Brazil, Paraguay, Uruguay, Argentina.
(8) L. neuwiedii (known as the Urutù in Brazil; fig. 70).—Snout obtusely pointed; supraocular large, separated from its fellow by 6-9 longitudinal series of scales; 8 or 9 supralabials; scales very strongly keeled, in 21-27 rows; 168-182 ventrals; 41-53 subcaudals.
Colour yellowish or pale brown, with dark brown black-edged spots; the spots on the back form a single series or a double alternating series; a dark spot on the snout; a pair of dark bands from the vertex to the nape, and another from the eye to the angle of the mouth; belly yellowish, more or less powdered with brown.
Total length, 770 millimetres; tail 120.
Habitat: Brazil, Paraguay, Argentina. 116

Fig. 70.—Lachesis neuwiedii (known as the Urutù in Brazil). (After Lacerda.) 117
(9) L. ammodytoides.—Snout turned up, in the shape of a wart; two series of scales between the eye and the labials; scales in 23 or 25 rows, very strongly keeled; 149-160 ventrals; 30-38 pairs of subcaudals.
Colour pale brown, with large brown black-edged spots or cross-bands, which may alternate and form a zigzag band; a dark streak behind the eye; belly yellowish, spotted with brown.
Total length, 460 millimetres; tail 55.
Habitat: North-eastern Patagonia and Argentina.
(10) L. xanthogrammus.—Head elongate, snout short; scales in 27 rows, feebly keeled; 196 ventrals; 54 subcaudals.
Colour very dark olive, with a yellow zigzag line on each side from the head to the base of the tail; the angular parts enclose rhombic spaces and lateral triangles; top of head black, with a pair of undulating yellow bands from the nape to the vertex; a bright golden band round the snout; labials bright yellow; ventral shields black, paler in the middle, with yellow triangular spots at their extremities.
Total length, 1,530 millimetres; tail 190.
Habitat: Eastern Ecuador, Andes of Colombia.
(11) L. castelnaudi.—Head narrow and elongate; scales on the head smooth or feebly keeled, small; body-scales strongly keeled, in 25 or 27 rows; 230-253 ventrals; 72-83 subcaudals, all or majority in a single row.
Colour greyish or brown, with dark or light-edged spots or cross-bands; head with dark spots, one of which occupies the middle of the snout; a dark band from the eye to the angle of the mouth; belly brown or blackish, spotted with yellow.
Total length, 1,220 millimetres; tail 180.
Habitat: Brazil, Ecuador, Eastern Peru.
(12) L. nummifer.—Snout broad, rounded; 10 or 11 supralabials; scales in 23-27 rows, strongly keeled; 121-134 ventrals; 26-36 subcaudals, all or the majority single.
Colour pale brown, with a dorsal series of rhomboidal spots, 118 which may form a zigzag band; belly whitish, uniform or spotted with dark brown.
Total length, 800 millimetres; tail 90.
Habitat: Mexico and Central America.
(13) L. godmani.—Snout broad, rounded; 9 or 10 supralabials; scales in 21 rows, strongly keeled; 135-142 ventrals; 22-34 subcaudals in a single row.
Colour brown, with or without a dorsal series of large darker spots; belly yellowish, more or less spotted with grey or blackish.
Total length, 610 millimetres; tail 60.
Habitat: Guatemala.
(14) L. lansbergii.—Snout pointed, turned up at the end, as in Vipera aspis; scales in 25-27 rows, strongly keeled; 152-159 ventrals; 29-35 subcaudals in a single row.
Colour yellowish-brown, pale brown, or grey, with a dorsal series of large rhomboidal or squarish spots, usually divided by a narrow yellow or orange vertebral line; cheeks blackish; belly powdered with brown, with or without whitish spots.
Total length, 575 millimetres; tail 70.
Habitat: from Southern Mexico to Colombia, Venezuela, and Brazil.
(15) L. brachystoma.—Similar to the foregoing species; scales in 23 (rarely 25) rows; 132-150 ventrals; 27-38 subcaudals.
Total length, 500 millimetres; tail 50.
Habitat: Southern Mexico and Central America.
(16) L. bilineatus.—Snout rounded; 7 or 8 supralabials; scales in 27-35 rows, strongly keeled; 198-218 ventrals; 59-71 subcaudals, all or majority in two rows. Tail prehensile.
Colour green, uniform or spotted with black; a lateral yellow streak; belly white; end of tail reddish.
Total length, 840 millimetres; tail 125.
Habitat: Brazil, Bolivia, Peru, Ecuador.
(17) L. undulatus.—Snout short, rounded; 11 supralabials; scales in 21 rows, the dorsals strongly or very strongly keeled; 149-171 ventrals; 41-49 pairs of subcaudals. Tail prehensile. 119
Colour olive or brown, sometimes speckled with black, with a dorsal series of rhomboidal dark spots or an undulous or zigzag band; belly yellowish or brownish, powdered with blackish.
Total length, 570 millimetres; tail 75.
Habitat: Mexico.
(18) L. lateralis.—Snout rounded; 9 supralabials; scales in 21 or 23 rows, rather strongly keeled; 171 ventrals; 59 subcaudals, in a single row. Tail prehensile.
Colour green, with a yellow line on each side of the body.
Total length, 485 millimetres; tail 75.
Habitat: Costa Rica.
(19) L. bicolor.—Very similar to the foregoing; scales in 21 rows; 164-167 ventrals; 62-67 subcaudals, in a single row.
Colour uniform green, yellowish on the belly.
Total length, 375 millimetres; tail 60.
Habitat: Guatemala.
(20) L. schlegelii.—Snout rounded; 8 or 9 supralabials; scales in 19-25 rows, more or less strongly keeled; 138-162 ventrals; 47-62 subcaudals, in a single row. Tail prehensile.
Coloration very variable, green or olive, spotted with black, or with pinkish, reddish, or purplish black-edged spots or cross-bars; belly yellow, spotted with green, or variegated; end of tail generally red.
Total length, 600 millimetres; tail 115.
Habitat: Central America, Colombia, Ecuador.
(21) L. nigroviridis.—Snout short, rounded; 9-11 supralabials; 19 rows of scales, rather feebly keeled; 134-146 ventrals; 49-54 subcaudals, in a single row. Tail prehensile.
Colour green or olive, spotted with black; head with a black streak on each side; belly yellowish.
Total length, 535 millimetres; tail 90.
Habitat: Costa Rica.
(22) L. aurifer.—Snout short and broad; 9 or 10 supralabials; scales in 19 rows, rather strongly keeled; 154-158 ventrals; 53-61 subcaudals, single. Tail prehensile. 120
Colour green, spotted with yellow; a black streak on the temple; belly greenish-yellow.
Total length, 825 millimetres; tail 145.
Habitat: Guatemala.
Head very distinct from neck, covered above with nine large symmetrical shields; eyes rather small, with vertical pupils. Body cylindrical; scales keeled, with apical pits; tail short, ending in a segmented horny apparatus (rattle), producing a special sound; subcaudals all or the majority in a single row.
(1) S. miliarius (Ground Rattle-Snake).—9-11 supralabials; scales in 21 or 23 rows, strongly keeled; 127-139 ventrals; 27-36 subcaudals; rattle short, consisting at the most of 10 segments.
Colour greyish, yellowish, or brown, the vertebral line often orange; two undulating dark stripes from between the eyes to the occiput, the enclosed space usually orange; belly whitish, spotted with dark brown or black.
Total length, 520 millimetres; tail 70.
Habitat: South-eastern North America, from North Carolina to Texas.
(2) S. catenatus (Prairie Rattle-Snake, or Massasanga, fig. 71).—Two or three series of scales between the eye and the labials; 11-14 supralabials; scales in 23 or 25 rows; 136-153 ventrals; 20-31 subcaudals.
Colour the same as in S. miliarius: a dark spot on the parietal shields.
Total length, 680 millimetres; tail 80.
Habitat: Great Lakes district; United States east of the Rocky Mountains and west of the Mississippi; Northern Mexico.
(3) S. ravus.—11 or 12 supralabials; scales in 21 or 23 rows; 147 ventrals; 26 subcaudals.
Colour yellowish-brown, with a dorsal series of dark brown 121 122 spots, longer than broad, and a series of transverse dark bars on each side; belly yellowish, spotted with blackish-brown.

Fig. 71.—Sistrurus catenatus (Prairie Rattle-Snake, or Massasanga). (After Holbrook and Stejneger.)
Total length, 200 millimetres; tail 22.
Habitat: Vera Cruz, Mexico.
These snakes differ from all others in that the end of the tail bears a series of large conical scales, forming rattles, each fitting into the next and movable in such a manner that when the reptile causes them to move they produce a strident sound (fig. 72).

Fig. 72.—A, Horny appendage (rattle) of Crotalus horridus (three-quarters natural size. After Garman). B, longitudinal section of the same. C, separated segments of the appendage: a, terminal point; h, basal segment (after Czermak).
The length of these animals often exceeds 2,000 millimetres. The head is flat, very large and expanded posteriorly, and terminated in front by a short, truncate snout; it is covered above with scales or small shields.
Rattle-Snakes are armed with enormous fangs enclosing a complete canal, which extends throughout almost their entire length. The poison-glands are of the size of a large almond.
The number of segments in the rattle is variable, but rarely exceeds 18 or 20. At the time of the shedding of the skin these 123 124 segments fall off and are at once replaced. Contrary to the belief which was long entertained, their number bears no relation to the age of the snake.

Fig. 73.—Crotalus terrificus (Dog-faced Rattle-Snake, Cascavella in Brazil). (After Stejneger.)
Rattle-Snakes are met with especially in stony and arid localities, or among brushwood near water. They hardly ever bite except when surprised or attacked.
(1) C. terrificus (Dog-faced Rattle-Snake, Cascavella in Brazil; fig. 73).—Snout very short; three or four series of scales between the eye and the supralabial shields; body-scales in 23-31 rows, dorsals very strongly keeled; 160-199 ventrals; 18-30 subcaudals.
Colour brown, with a series of darker, light-edged rhombs, often lighter in the centre; a dark streak from the eye to the angle of the mouth; belly yellowish-white, uniform or spotted with brown; tail generally brown or blackish.
Total length, 1,320 millimetres; tail 130.
Habitat: Arizona, New Mexico, and Texas to Southern Brazil, and Northern Argentina.
(2) C. scutulatus (Texas Rattle-Snake; fig. 74).—13-16 supralabials; scales in 25 or 27 rows; dorsals striated and strongly keeled; 167-170 ventrals; 18-20 subcaudals.
Colour yellowish or greyish-brown, with a series of large dark brown light-edged rhomboidal spots; an oblique dark streak below the eye; belly uniform yellowish-white.
Total length, 760 millimetres; tail 65.
Habitat: Arizona, New Mexico, Texas, North Mexico.
(3) C. confluentus (Pacific or Mottled Rattle-Snake; fig. 75).—Upper head-scales small, striated; 13-18 supralabials; body scales in 25-29 rows, striated and strongly keeled; 168-197 ventrals; 17-34 subcaudals.
Colour yellowish, greyish, or pale brown, with a dorsal series of large brown or red spots, usually rhomboidal or transversely elliptic in shape; a light streak or triangular marking across the supraocular shields; belly yellowish, uniform or spotted with brown. 125
Total length, 1,520 millimetres; tail 140.
Habitat: Western North America, from British Columbia to South California, eastwards to Assiniboia, Dakota, Nebraska, Kansas, and Western and Southern Texas; Northern Mexico.
According to Holbrook, this reptile feeds upon young rabbits, squirrels, rats and other small mammals. It lays its eggs in August, and the young are hatched at once, and are capable of seeking their own food.
In captivity as well as under natural conditions the Mottled Rattle-Snake is an excessively irritable species. “The noise of the wind,” says Brehm, “or even the distant view of a man or animal, are sufficient to irritate it. It then coils itself up in a spiral, and places its head and tail in the centre of the disc thus formed, in a state of absolute immobility. After a short interval the creature raises its head to a height of about 8 to 12 inches above the ground, curves its neck in the shape of an S, and elevates its tail into a vertical position and shakes it vigorously, whereupon the strident noise caused by the rattle is heard. So rapid are the movements communicated by the Crotalus to its tail that they can scarcely be distinguished. So long as the Crotalus believes itself menaced it remains in the position that we have just described, and continues to sound its rattle. If one withdraws from the irritated snake, the sound gradually lessens and ceases, to begin again more vigorously when the reptile is once more approached.”
The bite of these snakes is exceedingly dangerous. Pigs wage an inveterate war against them and devour them.
At the Pasteur Institute at Lille, I have kept several of these reptiles in captivity for eighteen months and longer. They invariably refused their food, and I always had to resort to artificial feeding. They are easily capable of withstanding a prolonged fast.
(4) C. durissus (Common Rattle-Snake).—7 or 8 longitudinal 126 127 series of scales between the supraoculars, 3 or 4 series of scales between the eye and the labials; 13-16 supralabials; scales in 25-29 rows, dorsals strongly keeled; ventrals 169-181; 24-32 subcaudals.

Fig. 74.—Crotalus scutulatus (Texas Rattle-Snake). (After Baird and Stejneger.)
Colour pale greyish or brownish, with a dorsal series of large blackish rhomboidal spots; a yellowish cross-line between the eyes; snout blackish; end of tail usually black; belly yellowish, more or less spotted with brown or black.
Total length may be as much as 8 feet (2,400 millimetres).
Habitat: South-eastern United States, from North Carolina to Florida and the mouths of the Mississippi.
(5) C. horridus.—Supraoculars considerably narrower than the space between them, which is covered by 3-8 longitudinal series of small scales; 12-16 supralabials; body scales in 23-29 rows, dorsals very strongly keeled; 165-178 ventrals; 19-29 subcaudals.
Colour greyish-brown, usually with a rusty vertebral stripe, and V- or M-shaped blackish cross-bands; head uniform above, with a dark band from the eye to the angle of the mouth; usually a pair of roundish or triangular dark spots on the nape; belly yellowish, uniform or spotted with blackish; end of tail blackish.
Total length, 1,340 millimetres; tail 135.
Habitat: United States, from Massachusetts and Iowa to Northern Florida and Texas.
(6) C. tigris.—13-15 supralabials; scales in 23 or 25 rows, dorsals strongly keeled; 166-181 ventrals; 26-46 subcaudals.
Colour yellowish or pale brown, with a dorsal series of brown spots and cross-bands posteriorly; sides with smaller dark spots; belly yellowish, spotted with brown.
Total length, 380 millimetres; tail 50.
Habitat: Southern California, Lower California, Nevada, Colorado, Arizona, and Northern Mexico.
(7) C. mitchelli.—14-16 supralabials; scales in 25 rows, striated, dorsals strongly keeled; 178-198 ventrals; 24-26 subcaudals. 128

Fig. 75.—Crotalus confluentus (Pacific or Mottled Rattle-Snake). (After Baird and Stejneger.) 129
Colour greyish-yellow to salmon-red, finely punctulated with brown, with a dorsal series of transverse darker spots; belly yellowish.
Total length, 1,020 millimetres; tail 90.
Habitat: Desert regions of Southern California, Lower California, and Arizona.
(8) C. triseriatus.—9-13 supralabials; scales in 21-25 rows, dorsals strongly keeled; 142-184 ventrals; 22-30 subcaudals.
Colour olive or brown, with a vertebral series of rather small dark brown spots edged with black and white; belly yellowish, spotted with dark brown, or dark grey-brown powdered with whitish.
Total length, 530 millimetres; tail 55.
Habitat: Mexico.
(9) C. polystictus.—Closely allied to the foregoing, but 4 internasals, 14 or 15 supralabials, and scales in 27-30 rows; 123-151 ventrals; 18-23 subcaudals.
Colour yellowish-brown, beautifully marked with 6 or 7 longitudinal series of alternating, elongate, dark brown, black- and white-edged spots, separated by narrow interspaces of the ground-colour; a pair of diverging dark bands on the top of the head, separated by a narrow pinkish-white streak; belly pinkish or yellowish, spotted with dark brown.
Total length, 600 millimetres; tail 60.
Habitat: Tableland of Mexico.
(10) C. lepidus.—12 supralabials; scales in 21 or 23 rows, dorsals strongly keeled; 153-169 ventrals; 24-31 subcaudals.
Colour brown or greenish-grey, with dark brown or jet-black light-edged cross-bands, narrowing on the sides; two dark spots, or a V- or heart-shaped black marking on the nape; belly dirty white, spotted with brown.
Total length, 350 millimetres; tail 60.
Habitat: Western Texas, New Mexico, Arizona, North Mexico.
(11) C. cerastes (Horned Rattle-Snake; fig. 76).—Supraocular 130 131 transformed into a raised horn-like process; 11-13 supralabials; scales in 21 or 23 rows; dorsals feebly keeled, each scale along the middle of the back with a central tubercular swelling; 146 ventrals; 17 subcaudals.

Fig. 76.—Crotalus cerastes (Horned Rattle-Snake.) (After Baird and Stejneger.)
Colour yellowish, with a dorsal series of rather indistinct brown blotches; a narrow brown streak from the eye to the angle of the mouth.
Total length, 250 millimetres; tail 20.
Habitat: Desert regions of Southern California, Nevada, Arizona, and Utah.
The Sea-Snakes, which are found in great numbers on the shores of the Indian Ocean, are common throughout the whole of the tropical zone of the China Sea and the Pacific. They are met with from the Persian Gulf to the west coast of Equatorial America, but are entirely absent from the east coast of the same continent and the west and east coasts of Africa.
They often travel in companies. All are poisonous, and very savage. They never come to land, and move with difficulty if taken out of the water, although they are excellent swimmers. It is impossible to keep them in captivity in aquariums, and they die in two or three days. Their food consists of fishes and crustacea. Their tail is prehensile, and they make use of it as an anchor to attach themselves to coral reefs when they wish to rest. They generally float on the surface of the waves, but can dive to great depths, thanks to the extreme dilatability of their lungs, which enables them to store up large reserves of air. They are viviparous.
In these snakes, the head, which is always very small, is scarcely distinct from the body. It is often covered with nine large shields. The body is laterally compressed, and the tail, which serves as a fin, is similarly flattened. The nostrils open 132 on the upper surface of the snout, between the nasal shields. The eyes are always very small.
The number of species at present known is considerable; they are divided into ten genera. We shall confine ourselves here to mentioning the principal diagnostic characters of these genera, and to describing the most common species.

Fig. 77.—Skull of Hydrus platurus. (After G. A. Boulenger, op. cit.)
Maxillaries longer than the ectopterygoids, not extending forwards as far as the palatines; poison-fangs grooved, rather short, followed, after an interspace, by 7 or 8 solid, backwardly 133 curved teeth. Snout long, bearing the nostrils on its upper surface; head-shields large, nasals in contact with each other. Body rather short; scales hexagonal or squarish, juxtaposed; no distinct ventral scales.

Fig. 78.—Hydrus platurus (syn.
Pelamis bicolor).
(After Krefft.)
The principal species of this genus is H. platurus (syn. Pelamis bicolor, fig. 78).
Coloration black or brown and yellow, with very variable markings.
Total length, 700 millimetres; tail 80.
Habitat: Indian Ocean, Tropical and Sub-tropical Pacific.
Poison-fangs followed by 5 small teeth. Snout short; nostrils superior, horizontal, between two nasal shields and an internasal; frontal and parietal shields large; præocular present. Body rather elongate; scales hexagonal, juxtaposed; no distinct ventral scales.
T. anomalus.—Body with dark annuli, wider on the back.
Total length, 810 millimetres; tail 84.
Habitat: Java.
Maxillaries longer than the ectopterygoids; frontal and parietal shields broken up into scales. Body rather elongate; scales subimbricate; no distinct ventrals.
A. peronii.—Greyish or pale olive, with dark cross-bands; belly whitish.
Total length, 890 millimetres; tail 115.
Habitat: Western Tropical Pacific. 134
Snout short; 6 teeth behind the poison-fangs; nostril in a single nasal shield; head-shields large. Body feebly compressed; scales imbricate; ventral scales small, but well developed.
H. darwiniensis.—Body with yellowish-white and blackish annuli, the black rings narrower on the belly; head dark olive spotted with black.
Total length, 435 millimetres; tail 43.
Habitat: North Australia.

Fig. 79.—Hydrophis coronatus. (After Sir Joseph Fayrer.)
Poison-fangs large, followed by a series of 7-18 solid teeth. Head small; nostrils on the upper surface of the snout, pierced in a single nasal shield; head-shields large; præocular present. Body long, often very slender anteriorly; scales on the anterior part of the body imbricate, rectangular, keeled or tubercular; ventrals more or less distinct, very small.
A considerable number of (at least 22) species of Hydrophis are known. Those most frequently met with are the following:— 135
H. spiralis.—Olive above, yellowish beneath, with black rings; head black above, with a horse-shoe-shaped yellow mark, the convexity of which rests on the præfrontal shields; end of tail black.
Total length, 400 millimetres. Grows to 1,800 millimetres.
Habitat: Coasts of India, and the Malay Archipelago.
H. cærulescens.—Grey above, with black cross-bands, which form complete rings, or are interrupted on the belly; head uniform black.
Total length, 665 millimetres; tail 75.
Habitat: Bombay Coast, Bay of Bengal, Straits of Malacca.
H. nigrocinctus.—Pale olive on the back, yellowish on the belly, with black annuli, which are broader on the back.
Total length, 1,000 millimetres; tail 100.
Habitat: Bay of Bengal and Straits of Malacca.

Fig. 80.—Hydrophis elegans. (After Krefft.)
H. elegans (fig. 80).—Yellowish-white, back with transverse rhomboidal black spots, separated by a series of small black spots; belly with black spots or cross-bars; head blackish, with a more or less distinct light crescentic marking across the snout, from above the eyes.
Total length, 710 millimetres; tail 60.
Habitat: North coast of Australia.
H. gracilis.—Bluish-black or greyish, olive above in the adult, with more or less distinct lighter cross-bands anteriorly. Young sometimes with rhombic black cross-bands extending to the belly, or sub-interrupted on the sides.
Total length, 1,020 millimetres; tail 90.
Habitat: Coasts of Persia, India, and Ceylon; Malay Archipelago.
H. cantoris.—Body dark olive or blackish anteriorly, with yellowish cross-bands above; posterior part of body olive above, yellowish on the sides; tail with olive vertical bars; a blackish streak along the belly. 136
Total length, 1,100 millimetres; tail 90.
Habitat: Bay of Bengal.
H. fasciatus.—Head and neck black, the latter with yellowish cross-bands; body pale, with black annuli, which are broader on the back.
Total length, 1,000 millimetres; tail 85.
Habitat: From the coasts of India to China and New Guinea.
H. obscurus (syn. H. stricticollis).—Olive or dark green above, with yellowish cross-bars, which form complete rings round the slender anterior part of the body; a yellow spot on the snout, and a yellow streak on each side of the upper surface of the head.
Total length, 970 millimetres; tail 105.
Habitat: Bay of Bengal, Malay Archipelago.
H. leptodira.--Black, with yellow cross-bars on the neck, and complete annuli on the body, the bars and annuli numbering 77.
Total length, 525 millimetres; tail 40.
Habitat: Mouths of the Ganges.
Poison-fangs large, followed by 4-10 grooved teeth. Head larger than in Hydrophis; body more or less elongate; scales on the anterior part of the body imbricate; ventrals more or less distinct, and always very small.
The species of this genus, 18 in number according to the British Museum Catalogue, are found in the Indian and Pacific Oceans, from the Persian Gulf to Japan and New Caledonia.
The most important are:—
D. ornata.—Uniform blackish-olive on the back, whitish on the belly.
Total length, 1,200 millimetres; tail 130. Habitat: From the Mouth of Persian Gulf, and the coasts of India and Ceylon to New Guinea, and North Australia. 137
D. subcincta.—Trunk with 41 broad dark cross-bands, about as broad as the interspaces, not extending downwards to the middle of the side; a series of small roundish, blackish spots along the lower part of the sides.
Total length, 1,070 millimetres; tail 100.
Habitat: Indian Ocean.

Fig. 81.—Skull of Distira.
(After G. A. Boulenger, op. cit.)
D. cyanocincta.—Greenish-olive above, with dark olive or black cross-bars or annuli, broader on the back, and sometimes joined by a black band along the belly, or yellowish, with a black vertebral stripe and a few bars on the neck.
Total length, 1,500 millimetres; tail 140.
Habitat: From the Persian Gulf and the coasts of India to China, Japan, and Papuasia.
D. jerdonii.—Olive above, yellowish on the belly, with black cross-bands forming complete rings in young and half-grown 138 specimens; a black spot sometimes present between each pair of annuli.
Total length, 910 millimetres; tail 100.
Habitat: Bay of Bengal, Straits of Malacca, Borneo.
Two large poison-fangs, and 2-4 small feebly grooved teeth. Body short and stout; scales hexagonal or squarish, juxtaposed, disappearing almost completely on the belly.
E. curtus.—Above with dark transverse bands, broadest in the middle; end of tail black.
Total length, 750 millimetres; tail 75.
Habitat Coasts of India and Ceylon.

Fig. 82.—Enhydrina valakadien (syn. E. bengalensis). 139
Two large poison-fangs, followed by 4 solid non-grooved teeth. Body moderately elongate; scales imbricate; ventrals distinct but very small.
E. valakadien (syn. E. bengalensis; fig. 82).—Colour olive or grey, with black transverse bands, usually less distinct in the adult; sides and belly whitish.

Fig. 83.—Skull of Platurus olubrinus.
(After G. A. Boulenger, op. cit.)
Total length, 1,300 millimetres; tail 190.
Habitat: From the Persian Gulf along the coasts of India and Burma, to the Malay Archipelago and Papuasia. 140
Maxillaries a little longer than the ectopterygoids; poison-fangs moderate, followed, after a short interval, by 8-10 grooved teeth; anterior mandibular teeth feebly grooved. Snout short; head shields large, or broken up into scales. Body moderate; scales imbricate; ventrals large, keeled in the middle.
A. australis.—Brown, or cream-colour, with brown spots forming more or less distinct cross-bars.
Total length, 930 millimetres; tail 110.
Habitat: Coasts of New Guinea and Australia.
Other species of Aipysurus (A. eydouxii, annulatus, and lævis) are found on the coasts of Singapore, Java, Celebes, and the Philippine and Loyalty Islands.

Fig. 84.—Platurus laticaudatus (syn. P. fischeri).
(After Sir Joseph Fayrer.)
Two large poison-fangs, and only one or two small solid teeth near the posterior extremity of the maxillary. Head shields large; 141 nostrils lateral, the nasal shields separated by the internasals. Body greatly elongate; scales smooth and imbricate; ventrals and subcaudals large.
Four species, distributed in the eastern parts of the Indian Ocean and in the Western Pacific.
P. laticaudatus (syn. P. fischeri; fig. 84).—Olive above, yellowish on the belly, with 29-48 black annuli.
Total length: 970 millimetres; tail 90.
Habitat: From the Bay of Bengal to the China Sea and the Western South Pacific Ocean.
P. colubrinus (fig. 83).—Olive above, yellowish on the belly, with 28-54 black annuli, some or all of which may be interrupted below.
Total length, 1,270 millimetres; tail 125.
Habitat: From the Bay of Bengal to the China Sea and the Western South Pacific Ocean.
P. muelleri.—62 black annuli, some of which are interrupted on the belly.
Habitat: Only found in the South Pacific Ocean (subtropical zone), as far as the New Hebrides and the shores of Tasmania.
P. schistorhynchus.—Coloration and size as in P. colubrinus: body with 25-45 annuli.
Habitat: Western Tropical Pacific. 142
| (1) EUROPE. | |||
|---|---|---|---|
| Families | Sub-Families | Genera | Geographical Area |
| Colubridæ | Dipsadomorphinæ | Cœlopeltis | Borders of the Mediterranean in France and Spain; Italy (only in Liguria). |
| Viperidæ | Viperinæ | Vipera | France, Italy, Switzerland, Austria-Hungary, Germany, Belgium, Sweden and Norway, Gt. Britain, Spain and Portugal, Bosnia and Herzegovina, Southern Russia, Turkey and Greece. |
| (2) ASIA. | |||
| Colubridæ | Hydrophiinæ |
Hydrus Thalassophis Acalyptophis Hydrelaps Hydrophis Distira Enhydris Enhydrina Aipysurus Platurus |
Persian Gulf, Indian Ocean, Bay of Bengal, Straits of Malacca, China Sea, Philippines, and Malay Archipelago. |
| Elapinæ | |||
| Bungarus | India, Ceylon, Burma, Indo-China, Southern China, Dutch Indies, Borneo. | ||
| Naja | India, Ceylon, Burma, Indo-China, Dutch Indies, Philippines. | ||
| Hemibungarus | South-eastern Asia, India, Japan, Philippines. | ||
| Callophis | South-eastern Asia, India, Burma, Indo-China, Formosa, Southern China. | ||
| Doliophis | Indo-China, Malay Peninsula.143 | ||
| Viperidæ | Viperinæ | Vipera | Turkestan, Ural, Siberia, Caucasus, Persia, Armenia, Western China, India, Ceylon, Himalayas. |
| Pseudocerastes | Persia. | ||
| Cerastes | Arabia, Palestine. | ||
| Echis | Persia, Arabia, India, Baluchistan, Afghanistan. | ||
| Crotalinæ | Ancistrodon | Transcaspia, Turkestan, Himalayas, Southern China, Formosa, Japan, Ceylon, Java. | |
| Lachesis | South-eastern Asia, India, Southern China, Indo-China, Formosa, Sumatra. | ||
| (3) AFRICA. | |||
| Colubridæ | Elapinæ | Boulengerina | Central Africa. |
| Elapechis | Central and South Africa. | ||
| Naja | Egypt, Central and West Africa, Morocco, Congo, Angola. | ||
| Sepedon | South Africa, Cape of Good Hope. | ||
| Aspidelaps | South and South-east Africa, Mozambique. | ||
| Walterinnesia | Egypt. | ||
| Homorelaps | South Africa, Cape of Good Hope. | ||
| Dendraspis | Central and South Africa, Angola, Great Lakes, Congo, Transvaal. 144 | ||
| Viperidæ | Viperinæ | Causus | West Africa, Gambia, Great Lakes, Congo, Angola, Transvaal. |
| Vipera | Morocco, Algeria, Tunis, Egypt, Mozambique. | ||
| Bitis | Zanzibar, Zambesia, Cape, Transvaal, Congo, the Gaboon, Benguella, Angola, Senegal, Nigeria. | ||
| Cerastes | North Africa, the Sahara. | ||
| Echis | North Africa, Lake Chad, Soudan, Egypt, Somaliland, Socotra. | ||
| Atheris | Tropical Africa, Dahomey, Lagos, the Cameroons, the Gaboon, Congo. | ||
| Atractaspis | Tropical and South Africa, Congo, Angola, Lake Chad, the Gaboon, Dahomey, Gold Coast, Zanzibar, Somaliland, Natal, and the Cape. 145 | ||
| (4) OCEANIA. | |||
| Colubridæ | Hydrophiinæ |
Hydrus Thalassophis Hydrelaps Hydrophis Distira Enhydris Enhydrina Aipysurus Platurus |
Equatorial and Sub-tropical Pacific Ocean, the Moluccas, Papuasia, New Guinea, Celebes, Timor, Australia, Tasmania, New Caledonia, New Hebrides. |
| Elapinæ | Ogmodon | Fiji Islands. | |
| Glyphodon | Northern Australia, New Guinea. | ||
| Pseudelaps | Australia, the Moluccas, Papuasia. | ||
| Diemenia | Australia, New Guinea. | ||
| Pseudechis | Australia, New Guinea. | ||
| Denisonia | Australia, Solomon Islands, Tasmania. | ||
| Micropechis | New Guinea, Solomon Islands. | ||
| Hoplocephalus | Australia. | ||
| Tropidechis | Australia. | ||
| Notechis | Australia, Tasmania. | ||
| Rhinhoplocephalus | Australia. | ||
| Brachyaspis | Australia. | ||
| Acanthophis | Moluccas, Papuasia, Northern Australia. | ||
| Elapognathus | Australia. | ||
| Rhynchelaps | Australia. | ||
| Furina | Australia. 146 | ||
| (5) AMERICA. | |||
| Colubridæ | Elapinæ | Elaps | Mexico, Central America, Bolivia, Ecuador, Peru, Colombia, Brazil. |
| Viperidæ | Crotalinæ | Ancistrodon | North America, Florida, Texas, Mexico, Guatemala. |
| Lachesis | Central and South America, Martinique, St. Lucia. | ||
| Sistrurus | North America east of the Rocky Mountains, Mexico. | ||
| Crotalus | Southern Canada, British Columbia, Central America, Guiana, Venezuela, Brazil, Uruguay, Northern Argentina. | ||
147
Non-poisonous as well as poisonous snakes possess parotid and upper labial glands capable of secreting venom. In the former the organs of inoculation are wanting, but we shall see later on that the toxic secretion of their glands is just as indispensable to them as to the snakes of the second category for the purpose of enabling them to digest their prey.
For the morphological, histological, and physiological demonstration of the existence of these glands in harmless reptiles we are indebted to Leydig (1873), whose discovery has since been confirmed and extended by the researches of Phisalix and Bertrand, Alcock, L. Rogers, and L. Lannoy.
The parotids of Grass Snakes are mixed glands of the sero-mucous type. The serous tubes are situate almost exclusively in the posterior portion of the gland. As we proceed towards the anterior portion, we find that these serous tubes are interspersed with others which are exclusively mucous or sero-mucous, and they become entangled with those of the upper labial gland, properly so-called. The substance of the gland is divided into several lobes by bands of connective tissue; the tubes are separated by septa of the same tissue, in extremely delicate layers (Lannoy).
In poisonous snakes these glands are much more developed, especially in their hinder portions, which sometimes assume 148 enormous dimensions. They may attain the size of a large almond (Crotalus, Naja), and they then occupy the spacious chamber already described (Chap. I., p. 10), which is situated behind the eye on each side of the skull.
Each gland is surrounded by a thick capsule of fibrous tissue, two prolongations of which, the one anterior, the other posterior, keep it in its place beneath the masseter muscle. A portion of the latter is inserted in the capsule itself, in such a way that when the snake closes its jaws to bite, the gland is forcibly compressed and the contained liquid is squeezed into its excretory duct.
Between the muscle and the envelope of the gland there is a serous pouch, which enables the one to slide over the other.
The excretory duct runs along the outer side of the upper jaw, and opens by a slit at the base of the poison-fang, with which it inosculates at right angles in a little muscular mass forming a sphincter.
In the normal position of repose, the poison-fang is always concealed by a gingival fold of mucous membrane, in the substance of which are buried a few fibres of the tendon of the internal pterygoid muscle. When the latter contracts, the tooth is almost completely exposed, and the efferent duct of the gland then assumes an oblique position, which allows of the direct discharge of the venom through the canal which runs along the greater portion of the length of the tooth.
When the poison-fangs are folded back in their sheath, the poisonous secretion can escape freely into the buccal cavity by the slit situated at the base of the fangs.
At the moment when the animal is about to bite, when it throws back its head and opens its jaws, directing its fangs forwards, the muscles that come into action (masseters, temporals, and pterygoids) compress the glands on each side, and cause the venom to be expelled in a sudden jet, as if by a sort of ejaculatory process. In the case of certain species the venom may be projected to a distance of more than a yard. 149
The quantity of venom secreted by the glands varies greatly, according to the length of time which has elapsed since the animal took its last meal, and in accordance with a number of other conditions not very easy to determine.
The Common Viper of Europe yields scarcely 10 centigrammes of poison, while an adult Indian Cobra may excrete more than 1 gramme.
Freshly collected venom is a syrupy liquid, citron-yellow or slightly opalescent white in colour.
When dried rapidly in vacuo or in a desiccator over calcium chloride, it concretes in cracked translucent lamellæ like albumin or gum arabic, and thus assumes a crystalloid aspect. In this condition it may be kept indefinitely, if protected from light, air, and moisture. It dissolves again in water just as readily as albumin or dried serums.
I regularly weighed the dry residue from eleven bites made on a watch-glass by two Naja haje, received at my laboratory from Egypt at the same time, and placed in the same case. Both snakes were approximately of equal length, 1,070 millimetres. Throughout the entire course of the experiment, which lasted one hundred and two days, neither of them took any food, but they drank water and frequently bathed.
The results that I obtained are shown in the table on next page.
It will be seen that in one hundred and two days, an adult Naja haje is capable of producing on an average 0·632 gramme of liquid venom, equal to a mean weight of 0·188 gramme of dry extract; and we may conclude that 1 gramme of liquid gives 0·336 gramme of dry venom.
In Australia it has been found by MacGarvie Smith, of Sydney, that Pseudechis porphyriacus yields at each bite a quantity of venom varying from 0·100 gramme to 0·160 gramme (equal to 0·024 gramme to 0·046 gramme of dry venom), and that a Hoplocephalus curtus (Tiger Snake) yields 0·065 gramme to 0·150 gramme of 150 liquid venom, with 0·017 gramme to 0·055 gramme of dry residue. In all the experiments of this physiologist, the proportion of dry residue varied from 9 to 38 per cent. of the liquid venom excreted by the reptile.
A Lachesis lanceolatus (Fer-de-lance) from Martinique, of medium size, when both of its glands were squeezed, furnished me with 0·320 gramme of liquid venom, and 0·127 gramme of dry extract.
| Number of bite |
Date | NAJA HAJE I. Weight of Venom |
NAJA HAJE II. Weight of Venom |
||
|---|---|---|---|---|---|
| Fresh | Dry | Fresh | Dry | ||
| Gramme | Gramme | Gramme | Gramme | ||
| 1 | April 20 | 0·119 | 0·031 | — | — |
| 2 | April 23 | — | — | 0·151 | 0·043 |
| 3 | May 14 | 0·124 | 0·035 | — | — |
| 4 | May 21 | — | — | 0·132 | 0·037 |
| 5 | May 28 | — | — | 0·091 | 0·019 |
| 6 | June 2 | 0·127 | 0·039 | — | — |
| 7 | June 19 | — | — | 0·121 | 0·043 |
| 8 | July 1 | — | — | 0·078 | 0·026 |
| 9 | July 2 | 0·122 | 0·048 | — | — |
| 10 | July 25 | — | — | 0·111 | 0·034 |
| 11 | July 26 | 0·079 | 0·021 | — | — |
| Totals ... | 0·581 | 0·174 | 0·684 | 0·202 | |
Two large Cerastes vipers, from Egypt, yielded me, one 0·123 gramme, the other 0·085 gramme of liquid venom, which, after desiccation, left respectively 0·027 gramme and 0·019 gramme of dry residue.
Under the same conditions, a magnificent Crotalus confluentus (Mottled Rattle-Snake), for which I was indebted to the kindness of Mr. Retlie, of New York, yielded, two months after reaching my laboratory, 0·370 gramme of liquid venom and 0·105 gramme of dry extract in a single bite. 151
The total quantity of liquid venom that I found contained in the two glands of the same reptile, when extirpated after death, and after the snake had been in the laboratory for five months, amounted to 1·136 gramme, which gave 0·480 gramme of dry extract.
We see, therefore, that the proportion of dry residue, including albumin, salts, the débris of leucocytes, and the toxic substance, oscillates between 20 and 38 per cent. Its strength varies with the length of time that has elapsed since the snake’s last bite or last meal.
From the histological standpoint, the process of the secretion of venom, in the cells of the glands, may be divided into two stages:—
(a) A stage of nuclear elaboration.
(b) A stage of cytoplasmic elaboration.
These two stages are superposed and successive.
In addition to the passive exchanges between the nucleus and the cytoplasm, the nuclear mass actively participates in the secretion. This participation is rendered evident:—
(1) By the difference of chromaticity in the granules of chromatin.
(2) By the emission of formed granules into the cytoplasm, granules which are spherical and of equal bulk, with the chromatic reactions of differentiated intranuclear chromatin.
(3) By the exosmosis of the dissolved nuclear substance, accessorily formed in an ergastoplasmic shape.
These formations constitute, on the one hand, the granules of venogen; on the other, the ergastoplasmic venogen. In the poison-cell of Vipera aspis, and in the serous cell of the parotid glands of Tropidonotus natrix (Grass Snake) the venogen is elaborated chiefly in granular form.
On entering the perinuclear cytoplasm, the granule of venogen and the ergastoplasmic venogen may either disappear immediately, 152 as happens in periods of cellular stimulation, or else continue to exist for some time within the cell, indicating a period of saturation by the elaborated material.
During cytoplasmic activity the granule of venogen and the ergastoplasmic venogen disappear.
Nuclear elaboration and cytoplasmic elaboration constitute two different cycles of secretion. The effect of the nuclear cycle is to furnish the cytoplasm with the elements necessary for the work of secretion properly so-called. Cytoplasmic elaboration is not confined to the basal protoplasm, but takes place throughout the entire cell: it is especially active in the perinuclear cytoplasm.
The granule of venogen is distinguished from the granule of elaborated venom by its affinity for Unna’s blue, safranin, and fuchsin. The granule of venom has an affinity for eosin; it is never excreted in granular form, but after intracellular dissolution.
Venogen is never met with in the lumen of the gland-tube.6
Venom can be extracted from the poison-glands of either freshly killed or living snakes.
In cases in which the venom of dead snakes has to be collected, the best method of extraction consists in fixing the head of the animal to a sheet of cork and carefully dissecting out the gland on each side. The reptile being placed on its back, the lower jaw is removed with a pair of scissors; two strong pins or two tacks are thrust through the skull, in the median line, in order to keep the head from moving. The poison-fangs are next drawn out of their sheaths, and, without injuring them, the two poison-ducts, which open at their bases, are isolated and tied with a thread in order to prevent the poison from running out.
The dissection of the glands is then very easy; they are lifted 153 out and placed in a saucer. The end of the duct is cut between the gland and the ligature, and with a pair of fenestrated or polypus forceps the whole of the glandular mass is gently squeezed from behind forwards, the liquid which flows out being received in a large watch-glass.
If pressed for time, a more simple method of operating is to hold the head of the snake in the left hand, with the mouth open and the lower jaw directed downwards. A watch-glass, capsule, or receptacle of some sort, such as a cup or plate, is then introduced by an assistant between the jaws, and, with the index finger and thumb of the right hand, the whole of the region occupied by the glands on each side of the upper jaw is forcibly compressed from behind forwards; the poison flows out by the fangs.
The extraction of the venom from living snakes is effected in the same manner. The animal being firmly held by the neck, as close as possible to the head, so that it cannot turn and bite; it can be made to eject the greater portion of the liquid contained in its two glands by compressing the latter with force from behind forwards, as one would squeeze out the juice from a quarter of an orange (fig. 85).
It is necessary to take care that the reptile cannot coil itself round furniture or other objects in the vicinity of the operator, for if this should happen there would be the greatest difficulty in making it let go, especially if dealing with a strong animal such as a Cobra, Rattle-Snake, or Fer-de-lance.
Snakes of the last-mentioned kind are especially difficult to manage. In order to avoid the risk of being bitten, it is always wise to begin by pinning down the head of the animal in a corner of its cage by means of a stick, and to seize it with a pair of long fenestrated tongs shaped like forceps. The operator then easily draws the reptile towards him and grasps it firmly by the neck with his left hand, always as close to the head as possible, at the same time raising the body quickly in order to prevent it from taking 154 155 156 hold of anything. In this way the most powerful snake is perfectly under control.

Fig. 85.—Collecting Venom from a Lachesis at the Serotherapeutic Institute at São Paulo (Brazil).

Fig. 86.—Chloroforming a Cobra in order to Collect Venom, at the French Settlement of Pondicherry, in India (Stage I.).
At Pondicherry, where is collected the greater portion of the venom of Naja tripudians used by me for the vaccination of the horses that produce antivenomous serum, it is customary to chloroform the snakes in order to render them easier to manipulate.
The reptile is placed in a large covered jar, containing a pad of absorbent wool impregnated with chloroform (figs. 86, 87), and in a few minutes it is stupefied. It is then grasped by the neck with the hands, and the edge of a plate is slipped between its jaws. On compressing the two poison-glands with the fingers, the venom dribbles out on to the plate.
A detailed description of this technique will be found in a note kindly drawn up for me by my friend Dr. Gouzien, late head of the Medical Staff of the French Settlements in India, and reproduced further on in the section of this book devoted to documents. The note in question was accompanied by figs. 17, 18, 19, 86, 87, and 88, which are reproduced from photographs, for which I am indebted to the kindness of M. Geracki, Engineer of the Savanna Spinning Mill at Pondicherry, Dr. Lhomme, and M. Serph, Assistant Surgeon-Dispenser.
The collection of the venom having been completed, the snake is put back into its cage again, the tail and the body being introduced first, and then the head. The lid or trap-door is half closed with the left hand, and, with a quick forward thrust, the right hand releases its grasp of the reptile and is immediately withdrawn; at the same time the left hand completes the closure of the cage. The snake is temporarily dazed, as though stunned, and it is only after the lapse of a moment that it thinks of darting open-mouthed at the walls of its prison.
When it is desired to procure large quantities of venom, as is indispensable in laboratories where antivenomous serum is prepared, the endeavour must be made to keep the snakes alive for the longest possible time. It then becomes necessary to resort 157 to artificial feeding in the manner previously described (see p. 17), for they very often refuse to feed themselves.

Fig. 87.—Chloroforming a Cobra in order to Collect Venom, at the French Settlement of Pondicherry, in India (Stage II.).
Except when a snake is moulting, the venom can be extracted from its glands about every fortnight; and it is better that the extraction be not performed concurrently with artificial feeding, since, owing to the fact that the venom serves the animal as digestive juice, the reptile will soon perish if deprived of the means of digesting the food that it is obliged to receive. It is best, therefore, to select one day of the week for artificial feeding, and the corresponding day of the following week for the extraction of the venom.
When the venom has been collected, it must immediately be 158 placed in a desiccator over calcium chloride or sulphuric acid, in order to dry it rapidly. In hot countries, and where no laboratory specially equipped for the purpose exists, it will suffice to dry the venom in a current of air, or even in the sun. It then concretes in scales of a citrin colour, more or less dark, according to the concentration of the liquid. In this dry condition, placed in well-corked bottles, protected from damp air, it may be kept almost indefinitely without losing anything of its original toxic power. On the contrary, if the desiccation be imperfect it undergoes a somewhat rapid change, and assumes a disagreeable odour of meat peptone. I have kept samples of various venoms, dried as described, for fifteen years without any sensible diminution of their activity.

Fig. 88.—Collecting Cobra Venom at Pondicherry (Stage III.). 159
In the condition in which they are received on issuing from the glands, venoms always present the appearance of a thick saliva, of an oily consistency and more or less tinged with yellow, according to the species of snake by which the poison has been produced. They are entirely soluble in water, the addition of which renders them opalescent. Tested with litmus they exhibit a slightly acid reaction; this acidity, which is due to the presence of a very small quantity of an indeterminate volatile acid, disappears on desiccation, so that solutions of dried venom are neutral. The taste of venoms is very bitter. Their density, which is slightly greater than that of water, varies from 1030 to 1050.
Venoms are composed of a mixture, in variable proportions, of proteid substances, mucus and epithelial débris, fatty matters and salts (chlorides and phosphates of lime, ammonia and magnesia), with from 65 to 80 per cent. of water.
The elementary analysis of Cobra-venom made by H. Armstrong7 gave the following results:—
| Carbon | 43·04 | per cent. |
| Hydrogen | 7·00 | “ |
| Nitrogen | 12·45 | “ |
| Sulphur | 2·50 | “ |
| Residue | Small quantities. | |
Not much is to be learnt from these figures; it would be of far greater importance to know the exact constitution of the 160 proteid substances to which venom owes its physiological properties. Unfortunately, our knowledge of the chemistry of the albuminoid matters is still too imperfect for it to be possible for us to determine their nature.
As early as 1843 it was pointed out by Lucien Bonaparte that in the venom of Vipera berus the most important principle is a proteid substance to which he gave the name of viperin or echidnin, and which he compared to the digestive ferments. Later on Weir Mitchell and Reichert, and subsequently Norris Wolfenden, Pedlar, Wall, Kanthack, C. J. Martin, and MacGarvie Smith, showed that venoms, like diastases, exhibit a great complexity in composition; that all their characteristic toxic constituents are precipitable by absolute alcohol, and that the precipitate, when redissolved in water, recovers the properties possessed by the venom before precipitation.
According to Armand Gautier,8 venoms contain alkaloids. The latter may be obtained, in very small amounts, however, by finely pulverizing dried venom with carbonate of soda, and systematically exhausting the mixture with alcoholic ether at a temperature of 50° C. These alkaloids have yielded crystallized chloraurates and chloroplatinates, and slightly deliquescent crystallized chlorhydrates. The latter produce Prussian blue when treated with very dilute ferric salts, and mixed with a little red prussiate. They therefore represent reductive bodies analogous to ptomaines.
Norris Wolfenden did not succeed in extracting these alkaloids from Cobra-venom, whence they had nevertheless been isolated by Armand Gautier. Wolcott Gibbs, and afterwards Weir Mitchell and Reichert, likewise failed to find them in Crotalus-venom. The toxicity of these bases is, moreover, but very slight, for the totality of the alkaloids extracted by A. Gautier from 0·3 gramme of Cobra-venom did not kill a small bird.
It is therefore to the toxalbumins that the toxic properties of venoms are essentially due. 161
All venoms are not equally affected by heat. The venoms of Colubridæ (Naja, Bungarus, Hoplocephalus, Pseudechis) and those of the Hydrophiidæ are entirely uninjured by temperatures approaching 100° C., and even boiling for a short time. When the boiling is prolonged, or when venoms are heated beyond 100° C., their toxic power at first diminishes, and then disappears altogether. At 120° C. it is always destroyed.
The venoms of Viperidæ (Lachesis, Crotalus, Vipera) are much less resistant. By heating to the coagulating point of albumin, i.e., to about 70° C., their toxic properties become attenuated, and they are entirely suppressed between 80° and 85° C. Lachesis-venoms are the most sensitive; their toxicity is lost if they be heated beyond 65° C.
On separating the coagulable albumins of the venoms of Colubridæ, by heating to 72° C., followed by filtration, we obtain a perfectly limpid liquid, which is no longer injured by boiling, and in which the toxic substance remains wholly in solution. The albuminous precipitate, when separately collected and washed, is no longer toxic. The clear liquid, after being filtered, is again precipitated by absolute alcohol, and the precipitate, redissolved in an equal quantity of water, is just as toxic as the original filtered liquid.
The venoms of Viperidæ, when coagulated, by heating them to a temperature of 72° C., and filtered, are almost always inert. The albuminous coagula, if washed, redissolved in water, and injected into the most sensitive animals, produce no harmful effect whatever.
The results of dialysis likewise differ when we experiment with the venoms of Colubridæ and Viperidæ. The former pass slowly through vegetable membranes, and with greater difficulty through animal parchment. The latter do not dialyse.
Filtration through porcelain (Chamberland candle F) does not sensibly modify the toxicity of the venoms of Colubridæ; 162 on the contrary, it diminishes that of the venom of Viperidæ by nearly one-half.
By using a special filter at a pressure of 50 atmospheres, C. J. Martin has succeeded in separating from the venom of an Australian Pseudechis two substances: a non-diffusible albuminoid, coagulable at 82° C., and a diffusible, non-coagulable albumose. The former produces hæmorrhages; the second attacks the nerve-cell of the respiratory centres.
All venoms exhibit most of the chemical reactions characteristic of the proteids:—
Millon’s reaction.
Xantho-proteic reaction (heating with nitric acid and subsequent addition of ammonia = orange coloration).
Biuret reaction (caustic potash and traces of sulphate of copper).
Precipitation by picric acid, disappearing on being heated, reappearing when cooled.
Precipitation by saturation with chloride of sodium.
Precipitation by saturation with sulphate of magnesium.
Precipitation by saturation with ammonium sulphate.
Precipitation by a 5 per cent. solution of sulphate of copper.
Precipitation by alcohol.
According to C. J. Martin and MacGarvie Smith, the albumoses of the venoms of Colubridæ are hetero-albumoses, proto-albumoses, and perhaps deutero-albumoses in small quantities. They can be separated in the following manner:—
The solution of venom is heated to 90° C., and filtered in order to separate the albumins coagulable by heat. The filtrate, saturated with sulphate of magnesium, is shaken for twelve hours. By this means there is obtained a flocculent precipitate, which is placed upon a filter and washed with a saturated solution of sulphate of magnesium. The filtrate is dialysed for twenty-four hours in a stream of distilled water, and then concentrated, likewise by dialysis, in absolute alcohol. Thus we obtain a few cubic centimetres 163 of liquid, which contains a small quantity of proteids in solution. These proteids can be nothing but a mixture of proto- and deutero-albumoses with peptones. That there is actually no trace of the latter can easily be ascertained.
Neumeister9 has shown that it is impossible to precipitate all the proto-albumoses of a solution by saturation with neutral salts, and, since the filtrate becomes slightly turbid when a few drops of a 5 per cent. solution of sulphate of copper are added to it, we must conclude that it contains a small proportion of these proto-albumoses.
The deposit retained upon the filter after washing with sulphate of magnesium is redissolved in distilled water, and dialysed for three days. An abundant precipitate then becomes collected in the dialyser. This is centrifuged. The clear liquid is decanted with a pipette, then concentrated by dialysis in absolute alcohol, and finally evaporated at 40° C. until completely desiccated. The solid residue is washed and centrifuged several times in distilled water, after which it is dried on chloride of sodium.
This method enables us to separate two albumoses, both precipitable by saturation with sulphate of magnesium, and belonging to the class of primary albumoses: one of these, proto-albumose, is soluble in distilled water, the other, hetero-albumose, is insoluble; but the latter can be dissolved in dilute solutions of neutral salts. These bodies are respectively identical with those obtained by the pepsic digestion of proteids.10
In order to study separately the local and general effects of these different albumoses, C. J. Martin and MacGarvie Smith performed the following experiment:—
They introduced beneath the skin of the belly of a guinea-pig, previously shaved and rendered aseptic, two small pieces of sterilized sponge, about 2 c.mm., one of which was impregnated 164 with the solution of proteid, while the other served as control. The two small incisions, one on either side of the median line, were then sutured and covered with collodion. In this way the maximum of local effect and the minimum of general effects was obtained. The solutions of albumoses introduced by this method into the organism produced an enormous œdema, which, in from six to eight hours, extended along the whole side of the abdomen containing the sponge charged with poison.
To test the general toxic effects, the solutions were injected into a vein or into the peritoneal cavity. It was thus found that the proto- and hetero-albumoses killed the animals in a few hours.
It must therefore be concluded from these facts that the active principles of venom are proto- and hetero-albumoses, the albumins that it contains being devoid of all toxic power.
Many chemical substances modify or destroy venoms, and we shall see in another chapter that several of them, by reason of their properties, may be very usefully employed for the destruction, in the actual wound resulting from a venomous bite, of the venom that has not yet been absorbed in the circulation.
Among these substances the most important are:—
A 1 per cent. solution of permanganate of potash (Lacerda).
A 1 per cent. solution of chloride of gold (Calmette).
Chloride of lime or even hypochloride of calcium (Calmette), in a solution of 1 in 12, which is augmented, at the moment of use, by 5 to 6 volumes of distilled water, so as to bring it to the standard strength of about 850 cubic centimetres of active chlorine per litre of solution.
A 1 per cent. solution of chromic acid (Kaufmann).
Saturated bromized water (Calmette).
A 1 per cent. solution of trichloride of iodine (Calmette).
All these chemical bodies also modify or destroy the diastases and the microbic toxins. The venoms, although more resistant to the influence of heat, behave, therefore, like these latter, and exhibit the closest affinity with them. Moreover, like all the 165 normal glandular juices, they possess very manifest zymotic properties, which singularly complicate their physiological action, and upon which we shall dwell later on.
Electricity, employed in the form of continuous electrolytic currents passing through a solution of venom, destroys the toxicity of the latter, because under these conditions there is always formed, at the expense of the salts accompanying the venom, a sufficient quantity of chlorinated products (hypochlorites, chlorates, &c.), and a small amount of ozone, the oxidizing action of which is extremely powerful.
With alternating currents of high frequency, Phisalix, repeating the experiments that Arsonval and Charrin had performed upon diphtheria toxin, thought that he had succeeded in attenuating venom to the point of transforming it into vaccine.11 But it has been shown by Marmier that this attenuation was simply the result of thermic actions. When, by means of a suitable arrangement, any rise of temperature was carefully avoided, no modification of toxicity was obtained.12
The influence of light, which has no effect upon venom preserved in a dry state, is, on the contrary, very marked upon venom in solution. Solutions of venom that are destined for physiological experiments should therefore not be employed without controls, if they be several days old. Apart from the fact that, if care be not taken to render them aseptic, they very soon become contaminated with the germs of all kinds of microbes, it is found that they gradually lose a large part of their activity, especially when they remain in contact with the air. By filtering them through a Chamberland candle and keeping them in the dark, in a refrigerator, in perfectly closed phials, they may be kept unimpaired for several months. 166
The addition of glycerine in equal parts to a concentrated solution of venom is also an excellent means of preservation.
Phisalix has shown that the emanations from radium attenuate and then destroy the virulence of Cobra- and also of Viper-venom.
“Dry Viper-venom, dissolved in aqua chloroformi in the proportion of 1 in 1,000, is put up in four tubes, three of which are irradiated, the first for six hours, the second for twenty hours, and the third for thirty-six hours. Three guinea-pigs, of equal weight, are inoculated with equal quantities of the irradiated venom; a control receives the non-irradiated venom. The latter dies in ten hours; the animal inoculated from the first tube dies in twelve hours; the one inoculated from the second tube in twenty hours, and the third proves resistant without any symptom of poisoning. A second inoculation produces a transitory lowering of the animal’s temperature by half a degree. At the end of four days it dies after inoculation with a lethal dose.”
The nature of the solvent exerts a great influence upon the action of the emanations from radium: if the same experiment be performed with venom dissolved in a 50 per cent. mixture of glycerine and water, the attenuation is merely relative after six hours.
Auguste Lumière and Joseph Nicolas, of Lyons, conceived the idea of studying the effect upon venom of the prolonged action of the intense cold produced by the evaporation of liquid air.13 The Cobra-venom employed by these investigators was in solution at a strength of 1 in 1,000. It was submitted to the action of liquid air, partly for twenty-four hours and partly for nine days at -191° C. Its toxicity was in no way diminished.
Lastly, I must mention the recent researches of Hideyo 167 Noguchi,14 with reference to the photodynamic action of eosin and erythrosin upon the venoms of the Cobra, Vipera russellii, and Crotalus. It was found by the scientist in question that the toxicity of these various venoms is more or less diminished in the presence of these aniline colours, when the mixtures are insolated. Cobra-venom is the most resistant, just as it is in regard to the other physical or chemical agents. That of Crotalus, on the contrary, is the least stable. 168
The bites of poisonous snakes produce very different effects according to the species of snake, the species to which the animal bitten belongs, and according to the situation of the bite. It is therefore necessary to take these various factors into account, in describing the symptoms of poisoning in different animals.
When the quantity of venom introduced into the tissues by the bite of the reptile is sufficient to produce fatal results—which is happily not always the case—the venom manifests its toxic action in two series of phenomena: the first of these is local and affects only the seat and surroundings of the bite; the second, or general series, is seen in the effects produced upon the circulation and nervous system.
It is remarkable to find how great is the importance of the local disorders when the venomous reptile belongs to the Solenoglypha group (Viperidæ), while it is almost nil in the case of the Proteroglypha (Colubridæ and Hydrophiidæ).
The effects of general intoxication, on the contrary, are much more intense and more rapid with the venom of Proteroglypha, than with that of Solenoglypha.
In considering the usual phenomena of snake-poisoning in man, we must therefore take this essential difference into account, and 169 draw up separately a clinical description of the symptoms observed after a bite from a Cobra (Colubridæ), for instance, and another list of those that accompany a bite from Lachesis or Vipera berus (Viperidæ).
The bite of a Cobra, even of large size, is not very painful; it is characterized especially by numbness, that supervenes in the bitten part, rapidly extends throughout the body, and produces attacks of syncope and fainting. The patient soon experiences a kind of lassitude and irresistible desire to sleep; his legs scarcely support him; he breathes with difficulty and his respiration becomes of the diaphragmatic type.
By degrees the drowsiness and the difficulty of breathing become greater; the pulse, which at first is more rapid, becomes slower and gradually weaker, the mouth contracts, and there is profuse salivation, the tongue appears swollen, the eyelids remain drooping, and, after a few hiccoughs frequently accompanied by vomiting and involuntary emissions of urine or fæcal matter, the unfortunate victim falls into the most profound coma and dies. The pupils react to luminous impressions up to the last moment, and the heart continues to beat sometimes for two hours after respiration has ceased.
All this takes but a few hours, most frequently from two to six or seven, rarely more.
When the reptile by which the bite is inflicted is one of the Solenoglypha, such as a Lachesis for example, the seat of the bite immediately becomes very painful and red, then purple. The surrounding tissues are soon infiltrated with sanguinolent serosity. Sharp pains, accompanied by attacks of cramp, extend towards the base of the limb. The patient complains of intense thirst, and extreme dryness of the mouth and throat; the mucous membranes of the eyes, mouth, and genitalia become congested.
These phenomena often continue for a very long period, even for more than twenty-four hours, and are sometimes accompanied by hæmorrhages from the eyes, mouth, stomach, intestines, or bladder, and by more or less violent delirium. 170
If the quantity of venom absorbed be sufficient to cause death, the patient exhibits, a few hours after being bitten, stupor, insensibility, and then somnolence, with increasing difficulty of respiration, which ends by becoming stertorous. Loss of consciousness seems complete a good while before coma appears. Asphyxia then ensues, and the heart continues to beat for nearly a quarter of an hour after respiratory movements have entirely ceased.
In certain exceptional cases death is very rapid; it may supervene suddenly in a few minutes, even before the local phenomena have had time to manifest themselves; in this case the venom, having penetrated directly into a vein, has produced almost immediate coagulation of the blood, thus causing the formation of a generalized embolism.
If the venom be introduced in a highly vascular region, or directly into a vein, the result is almost invariably fatal. On the contrary, if the derm be scarcely broken, or if the clothing has acted as a protection, scarcely any absorption will take place. We are here confronted with the same factors of gravity as in the case of bites inflicted upon human beings by animals suffering from rabies.
In experiments we are able to eliminate all these factors, and to follow in an animal inoculated with a known quantity of venom the whole series of phenomena of poisoning, the intensity of which can be graduated. Let us see, then, how the various animals that it is possible to make use of in laboratories behave with regard to venoms of different origins.
In the monkey, the first apparent sign of the absorption of Cobra-venom, or of the venom of any other species of Colubridæ, is a sort of general lassitude; the eyelids next become half closed. The animal appears to be seeking a suitable spot in which to rest; it gets up again immediately, and walks with a jerky action; its 171 limbs have a difficulty in supporting it. It is soon attacked by nausea, vomiting and dyspnœa; it rests its head upon the ground, raises it, trying to get breath, and carries its hand to its mouth as if in order to pluck a foreign body from its throat. It totters upon its limbs, and lies down upon its side with its face against the ground. Ptosis increases, and complete asphyxia soon supervenes. The heart continues to beat for some time after respiration has ceased, and then stops in diastole.
Cadaveric rigidity very rapidly sets in, and persists for a long time, even after putrefaction has commenced. During the last moments of life the pupil remains very sensitive; the animal appears to retain unimpaired its sense of hearing and sensibility to pain. The electric excitability of the muscles of the face persists, but that of those of the limbs and body almost entirely disappears. The application of volta-faradic currents from the nape to the diaphragm produces no respiratory movement when asphyxia begins to manifest itself. The sphincters of the bladder and anus relax after a few spasms, which, in case of males, frequently provoke the ejaculation of semen; the urine and fæces immediately escape.
The autopsy reveals slight hæmorrhagic œdema at the point of inoculation, and hyperæmia of all the viscera, especially of the liver and spleen, with, very frequently, small hæmorrhagic patches on the surface of these organs, and on that of the intestine and kidneys. The serous membranes, especially the meninges, endocardium, pleuræ, and peritoneum, exhibit ecchymoses; the lungs are besprinkled with small infarcts, the more numerous the slower the intoxication. The blood remains fluid and laccate.
In poisoning by the venoms of Viperidæ, the hæmorrhagic phenomena appear at the outset, and are more intense. Death is always preceded by a period of asphyxia, indicating that the bulbar nuclei of the pneumogastric nerve have become affected. At the autopsy, however, the blood, instead of remaining fluid, is always 172 found to be coagulated into a mass in all the vessels; it afterwards gradually becomes redissolved in six or eight hours, and then appears laccate, as after poisoning by Cobra-venom, but darker.
All mammals exhibit the same symptoms after inoculation with lethal doses of venom. The same applies to birds; but in the latter the period of asphyxia is much longer, probably on account of the reserves of air accumulated in their air-sacs and pneumatic bones. They gape like pigeons that are being suffocated, rest the tip of the beak on the floor of the cage, and frequently have convulsive spasms of the pharynx, accompanied by flapping of the wings. Small birds and even pigeons are extremely sensitive to venom; fowls are more resistant.
Frogs, thanks to their cutaneous respiration, succumb very slowly. I have seen some survive for thirty hours after being inoculated with a quantity of venom which, when subcutaneously injected into a rabbit, causes death in ten minutes.
Lizards and chameleons succumb very rapidly. Grass Snakes and non-venomous snakes in general withstand doses of venom that in proportion to their weight are fairly large; nevertheless, as indeed we shall see in the sequel, they do not possess any real immunity. It is only poisonous snakes that are unaffected by enormous doses of their own venom, as has already been shown by Fontana, Weir Mitchell, and Viaud Grand Marais. They are, however, quite capable of being poisoned by snakes belonging to altogether different species; strong doses of Crotalus- or Lachesis-venom are fatal to Cobras or Kraits, and, when several poisonous snakes are shut up together in the same cage, they are not infrequently seen to kill each other as the result of repeated bites.
Fishes, which are particularly sensitive to the venom of Hydrophiidæ, readily succumb to inoculation with other venoms, such as that of the Cobra. At Saigon, in 1891, I made experiments upon the action of this latter venom on two specimens of the 173 fighting fishes, that the natives of Annam rear in aquariums in order to witness their combats and make bets on them. The fishes died five hours after intramuscular inoculation with a dose which kills a pigeon in twenty minutes.
Many invertebrates, such as leeches, crayfish, and gastropod molluscs (snails), are killed by inoculation with very small quantities of venom.
It is very difficult to specify, even within broad limits, the dose of venom necessary to kill a human being. The quantity of poison introduced by the bite of a venomous snake depends, as has already been stated, upon a large number of factors, and, very fortunately, this quantity is not always sufficient to cause death. Thus in India, that is to say in the region in which snakes are most numerous and most dangerous, the mean mortality seems scarcely to exceed 35 to 40 per cent., so far as it is possible to judge from official statistics. But, by experimenting upon animals, and commencing with known doses of venom, which has first been dried and then dissolved again in always the same quantity of physiological saline solution or sterile distilled water, we can determine exactly, for each kind of venom, and for each species of animal, the minimum lethal dose per kilogramme of animal.
The entire series of data collected by investigators who have devoted themselves to this study may be summed up as follows:—
Minimal doses lethal in twenty-four hours for a guinea-pig weighing from 600 to 700 grammes:—
| Colubridæ. | |||
|---|---|---|---|
| Venom of | Naja tripudians | 0·0002 | gramme |
| “ | Bungarus cæruleus | 0·0006 | “ |
| “ | Naja haje | 0·003 | “ |
174
| VIPERIDÆ. | |||
|---|---|---|---|
| Venom of | Vipera berus | 0·04 | gramme |
| “ | Vipera russellii (Daboia) | 0·001 | “ |
| “ | Lachesis lanceolatus | 0·02 | “ |
| “ | Lachesis mutus (Surucucu) | 0·02 | “ |
| “ | Lachesis neuwiedii (Urutù) | 0·02 | “ |
| “ | Lachesis flavoviridis | 0·007 | “ |
| “ | Ancistrodon contortrix | 0·015 | “ |
Cobra-venom. Dose lethal in twenty-four hours for different animals:—
| Dog | 0·0008 | gramme per | kilogramme | |
| Rabbit | 0·0005 | “ | “ | |
| Guinea-pig | 0·0004 | “ | “ | |
| Rat | 0·0001 | “ | 150 | grammes |
| Mouse | 0·000003 | “ | 25 | “ |
| Frog | 0·0003 | “ | 30 | “ |
Venom of Bungarus cæruleus (Common Krait), according to Elliot, Sillar, and Carmichael.15 Minimal lethal doses for:—
| Frog | 0·0005 | gramme | |
| Rat | 0·001 | “ | |
| Rabbit (by subcutaneous injection) | 0·00008 | “ | per kilogramme |
| Rabbit (by intravenous injection, according to G. Lamb) | 0·00004 | “ | “ |
Venom of Enhydrina valakadien (according to Elliot and Fraser).16 Minimal lethal doses per kilogramme:—
| Rat | 0·00009 | gramme |
| Rabbit | 0·00006 | “ |
| Cat | 0·0002 | “ |
Venom of Enhydris curtus:—
Rat 0·0005 to 0·0006 gramme per kilogramme 175
Venom of Notechis scutatus (syn. Hoplocephalus curtus; the Tiger Snake of Australia):—
| Rabbit (by intravenous injection, according to Tidswell) | 0·00006 gramme per kilogramme |
Venom of Vipera russellii (Daboia):—
| Rabbit (by intravenous injection, according to G. Lamb) | 0·00005 gramme per kilogramme |
Venom of Lachesis gramineus (Green Pit-Viper, India):—
| Rabbit (by intravenous injection, according to G. Lamb) | 0·002 gramme per kilogramme |
Venom of Crotalus adamanteus (Californian Rattle-Snake):—
| Rabbit (by intravenous injection, according to McFarland, G. Lamb, and Flexner and Noguchi) | 0·00025 gramme per kilogramme |
It will have been seen from the foregoing figures, that the respective sensitiveness of the dog, cat, rabbit, guinea-pig, rat, mouse, and frog, with regard to the same venom, is in no way proportional to the weight of these animals.
The species mentioned are, per unit of weight, more or less resistant to intoxication; and, on experimenting with other animals, as for instance the monkey, pig, ass, and horse, we find that the monkey is much more susceptible to intoxication than the dog, and that the ass is extremely sensitive (0·010 gramme of Cobra-venom is sufficient to kill it), while the horse is less so, and the pig is by far the most resistant.
The same weight of dry Cobra-venom, let us say 1 gramme to be precise, will enable us to kill 1,250 kilogrammes of dog, 2,000 kilogrammes of rabbit, 2,500 kilogrammes of guinea-pig, 1,430 kilogrammes of rat, or 8,333 kilogrammes of mouse. 176
The lethal dose for a horse being, as I have ascertained by my own experiments, about 0·025 gramme, 1 gramme of dry Cobra-venom will therefore suffice to kill 20,000 kilogrammes of horse.
Assuming that man, in proportion to his weight, possesses a resistance intermediate between that of the dog and that of the horse, we may consider that the lethal dose for a human being is about 0·015 gramme. It follows, therefore, that 1 gramme of venom would kill 10,000 kilogrammes of man, or, let us say, 165 persons of an average weight of 60 kilogrammes.
Another extremely important fact, which must not be lost sight of, is that differences of toxicity, which are often considerable, are exhibited by the venoms of different specimens of the same species of snake, or by the venom of the same snake collected at different times. I have found, for instance, in the case of the specimens of Naja and Lachesis reared in my laboratory, that, according to the length of time that the animals had been without food, and to the nearness or otherwise of the moulting period, the venom was more or less active, and that on evaporation it left behind a more or less considerable quantity of dry extract. In certain cases, immediately after the moult and after a prolonged fast, the venom was ten times more active than after a plentiful meal or before the moult.
The figures given above must therefore not be regarded as determining the minimal lethal doses of the different venoms, except in a purely comparative way, and they must be considered only as data useful to know when it is desired to experiment upon animals with these substances.
Variations of this kind are observed in the case of all species of snakes. Thus Phisalix rightly insists upon the necessity of always noting, besides the species of snake, the place of origin and the season; for he has himself seen that, as regards French vipers, those of the Jura, for example, produce in the spring a venom almost devoid of local phlogogenic action; while vipers from the vicinity of Clermont-Ferrand, though less toxic, produce much more serious local effects. 177
On the other hand, it has been shown by Th. Madsen and H. Noguchi, in a very interesting study of venoms and anti-venoms,17 that, when we examine the relation between dose and toxicity, we find that the interval separating the moment of inoculation from that of death diminishes only up to a certain point in proportion as the dose is increased. In the case of the guinea-pig, with 0·0005 gramme of Cobra-venom the interval is 3 hours 75 seconds; but after this, an increase in the dose produces only a relatively inconsiderable acceleration of death. There is therefore no strict ratio between the dose inoculated and the time that elapses until death supervenes.
When the quantity of venom introduced into the organism is insufficient to cause death, the phenomena that precede and accompany recovery differ very greatly according as the snake from which the venom was derived belongs to the Colubridæ or Viperidæ.
After a non-lethal bite from a Cobra or Krait, for example, convalescence usually takes place very rapidly, and, apart from the local œdema of the subcutaneous tissue surrounding the wound, which in very many cases leads to the formation of a suppurating abscess, no lasting injury to health is observed. The venom is eliminated by the kidneys, without even causing albuminuria, and sensation gradually returns, in twenty-four or forty-eight hours, in the part affected by the original lesion.
If the bite has been inflicted by a Viperine snake, the local lesion, which is much more extensive, almost always results in the formation of a patch of gangrene. Hæmorrhages from the mucous membranes, and sanguineous suffusions into the serous cavities, 178 such as the pleura or pericardium, may supervene more or less slowly. Pulmonary infarcts are sometimes produced, as well as desquamation and hæmorrhage from the kidneys, albuminuria, or hæmaturia. These lesions, which are more or less severe, last for several days, and then slowly disappear after a period of true convalescence. In many cases they leave behind them traces which last for months and even years, and they then more or less affect the health of the subjects according to the organs that were most seriously affected.
In certain cases, in domestic animals such as dogs, and more rarely in man, after recovery from the bite of a viper, total or partial loss of sight, smell, or hearing, has been observed. Such results, however, are fortunately exceptional. 179
The physiological effects of the various venoms are very different from those that we have just described, when these toxic substances are introduced into the organism otherwise than subcutaneously.
Their direct penetration into the blood-stream, whether by the bite of the snake itself or by experimental intravenous injection, always produces immediate results. With the venoms of Viperidæ, coagulation of the blood and, consequently, death are almost instantaneous. With the venoms of Colubridæ, which, on the contrary, destroy the coagulability of the blood, the toxic effects are less rapid, but after the lapse of only a few minutes asphyxia ensues and the death-struggle is very short.
Absorption by the serous membranes is slower, but is nevertheless effected much more quickly than when it takes place in the subcutaneous cellular tissue. When cobra-venom is injected into the peritoneal cavity of a rabbit or a guinea-pig, the local effects upon the serous membrane are almost nil. No leucocytic exudation is observed; death supervenes before this has had time to take place. The venoms of Viperidæ, on the contrary, produce, directly after their introduction into the peritoneum, an enormous afflux of sanguinolent serosity; the capillary vessels of the serous membrane, immediately becoming distended, allow the blood to filter through their walls, and the animal succumbs 180 after a few minutes, or a few hours, according to the dose injected, with the peritoneum full of blood.
When deposited upon the mucous membranes of the eye, vagina, or urethra, all venoms, those of Colubridæ like those of Viperidæ—but the latter with greater intensity—cause very acute inflammation, comparable to that produced by jequirity; the capillaries become distended, allow leucocytes to exude en masse, and, as for instance upon the eye of the rabbit, a purulent ophthalmia soon establishes itself.
Certain species of Sepedon (Colubridæ), common on the West Coast of Africa, especially in Senegambia and in the hinterland of Dahomey, and to which the name Spitting Snakes has been given, possess the faculty of projecting little drops of venom to a distance by forcibly expelling the air from their lungs, and the natives assert that this venom, when it happens to come into contact with the eyes, causes blindness. This is true to a certain extent, in so far as it produces attacks of purulent ophthalmia which are often serious; but these attacks, like those provoked experimentally in animals, can be cured in a few days when properly treated.
When absorbed by the digestive tract, the venoms of Colubridæ often produce no ill-effects. It is otherwise with those of Viperidæ. The venom of Lachesis, for example, if administered in sufficient doses, sets up acute inflammation of the gastric mucous membrane, and the animals speedily succumb with attacks of gastro-intestinal hæmorrhage, even before it has been possible for the toxic effects upon the nerve-cells to become apparent.
These facts explain the contradictions that are to be found in the works of different investigators upon this subject. It is affirmed by some writers that venom can be swallowed without danger, and they even advise the sucking of venomous wounds in order to hinder its absorption. Others, including Sir Joseph Fayrer, Richards, and Weir Mitchell, have killed pigeons and fowls by making them ingest venom of Vipera russellii, or Crotalus. 181 C. J. Martin, in experimenting upon rats with the venom of Pseudechis (Colubridæ), has succeeded in keeping these animals alive for a whole week by providing them every day with a ration of bread and milk mixed with a dose of venom one hundred times greater than the lethal dose for a subcutaneous injection. This innocuousness of the venoms of Colubridæ, which I have frequently been able to establish by causing them to be ingested by different animals, is explained by the fact that the pancreatic juice and the ptyalin of the saliva very rapidly modify the proteic substances to which these venoms owe their toxicity, so that this disappears. No trace of them is found in the fæces.
The glandular secretions of persons bitten by venomous snakes, and those of animals inoculated with doses of venom calculated to kill only after a few hours, are not infrequently found to be toxic. In the case of the urine in particular this has been shown to be so.
Observations have also been recorded by C. Francis18 and Sir Joseph Fayrer with reference to the passage of venom through the mammary gland. In the year 1893 a poor Mussulman woman died at Madras from the bite of a Cobra. She was nursing her child at the time, and the latter succumbed in its turn a few hours later, with all the symptoms of poisoning, although it had not itself been bitten, and had been suckled by its mother only once since the bite.
The histological lesions produced by snake poisoning have been particularly well studied by Hindale,19 Karlinski,20 Nowak,21 Louis Vaillant-Hovius,22 and Zeliony.23 182
Whether we are dealing with the venoms of Viperidæ or Colubridæ, the anatomo-pathological processes are alike, and the changes produced are more or less profound, according to the degree or the slowness of the intoxication.
The liver is more affected than any other organ. In cases in which death has quickly followed the injection of the venom, the protoplasm of the cells is merely cloudy, or granular, and the granulations readily take a stain in their periphery, though the interior remains uncoloured. If, on the contrary, the animal has survived for some hours, the protoplasm becomes condensed in certain parts of the cell, leaving vacuoles, the limits of which are not well defined. A portion of the cellular protoplasm is necrosed and destroyed. In these cases the nuclei have already undergone a change; although their contours may be well defined, we discover in their interior only a very little chromatin in the form of small granulations, and the nuclear fluid takes a feeble stain with basic colours, since it contains a little chromatin in solution.
When the protoplasm of the hepatic cells has suffered more pronounced lesions, the changes in the nuclei are also more marked; the quantity of nuclear chromatin diminishes and slowly loses its property of taking stains, in proportion as the protoplasm of the hepatic cells undergoes necrosis; finally, in the hepatic cell, there remains nothing more than a small quantity of granular protoplasm without a nucleus (Nowak).
In certain cases we find extensive areas of fatty degeneration, or small foci in which the hepatic tissue is absolutely destroyed. In the case of the dog it may even happen that the microscopic structure of the parenchyma has entirely disappeared. The arrangement of the hepatic cells in lobules can no longer be distinguished; the trabeculæ are ruptured and broken asunder, and we find nothing more than a confused agglomeration of cells floating in the extravasated blood. 183
In animals which have lived for a long time after being poisoned, lesions of the bile-ducts are also found. The epithelial cells have undergone fatty degeneration, or else, in the case of small animals, the ducts appear infiltrated with small mononuclear cells, which penetrate between the epithelial cells of the canaliculi. Sometimes also the latter cells are distended, and enclose large vacuoles.
Venom thus produces in the liver lesions of fatty degeneration, or necrosis, and an infiltration of the bile-ducts by lymphatic cells.
The changes in the kidney are also very extensive. The three portions of the glomerulus often exhibit lesions; the vessels of the tuft show ectasia; their walls are sometimes ruptured, and the blood is extravasated into the capsular cavity. The latter is filled with a granular exudation, which varies in amount with the slowness of the intoxication. The epithelial lining of Bowman’s capsule is swollen; the nucleus stains badly (Vaillant-Hovius).
In the tubuli contorti the lesions in the cells greatly resemble those seen in the liver. Granulations and vacuoles appear, and the nucleus becomes diffuse. The lumens of the tubules are filled with necrosed cells, and the branches of Henle are found to be similarly obliterated.
In the straight tubes and in the collecting tubes the epithelium is sometimes detached in its entirety. Some of these canals are obliterated by granular cylinders or by accumulations of epithelial cells.
The vessels met with in the parenchyma of the kidney are always greatly distended, and sometimes they are torn, whence there results the formation of small foci of interstitial hæmorrhage. In many cases the extravasated blood also destroys the parenchyma.
In the spleen, Nowak merely found a little fatty degeneration, and only in cases in which the lesions in the liver and kidneys 184 were very far advanced. The same applies to the muscular fibres of the heart. This organ exhibits, above all, hæmorrhagic infiltrations in its peripheral portion, rarely in its substance.
The lungs are the seat of more important lesions. We find in them a multitude of little infarcts. Around these the capillary vessels are extremely dilated, and the pulmonary vesicles have become very small.
All these lesions of the visceral organs strangely resemble those observed in the case of individuals who have died from yellow fever. This observation has been made by several scientists, among others by Sanarelli, and it is this perhaps that has suggested to some (Dyer, of St. Louis, R. Bettencourt, of São-Paulo24) the idea of treating—without much success, however—yellow fever by the antitoxin of venom.
The changes in the striated muscles in places at which venom has been injected do not present any specific character. The muscular fibres already become necrosed half an hour after the injection; the diseased tissue becomes permeated with an albuminous mass rich in fibrin, and the blood is extravasated. A few hours later we observe, between the bundles of degenerate muscle fibres, polymorphous leucocytes. The number of these latter constantly increases, and attains its maximum after one or two days. The muscular nuclei become distorted, appear long or angular, and assume the aspect of myoblasts (sarcoblastic muscle cells). In the protoplasm of the myoblasts we frequently find particles of broken-down muscle, and globules of fat.
All these changes resemble those observed as the result of the action of a host of other muscle poisons, especially the irritant or caustic chemical substances. 185
It is extremely difficult to determine with any degree of precision the nature of the lesions produced by venoms in the nervous system. The intensity of these lesions depends in the first place upon the length of time that has elapsed between the introduction of the venom into the organism and death. It depends, secondarily, in a large measure, upon the origin of the venom. That of the Viperidæ acts almost exclusively upon the blood by coagulation, and exhibits only a very slight degree of toxicity as regards the nerve-cell. That of the Colubridæ, on the contrary, produces manifest changes in the chromatic substance. Nissl’s bodies are completely disintegrated, and transformed into a granular mass. In the majority of the stichochromes neither the form of the bodies nor even the reticulum is distinguishable. The nuclei are opaque, the nucleoli swollen and broken up. The dendrites often become irregular and contracted (Ewing and Bailey,25 G. Lamb26).
It was found by Bailey that the majority of the cells of the anterior cornua of the medulla are normal, but that a small number of them exhibit indications of acute granular degeneration; a few cells were found to have lost almost all their chromatic substance.
From the physiological point of view it is perfectly clear that Cobra-venom especially affects the bulbar centres, and particularly the nuclei of origin of the pneumogastric nerve. We observe in the first instance the gradual suppression of the functions vested in the nerve-cells that are found in connection with the vagus nerve, the spinal accessory, and the hypoglossal. Later on the excitability of the nerve-endings in the muscles is found to have been destroyed, and this action presents great similarity to that of curare.
The venoms of Viperidæ, when injected in very weak doses, exercise a paralysing action upon the reflex excitability of the 186 medulla. But it is open to question whether these effects are not exclusively due to the lesions of the blood, which are here all-predominant; for no histological modification is observed in the cells of the central nervous system.
I have made a number of experiments with a view to discovering whether the cerebral, bulbar, or medullary substance of animals susceptible to the action of Cobra-venom (rabbit, guinea-pig, fowl) possesses the property of fixing this venom as it fixes the toxin of tetanus (Wassermann and Takaki). I found that, on pounding up a little of the pulp of the cerebral hemispheres or bulb with doses of venom lethal in two hours for the control animals, the injection of the mixture, well washed and centrifuged in order to free it from all excess of non-fixed venom, always caused death, but with a retardation of from four to ten hours. We see, therefore, that partial fixation of the venom upon the nervous elements really takes place, but we cannot conclude from this that these elements exercise an antitoxic function, any more than in the case of tetanus, for animals that receive cerebral emulsions in one thigh and the dose of venom lethal in two hours in the other thigh, succumb at the same time as the controls.
Major Rogers has made similar experiments with the venom of Enhydrina (Hydrophiidæ), and has obtained the same result on employing the cerebral hemispheres of the pigeon.27
Flexner and Noguchi,28 on their part, have compared, by aid of the method of intra-cerebral injections, the toxicity of the venom of Crotalus with that of the venom of the Cobra. On employing Cobra-venom heated to 75° C., they found that the convulsive and paralytic effects were immediate, contrary to what takes place after subcutaneous or intraperitoneal injections, but that the dose of venom necessary to produce death was the same 187 (0·1 milligramme for the guinea-pig) as when the injection is made in the peritoneum or beneath the skin.
With the venom of Crotalus heated for half an hour at 75° C., which contains but very little neurotoxin and has lost all its hæmorrhagic properties, 0·5 milligramme introduced directly into the brain of the guinea-pig only produces transitory and non-lethal effects; while, if fresh venom be employed, 0·05 milligramme is sufficient to cause death in three hours, with severe hæmorrhagic lesions. Now this dose is twenty times smaller than the minimal lethal dose for a subcutaneous injection.
It is evident that the harmful matter, in the particular case of Crotalus-venom, is not the neurotoxin, but an altogether different substance, termed by Flexner and Noguchi hæmorrhagin, which acts upon the elements of the blood and upon the endothelium of the blood-vessels.
We shall meet with this substance again in almost all Viperine venoms, and shall study it further on. 188
On making an autopsy of an animal which has succumbed to intoxication by snake-venom, we find that the blood in the heart and large vessels is sometimes coagulated into a mass, sometimes entirely fluid, and that, in certain cases, it is as black as prune-juice, while in others it is of a fine transparent red colour.
These differences in the effects of venom upon the blood are due to the fact that the various venoms contain in variable proportions, besides the neurotoxic substance which represents the true venomous toxin, other substances which act, some upon the plasmasia or fibrin-ferment, or upon the fibrin, others upon the red corpuscles, others on the leucocytes, and others again on the endothelium of the blood-vessels.
It was observed long ago by Fontana29 that after viper-bites the blood remains fluid, and Brainard30 on the contrary, pointed out that, in the case of animals that succumb very rapidly after having been bitten by a Crotalus, the blood was always found coagulated into a mass, while, when a certain interval of time had elapsed since the bite, it remained fluid. Weir Mitchell31 189 explained these differences by the hypothesis that, in cases of rapid death, the blood had not had time to become modified by the venom.
Later on it was found by Sir Joseph Fayrer, and subsequently by Halford,32 in Melbourne, C. J. Martin,33 in Sydney, G. Lamb,34 in Bombay, and recently by Noc, in my laboratory, that the venoms of Colubridæ, especially those of Naja tripudians and Australian species of this family, always leave the blood fluid after death, while the venoms of Viperidæ, on the contrary, are usually coagulant.
On the other hand, it was observed by Phisalix,35 and at an earlier date by Mosso, of Turin, that the venom of Vipera berus causes the blood of the dog to lose its coagulability, while, on the contrary, the same venom is actively coagulant as regards the blood of the rabbit.
How are these differences of action to be explained? It was found by Delezenne,36 who made an excellent study of the phenomena following the injection of peptone, extracts of organs, and other anticoagulant substances into the organism, that those of these substances that render the blood non-coagulable always dissolve the leucocytes, and thus set at liberty two antagonistic bodies which they contain. One of these substances is coagulant and is found retained by the liver, while the other remains in solution in the plasma, and keeps the blood fluid after issuing from the vessels.
Now, certain extracts of organs, ricin, abrin and certain venoms in weak doses, retard coagulation, while in large doses, on the contrary, they produce partial or general intravascular clotting.
It is believed by Delezenne that the explanation of this 190 phenomenon may be that the doses, which are weak but sufficient to produce the disintegration of the leucocytes, injure the red corpuscle in only a slight degree, while the stronger doses are equally destructive to the two kinds of blood corpuscles.
It follows that we must understand that there are two phases in the action of venoms: one negative, when the dose absorbed does not injure the leucocytes; the other positive, when the leucocytes are destroyed.
If the blood of the dog remains non-coagulable when mixed with doses of venom which, on the contrary, are actively coagulant for the blood of the rabbit, the reason would be that the leucocytes of these animals are not equally resistant to venom.
This conception, however, does not conform to the facts that I have myself observed. I have always found that viper-venom, mixed with citrate- or oxalate-plasma of the dog, rabbit, or horse, coagulates these various plasmas when the venom is in weak doses, while with strong doses coagulation is not produced. To be quite accurate, it should be stated that the quantity of venom necessary to render the plasma of the dog, or of the horse, non-coagulable is less than that which must be employed in the case of the plasma of the rabbit.
I have caused Noc to take up anew the study of this question in my laboratory, with venoms of nine different origins, and I here give a résumé of the results of his researches.37
The venoms of Viperidæ studied range themselves as follows according to their coagulant power:—
| Crotalinæ: | Lachesis lanceolatus (Fer-de-lance, Martinique). |
| Lachesis neuwiedii (Urutù, Brazil). | |
| Lachesis mutus (Bushmaster, or Surucucu, Brazil). | |
| Lachesis flavoviridis (Japan). | |
| Viperinæ: | Vipera russellii (Daboia, India). |
191
The venoms of Ancistrodon contortrix and A. piscivorus (Crotalinæ) proved entirely inactive.
No Colubrine venom exhibited coagulant power, whatever the dose employed.
There is, therefore, a very decided difference between venoms of divers origins as regards their effects upon the coagulation of the blood.
Noc has determined more especially the coagulant action of the venom of Lachesis lanceolatus (Fer-de-lance of Martinique) upon 1 per cent. citrate-plasmas, 1 per cent. oxalate-plasmas, 4 per cent. chloridate-plasmas, and upon blood rendered non-coagulable by extract of leeches’ heads. He found that, while weak doses of venom (1 milligramme per cubic centimetre of horse- or rabbit-plasma) produce coagulation in a few minutes in the citrate-plasmas, chloridate-plasmas, or those treated with extract of leeches, the doses of the same venom greater than 4 milligrammes on the contrary suppress the coagulability of these plasmas, even when there be added to them doses of chloride of calcium (for the citrate- and oxalate-plasmas), or of distilled water (for the chloridate-plasma), or of fibrin-ferment (for the plasma treated with leech-extract) sufficient to cause rapid coagulation in the control tubes that do not contain venom.
Noc also observed that the venom of the same species of snake (Lachesis lanceolatus), when heated to 75° C., entirely loses its coagulant properties; and that, with a temperature of 58° C., its coagulant power already commences to diminish. When heated for thirty minutes at a temperature of 65° C., a dose of 1 milligramme does not coagulate more than 1 c.c. of citrate-plasma in one hour. G. Lamb has likewise found that the venom of Vipera russellii loses its coagulant power when heated to 75° C.
The coagulant substance in these venoms is precipitable by alcohol at the same time as the neurotoxin and other active substances. The precipitate, when dissolved again in physiological water, preserves all the properties of the original solution. 192
Antivenomous anticolubrine serum, that is to say, that furnished by horses vaccinated against the venoms of the Cobra and the Krait, does not prevent coagulation by coagulant venoms. This need not surprise us, since the coagulant substances in venoms are destroyed by heating, and the animals vaccinated in order to obtain antitoxic serum are usually inoculated exclusively with heated venoms.
It is easy, however, to obtain active serums specific against the coagulant venoms; it is sufficient to treat these animals by inoculation with progressively increasing doses of the same venoms unheated. I have had no difficulty in achieving this result with small laboratory animals (guinea-pigs and rabbits) and also with the horse, but I have never had at my disposal a sufficient amount of the venoms of Lachesis or Vipera russellii to undertake with them the regular acquisition of large quantities of horse-serum, at once antineurotoxic and anticoagulant. The preparation of such a serum, nevertheless, presents much interest for certain countries, such as Burma, where the Daboia (Vipera russellii) is almost as common as the Cobra, and Brazil, where nearly all the casualties due to venomous snakes are produced by Lachesis.38
Contrary to what is observed with the venoms of Viperidæ in general, all the venoms of Colubridæ and, as exceptions to the rule, the venoms of some North American Crotalinæ (Ancistrodon contortrix and A. piscivorus) suppress the coagulability of the blood in vivo and in vitro. It is, however, important to observe that, in vivo, the blood remains fluid after death only if the dose of venom absorbed has been sufficient. In vitro this phenomenon is easier to study, and has been the subject of several important memoirs. 193
Halford,39 Sir Joseph Fayrer,40 C. J. Martin,41 Delezenne,42 Phisalix,43 and lastly Noc,44 have shown that the venoms of Colubridæ exert a manifestly anticoagulant action upon citrate-, chloridate-, or oxalate-plasmas, and also upon blood mixed with venom on issuing from the vessels.
On adding 1 milligramme of Cobra-, Bungarus-, Australian Pseudechis-, or Ancistrodon-venom to 1 c.c. of citrate-, oxalate-, or chloridate-plasma, and supplementing the mixture, after varying periods of contact, with a quantity of chloride of calcium (for the citrate- or oxalate-plasmas), or distilled water (for the saline plasma) sufficient to produce coagulation in a few minutes in the control tubes without venom, we find that coagulation no longer takes place after one hour in the tubes containing Cobra- or Bungarus-venom, and after ten minutes in those that contain the venom of Ancistrodon.
In doses less than 1 milligramme for 1 c.c. of plasma, these venoms by themselves never produce coagulation as do those of Lachesis or Vipera russellii. They are thus sharply differentiated in this respect.
If fresh blood issuing from the arteries of an animal be received in a vessel containing a sufficient quantity of Colubrine-venom (that of the Cobra for example), and steps be immediately taken to ensure the perfect mixture of the venom and the blood, we find that the latter has entirely lost its coagulability, just as though it had been mixed with peptone or extract of leeches’ heads.
Again, if a mixture be made in vitro of coagulant venoms, such 194 as that of the Lachesis, with anticoagulant venoms such as that of the Cobra or of Ancistrodon, it is found that these mixtures, when properly effected, become neutral, so that the respective effects of the component venoms are entirely destroyed. Assuming, for instance, that 1 milligramme of Lachesis-venom coagulates in two minutes 1 c.c. of 1 per cent. citrate rabbit-plasma, if we add to the plasma firstly 1 milligramme of Ancistrodon-, or 1 milligramme of Cobra-venom, and then 1 milligramme of Lachesis-venom, the plasma remains non-coagulated, yet coagulates perfectly on the subsequent addition of 1 c.c. of a ½ per cent. solution of chloride of calcium.
There is, therefore, a real antagonism between the actively coagulant substance contained in certain Viperine venoms and the anticoagulant substance comprised in the venoms of certain other Viperidæ (Ancistrodon), belonging to the subfamily Crotalinæ, and in those of all the Colubridæ.
The conclusion to be deduced from the foregoing facts is that the venoms of Colubridæ and those of certain Viperidæ are decidedly anticoagulant, while the majority of the venoms of Viperidæ, on the contrary, possess strong coagulant properties, even when mixed with blood in infinitesimal doses.
The question therefore arises why these coagulant Viperine venoms suppress the coagulability of the blood when mixed with it in vitro in strong doses (for example, in doses beginning from 4 milligrammes of Lachesis-venom, or 7 milligrammes of the venom of Vipera russellii for 1 c.c. of 1 per cent. citrate rabbit-plasma).
The explanation of this apparently contradictory phenomenon is furnished by the intense proteolysis that these Viperine venoms exert upon fibrin, in solution or coagulated. This proteolysis actually manifests itself with weak coagulant doses, for the compact clots formed at the outset soon become soft and then dissolve, like a cube of egg-albumen in an experiment in artificial digestion by trypsin. We shall revert to the subject later on. 195
The anticoagulant action of the venoms of Colubridæ and of Ancistrodon upon the blood appears to take effect in the first place upon the fibrin-ferment, and afterwards upon the fibrin by proteolysis. The action on the fibrin-ferment seems manifest when we experiment with anticoagulant venoms which are feebly proteolytic, like the venom of the Cobra.
I have already stated that a mixture of fresh blood with a sufficient dose of Cobra-venom is non-coagulable, as though the blood on issuing from the animal had been mixed with peptone or leech-extract. But, while blood when peptonised or mixed with leech-extract coagulates readily on the subsequent addition of fibrin-ferment, blood mixed with venom remains positively non-coagulable. It is the same with citrate- or oxalate-plasmas, which no longer coagulate when chloride of calcium is added to them, and with 4 per cent. saline plasma on the addition of distilled water.
The anticoagulant substance in the venoms of Colubridæ and Ancistrodon is precipitable by alcohol, like the coagulant substance in the venoms of Viperidæ and like the neurotoxins, from which it is difficult to separate them. The separation can nevertheless be effected by the aid of heat, if we make use of certain venoms that are particularly resistant to high temperatures, such as those of the Cobra or the Krait. These latter venoms, when heated for one hour at 70° C., cease to be anticoagulant, and preserve their toxicity unimpaired. It is, however, impossible to suppress the toxicity without at the same time destroying the anticoagulant substance.
Antivenomous serum completely protects citrate- or chloridate-plasmas against the anticoagulant action of venoms. It is sufficient to mix ½ c.c. of 4 per cent. saline antivenomous serum with 1 c.c. of 4 per cent. saline plasma to ensure that the subsequent addition of 1 milligramme of Cobra-venom to this mixture remains without effect upon the coagulability of the latter. If, 196 after a contact of two hours or more, 2 c.c. of distilled water be added, coagulation is produced just as in saline plasma without venom.
(1) Hæmolysis.—The hæmolytic properties of venoms, that is to say, their faculty of dissolving the red corpuscles, have been the subject of very important researches on the part of a number of investigators during the last few years (W. Stephens,45 Flexner and Noguchi,46 Calmette,47 Phisalix,48 Preston Kyes and Hans Sachs,49 Noc50).
The different venoms are all hæmolytic, but in very variable doses. It is possible to make a very precise comparative study of them from this special point of view by taking as a base for each venom, as was done by Noc, the unital dose of 1 milligramme (or one-tenth of a cubic centimetre of a 1 per cent. solution freshly prepared and not filtered, the filtration through porcelain retaining an appreciable part of the active substance), and noting the time strictly necessary for this dose of 1 milligramme to dissolve completely, in vitro, 1 c.c. of a 5 per cent. dilution of red corpuscles of the horse in physiological saline solution.
It is very important, before allowing the venom to act on the red corpuscles, to first wash the latter by means of several successive centrifugings with 8 per 1,000 physiological saline solution.
It is also better to choose the corpuscles of the horse in preference to those of other species of animals, since they exhibit 197 a nearly constant mean sensitivity. The corpuscles of the ox, goat, sheep, and rabbit are less sensitive. Those of man, the guinea-pig, and the rat, on the contrary, are more so.
On experimenting with washed corpuscles, it is found that venom alone is incapable of dissolving them. In order that dissolution may take place, we are obliged to add to the mixture either a small quantity of normal horse-serum, preferably heated, and, consequently, deprived of alexin (Calmette), or ½ c.c. of a 1 in 10,000 solution of lecithin in physiological saline water (P. Kyes).
Venom, therefore, is capable of hæmolysing red corpuscles only when it is quickened, either by heated normal serum, or by lecithin. The solution of lecithin employed for this purpose should be prepared by dissolving 1 gramme of lecithin in 100 grammes of pure methylic alcohol. Taking 1 c.c. of this dilution we add it to 9 c.c. of 8 in 1,000 saline solution, and make a second dilution of 1 c.c. of the foregoing mixture in 9 c.c. of saline water. This latter dilution of 1 in 10,000 is utilised as the reagent.
Let us now see how the serum or lecithin acts. It has been shown by P. Kyes that with either of these substances the mechanism of the hæmolytic action is the same, for the serum quickens the venom only through the agency of the free lecithin it contains. The lecithin takes part in the reaction by combining with the venom to form a hæmolysing lecithide more resistant to heat than its two components, for it may be heated for several hours at a temperature of 100° C., without the loss of any of its properties.
When venom is brought into contact with certain kinds of highly sensitive red corpuscles, those of the rat for example, these corpuscles, although washed and freed from serum, may undergo hæmolysis. This result is due to the fact that these corpuscles contain sufficient quantities of lecithin, which becomes liberated from their protoplasm and, uniting with the venom, constitutes the active lecithide.
It was already known that lecithin is capable of combining with various albuminoid matters and with sugars to form lecithides. 198 We must not, therefore, be surprised to find that such a combination may take place with the proteic substances in venom. The combination in this case is a truly chemical one. Lecithin in its natural state, or that which is normally found in serums which quicken venom, such as horse-serum, even when heated to 65° C., therefore plays the part of complement according to Ehrlich’s theory, or that of alexin according to the theory of Bordet, while venom itself would be an amboceptor or sensitiser.
This is not, however, the way in which the phenomenon should be understood, for it is impossible to admit the identification of heated serum or lecithin with the complementary substances or alexins, seeing that the essential characteristic of the latter is that they are intolerant of heat and become entirely inactive on being raised to a temperature of 58° C., or even by simply being kept for a few days exposed to the air and light. We must therefore suppose, with P. Kyes and H. Sachs, that the red corpuscles themselves contain substances capable of playing the part of complements (endo-complements), and that it is with these that the venom combines when quickened by the presence of lecithin or heated serum, the latter only acting because it contains free lecithin.
All substances that contain lecithin, such as bile, hot milk, or cephalin, are capable of exerting the same quickening action, but do not themselves possess any inherent hæmolytic power.
Cholesterin, on the contrary, represents a kind of antidote to lecithin, as also to normal serums. It prevents hæmolysis of the red corpuscles in a mixture of washed corpuscles and venom, yet it does not in any way modify the properties of true alexins or complements.
Moreover, no correlation exists between lecithides and the neurotoxin in venoms. The combination lecithin + venom possesses hæmolytic action, but is in no way neurotoxic. Conversely, venom can be freed from its groups of molecules combinable with lecithin, and remain neurotoxic.
Lecithide is insoluble in ether and acetone, but soluble in 199 chloroform, alcohol, toluene, and water. Its properties are therefore entirely distinct from those of its two components. It precipitates slowly from its aqueous solutions, without losing its hæmolytic power; it does not show biuret-reaction; it dissolves with equal readiness the red corpuscles of all species of animals, and its effects, like those of venom, are impeded by cholesterin.
Kyes has succeeded in obtaining lecithides with all the hæmolytic venoms that he was able to study: thus he has prepared lecithides from Lachesis lanceolatus, Naja haje, Bungarus, Lachesis flavoviridis, and Crotalus. It is therefore probable that the lecithinophile group exists in all venoms, even when these differ as regards their other properties.
A wide range of difference is exhibited by the various venoms, as regards their hæmolysing power in the presence of normal heated serum or lecithin. The venom of Naja and that of Bungarus are the most active. The action of the venoms of Viperidæ, and especially of those of Crotalus, is very weak. For example, while 1 milligramme of Cobra-venom dissolves in from five to ten minutes 1 c.c. of a 5 per cent. dilution of red corpuscles in the presence of lecithin or normal heated serum, the same dose of the venom of Vipera russellii takes thirty minutes to effect the dissolution, and the venom of Lachesis lanceolatus takes three hours.
P. Kyes and H. Sachs have discovered the apparently paradoxical fact that, if to the red corpuscles of certain species of animals Cobra-venom be added in increasing doses, hæmolysis augments up to a certain point, beyond which the destruction of the corpuscles shows progressive diminution. In a large dose Cobra-venom no longer produces any effect upon the corpuscles of the horse, for example, even when the venom is added in presence of a great excess of lecithin or heated serum. It would seem, then, that, according to the theory of Ehrlich, under the influence of an exaggerated amount of venom-amboceptor there is produced a deviation on the part of the complement (serum or 200 lecithin), and that the latter, instead of fixing itself upon the corpuscles, becomes united with the surplus fraction of the amboceptors, which has remained free in the liquid.
Noguchi,51 resuming the study of this extremely curious action of strong doses of venom, observed that the red corpuscles of certain species of animals (such as the horse for example), when previously washed and held in suspension in a physiological solution of sea-salt containing 4 per cent. of Cobra-venom, acquire a considerable augmentation of resisting power with regard to various physical and chemical agents. In consequence of this they are no longer hæmolysed by distilled water, ether, or saponin.
Nevertheless, acids or alkalies, except ammonia, destroy corpuscles treated with venom more easily than those in their normal condition.
If corpuscles, previously treated with a strong dose of venom, are subjected to repeated washings in physiological saline solution, the special resistance acquired by them in the presence of the venom disappears; they even become more sensitive to the action of destructive agents, such as water, ether, or saponin.
The principle contained in venom, to which must be attributed the protective action, is not destroyed by heating to 95° C., although at this temperature Cobra-venom becomes partially coagulated. Moreover, the protective substance is contained in the coagulum, while the hæmolysin remains entirely in the filtrate. The agglutinin of venom, on the other hand, is destroyed at a temperature of 75° C. The protective substance, therefore, can be identified neither with the hæmolysin nor with the agglutinin.
It follows that it is impossible to accept the hypothesis of the “deviation of the complement” suggested by Kyes and Sachs to explain the innocuousness of strong doses of venom. Besides, it would be difficult to reconcile this hypothesis with the fact, observed by Noguchi, that venom in a strong dose protects corpuscles, 201 not only against the action of lecithin (complement), but also against distilled water, ether, &c.
Noguchi, seeking more thoroughly to elucidate the mechanism of this protective action, finds that Cobra-venom forms a precipitate with blood-serum, when the latter is relatively poor in salts or when it is dilated with water. It likewise forms a precipitate with the aqueous extract of red corpuscles, and precipitates the globulins, hæmoglobin, or globin of the corpuscle, when treated separately. The precipitates are insoluble in water, but dissolve with the assistance of a small quantity of acid or alkali, and also in a great excess of saline solution.
Noguchi supposes that red corpuscles, when treated with strong solutions of venom, are protected against destructive agents on account of the formation by the venom and certain constituents of the corpuscle (chiefly hæmoglobin) of a compound insoluble in water. When this compound is removed by repeated washings in physiological solution, the corpuscles can easily be hæmolysed afresh by the ordinary destructive agents. Venom, none the less, exerts a noxious influence upon the corpuscles in all cases; but when strong solutions are employed, this effect is masked by the protective action.
All kinds of red blood corpuscles are not equally sensitive to the protective action of strong doses of venom. In this respect all degrees are observed in the action of venom. Thus the corpuscles of the dog are not protected at all by Cobra-venom. But it is interesting to observe that this venom in no way precipitates either the aqueous extract of dog’s corpuscles, or the hæmoglobin, or the globin of this animal.
The venom of Crotalus and that of Ancistrodon likewise possess protective power, which is, however, less marked than in the case of Cobra-venom.
Noguchi finally points out that corpuscles treated with venom are not hæmolysed by fluorescent substances such as eosin. They are also refractory to the hæmolysing action of tetanolysin. 202
The resistance of the hæmolysins of venom to heat (which, according to Morgenroth, may extend to heating for thirty minutes at a temperature of 100° C.) explains how it is that the serum of horses immunised by means of venoms heated to 72° C. is distinctly antihæmolysing, and capable of perfectly protecting the red corpuscles in vitro and in vivo.
I have been able to prove that the antineurotoxic property of antitoxic serums with regard to the venoms of Colubridæ is pretty much on a par with their antihæmolysing property, so that it is possible to measure in vitro the antitoxic activity of a serum by establishing the degree of its antihæmolysing activity. Thus we see that a serum, which is antitoxic and antihæmolytic with respect to the venom of Naja, is likewise antihæmolytic as regards the other Colubrine-venoms, and even certain venoms of Viperidæ. Here we have a very important fact, for it enables us to measure in vitro the activity of antivenomous serums.
(2) Precipitins of Venoms.—The serum of rabbits treated with increasing doses of Cobra-venom precipitates the latter in more or less concentrated solution. It has no effect as regards other venoms. On the other hand, the serum of a strongly immunised horse, the antivenomous power of which was pretty considerable, gave no precipitate with Cobra-venom; the formation of precipitate is therefore entirely independent of that of antitoxins (G. Lamb).52
(3) Agglutinins of Venoms.—Besides their hæmolytic action, it is easy to observe that certain venoms, especially those of Viperidæ, agglutinate the red corpuscles, and that the substance that produces this agglutination is different from the hæmolysing substance; for it acts rapidly at a temperature of O° C., at which hæmolysin manifests its effects only with extreme slowness. Heating to 75° C. destroys this agglutinant property (Flexner and Noguchi). 203
The white corpuscles themselves do not escape the action of venom. It is possible in vitro to prove this action upon leucocytic exudations obtained, e.g., by injecting sterilised cultures of Bacillus megatherium into the pleura or peritoneum of the rabbit. After a few hours this exudation is removed by means of capillary tubes, and, on mixing these with weak doses of venom, we see, in the course of a microscopic examination, that the large mononuclear cells are the first to be dissolved, then the polynuclears, and lastly the lymphocytes. The leucolysis is much more intense and more rapid with Cobra-venom than with that of Crotalus (Flexner and Noguchi, Noc). 204
Proteolytic, Cytolytic, Bacteriolytic and various Diastasic Actions of Venoms: Diastasic and Cellular Action on Venoms.
The proteolytic action of venoms on gelatine, fibrin, and egg-albumen has been studied by Flexner and Noguchi,53 Delezenne,54 and subsequently by Noc55 in my laboratory. It was already known that in vivo certain venoms exert a manifestly dissolving action on the endothelium of blood-vessels and on the muscular tissues themselves.
Delezenne, on his part, has established the existence in snake-venoms of a kinase analogous to the kinase of leucocytes and enterokinase. Venom alone does not attack egg-albumen coagulated by heat, but it confers an exceedingly strong digestive power on inert pancreatic juices.
Lachesis-venom has been found to be much the richest in kinase. It digests gelatine perfectly, and when this substance has been subjected to its action it is no longer capable of being solidified.
Lannoy,56 on the other hand, experimenting upon albuminoid 205 substances (casein, albumins of ox-serum) in solution, has shown that Cobra-venom and that of Vipera disintegrate the albuminoid molecule; but the latter remains soluble after the addition of formol and is no longer precipitable by acetic acid. The hydrolysis never leads to the stage of peptone, but only to the formation of albumoses which give biuret-reaction.
The action of venoms upon fibrin may be demonstrated in vitro by bringing sufficient quantities of venom, 1 centigramme, for example, into contact with small fragments of non-heated fibrin, derived from blood clots from an ox, rabbit, or birds, and carefully washed. These fragments soon separate from each other, and become dissolved in a space of time which varies according to the venom used. The Viperine-venoms, especially those of Lachesis and Ancistrodon, are the most active. Viper-venom is much less so, and the venoms of Colubridæ are the slowest.
This proteolytic action of the various venoms corresponds pretty exactly to their coagulant and decoagulant action on rabbit- or horse-plasma, so that, as I have already stated, we must suppose that the property possessed by Viperine-venoms of more or less rapidly dissolving blood which they have caused to coagulate, results from the fact that these venoms contain, in addition to a coagulant substance, another substance which is strongly proteolytic.
The latter is destroyed by heating. Lachesis-venom, when heated to 70° C., no longer has any dissolving action on either gelatine or fibrin. Moreover, antivenomous serum furnished by horses vaccinated against heated venoms does not prevent proteolysis by non-heated venoms. On the other hand, the serum of animals vaccinated against Viperine-venoms, simply filtered by the Chamberland process and non-heated, affords perfect protection to gelatine and fibrin against the dissolving action of these venoms. 206
Simon Flexner and Noguchi57 have observed that the venoms of Naja, Ancistrodon, Crotalus, Vipera russellii, and Lachesis flavoviridis, contain substances which possess the property of dissolving a large number of the cells of warm-blooded and cold-blooded animals, and that these cytolysins are very markedly resistant to high temperatures.
They employed for their experiments 5 per cent. emulsions of organs, spermatozoids, or ova in physiological saline solution. The solution of venom at a strength of 1 per cent. was kept in contact with the different kinds of cells for three hours at a temperature of 0° C.; the liquid was then centrifuged and examined with the naked eye and under the microscope.
The venoms experimented upon dissolved more or less rapidly the parenchymatous cells of the liver, kidney and testicle of the dog, guinea-pig, rabbit, rat and sheep. The most active venoms in this respect were those of Vipera russellii, Ancistrodon and the Cobra; the venom of Crotalus was the least active.
With regard to the nerve-cells, spermatozoids and ova of cold-blooded animals (frogs, fish, arthropods, worms, and echinoderms) Cobra-venom proved to be the most active; then that of Ancistrodon, and lastly that of Crotalus.
These cytolysins are not destroyed by heating for thirty minutes at 85° C. in a damp medium, nor by dry heating for fifty minutes at 100° C.
If we bring into contact with a 1 per cent. solution of Cobra-venom, rendered aseptic by filtration through porcelain, sensitive micro-organisms, such as the cholera vibrio, or the bacterium of anthrax in a very young non-sporulated culture, or in its 207 non-spore-producing variety, we find that these microbes are dissolved by the solution of venom in varying periods of time.
On making a direct microscopical examination we see that Koch’s vibrios become immovable, then break up into granulations and disappear in the liquid. The bacteriolysis is even more distinct in the case of the bacterium. The enveloping membrane seems to dissolve, and the microbe appears as though composed of a series of granulations placed end to end, which finally disperse and disappear.
By my instructions this bacteriolytic property of venom with respect to different species of micro-organisms was studied by Noc. It was especially clearly seen with the non-spore-producing bacterium of anthrax, the cholera vibrio, Staphylococcus aureus, the bacillus of diphtheria, and B. subtilis in a young culture; it is less distinct with B. pestis, B. coli, and B. typhi, is almost nil with the pyocyanic bacillus and B. prodigiosus, and nil with B. tuberculosis.
Investigations have likewise been made by Noc, and subsequently by Goebel,58 in order to determine whether cobra-venom dissolves Trypanosomes. These hæmatozoa are more resistant than bacteria, but they nevertheless end by being dissolved after twenty to thirty minutes’ contact in the 1 per cent. solution.
The bacteriolytic substance in venom is distinct from that which produces proteolysis, for the latter is destroyed at 80° C., while the former only disappears with a temperature of and beyond 85° C. maintained for half an hour. It is likewise distinct from the hæmolysin, for this resists temperatures considerably higher than 85° C. Moreover, venom which has dissolved microbes until the saturation point has been reached, is found to have preserved in its entirety its hæmolytic power upon the red corpuscles of the horse. Neither does it act upon the microbes owing to the presence of a cytase or alexin. The well-known characteristics of alexins are not met with here—destruction at 55° to 56° C., sensitivity to light, rapid alteration at ordinary temperatures, &c. 208
We cannot, again, compare the bacteriolytic action of venom to that of rat-serum, which dissolves B. anthracis by aid of a substance distinct from vibrionicide alexin. According to the researches of Malvoz and Y. Pirenne, the lysin of rat-serum appears to be a basic substance, the neutralisation of which destroys its activity. Now Cobra-venom in a very active solution is perfectly neutral to sensitive litmus papers, while these are turned blue by rat-serum. Moreover, venom acts not only upon microbes of the same kind, but also on very different species which are not affected by rat-serum, especially upon B. pestis, for which, on the contrary, this serum, when fresh, proves a favourable culture medium. The bacteriolytic power of Cobra-venom therefore constitutes a special property of venom.
“In their work on the cytolysins of venom, S. Flexner and Noguchi have shown that animal cells, when heated to 55° C. and rendered inactive, do not undergo complete dissolution under the influence of venoms which destroy the fresh cells. The authors in question infer the existence of cellular receptors (endo-complements, according to the theory of Ehrlich), which fix the amboceptors of venom. Pursuing the same order of ideas, I have observed that bacteria killed by heating for one hour at 60° C. do not undergo total disintegration as do living bacteria. But, while Flexner and Noguchi infer the plurality of the cytolysins in venom for different animal cells, I have not been able to prove the same thing with regard to the bacteriolysin; venom saturated with cholera vibrios to such an extent that vibrios added at repeated intervals are no longer dissolved, is incapable of dissolving another highly sensitive species of microbe, such as the asporogenous bacterium, and vice versâ. Besides, it would be difficult to understand the existence in venom of cytolysins specific for a whole series of species of micro-organisms” (Noc).59
Antivenomous serum, in a dose of 0·01 or 0·05 c.c., neutralizes the bacteriolytic action of 1 milligramme of Cobra-venom, 209 while normal serum heated, even in larger doses, is without effect. The lysin and the antivenomous serum appear also to enter into stable combination; by heating to 80° C., after dilution of the mixture neutral antivenomous serum + venom, the property of dissolving is not restored to the latter.
Pursuing his researches upon the bacteriolytic actions, Noc has also shown that the fresh serums of the rabbit, horse, guinea-pig, rat, and man are capable of destroying them completely. We must conclude from this that venom has the property of fixing the alexin of these fresh serums, and in fact it is easy to show that this fixation takes place by experimenting with hæmolytic alexin, which is much more easy to study; it is sufficient to eliminate the intervention of the hæmolysin proper to Cobra-venom.
With this object, Noc employed horse-corpuscles (which are readily dissolved by fresh rat-serum), and neutralised the hæmolysin proper to the venom by antivenomous serum, which has no effect upon fresh horse-corpuscles and upon the alexin of rat-serum.
For experimental purposes six tubes are prepared with contents as follows:—
(1) 0·5 c.c. of fresh rat-serum.
(2) 0·5 c.c. of fresh rat-serum + 0·5 milligramme of Cobra-venom (0·5 c.c. of a solution of 1 in 1,000).
(3) 0·5 c.c. of fresh rat-serum + 1 milligramme of venom (after fifteen minutes’ contact of the venom with the alexin in tubes 2 and 3 the venom is neutralised by 1 c.c. of antivenomous serum in the case of tube 2, and by 2 c.c. in that of tube 3).
(4) 1 milligramme of venom.
(5) 1 c.c. of antivenomous serum.
(6) 0·5 c.c. of fresh rat-serum + 1 c.c. of antivenomous serum.
To each tube 2 drops of defibrinated horse-blood are added, and the tubes are placed in the stove at a temperature of 35° C.
In tubes 1 and 6, which contain fresh rat-serum alone, and fresh serum + antivenomous serum, hæmolysis appears in a few minutes. 210 In tube 4, which received venom alone, hæmolysis is also produced in one hour. It is not produced at all in tubes 2 and 3, which received the neutral mixture of fresh serum and venom, proving that the hæmolytic alexin has been fixed by the venom. The latter, therefore, here plays the part of a true fixator or amboceptor.
Venom behaves, in short, after the manner of extracts of organs. The fixation of hæmolytic alexin by extracts of organs, the tissues, and animal cells (liver, spleen, spermatozoids, &c., &c.), has already been demonstrated by V. Dungern, P. Müller, Levaditi, and E. Hoke. The same fact is also observed with solutions of peptone. The fixation of alexin is therefore a general property of certain albuminoid molecules.
It was interesting to endeavour to reproduce, with Cobra-venom, J. Bordet’s experiments upon alexins and anti-alexins. It was to be hoped that we had in this substance an anti-alexic body capable of being preserved for an indefinite time and constant in its activity, which would enable us easily to measure the dose of alexin contained in a small quantity of a serum, or other liquid of leucocytic origin.
The experiment proved to Noc that, contrary to the ideas of Ehrlich and his pupils, and conformably to the results obtained by Bordet with serums and toxins, the neutralisation of venom takes place in a variable ratio.
If a dose A of fresh serum is capable of neutralising exactly 5 milligrammes of Cobra-venom with regard to a sensitive microbe, on employing a dose of the strength of 2 A we ought to find a bactericidal dose, 1 A, in the excess of serum, according to the theory of definite proportions. No such bactericidal action is seen, however; the serum, on the other hand, acts in the contrary direction by means of its nutritive substances, and in the mixture 2 A + venom we obtain a larger number of colonies of micro-organisms than in the mixture A + venom.
We see, then, that the property of cells of fixing in excess the active substance in serums, discovered by Bordet for the hæmolysins 211 (staining phenomena), is met with again in the case of extracts of organs, at least with regard to the bacteriolytic substance of Cobra-venom.
It results, then, from the foregoing facts that Cobra-venom contains a cytolysin, which acts upon micro-organisms and is capable of fixing the alexin of normal serums.
The application of these data to the living animal is evidently full of difficulties, by reason of the complexity of the substances that come into play. Let us see, however, to what extent they are capable of serving to explain the phenomena that are produced as the result of poisoning.
It was observed by Kaufmann that the cadavers of animals which have died from snake-bite are very rapidly invaded by the bacteria of putrefaction. Welch and Ewing, referring to these phenomena of rapid putrefaction in cases of death from venom, explained them as being due to the loss of the bactericidal power of the serum. In hot countries, even when snake-bites are not fatal, they are frequently complicated by local suppuration or gangrene, occasioned by micro-organisms introduced at the time of the bite. The minute analysis of the phenomena of poisoning shows, in reality, that the organism undergoes different modifications according to the quantity of venom injected and its channel of penetration.
When the dose of venom is rapidly lethal, whether because it penetrates into the veins or because a larger amount of it is diffused beneath the skin, it occasions a transient hypoleucocytosis, which is, moreover, a reaction common to injections of venom, pro-peptone, extracts of organs, and microbic toxins (Delezenne, Nolf). It follows that blood collected a short time after the injection may be totally bereft of its bactericidal power, in consequence of the disappearance of the leucocytes, which have migrated into the organs.
Thus it was observed by S. Flexner and H. Noguchi that the serum of a rabbit, treated with 10 milligrammes of Cobra-venom, showed, fifty-seven minutes after the injection, a great loss of bactericidal properties. But it is impossible to conclude, from 212 the diminution of bactericidal power in this experiment, that the alexin becomes fixed by the venom. Since the secretion of alexin is connected with the presence of leucocytes, the hypoleucocytosis due to the venom is sufficient to explain the loss of bactericidal power.
Nevertheless, the action of venom is not confined to these physiological phenomena; in diffusing itself through the organism it stays more especially in parts where the circulation has become slower, in the capillaries of the organs where the leucocytes that have disappeared from the general circulation are already to be found agglomerated and altered. Here the cytolysins of the venom, continuing their effects, are capable of neutralising the alexins set at liberty by the destruction of the leucocytes, and thus the rapid multiplication of the bacteria of putrefaction, which have come from the intestine or were carried in with the bite, is easily explained. In the same way, we can account for the suppuration that is met with as a complication of non-lethal bites, in spite of the hyperleucocytosis consequent upon the penetration of a weak dose of venom; immediate neutralisation of the alexin set at liberty at the level of the wound has sufficed to enable micro-organisms to multiply.
So long ago as 1884, de Lacerda, in his “Leçons sur le venin des serpents du Brésil,” described the results of his researches upon the diastasic actions of venom. He proved that venom emulsifies fats, causes milk to curdle, and does not saccharise starch. But the solutions of venom employed by this author were not sterile, so that putrefactive phenomena may be believed to have occurred in the course of his experiments.
The subject has been studied afresh by Wehrmann60 in my laboratory, and afterwards by Lannoy.61 These two investigators 213 have shown that venoms do not hydrolyse either starch or inulin. Cobra-venom and that of Vipera change saccharose very slightly. They do not modify the glucosides (amygdalin, coniferin, salicin, arbutin, and digitalin); they therefore do not contain emulsin.
On the other hand, these venoms possess, as I have already stated, very interesting kinasic properties, which have been pointed out by Delezenne.62 They consist in the fact that while venom alone is incapable of digesting cooked albumin, we have only to add to a pancreatic juice, in itself devoid of effect upon albumin, a trace of venom, to see this albumin immediately become digested.
Lachesis-venom is especially active in this respect. In Delezenne’s experiments it was generally sufficient to add to 1 c.c. of inactive pancreatic juice, 0·5 to 1 c.c. of a 1 in 1,000 solution, that is 0·5 to 1 milligramme of venom, to obtain the digestion of a cube of albumin weighing 0·5 gramme in the space of from ten to twelve hours. Much weaker doses, ⅕, ⅒, sometimes even 1/80 of a milligramme still gave the same result, with this sole difference that digestion took twenty-four, forty-eight, and even seventy-two hours to become complete.
Cobra-venom was found to be a little less active than the foregoing, but still its action was usually evident enough when it was employed in a dose of 0·5 milligramme or even 0·1 milligramme. As for the venom of Vipera berus, it was often necessary to employ it in doses from five to ten times stronger in order to obtain the same result.
Delezenne has ascertained, on the other hand, that these venoms entirely lose their kinasic power when they are subjected to ebullition for fifteen minutes.
This kinase or diastase, capable of quickening the inert pancreatic juice, must evidently be of very great utility to the reptile in enabling it to digest its prey. Venom, therefore, is not, as has long been believed, a purely defensive secretion; it corresponds to a physiological necessity, like that of the intestinal or 214 pancreatic juice. Herein is to be found the explanation of the fact that the non-poisonous snakes, although destitute of organs of inoculation, possess supralabial or parotid glands which produce venomous saliva.
Experiments have been made by Ch. Féré63 to determine the effect upon the development of the embryo of the introduction of venom into the albumen of the egg of the fowl. He found that 83 per cent. of the embryos, developed in eggs intoxicated with 0·05 milligramme of viper-venom and opened after being incubated for seventy-two hours, exhibited various anomalies in development.
Venoms are modified or destroyed by certain normal diastases of the organism. It was shown long ago by Lacerda, Weir Mitchell, Sir Joseph Fayrer, and Lauder Brunton, that it is possible to introduce without danger into the stomachs of adult animals amounts of venom many times greater than the lethal dose. I have repeatedly verified this, but have nevertheless observed that young mammals, while being suckled, readily absorb venom by their alimentary canal, and succumb to the ingestion of doses scarcely larger than those which kill when subcutaneously injected. Here we have a very important fact, which once more proves how easily the intestinal mucous membrane of young animals is permeated by toxins. By my instructions Wehrmann64 and Carrière,65 in my laboratory, have studied the modifications undergone by venoms in the alimentary canal of rabbits. We have seen that these animals can ingest without inconvenience doses of venom 600 times greater than the lethal dose, and that, if we cause these ingestions to be repeated several times, contrary to the assertion of Fraser66 (of 215 Edinburgh), we never succeed in obtaining immunity to the subcutaneous injection of a simple lethal dose, and no antitoxin is formed in the blood.
The ptyalin of the saliva, pancreatic juice, and bile destroy Cobra-venom in vitro. We must, therefore, assume that these diastases are veritable agents of destruction for ingested venom. The intestinal microbes play no part, any more than the intestinal juice by itself. The gastric juice has very little effect. Papain is almost as active as the pancreatic juice.
It had already been proved by Fraser, so long ago as 1895, that bile, after prolonged contact and in a sufficient dose, has a strongly destructive effect upon Cobra-venom; but, contrary to the opinion of this investigator, it is not antitoxic, for it possesses no preventive or curative property, and its effects are produced only in vitro.
We see from what has been stated above, that venoms introduced into a sensitive organism are capable of producing extremely complex effects upon the various tissues or humours. They act on the nerve-cells by their neurotoxin, on the endothelium of the blood-vessels by their hæmorrhagin (Flexner and Noguchi), on the red corpuscles by their hæmolysin, on the fibrin of the blood and muscles by their proteolytic diastase, and on the fibrin-ferment itself by their thrombase.
They also act on the leucocytes, according to the experiments of Chatenay,67 performed under the direction of Metschnikoff, and according to those of Flexner and Noguchi,68 already cited.
Thus we understand how complex must likewise be the means of defence that have to be employed in order to afford an effective protection against such poisons.
The slightly intoxicated organism at first reacts by the intervention of the leucocytes; a hyperleucocytosis is produced, accompanied 216 by a more or less considerable rise of temperature. After a few hours everything returns to its normal condition, and if the injection of a lethal dose of venom is repeated several times, at intervals of a few days, it is not long before antitoxic substances are found to appear in the serum.
When the dose of venom injected is sufficient to cause death, we observe, a very few moments after the injection, a lowering of temperature and a hypoleucocytosis, which is the more pronounced in proportion to the nearness of the dose of venom to the minimal lethal dose. With very strong doses the hypoleucocytosis has not time to manifest itself.
It is therefore probable that, in intoxication by venoms as in that by the toxins of micro-organisms, the protective rôle of the leucocytes is all-important, not only because these cells are capable of digesting venoms owing to their protoplasmic digestive juices, but also because they constitute if not the only, at any rate the principal source of the antitoxic substances or amboceptors. 217
Several physiologists, among whom it is right to mention Fontana,69 Leydig,70 Reichel,71 Raphael Blanchard,72 Phisalix and Bertrand,73 and S. Jourdain,74 have pointed out the presence of poison-glands in Tropidonotus natrix or other non-venomous snakes, and have explained the immunity enjoyed by these animals with regard to venom as being due to the existence of an internal secretion of this poison.
We also know, from the writings of Phisalix and Bertrand, that the blood of the viper, and that of the salamander and toad are toxic. For my part I have found75 that the blood of Naja, Bungarus, Lachesis, and Cerastes possesses the same properties, and a comparative study has been made by Wehrmann,76 in my laboratory, of the toxicity of the blood of the viper and of that of the blood of the eel, already established by Mosso (of Turin).77
It is remarkable to find that the blood of the various venomous or non-venomous snakes, like that of certain fishes, such as eels, produces, when injected beneath the skin or into the peritoneum, local and general effects very similar to those of venoms. Injections of 0·5 c.c. to 1 c.c. of the blood of the viper or of the common 218 snake, beneath the skin of the guinea-pig, provoke an intense local reaction, which always results in the formation of an eschar. The injection of slightly stronger doses, 1 c.c. to 2 c.c., into the peritoneum, almost always kills these animals, like venom, with symptoms of respiratory asphyxia.
The blood of Naja tripudians, injected subcutaneously, is lethal to the mouse in a dose of 0·25 c.c.
When this blood is heated, after having been suitably diluted with three or four parts of distilled water, in order to prevent it from coagulating, it is found that a temperature of 70° C. maintained for fifteen minutes is sufficient to cause it to lose all toxic effect. The same applies to the blood of the other poisonous or non-poisonous snakes, and to that of the Murænidæ.
Now, since the majority of venoms resist even prolonged heating at this temperature, it cannot be supposed that the toxicity of the blood is due to its containing venom derived from the internal secretion of the poison-glands, as was thought by Phisalix and Bertrand. On the contrary, it is probable that the toxicity results from the fact that the blood contains diastasic substances of cellular origin, which themselves represent certain of the constituent elements of venoms.
These substances, moreover, possess some of the properties of venoms, as, for instance, the faculty of producing hæmorrhages and of being influenced by antivenomous serum, which causes them to lose a large portion of their toxic qualities.
I have found that they can even be utilised to vaccinate animals against venom; by injecting weak, non-lethal, and repeated doses of dilute Cobra-blood into guinea-pigs and rabbits, I have succeeded in rendering them immune to doses of Cobra-venom several times greater than the lethal dose.
There is no doubt that it is to these substances that the poisonous and non-poisonous snakes owe the partial immunity that they themselves enjoy with respect to venoms. We know, in fact, that common snakes suffer without danger many bites from 219 vipers (Phisalix and Bertrand78), and that the Cobra is relatively little affected by inoculation with its own venom or with that of other Colubridæ, such as Bungarus, or even of Viperidæ, such as Vipera russellii.
This immunity, however, is far from being absolute; I have killed common snakes (Tropidonotus natrix) with doses of viper-venom ten times greater than the lethal one for the rabbit, and Lachesis lanceolatus (from Martinique) with 0·02 gramme of the venom of Naja tripudians.
Phisalix,79 on his part, has shown that, while it was necessary to inject from 100 to 200 milligrammes of viper-venom into other vipers or common snakes, beneath the skin or into the peritoneum, in order to cause death, the introduction of only 2 to 4 milligrammes of this venom into the brains of these reptiles was sufficient to kill them with the same symptoms of intoxication. This dose, however, is only twenty-five to thirty times greater than the lethal one for the guinea-pig.
The practical lesson to be learnt from the establishment of the foregoing facts is that poisonous snakes of different species must never be placed in the same cage, for these animals sometimes bite each other, and may thus kill one another.
Simon Flexner and Noguchi80 have studied the action of the serums of Crotalus, Ancistrodon, and a non-poisonous species, the pine snake (Pituophis catenifer), on the venoms of Naja, Ancistrodon, and Crotalus. They found that the serum of Crotalus rapidly dissolves the red corpuscles of man, the dog, rabbit, guinea-pig, sheep, rat, pigeon, and horse.
The serum of the pine snake affects the same red corpuscles, but in a lesser degree. Heating to 58° C. suppresses the hæmolytic power of these serums, but they can be restored to activity by the addition of a very small quantity of the same serum in a fresh 220 condition, of fresh serum derived from other snakes, or of fresh serum from the guinea-pig.
Antivenomous serum also, when added in a suitable dose, entirely suppresses the hæmolytic action of snake-serums; it has, however, greater effect upon the hæmolysin of Cobra-blood than upon that of the blood of other snakes. This observation had previously been made by W. Stephens,81 and it has been verified by Noc in my laboratory.
Crotalus-serum dissolves the red corpuscles of the mongoose (Herpestes ichneumon) of Jamaica, whose extraordinary resistance to venom is well known. But if variable doses of Ancistrodon-venom and Crotalus-serum be made to act simultaneously upon these corpuscles, the latter are no longer dissolved. Again, if, instead of red corpuscles which are but little sensitive, like those of the mongoose, we employ the highly sensitive corpuscles of the guinea-pig, the result is the same. These experiments are regarded by Flexner and Noguchi as proving that the amboceptors of the toxic serum become fixed, in conformity with Ehrlich’s theory of the lateral chains, upon the receptors of the sensitive erythrocytes, and leave no more receptors free for the fixation of the venom.
The same investigators have endeavoured to determine the respective toxicity of the tissues of the different organs of Crotalus. They found that the most toxic organs are the spleen and the liver; the toxicity of the spinal cord, kidney and muscles is much less. It appears that this toxicity is intimately connected with the quantity of blood that the tissues retain, for the physiological effects observed are identical with those that follow the injection of blood or serum alone.
They also ascertained that the contents of the eggs of Crotalus are especially rich in poison, and this poison appears to consist for the most part of neurotoxin, since it does not cause hæmorrhages. Phisalix has observed that the ovules of the viper exhibit analogous toxicity.82 221
Summing up what has been stated above, we find that the blood of both poisonous and non-poisonous snakes contains toxic substances, destructible by heating to 68° C., and physiologically distinct from venoms, but like the latter possessing the property of dissolving the red corpuscles of the majority of vertebrates and of producing hæmorrhages. 222
It was long ago pointed out that certain warm-blooded animals, including the mongoose (Herpestes ichneumon, Family Viverridæ), hedgehog (Erinaceus europæus, Family Erinaceidæ), pig (Sus scrofa, Family Suidæ), and some herons (Ajaja, Subfamily Plataleinæ; Cancroma, Subfamily Cancrominæ; Botaurus, Subfamily Ardeinæ; Mycteria, Subfamily Ciconiinæ), known in Colombia under the names Culebrero and Guacabo, exhibit a natural immunity with respect to snake-bites.
Pigs devour vipers with great readiness, and in the region of North America which adjoins the Mississippi and its tributaries they are even trained to destroy the young rattle-snakes and other poisonous serpents with which the valleys of these watercourses are infested.
During my stay in Indo-China I inoculated a young pig, beneath the skin of the back, with a dose of Cobra-venom (10 milligrammes) capable of killing a large-sized dog. The animal withstood the injection, but I am inclined to think that this is not a case of true immunity; it is probable that the pig owes its resistance to venom to the fact that its skin is lined with an enormous layer of adipose tissue, which is but very slightly vascular, and in which absorption takes place very slowly. This opinion is corroborated by my discovery that the serum of this animal is entirely destitute of any antitoxic substance. I mixed a dose of Cobra-venom, lethal for the rabbit, with 3·5 and 8 c.c. of pig-serum. These mixtures killed rabbits in the same time as the controls that received the venom 223 diluted with equal quantities of rabbit-serum or physiological saline solution.
The natural immunity of the mongoose and the hedgehog rests upon more scientifically established facts.
My own experiments upon the immunity of the mongoose were made with six specimens of these little carnivores captured in Guadeloupe (French West Indies), an island in which no poisonous snakes exist; consequently their immunity could not have arisen from their having become accustomed to the bites of venomous reptiles.
I first introduced a mongoose into a cage containing a Naja bungarus (Ophiophagus) of large size. The snake rose up immediately, dilated its hood, and struck savagely at the little animal, which, darting nimbly out of the way, escaped being seized and, frightened for a moment, took refuge in a corner of the cage. Its stupor, however, was but of brief duration, for at the very moment when the hamadryad was preparing to strike at it again, the mongoose, with open mouth and snarling, sprang upon the reptile’s head, bit it hard in the upper jaw and crushed its skull in a few seconds. This scene is in every respect reminiscent of the admirable description given by Rudyard Kipling, in his celebrated “Jungle Book,” of the great war that Rikki-tikki (the Mongoose) fought with Nag (the Cobra) “through the bathrooms of the big bungalow in Segowlee cantonment”:—
“Nag was asleep, and Rikki-tikki looked at his big back, wondering which would be the best place for a good hold. ‘If I don’t break his back at the first jump,’ said Rikki, ‘he can still fight; and if he fights—O Rikki!’ He looked at the thickness of the neck below the hood, but that was too much for him; and a bite near the tail would only make Nag savage.
“’It must be the head,’ he said at last; ‘the head above the hood; and when I am once there, I must not let go.’
“Then he jumped. The head was lying a little clear of the water-jar, under the curve of it; and, as his teeth met, Rikki braced 224 his back against the bulge of the red earthenware to hold down the head. This gave him just one second’s purchase, and he made the most of it. Then he was battered to and fro as a rat is shaken by a dog—to and fro on the floor, up and down, and round in great circles; but his eyes were red, and he held on as the body cart-whipped over the floor, upsetting the tin dipper and the soap-dish and the flesh-brush, and banged against the tin side of the bath. As he held he closed his jaws tighter and tighter, for he made sure he would be banged to death, and, for the honour of his family, he preferred to be found with his teeth locked. He was dizzy, aching, and felt shaken to pieces, when something went off like a thunderclap just behind him; a hot wind knocked him senseless, and red fire singed his fur. The big man had been wakened by the noise, and had fired both barrels of a shot-gun into Nag just behind the hood.”83
From the experimental point of view, these stirring battles between mongooses and cobras only show that a mongoose of the size of a large squirrel makes a plucky and victorious attack upon a venomous reptile of the most dangerous species and of very large dimensions; but it is impossible to tell with certainty whether the mongoose has been bitten.
I therefore inoculated a second mongoose with 2 milligrammes of venom, a lethal dose for 4 kilogrammes of rabbit. The animal did not experience the slightest malaise.
I then took blood from three other mongooses, by tying a carotid without killing the animals. This blood, mixed with venom or injected as a prophylactic into rabbits, exhibited an antitoxic power, which, though evident, was of little intensity, and insufficient in all cases as a certain preventative of death. All the rabbits that received a preventive dose varying from 2 to 7 c.c. of mongoose-serum succumbed to inoculation with venom, but with a considerable retardation (from two to five hours) as compared with the controls. 225
I endeavoured to determine the limit of tolerance of the mongoose with respect to venom. Two of these animals, which had never been inoculated, received doses of venom respectively four times and six times lethal for the rabbit. The first mongoose remained perfectly well; the second was ill for two days, and then recovered. A third mongoose, into which I injected a dose eight times lethal for the rabbit, succumbed in twelve hours.

Fig. 89.—Mongoose seized by a Cobra.
(For this illustration I am indebted to the kindness of M. Claine, late French Consul
at Rangoon.)
It must be concluded from these facts that the West Indian mongoose is but little sensitive to venom; that it is capable of withstanding, without malaise, doses which are considerable in proportion to its size, but that its immunity is far from being absolute. If it is generally the victor in its combats with poisonous snakes, the result is mainly due to the extreme agility with which it is endowed. 226
A number of experiments have been made by Lewin,84 and by Phisalix and Bertrand,85 upon the immunity of the hedgehog to the venom of Vipera berus.
It had long been known that hedgehogs are inveterate hunters of vipers, which they devour with avidity. Thanks to the long and sharp spines by which their bodies are protected, they avoid being bitten and contrive to catch the reptiles very cleverly, but it occasionally happens that they do not escape being struck. However, even in these cases they rarely succumb.
Inoculation with fairly large quantities of venom does not make them ill: the dose of viper-venom lethal for these small animals is about forty times greater than that which kills the guinea-pig. Their power of resistance is therefore beyond doubt.
It may be asked whether this is due to their blood normally containing antitoxic substances. In order to elucidate this question, Phisalix and Bertrand first proved that the blood of normal hedgehogs is toxic to laboratory animals, especially to the guinea-pig. A mixture of this blood with viper-venom cannot therefore be harmless. But it sufficed to heat hedgehog blood to 58° C. to cause it to lose its toxicity, and it was found that it then became antitoxic. Guinea-pigs inoculated in the peritoneum with 8 c.c. of heated hedgehog-serum were able to withstand, immediately afterwards, twice the lethal dose of viper-venom.
It really seems, therefore, that the resistance of the hedgehog to venom is due to the presence of antitoxic substances in its blood. But, as in the case of the mongoose, there is no question here of genuine immunity.
The same is probably true with respect to the herons of Colombia, the Culebrero and Guacabo, which eagerly search after 227 young snakes for food. No investigations, however, have yet been made upon this subject.
These birds, moreover, are few in number; hunters pursue them for the sake of their brilliantly coloured plumage, and it is to be regretted that no attempt is made to prevent their destruction or to acclimatise them in countries in which poisonous snakes constitute a veritable calamity, such as Martinique, St. Lucia, or India. 228
In all the countries of the globe where poisonous snakes are formidable to man, there are certain individuals who profess to be secure from all ill-effects from the bites of these reptiles, whether because they are immune to venom, or because they possess secrets which enable them to cure themselves when they happen to have been bitten. Not unnaturally these secrets are sometimes turned to profitable account, and the possessors of them generally enjoy considerable popular influence, and are very highly venerated. Intimate relations with the divinities are freely attributed to them.
Among the Romans the jugglers who carried on the profession of snake-charmers and healers of snake-bites were known as Psylli. Plutarch tells us that Cato, who loved not doctors because they were Greeks, attached a certain number of them to the army of Libya. They were accustomed to expose their children to serpents as soon as they were born, and the mothers, if they had failed in conjugal fidelity, were infallibly punished by the death of their offspring. If, on the contrary, the children were lawful, they had nothing to fear from the bites of the reptiles. “Recens etiam editos serpentibus offerebant; si essent partus adulteri, matrum crimina plectabantur interitu parvulorum; si pudici, probos ortus a morte paterni privilegium tuebatur” (Solinus).
The Libyian Psylli of antiquity still have their representatives in Tunis and in Egypt. Clot Bey writes as follows with reference to the Egyptian Psylli:—
“The Ophiogeni, or Snake-charmers, have been renowned from 229 all time. Strabo speaks of them, and Prosper Alpinus was a witness of the singular effects of their art. The majority of modern travellers who have visited Egypt have been equally struck with the freedom with which they handle poisonous reptiles and animals.
“The Psylli go from house to house, calling forth and charming the snakes that they may happen to contain. They claim to attract them by means of a particular power. Armed with a short wand, they enter the chamber to be purged from these venomous guests, make a smacking noise with their tongue, spit upon the ground, and pronounce the following incantation: ‘I adjure you, by God, if you are without or within, to appear; I adjure you, by the greatest of names; if you are obedient, appear! If you disobey, die! die!’ The snake, submissive to this command, departs forthwith, issuing from a crack in the wall or floor.”86
India is pre-eminently the country of snake-charmers. There exists an entire caste of Hindus, called Mal, who are professional catchers and vendors of snakes, but do not perform tricks with them.
The snake-charmers are recruited from among another caste, that of the Sangis or Tubriwallahs of Bengal.
These men, who are usually clothed in yellow robes and wear large turbans, manage the Cobra with really marvellous skill. All travellers who have had the opportunity of crossing India or of touching at a port on the coast or on that of Ceylon have witnessed scenes similar to that described by Natalis Rondot (figs. 90 and 91):—
“Towards six o’clock in the evening a Hindu juggler comes on board. He is poorly clad, and wears a turban decorated with three feathers, and several necklaces of those amulet-sachets called gris-gris in Senegal. In a flat basket he carries a spectacled Cobra-di-Capello. 230

Fig. 90.—Indian Snake-charmer at Colombo (Ceylon). 231

Fig. 91.—Indian Snake-charmer at Colombo (Ceylon). 232
“This man instals himself on deck; we sit down on the seat provided for the officer of the watch, and the sailors form a circle. The basket is placed on the deck and uncovered; the Cobra is coiled up at the bottom of it. The juggler squats a few paces off and commences to play a slow, plaintive, and monotonous air, with a kind of small clarinet (fig. 92), the sounds of which recall those of the Breton biniou.

Fig. 92.—Musical Instrument used by Indian Snake-charmers to Charm Cobras.
(For this figure I am indebted to the kindness of Dr. Pineau.)
“By degrees the snake moves, extends itself, and then assumes an erect posture, but without quitting the basket. It begins to appear uneasy and endeavours to recognise its surroundings; it becomes agitated and irritated, expands and spreads out its hood, breathes hard rather than hisses, and frequently and quickly shoots out its slender forked tongue; several times it makes a violent dart as though to attack the juggler; it frequently trembles, or rather gives sudden starts. The juggler keeps his eyes always fastened upon the snake, and gazes at it with a singular fixed stare. After some time, about ten or twelve minutes, the Cobra becomes less animated, grows quiet, and then sways as though influenced by the slow and monotonous rhythm of the musician; it keeps incessantly darting out its tongue. Little by little it is brought to 233 a sort of somnolent condition. Its eyes, which at first watched the juggler as though in order to take him by surprise, are, to a certain extent, fixed and fascinated by the latter’s gaze. The Hindu takes advantage of this moment of stupefaction on the part of the snake by approaching it slowly without ceasing to play, and touches the head of the Cobra, first with his nose and then with his tongue. Although this takes but an instant the reptile starts out of its sleep, and the juggler has barely time to throw himself backwards so as not to be struck by the snake, which makes a furious dart at him.
“We doubt whether the Cobra still has it fangs, and whether the Hindu incurs any real danger in approaching it. Accordingly we promise our man a Spanish piastre if he will make the snake bite a couple of fowls. A black hen, which struggles violently, is taken and offered to the Cobra, which half rises, looks at the bird, bites it, and lets it go. The fowl is released and runs off terrified. Six-minutes later, by the watch, it vomits, stretches out its legs, and dies. A second fowl is placed in front of the snake, which bites it twice, and the bird dies in eight minutes.”87
Certain jugglers exhibit snakes from which they have taken care to extract the fangs; they offer the animal a piece of cloth or soft stuff into which it drives its poison-teeth, and the fabric is then quickly snatched away in order by this means to break off the poison-fangs that have penetrated it. This operation is repeated at certain intervals with a view to preventing the reserve fangs from coming into use, and the reptiles can then be handled without any danger.
It is unquestionable, however, and I have personally satisfied myself of the fact, that many genuine snake-charmers go through their performances with Cobras whose poison-apparatus is absolutely intact. That they almost always avoid being bitten is due to a perfect knowledge of the habits and movements of these reptiles. 234 Nevertheless, accidents sometimes happen to them, and every year a few of them succumb in pursuit of their calling (see p. 370). Still, it may be asserted that some of them really know how to vaccinate themselves against venom, by making young Cobras bite them from time to time.
It is stated by E. C. Cotes,88 formerly of the Calcutta Museum, that the Indian snake-charmers do not extract the poison-fangs from their snakes. Even though deprived of its fangs, the snake would still be dangerous on account of its other teeth, the punctures of which would provide another channel for the penetration of the venom.
Snake-charmers pretend that they owe their immunity to graduated inoculations. This is not yet conclusively proved; what is better established is that they take the greatest care to avoid being bitten, and that in so doing they display the most remarkable skill.
Even in France we are acquainted with professional viper-catchers, who employ the method of graduated inoculations in order to render themselves immune to the bites of indigenous reptiles. One of these men, who lives near Arbois (Jura), takes good care to get himself bitten, at least once a year, by a young viper; when he forgets this precaution and happens to be bitten, he always feels the effects much more severely.
Fraser89 (of Edinburgh) thinks that the repeated ingestion of small quantities of venom may suffice to confer immunity, and he mentions a certain number of experiments performed by him upon white rats and kittens, from which it would appear that the ingestion of venom, continued for a long time, finally renders these animals absolutely refractory to subcutaneous inoculation with doses of the same venom several times greater than the lethal one. He therefore concludes that this process of vaccination may probably be in use among snake-charmers. 235
I have submitted this hypothesis to the test of experiment. I succeeded in making adult rabbits, guinea-pigs, and pigeons absorb enormous doses of Cobra-venom by way of the alimentary canal. In this manner I have administered doses as much as a thousand times greater than the lethal one, yet I have never been able to prove that the serum of these animals became antitoxic.
On the other hand, I have succeeded in vaccinating very young guinea-pigs and young rabbits which were still being suckled, by making them absorb, every second day, minimal and certainly innocuous doses of very dilute venom. In the case of young animals, venom is not modified by the digestive juices, and a portion of it is absorbed by the mucous membrane of the intestine. When the dose ingested is suitably reduced they withstand it, and when these ingestions are repeated every second or third day during the first weeks of life, the animals become perfectly vaccinated against doses certainly lethal for controls of the same age and weight. But it is always difficult to push the vaccination far enough for the serum to acquire antitoxic properties, and I have never been able to prove the appearance of the latter.
I think, however, that it ought to be possible to arrive at this result by experimenting upon animals such as lambs, kids, calves, or foals, the intestine of which remains permeable to toxins for a sufficiently long period.
It may be that certain snake-charmers, who claim to possess family secrets which they transmit from father to son, employ an analogous method in order, in their infancy, to confer immunity to venoms upon those of their male children who are to inherit their strange and lucrative profession.
In Mexico, certain Indians called Curados de Culebras know how to acquire the privilege of being able to be bitten by poisonous snakes without the least danger to life, by inoculating themselves several times with the teeth of rattle-snakes.
Dr. Jacolot,90 a naval surgeon, while staying at Tuxpan, made 236 enquiries as to these Curados de Culebras, and was able to satisfy himself that their immunity is an actual fact.
The process of vaccination employed by the natives of Tuxpan is as follows:—A preparatory treatment is necessary. On the very day on which a man is to inoculate himself or get himself inoculated, he takes from 5 to 15 tubers of a plant known by the name of Mano de Sapo (i.e., Toad’s hand, Dorstenia contrayerva, Family Urticaceæ). These tubers must—and this is absolutely necessary—be administered on a Friday, and always in an odd number, 5, 7, 9, &c., up to 15, according to the tolerance of the subject.
If the plant be gathered on the first Friday in March it possesses its marvellous properties in the highest degree; in this case, even if it be dry, it is still excellent for the preparatory stage of the inoculation.
The physiological effects of mano de sapo are not very marked: the circulation is slightly diminished and a sensation of cold is experienced, but there are no nervous troubles. The subject frequently has attacks of vomiting or nausea. The inclination to vomit must be fought against, for if the plant should happen to be rejected it would be dangerous to submit to the inoculation.
The root of the mano de sapo is usually taken fresh. There is another indispensable precaution: while undergoing this treatment it is necessary to abstain from all sexual intercourse for three days after the first inoculation, for two days after the second, and for one day after the third.
For the inoculation a large snake’s tooth, that is to say, one of the fangs, is employed, and the fangs of the most poisonous snakes, such as the rattle-snake (cuatro narices), are selected. The snake must be killed on a Friday, and the fangs extracted the same day. The same fang may serve for several years!
The inoculation is commenced on the dorsal surface of the left foot; care must be taken to avoid coming into contact with a vein. The skin is torn with the point of the fang, so that it bleeds a little, and the incision is in the shape of a square. 237
From the left foot the operator passes to the right wrist (anterior surface), then to the right foot (dorsal surface), and left wrist (anterior surface), always changing from one side of the body to the other.
Operations are continued on the left thigh, then on the right arm, right thigh, and left arm; in this way all the limbs are inoculated. On the body an inoculation is made in the centre of the sternum; another is made in the nape, and a final one in the centre of the forehead. The finishing touch is given with the semblance of a square incision in the tongue.
At least seven series of similar inoculations are necessary to protect a man from the spells of the serpent, and at the same time to confer upon him the faculty of curing by suction the bites of the venomous snakes that are most dreaded.
During the whole of the period in which the Indian thus submits to successive inoculations, his health shows no noteworthy derangement. He feels a slight headache and a strange inclination towards alcoholic drinks. But when the moon is at the full, then indeed, an excitement which is dangerous in another way takes possession of him. His cerebral faculties become over-excited, and he feels that his senses are deserting him; his eyes become bloodshot, and he is pursued and tormented by an irresistible impulse to bite. He has itching sensations in his gums, his mouth burns, and salivation is greatly increased. He feels that he is going to give way to the necessity to bite, and then he flees to the woods, where he bites the trees viciously, tears their bark and discharges his venom. His poisonous saliva mingles with the sap, and, surprising phenomenon, the tree withers and dies!
Woe to the man or animal who happens to be bitten by a Curado de Culebra in a fit of passion. The victim will die as quickly as if he had been bitten by a snake!
Almost all the semi-savage people of Guiana, and of the valleys of the Orinoco and the Amazons, as also the tribes of Central Africa and the races of India, possess witch-doctors, who pretend 238 to be in possession of means to preserve themselves from snake-bites, which are just as ridiculous and infallible as the procedure described above.
The archives of a criminal anthropology contain the story of a Lyonnese gold-seeker, who had himself immunised against venom by an aboriginal native of Guiana:91—
“The Indian took, from a bottle which contained several of them, a tooth of the Grage (Lachesis atrox), an extremely poisonous snake, and with it made upon my instep three incisions about 3 centimetres in breadth. He allowed the wounds to bleed for a minute. I then experienced a fainting sensation, and large drops of sweat rolled from my forehead. The wounds were next rubbed with a blackish powder. I have since learnt that this powder was composed of the liver and gall of the animal, dried in the sun and pounded up with the poison-glands. The blood immediately ceased to flow. The Indian chewed some leaves of a tree mixed with this powder, and, applying his lips to the sore, injected into it as much saliva as he could, making an effort as though to inflate a balloon. This completed the operation.
“Since then I have been bitten seven times by different species of very dangerous snakes, such as the Grage, coral-snake, &c., and have never even had an attack of fever. The Galibi, Boni, and Emerillon Indians, the Bosse negroes, and all the aboriginal natives of Guiana employ the same method of procedure. They even pretend that this kind of vaccination is transmissible to their offspring, and that the hereditary immunity is maintained through several generations.”
Some years ago Mons. d’Abbadie communicated to the Académie des Sciences92 a note from Colonel Serpa Pinto relating to another method of vaccination employed by the natives of Mozambique, which the Colonel himself consented to undergo. 239
“I was vaccinated,” writes Colonel Serpa Pinto, “at Inhambane (on the East Coast of Africa), among the Vatuas. These people extract the poison of a snake which is known in Portuguese as the Alcatifa (i.e., carpet), and is so called on account of the variegated colour of its skin, which resembles a carpet. I am not acquainted with the means employed in order to obtain the poison, which is mixed with vegetable substances, and forms with the latter a dark brown viscid paste.
“Two parallel incisions, 5 millimetres in length, are made in the skin, and into these is introduced the paste containing the poison. These incisions are made on the arms, near the junction of the radius and ulna with the carpal bones, on the back of the hand, on the back, on the shoulder-blades, and on the feet, near the great toes. After the operation the natives exact an oath that the vaccinated one will never kill a poisonous snake, because they say that henceforth the snake is his intimate friend, and they throw upon him an Alcatifa snake, which does not bite him.
“After undergoing this operation my whole body was swollen up for a week, and I underwent every possible kind of suffering.
“I have never been bitten by any snake, and cannot vouch for the infallibility of this remedy. The Vatuas do so, however, and they never kill a snake.
“A short time after having been vaccinated, I was stung, when in the Seychelle Islands, by a scorpion, which did me no harm. Ten years later, at the time of my journey across Africa, I was stung by another scorpion which hurt me dreadfully, and for a week I thought that I was going to die or lose my arm.”
Mystification and superstitious ideas play, as we see, a very great part in this preventive treatment, which is undergone by the natives of certain countries and snake-catchers or charmers. But it is not very surprising that, thanks to successive and repeated inoculations, a man can succeed in acquiring sufficient immunity to preserve himself from snake-bites.
In ancient times it was even pretended that it was possible for 240 this immunity to be transmitted in certain cases by heredity, and thus we can understand how the profession of snake-charmer was hereditary in certain native families in India or Egypt.
With reference to this subject, Professor Landouzy, in his fine work on serum therapeutics, quotes a passage from “The Pharsalia” of Lucan describing, in the year 60 A.D., the customs of the Psylli, a people encountered by the army of Cato during its sojourn in Africa. This passage is so interesting that I cannot refrain from reproducing it:—
The only scientific conclusion to be drawn from the facts and statements that we have just set before the reader is that, under certain circumstances, man can unquestionably acquire the faculty of resisting intoxication by snake-venom, by conferring upon himself a veritable active immunity by means of repeated inoculations of venom. We shall shortly see that the case is the same with regard to animals. 241
So long ago as the year 1887 it was shown by Sewall, in an important paper on “Rattlesnake-Venom,”94 that it is possible to render pigeons gradually more resistant to the action of this venom by injecting them with doses at first very small, and certainly incapable of producing serious effects, and then with stronger and stronger doses. In this way, although these little animals are very sensitive, he succeeded in making them withstand doses ten times greater than the minimal lethal dose.
A little later Kaufmann95 obtained the same result with the venom of French vipers. He did not, however, succeed in producing tolerance of doses more than two or three times greater than the lethal one.
In 1892, at the time of my first experiments with cobra-venom at Saigon,96 I arrived at the conclusion that it was possible, by 242 means of successive inoculations with heated venoms, to confer on animals a certain degree of resistance to doses invariably lethal to the controls.
From 1894 onwards, the investigations pursued simultaneously at the Paris Natural History Museum, by Phisalix and Bertrand, upon viper-venom, and at the Paris Pasteur Institute by myself, upon that of the cobra, and subsequently upon other venoms of various origins, led to much more definite results. These investigations show, on the one hand, that by vaccinating guinea-pigs or rabbits, and taking certain precautions, it is possible to confer upon these small animals a really strong immunity to venom; on the other hand, that animals vaccinated against cobra-venom are perfectly immune to doses of viper-venom or that of other snakes (Bungarus, Cerastes, Naja haje, Pseudechis) certainly lethal to the controls; and lastly, that the serum of the vaccinated animals contains antitoxic substances capable of transmitting the immunity to other animals.97
According to Phisalix and Bertrand, who, as we have stated, experimented only with viper-venom, the best method of vaccinating the guinea-pig consists in inoculating a dose of 0·4 milligramme of this venom heated for five minutes at 75° C., and, forty-eight hours afterwards, the same dose of non-heated venom. The latter is always lethal to the control guinea-pigs in from six to eight hours.
Vaccination against cobra-venom, which is much more toxic, is most surely effected by the method recommended by me, which consists in at first injecting small doses of this venom mixed with an equal quantity of a 1 per cent. solution of hypochlorite of lime. By degrees the quantity of venom is increased and that of the hypochlorite progressively diminished, and the injections are 243 repeated every three or four days, while attentively following the variations in the weight of the animals. The inoculations are suspended as soon as emaciation supervenes, and resumed when the weight becomes normal again. After four injections of chloridated venom the chloride is omitted, and a direct inoculation made with one-half the minimal lethal dose of pure venom; then, three or four days afterwards, the injection is increased to three-fourths of the minimal lethal dose; and finally, after the lapse of another three or four days, a lethal dose is injected.
If the animals prove resistant, the vaccination can thenceforth be pushed on rapidly, and the quantity of venom injected each time can be increased, testing the susceptibility of the organism by the variations in weight.
As a rule, three months are necessary for the vaccination of a rabbit against twenty lethal doses. In six months we can succeed in making it very easily withstand 100 lethal doses.
The serum of rabbits thus treated soon, i.e., after they have received from five to six lethal doses, exhibits antitoxic properties in vitro; these, however, are not very pronounced until after prolonged treatment. They gradually become just as intense as those observed in the case of animals vaccinated against diphtheria or tetanus.
In 1895 Fraser confirmed these results,98 and on May 15 in that year exhibited before the Medico-Chirurgical Society of Edinburgh a rabbit vaccinated against a dose of cobra-venom fifty times lethal.
At once considering the possibility of obtaining serums highly antitoxic against snake-venoms, and of practical utility in the therapeutics of snake-bites, I prepared to vaccinate a certain number of large animals, horses and donkeys, in order to procure great quantities of active serum. I at first experienced some difficulties in providing myself with a sufficient store of venom. But thanks on the one hand to the obliging collaboration of some of my old pupils 244 or colleagues, and on the other to the valuable co-operation of the Colonial Governments of Indo-China, the French Settlements in India, and Martinique, I soon received poisonous snakes and dried venom in abundance.

Fig. 93.—Vaccinating a Horse against Venom at the Pasteur Institute at Lille.
After this I was not long in pushing the vaccination of a few horses until I made them resist, in a single injection, 2 grammes of dry cobra-venom, a dose about eighty times lethal; for I was able to satisfy myself that about 0·025 gramme of cobra-venom was sufficient to kill fresh horses in from twelve to twenty-four hours.
The immunisation of horses to this very high degree of tolerance of venom is not obtained without difficulties; many animals succumb 245 in course of treatment from endocarditis or acute nephritis; in the case of others, each injection of venom leads to the formation of enormous aseptic abscesses, which have to be opened and drained. It may be said that on an average an interval of sixteen months is necessary in order to obtain a serum sufficiently antitoxic.

Fig. 94.—Aseptically Bleeding a Horse Vaccinated against Venom in order to obtain Antivenomous Serum at the Pasteur Institute at Lille.
When a horse is well vaccinated and tolerates without a reaction 2 grammes of dry cobra-venom in a single subcutaneous injection, it may be bled on three consecutive occasions in the space of ten days, and in this way 20 litres of blood may be drawn from it (fig. 94). 246
The bleeding is arranged in the following manner: Twelve days after the last injection of venom the horse is bled for the first time to the extent of 8 litres; five days later it is bled for the second time to the extent of 6 litres; five days later still the third bleeding takes place, when 6 litres are again withdrawn.
The animal is then allowed to rest for three months and supplied with strengthening food, and during this period 2 grammes of venom are again injected on two occasions at the end of a month, followed, a month and a half later, by the injection of 2 more grammes. The antitoxic power of the serum is thus maintained approximately at the same standard.
The serum drawn off at each bleeding must be severely tested, which is done by gauging its antitoxic power in vitro, when mixed with venom, and also its preventive effect.
An antivenomous serum may be considered to be utilisable when a mixture of 1 c.c. of serum with 0·001 gramme of cobra-venom produces no intoxicating effect in the rabbit, and when a preventive subcutaneous injection of 2 c.c. of serum into a rabbit of about 2 kilogrammes enables it to resist, two hours later, subcutaneous inoculation with 1 milligramme of venom.
The preventive power may be very quickly tested by injecting a rabbit, in the marginal vein of the right ear for example, with 2 c.c. of serum, and injecting, five minutes afterwards, in the marginal vein of the left ear, 8 milligramme of venom. This dose of 1 milligramme generally kills the control rabbits in less than thirty minutes when introduced into the veins, and in from two to three hours when injected beneath the skin.
This rapid proof by intravenous injection is extremely striking and demonstrative; it can be effected in public during a class or lecture in less than an hour, and enables an immediate estimate to be formed of the value of an antivenomous serum. When it is intended to adopt this method, it is essential to make use of a recent solution of venom, for solutions from a week to a fortnight old, although sterile, have already lost a large portion of 247 their toxicity, and, if these be employed, the dose of venom calculated to kill the control animals in thirty minutes, for example, takes an hour or more to do so.
I always prepare my test solutions of venom in the following manner:—
Ten milligrammes of dry cobra-venom are weighed in a delicate balance. The venom is dissolved in 10 c.c. of 0·8 per cent. physiological salt solution, which takes a few minutes. When the venom is thoroughly dissolved it is transferred to a test-tube, which is immersed for three-quarters of an hour in a water-bath heated to + 72° C. In this way the non-toxic albumins are coagulated without modifying the neurotoxic substance. The solution is poured on to a filter of sterilised paper, and the clear liquid which is collected is immediately put up in glass phials, which are hermetically sealed, or in small sterilised bottles. Its toxicity is tested upon control animals, and it may be kept for five or six days if protected from light, or for several weeks in a refrigerator at about 0° C.
One-tenth of this solution corresponds exactly to 1 milligramme of dry venom.
As for the antivenomous serum, as soon as its antitoxic value has been ascertained by the methods that I have just described, and it has been separated from clots and red corpuscles by suitable decantation, it is portioned out, with the usual aseptic precautions, into small sterilised bottles of 10 c.c. capacity, without the addition of any antiseptic.
In order to ensure that it will keep for a long time, care is then taken to heat the hermetically sealed bottles in a water-bath at a temperature of 58° C. for one hour, and this operation is repeated for three days in succession.
Serum prepared in this way preserves its antitoxic power unimpaired for about two years, in all climates. I have had occasion at various times to receive bottles which had been sent eighteen months and two years previously to India and Indo-China, 248 and I was able to show that their standard had not perceptibly deteriorated. It was only the appearance of the contained liquid that was slightly changed; it was discoloured, and when shaken small white flakes were seen floating through it. These flakes are not a sign of deterioration; they are composed of deposits of precipitated albumin. They can be partly dissolved again by violent shaking, or they may be separated before use by filtration through sterilised paper.
In a dry state, antivenomous serum may be kept for an almost indefinite period, in hermetically sealed glass tubes. In this condition it is usually divided into doses of 1 gramme, and when it is desired to make use of it, it is sufficient to dissolve a dose in 10 c.c. of water which has been boiled and allowed to cool, which takes two or three minutes. This solution is then injected beneath the skin, as though it were liquid serum.
The Pasteur Institute at Lille prepares in this way large quantities of antivenomous serum, which are sent all over the world to those countries in which poisonous snakes are most dangerous.
Recently, special laboratories for the production of this preparation have been instituted at Bombay and at Kasauli, in the Punjab, by Drs. G. Lamb and Semple; at Philadelphia, by Professor McFarland; at São-Paulo, in Brazil, by Dr. Vital Brazil; and at Sydney, by Dr. Tidswell.
Specificity and Polyvalence of Antivenomous Serums.—By means of a large number of experiments I have proved that snake-venoms, whatever their origin, contain two principal substances: neurotoxin, which exerts its effects upon the elements of the nervous system, and hæmorrhagin (Flexner and Noguchi), or proteolytic diastase, the effects of which remain exclusively local when the venom is introduced subcutaneously into the cellular tissue, but which produces coagulation of the blood when the venom is injected directly into the blood stream. 249
The venom of Colubridæ in general is characterised by the constant predominence of neurotoxin, to which it owes its extreme toxicity, which is especially intense in the case of cobra-venom. It contains no, or scarcely any hæmorrhagin; for this reason the local symptoms of poisoning by Colubrine venom are almost nil. This neurotoxin, as we have seen, shows itself very highly resistant to heat.
The venom of Viperidæ, on the contrary, especially that of Lachesis, is characterised by the almost total absence of neurotoxin, while its richness in hæmorrhagin is considerable. Consequently, heating for a few minutes at + 75° C. renders it almost entirely inactive, since hæmorrhagin is very sensitive to heat.
Given venom of some kind or other, the origin of which is unknown, it is therefore possible to ascertain whether the snake from which it was extracted belonged to the Colubridæ or Viperidæ, by determining its richness in neurotoxin resistant to heating at + 85° C.
Certain Viperine venoms, such as those of the European Vipera berus and Vipera aspis, the African Cerastes and American Crotalus contain at the same time a small proportion—varying greatly in amount according to the species—of neurotoxin, and a much larger proportion of hæmorrhagin. It is for this reason that these venoms, although greatly attenuated and deprived of their local action by heating, still remain toxic when injected in large doses into animals after having been heated to + 75° C.
On the other hand, some Colubrine venoms, such as those of Bungarus cæruleus, which are very rich in neurotoxin, contain a quantity of hæmorrhagin sufficient to differentiate their effects in appearance from those produced by cobra-venom, when they are injected, not beneath the skin, but directly into the veins. In this case their effects upon the blood are added to those of their neurotoxin.
It would seem, too, that the venoms of Australian Colubridæ 250 (Hoplocephalus, Pseudechis) form a special group, which is richer in hæmorrhagin than are those of the Colubridæ of the Old World.99
On studying, in the case of these various venoms, the action in vitro and in vivo of a purely antineurotoxic antivenomous serum, such as, for example, that of an animal vaccinated against cobra-venom heated to + 75° C., it is found that this serum has a very decided effect upon cobra-venom, and likewise upon that of snakes belonging to allied species (Naja bungarus, Naja haje), and that its action upon the other venoms is less in proportion as they contain less neurotoxin. It prevents hæmolysis in vitro, and suppresses the effects of intoxication on the nervous system, but does not modify in any way the phenomena of coagulation or of proteolysis.
If this serum be made to act in vitro on those Viperine venoms that, when heated to + 75° C. and deprived of their hæmorrhagin, remain neurotoxic, like the venom of the common viper, it is found that it renders them entirely innocuous. Therefore, in the case of all species of poisonous snakes, and perhaps also in that of other poisonous animals (such as scorpions), it appears that the neurotoxic substance is one and the same, and always neutralisable by an antineurotoxic serum like that of animals vaccinated against cobra-venom.
Neurotoxin being the essentially active substance in venoms, and that to which the dangerous properties of poisonous snakes, as regards man and domestic animals, are especially due, it is the effects of this that it is most necessary to prevent. Consequently, 251 the first quality that an antivenomous serum ought to exhibit, in order to be capable of being used in the therapeutics of poisoning, is the possession of an antineurotoxic power as high as possible. This antineurotoxic power is easily obtained by employing cobra-venom for the fundamental immunisation of the horses destined for the production of the serum.
Antineurotoxic serum thus prepared shows itself perfectly capable of preventing all effects of intoxication from cobra-bites, which are much the most frequent in India. In the same way it shows itself quite sufficiently efficacious with regard to Colubrine and Viperine venoms, the neurotoxic activity of which may cause death. But it does not possess any preventive action upon the local effects of hæmorrhagin, to which the noxiousness of certain Viperine venoms—such as those of Lachesis—are almost exclusively due.
In countries in which Viperidæ are very common, we must therefore not confine ourselves to vaccinating the animals that produce serum solely against the neurotoxin of cobra-venom, for instance; we must prepare these animals, after having immunised them to cobra-venom, by injecting them with progressively increasing doses of the various venoms derived from the snakes that are most frequently met with in the district.
Nothing, moreover, is easier than to train animals vaccinated against cobra-venom to tolerate strong doses of the venoms of Lachesis, Vipera russellii, Crotalus, Hoplocephalus, or Pseudechis. In a few months we succeed in obtaining serums very active against these different venoms.
Utilising the horse as producer of antitoxin, I have prepared by this method polyvalent serums capable of preventing the local action of Viperine venoms, and of suppressing in vitro their coagulant and proteolytic effects upon the blood.
Unfortunately, great as has been the kindness of the many persons who have most obligingly given me their assistance in the course of the fifteen years during which I have studied this question, 252 I have found it impossible to procure sufficient quantities of venoms of various origins to furnish each country with the polyvalent serums corresponding to its particular needs. I have therefore been obliged to confine myself to preparing for the most part antineurotoxins, which I have been able to do, thanks to the abundant provision of Cobra- and Bungarus-venoms, for which I am indebted to the liberality of the Government of the French Settlements in India, and to that of my pupils and friends who are at the present time in charge of the Colonial Laboratories of Indo-China. Moreover, the recent foundation of the Serum-Therapic Institutes of Bombay and Kasauli, Sydney, São-Paulo, and Philadelphia, to-day renders it very easy for each country to provide itself with antivenomous serum, either specific or polyvalent. Other institutes will doubtless be established for the purpose of extending the benefits of a method, the efficacy of which is sufficiently evident for its adoption to be incumbent upon all those who are concerned with safeguarding human existence. 253
It is difficult, in the present state of our knowledge on the subject of toxins and antitoxins, to determine the precise nature of the reactions that are produced in the living organism as the result of serum injected for the purpose of preventing the toxic action of venom.
I maintained, some years ago,100 that the phenomenon in this case was a purely physiological one, which I considered to be proved by the fact that, if we mix in vitro, in determinate proportions, venom and antivenomous serum, and if we heat this mixture at 68° C. for half an hour, the injection of the heated mixture kills animals as if they were inoculated with venom alone, although with a considerable retardation. I concluded from this that, in all probability, antitoxic serum does not modify the toxin with which it is mixed, but that it confines itself to displaying a parallel and opposite action by preventing the noxious effects. I therefore supposed that no chemical combination is produced between these two substances, or, at least, that the combination effected is very unstable.
My experiments were subsequently repeated by Martin and Cherry,101 who showed that the results as stated above were perfectly correct when the mixture of venom and antitoxin was heated less than ten minutes after it had been made, but that, if the heating 254 did not take place until twenty or thirty minutes later, the toxicity of the venom no longer reappeared.
On the other hand, the admirable researches of Kyes and Sachs, and subsequently those of Morgenroth, pursued under the direction of Ehrlich at the Laboratory of Experimental Therapy at Frankfort, have proved the readiness of venom to enter into chemical combination with certain elements of normal serums, in particular with lecithin, a combination which results in the formation of hæmolysing and non-toxic lecithides, the neurotoxin being left free.
It therefore seemed impossible to deny the existence of a chemical reaction between the venom and the serum, which was until quite recently considered as proved. We shall see presently that this is not the case. But let us first endeavour to determine the laws that govern the neutralisation of variable quantities of venom by antivenomous serum.
If, in a series of test-tubes, we bring the same quantity of cobra-venom (e.g., 0·00005 gramme, a dose which is invariably lethal to the mouse in two hours) into contact with progressively increasing quantities of an antivenomous serum (e.g., 0·01 c.c., 0·02 c.c., &c., up to 0·1 c.c.), and, after thirty minutes of contact, inject these different mixtures subcutaneously into a series of mice, we find that all those that have received the mixtures containing less than 0·05 c.c. of serum succumb after variable intervals, while all the rest survive. It is evident that, under these conditions, the serum experimented upon has shown itself capable of neutralising in vitro, in a dose of 0·05 c.c., 5 centimilligrammes of venom.
The same serum should therefore neutralise 1 milligramme of venom in a dose of 1 c.c., that is to say, that this mixture injected into a mouse ought to be entirely innocuous. Experiments show, however, that in reality it is necessary to mix 1·2 c.c. of serum with 1 milligramme of venom in order that the inoculated mouse may not succumb.
This proves that, in the initial mixture of 0·00005 gramme of venom + 0·05 c.c. of serum, there remained an exceedingly small 255 quantity of non-neutralised venom, and that this quantity of venom in a free state was insufficient to cause the death of the animal, or even any apparent malaise. When multiplied by twenty, however, it becomes capable of producing toxic effects; it is for this reason that, when it is desired to inoculate a mouse with twenty times the lethal dose of 0·00005 gramme neutralised, it is necessary to mix with this twenty times lethal dose a dose of serum a little larger than twenty times that which renders 0·00005 gramme of venom innocuous to the mouse, that is to say, 1·2 c.c.
If, instead of making use of the mouse as test animal, we employ the rabbit, it is found that the same serum, in a dose of 0·75 c.c., neutralises 0·001 gramme of venom sufficiently for the mixture to be innocuous when inoculated. It is clear that, in this mixture, the whole of the venom was not neutralised by the serum, but the small quantity left free is incapable of producing harmful effects.
By this method of employing mixtures of the same dose of venom with variable quantities of antivenomous serum, we are therefore enabled to determine with the greatest exactness the antitoxic power in vitro of each specimen of serum. But it must not be forgotten that the result obtained applies only to the species of animal into which the mixtures were injected.
I have already stated (Chapter VIII.) that a fairly close parallelism exists between the neurotoxic action of venoms and their hæmolytic action, and I have established that, in order that the sensitive red blood-corpuscles may be dissolved under the influence of venom, it is indispensable that the reaction take place in the presence of normal serum, since venoms have no effect upon red corpuscles freed from serum by several successive washings and centrifugings.
Preston Kyes has explained this phenomenon very well by showing that the venom combines with the lecithins in the serum, or with those contained in the stroma of the corpuscle, so as to constitute a hæmolysing lecithide.
The knowledge of this fact enables us to determine, by means 256 of a very neat and simple method, and with a sufficient degree of accuracy for practical purposes, the antitoxic power of an antivenomous serum by measuring its antihæmolytic power.102
To this end it is sufficient to cause variable doses of serum to act on a given quantity of defibrinated horse- or rat-blood, to which a constant dose of venom is then added. We employ, for example, a 5 per cent. dilution of defibrinated horse-blood, which is portioned out in doses of 1 c.c. into a series of test-tubes. To each of these tubes in succession is added a progressively increasing quantity of the serum for titration, starting with 0·01 c.c., and continuing with 0·02 c.c., 0·03 c.c., &c., up to 0·1 c.c. A control tube receives no serum. There are then introduced into all the tubes 1 decimilligramme of venom and 0·2 c.c. of normal horse-serum, deprived of alexin by previous heating for half an hour at 58° C. At a temperature of about 16° C. hæmolysis commences to manifest itself in the control tube in from fifteen to twenty minutes. It takes place in the other tubes with a retardation which varies with the dose of serum added. Tubes are to be noticed in which it does not occur even after the lapse of a couple of hours.
Experience shows that we may consider as good for therapeutic use serums which, in a dose of 0·05 c.c., completely prevent hæmolysis by 1 decimilligramme of Colubrine venom, such as that of Cobra, Krait, &c., and those that in a dose of 0·7 c.c., prevent hæmolysis by 1 milligramme of the venom of Lachesis or Vipera berus.
By a method calculated upon the foregoing, it is likewise possible to measure the antihæmorrhagic activity of an antivenomous serum, for the parallelism existing between the antineurotoxic and antihæmolytic actions of serums occurs again, as I have been able to establish in conjunction with Noc, between the antihæmorrhagic and antiproteolytic action of the same serums. 257
Now, the antiproteolytic action is easily determined by means of a series of test-tubes containing the same quantity of 20 per cent. gelatinised bouillon, rendered imputrescible by the addition of a small quantity of thymol. The gelatine being kept liquid in the incubating stove, a progressively increasing quantity of serum is poured into each tube. The same dose of venom, say 1 milligramme, is then added in each case. The tubes are placed in the stove for six hours at 36° C. They are then withdrawn and immersed in a bath of cold water. Those in which the gelatine solidifies are noted, and thus we establish the dose of antivenomous serum that inhibits the proteolysis of this substance.
These different methods of control enable us to verify the activity of antivenomous serums with great exactness, without the necessity of having recourse to experiments upon animals.
In a very important memoir on the reconstitution of the toxins from a mixture of toxin + antitoxin, J. Morgenroth103 has shown that the venom, after being naturalised by the antivenomous serum, can be dissociated from its combination by means of a method which consists in adding to the latter a small quantity of hydrochloric acid.
Previous experiments by Kyes had established:—
(1) That antivenomous serum, the antitoxic action of which is so manifest when it is mixed in vitro with cobra-venom, remains entirely inert when brought into contact with the combination lecithin + venom, that is to say, with cobra-lecithide.
(2) That the addition of lecithin to a neutral combination of venom + antivenomous serum does not set the venom free again, and that under these conditions no lecithide is formed.
If, in a neutral mixture of cobra-hæmolysin and antitoxin we could succeed in dissociating the two constituent elements, and in then making the cobra-hæmolysin combine with the lecithin, 258 we should have a toxin and antitoxin side by side; for the reasons indicated above, this toxin (lecithide) and antitoxin (antivenomous serum) would be no longer capable of combining; but the toxin (lecithide), thanks to its hæmolytic properties, could easily be demonstrated.
It is precisely this desideratum that J. Morgenroth has succeeded in realising, by means of hydrochloric acid, which renders it possible to dissociate the neutral mixture, toxin + antitoxin, into its constituent elements, and then to obtain a lecithide.
Experiments show that the quantity of lecithide thus restored absolutely corresponds to that of the cobra-hæmolysin originally added to the antitoxin, and that the antitoxin set free is not injured by the hydrochloric acid, even after twenty-four hours of contact. It is sufficient to add the quantity of soda or of ammonia necessary for the neutralisation of the acid, in order to see the antitoxin reappear in its original strength.
It is therefore possible, by causing hydrochloric acid (in a solution not stronger than 3 per cent.) to act on a neutral mixture of cobra-hæmolysin (toxin) and antitoxin, to set the former at liberty in the form of lecithide, to withdraw the latter from the action of the antitoxin, and to demonstrate its presence, owing to its hæmolytic properties.
It has been found by Kyes and Sachs that, under the influence of hydrochloric acid, cobra-hæmolysin becomes resistant to heat to such an extent that it is not destroyed even by prolonged heating at 100° C.
If to a neutral mixture of toxin + antitoxin we add a small quantity of hydrochloric acid, and then heat the mixture at 100° C., the antitoxin being in this case destroyed, we shall recover the whole of the toxin originally employed.
Therefore, as was shown by me so long ago as 1894, if the mixture of toxin + antitoxin produces a chemical combination between the two substances, this combination is unstable, and can be effectively broken up into these two constituent elements by various influences. 259
In all countries the remedies recommended for the bites of poisonous snakes are innumerable, and native pharmacopœias abound in so-called infallible recipes.
Pliny himself wrote on this subject as follows:—
“For poisonous bites, it is customary to employ a liniment made of fresh sheep-droppings, cooked in wine. Rats cut in two are also applied; these animals possess important properties, especially at the epoch of the ascension of the stars, seeing that the number of a rat’s fibres wax and wane with the moon.
“Of all birds, those that afford most assistance against snakes are vultures. The black ones are the weaker. The odour of their feathers when burnt puts snakes to flight. Provided with a vulture’s heart one need not fear encounters with snakes, and can also defy the wrath of wild beasts, robbers, and princes.
“Cock’s flesh, applied while still warm, neutralises the venom of snakes. The brains of the bird, swallowed in wine, produce the same effect. The Parthians, for this purpose, make use of chicken’s brains. The fresh flesh of the pigeon and the swallow, and owls’ feet burned, are good against snake-bites.
“If one has been bitten by a snake or by any venomous animal, another method of cure is to take salt fish and wine from time to time, so as to vomit in the evening. This remedy is chiefly 260 efficacious against the bite of the Chalcis, Cerastes, Seps, Elaps and Dipsas.”
In Equatorial America, and especially in India, a multitude of plants are credited with marvellous properties, which they possess only in the imagination of the snake-charmers or medicine-men by whom they are employed. None of them stand the test of experiment, any more than the more or less compound drugs, numbers of specimens of which from all sources have passed through my hands.
It cannot, however, be denied that certain chemical substances, of well-defined composition, are very useful, not as physiological antidotes to venoms, but as agents for their modification or destruction in the poisoned wounds, when they have not yet been absorbed. In this way permanganate of potash, chromic acid, chloride of gold, and the alkaline hypochlorites, especially hypochlorite of lime, may be extremely useful under many circumstances.
Permanganate of potash was recommended in 1881 by Professor de Lacerda,104 of Rio de Janeiro, as the result of experiments made by him with venoms of Brazilian snakes. When a few cubic centimetres of a 1 per cent. solution of permanganate of potash are quickly injected into the actual wound caused by the bite and around the point of inoculation, there can be no doubt that the venom not yet absorbed is destroyed. When mixed in vitro with venom, permanganate renders the latter innocuous.
Here, however, it is a case of actual destruction by direct contact. If we inject a lethal dose of venom into the right thigh of an animal, for example, and several cubic centimetres of permanganate solution into different parts of the body, or beneath the skin of the left thigh, neither the general intoxication nor the local effects of the venom are modified.
The same may be said with regard to chromic acid (1 per cent. 261 solution), recommended by Kaufmann105 for the bite of the common viper.
No other effect is produced by a 1 per cent. solution of chloride of gold, or the alkaline hypochlorites, which I have shown to possess a strong oxidising action on the different venoms, even on those that are most rapidly diffusible, such as cobra-venom (see Chapter V.). They possess, however, owing to their slight causticity, the advantage of not producing severe local disorders, and in this respect they are to be preferred.
The chemical reagent most to be recommended is hypochlorite of lime, in a fresh solution of 2 grammes per cent., and containing about 90 c.c. of chlorine per 100 grammes. It immediately and surely destroys the venom by simple contact, and the chlorine gas that it gives off, owing to its great diffusibility, acts at a fairly long distance from the point of inoculation on the venom which is already beginning to be absorbed.
Professor Halford, of Melbourne, advises the direct injection into the patient’s veins of from 10 to 20 drops of ammonia, diluted with an equal quantity of distilled water. This is a means of reviving nervous excitability in certain subjects at the commencement of intoxication; but torpor soon reappears, and, if the dose of venom inoculated is sufficient to cause death, a fatal ending takes place notwithstanding. Experimentally the effects of ammonia are nil.
No better results are obtained by injections of strychnine, as recommended by Dr. Mueller, in Australia. Moreover, the statistics published by Raston Huxtable106 positively condemn this therapeutic method. They show that, in 426 cases of snake-bite, out of 113 treated by strychnine 15 proved fatal, the ratio of mortality being 13·2 per cent., while the 313 cases not treated by strychnine only resulted in 13 deaths, or a mortality of 4·1 per cent. 262
In the case of animals intoxicated by venom, injections of strychnine, morphia, nicotine, or curare in small doses always prove ineffective; they even considerably assist the progress of the intoxication and hasten death. The use of these drugs in the case of human beings should therefore be absolutely forbidden.
It appears, on the other hand, that alcohol and coffee, or tea, absorbed by ingestion, are very often beneficial. Indeed, it was long ago observed that the swallowing of alcohol until symptoms of drunkenness appear retards or diminishes the phenomena of torpor and paresis that precede the ultimate phase of the intoxication. Its use may therefore be recommended when it is impossible to have recourse to the only treatment really specific that modern science places in our hands—antivenomous serum-therapy. It is important, however, to state that, when serum is used, alcohol must be forbidden. The latter hinders the effects of the former.
In practice, the rational treatment of the bite of a venomous snake must be directed towards:—
(1) Preventing the absorption of the venom.
(2) Neutralising, by the injection of a sufficient quantity of antitoxic serum, the effects of the venom already absorbed.
In order to prevent the absorption of the venom introduced into the wound, the first precaution to be taken is to compress the bitten limb by means of a ligature of some kind, such as a handkerchief, as close as possible to the bite, and between it and the base of the limb. The ligature must be tightly twisted, and, by compressing the tissues around the bite, an attempt should be made to squeeze out the venom that may have been introduced into them. The expulsion of the poison should be hastened, either by making an incision 2 or 3 cm. in length and 1 cm. in depth in the direction taken by the fangs of the reptile and also parallel to the axis of the bitten member, or by sucking the wound hard.
The ligature on the limb should not be applied for more than half an hour; if it were kept on longer it would interfere with the circulation to a dangerous degree, and would certainly injure the 263 vitality of the tissues. The period in question also usually affords sufficient time for taking the patient to a place where help can be obtained, and for the preparation of everything necessary for his subsequent treatment.
The wound should then be freely washed with a fresh 2 per cent. solution of hypochlorite of lime, or with a 1 in 1,000 solution of chloride of gold. In default of hypochlorite of lime or chloride of gold, either eau de Javel, diluted with tepid water to a strength of 1 in 10, or a 1 per cent. solution of permanganate of potash, may be employed. These reagents should be made to penetrate as deeply as possible into the tissues, and a few cubic centimetres of them should even be injected with a Pravaz syringe into the punctures caused by the bite and all round them.
The wound being then covered with a damp dressing by means of compresses saturated with hypochlorite of lime, or at least with pure alcohol, the next thing to be done is to prepare to apply the serum-therapic treatment in order to arrest the general intoxication, if this has already commenced to take effect, or to prevent it from setting in.
For the employment of serum it is necessary to be in possession of a sterilisable syringe of the capacity of 10 c.c., similar to those used in the treatment of diphtheria.
If the life of the patient be not immediately in danger, care should first be taken to have the syringe boiled, or at least to rinse it out with boiling water, making sure that the piston fits tightly, and that the syringe itself is in good working order.
Should a syringe of 10 c.c. not be available, any kind of Pravaz syringe, previously washed out with boiling water, may be employed, but in this case the use of so small an instrument renders it necessary to give several painful injections.
The entire contents of a bottle of serum (10 c.c. of liquid serum, or 1 gramme of dry serum dissolved in 10 c.c. of boiled water) should be injected into the subcutaneous areolar tissue of the abdomen, on the right or left side. There is no advantage in 264 making the injection at the actual spot bitten; the serum is best and most rapidly absorbed when injected into the loose tissues of the abdominal wall (fig. 95).

Fig. 95.—Technique of the Injection of Antivenomous Serum beneath the Skin of the Abdomen.
If it has been impossible to apply the treatment until several hours after the bite, and if the latter has been inflicted by a poisonous snake of large size or belonging to a very dangerous 265 species, such as the Cobra or Indian Krait, it is preferable to inject into the patient three whole doses of serum at once.
In cases in which the phenomena of serious intoxication have already appeared, and when asphyxia threatens, one must not hesitate to inject 10 or even 20 c.c. of serum directly into a vein. For such an injection it is most convenient to choose a superficial vein at the elbow or wrist, or on the back of the hand.
The introduction of serum into the veins is never dangerous if good care be taken not to allow either bubbles of air or particles of precipitated albumin to enter.
It is not advisable to repeat the injections beneath the skin or into the veins unless the general symptoms appear to become more acute.
In most cases the local pain, excitement, and attacks of cramp and nausea disappear within a few minutes after the first injection. Improvement progresses very rapidly, and by the following day the patient has recovered.
The administration of ammonia, alcohol, morphia, or ether by the mouth is entirely superfluous. These drugs, as I have already stated, may even be harmful to the patient and hinder the effects of the serum. All that should be done is to give copious hot drinks, tea or coffee, and to cover up the patient warmly in order to induce abundant perspiration.
The bitten member should not be cauterised with red hot iron or with chemical agents of any kind, since such cauterisations only lead to injuries which are too often prejudicial to the normal action of the affected organs.
Treatment of Poisonous Bites in the Case of Domestic Animals.—It often happens that dogs, horses, or cattle are bitten and succumb to the poisoning in a few hours or in two or three days. Such accidents are especially frequent among sporting dogs, even in Europe, in regions in which vipers are found.
In most cases, dogs, horses, and cattle are bitten on the nose, and such bites are immediately followed by a very painful swelling, 266 which arouses the suspicion of the owners of the animals. It is then necessary, as soon as possible, to inject subcutaneously in the right or left flank, or at the base of the neck, one or two doses of antivenomous serum, according to the gravity of the effects observed.
The injection of the serum and the dressing of the wound should be performed as in the case of poisonous bites in human beings.
Influence of the Doses of Antivenomous Serum injected, and of the Time that has elapsed since the Venomous Bite.—I have stated above that antivenomous serum possesses a preventive and curative power of such intensity, that it is capable in a few minutes of rendering animals into which it has been injected absolutely insensible to the most strongly neurotoxic venoms, such as those of Naja or Bungarus. On the other hand, I have established the fact that, the more sensitive are the animals to intoxication by venom, the greater is the quantity of antivenomous serum necessary to immunise them passively or to cure them.
In experimenting upon mice, guinea-pigs, and rabbits, it is found that in order to preserve, let us say, a mouse of 25 grammes against inoculation with half a milligramme of venom, which is ten times the lethal dose for this little animal, it is necessary to give a preventive injection of 1 c.c. of serum; while half a cubic centimetre of the same serum is sufficient to render the dose of half a milligramme of venom innocuous, when venom and serum are mixed in vitro before being injected.
In the case of the guinea-pig, it is likewise found that the dose of serum to be injected preventively, in order to protect the animal from intoxication by ten times the lethal dose of venom, is about twice as much as the quantity of the same serum that it is sufficient to mix in vitro with venom, in order to render ten times the lethal dose of venom innocuous.
If we inject into animals first venom, in doses calculated to kill the controls of the same weight in from two to three hours, and the serum fifteen minutes afterwards, it is found that the 267 quantity of serum that must be injected in order to prevent death is about thrice as great, as that which neutralises in vitro the dose of venom inoculated.
It is also found that the amount of curative serum that an animal intoxicated by venom must receive is inversely proportional to its weight.
The experiments upon dogs, performed at the Pasteur Institute at Lille by my collaborator C. Guérin, are highly demonstrative in this respect.107
A dog of 12 kilogrammes, inoculated with 9 milligrammes of venom (a dose lethal to controls of the same weight in from five to seven hours), is completely cured on receiving, two hours after inoculation with the poison, 10 c.c. of serum.
When the treatment does not take place until three hours after the injection of the venom, it is necessary to inject 20 cc. of serum in order to prevent the animal from dying. With a longer delay than this, death is inevitable, since the bulbar centres are already affected, and paralysis of the respiratory muscles commences to appear.
These facts show that:—
(1) The more sensitive animals are to venom, the greater is the quantity of serum necessary in order to prevent their intoxication by a given dose of venom.
(2) For a given species of animal and a given dose of venom, the longer the delay in applying the remedy, the greater is the quantity of serum that must be injected in order to arrest the poisoning.
It will be understood from what has been already stated, that a man weighing 60 kilogrammes, if bitten by a snake which injects, let us say, what would amount to 20 milligrammes of venom if collected in the dry state (the mean quantity that a 268 Naja is able to inoculate in a single bite), would only require, in order to escape death, to receive the quantity of antivenomous serum sufficient to neutralise the portion of venom in excess of the amount that he could tolerate without dying.
Let us suppose, for the sake of example, that the man of 60 kilogrammes can withstand intoxication by 14 milligrammes of Naja-venom. It follows that, in the case with which we are dealing, we must inject sufficient serum to neutralise 20-14 (=6) milligrammes of venom; that is to say, the injection of serum being made immediately after the bite, 6 c.c., if the serum employed neutralises in vitro 1 milligramme of venom per cubic centimetre.
Of course, if the serum is more powerful, less of it will be necessary, while more will be required if the remedy is applied later, or if the quantity of venom inoculated by the snake is supposed to have been greater.
For this reason, in practice, but very little serum is usually necessary in order to augment the natural resistance of a man of average weight or of a large animal; it is sufficient in most cases to give an injection of 10 or 20 c.c. in order to cure human beings who have been bitten. The clinical proof of this is, moreover, to be found in the cases, already very numerous, that have been published in the course of the last few years in the scientific journals of all countries. I have gathered together a few of these in the concluding pages of this book, and I would beg the reader to be good enough to refer to them. 269
VENOMS IN THE ANIMAL SERIES.
Besides reptiles, many other animals possess poison-glands and inoculatory organs which they employ, either to defend themselves against their natural enemies, or to capture the living prey upon which they feed.
The venoms that they produce are still, for the most part, but little understood. A few of them, however, have excited the curiosity of physiologists, especially those secreted by certain batrachians, such as the Toad, and certain fishes, such as the Weever. Some of them exhibit close affinity to snake-venom, and are composed, like the latter, of proteic substances modifiable by heat and precipitable by alcohol; others possess altogether special characters, and resemble alkaloids.
The lowest animal group in which these secretions begin to be clearly differentiated is that of the Coelenterates.
It has been shown by Charles Richet108 that the tentacles of sea-anemones (Anemone scultata) contain a toxic substance which has 270 the carious property of causing intense itching, pruritus, and even urticaria. This poison is perfectly soluble in alcohol, and can be prepared in the following manner:—
The tentacles are cut off close to the body of the animal, and immersed for a few days in an equal weight of alcohol at 95° C. The red liquid that results is decanted, and then filtered. The insoluble material is compressed, and yields large quantities of fluid, which is filtered and mixed with the previous liquid.
The whole is then evaporated in vacuo until there remains a thick oily liquid, which forms a red deposit. Filtration through paper is again employed, in order to separate this colouring matter, and to the filtered liquid is added an equal amount of alcohol at 95° C. By this means there is precipitated a blackish, gummy matter, insoluble in alcohol. The remaining liquid is decanted and once more evaporated until it is reduced to a smaller volume than before. It is again treated with twice its volume of absolute alcohol, when it precipitates, in addition to salts and gummy matter, a white flocculent substance, which is crude thalassin. This can be purified by redissolving it in alcohol at a temperature of 98° C. On cooling it separates from the fluid in the form of crystals, which are placed on a filter and can then be redissolved in a small quantity of water. Absolute alcohol, added to this solution, precipitates the thalassin in the shape of very pure crystals, which contain 10 per cent. of azote, and melt at 200° C.
This substance, in aqueous solutions, rapidly deteriorates owing to ammoniacal fermentation. When injected intravenously into dogs it produces pruritus, sneezing, and erythema, with intense congestion of the mucous membranes; 1 decigramme per kilogramme is a dose sufficient to produce these symptoms. It is not very toxic, since 1 centigramme is not lethal.
One kilogramme of anemones is capable of furnishing about 3 grammes of pure crystallised poison.
In addition to thalassin, Richet succeeded in isolating from the tentacles of the same sea-anemones another poison insoluble in 271 alcohol at 50° C., and richer in azote (14 per cent.), to which he has given the name congestin. This is not destroyed by heating to 107° C. It is prepared by precipitating, by four times its volume of alcohol, a solution of anemone-tentacles in 5 per cent. fluoride of sodium. The solid matter, after being precipitated and dried, is redissolved in six times its volume of water, and then filtered. On adding to the filtered and fluorescent liquid its volume of alcohol at 90° C., the congestin is precipitated. It is purified by redissolving it in water, and freeing it by dialysis from the fluoride of sodium that it has retained. In this way there is obtained, after evaporation, a product sufficiently toxic to kill dogs in twenty-four hours in a dose of 2 milligrammes per kilogramme.
Congestin exerts a sensitising or anaphylactic effect upon animals as regards thalassin, and is lethal in a dose of about 5 milligrammes per kilogramme of animal, and sometimes even in a dose of 7 decimilligrammes. It is therefore a very active poison.
Dogs, on the other hand, into which is injected first thalassin, and then, some time afterwards, congestin, are perfectly resistant to inoculation by the latter. Thalassin is therefore antitoxic or antagonistic to congestin.
The latter, on the contrary, if injected first of all in non-lethal doses, renders animals so sensitive to inoculation with thalassin, that from 4 to 5 milligrammes are sufficient to cause death.
The tentacles of these anemones therefore contain two toxic substances antagonistic to each other, which can easily be separated, since one (thalassin) is soluble in concentrated alcohol, while the other is completely insoluble in this reagent.
These poisons are not only extremely interesting from a physiological point of view, but also possess a practical interest, since it is at the present time almost a matter of certainty that they are the cause of a malady which specially affects sponge-divers in the Mediterranean. 272
A good description of the disease has been given by Dr. Skévos Zervos, of Athens.109 It is observed exclusively in men who dive quite naked, without a diving-dress. Now, beside the bases of the sponges and sometimes on their surface there live numbers of anemones which secrete a viscid substance, which is extremely virulent, especially in the month of August.
The first symptoms that supervene after contact with these Cœlenterates are an intense itching and burning sensation; a papule of a horny consistency appears at the outset at the spot at which contact took place; this is soon surrounded by a red zone, which becomes bluish and then black, and spreads to a greater or lesser extent, according to the region attacked and the virulence of the venom. After a few days the skin sloughs and leaves a deep ulcer, which suppurates in spite of antiseptic treatment. The onset of the disease is marked by a febrile attack with shivering, which is soon accompanied by cephalalgia, thirst, and pains in the back and limbs.
Zervos reproduced these disorders experimentally by rubbing an anemone, held with forceps, on the shaven abdomen of a dog. In a few minutes the region affected became quite red and pruriginous; twenty-five minutes later phlyctenæ full of serum appeared; three days afterwards five abscesses of different sizes developed, while at the place where it had been touched by the venom the skin assumed a deep blue colour; on the fifth day an area 2 cm. in diameter was completely gangrenous.
When ingested, anemones possess toxic properties which are well known to the sponge-fishers, for they frequently make use of them for the purpose of poisoning domestic animals. With this object they cut them up into small fragments, and mix them with bread or meat, which is given to the animals to eat; the latter die in convulsions in a few minutes.
In order to preserve the divers from the harmful effects produced 273 by contact with the anemones, they should be advised to cover their bodies with a layer of grease, a simple artifice which constitutes an efficient protection.
The Echinoidea (Sea-urchins) are provided with soft prehensile organs, the pedicellariæ, of which four kinds are distinguished: gemmiform, tridactyle, trifoliate, and ophiocephalous.
These pedicellariæ contain a special venom, which causes the paralysis and death of animals into which it is injected. Uexkull, who was the first to mention it, considered that the gemmiform pedicellariæ alone are toxic.
From this point of view various species of sea-urchins, Strongylocentrotus lividus, Arbacia æquituberculata, Sphærechinus granularis and Spatangus purpureus, have recently been studied by V. Henri and Mdlle. Kayalof.110
The pedicellariæ were removed and pounded up in sea-water, and the pulp was injected into crabs, holothurians, star-fish, cuttle-fish, frogs, lizards, and rabbits; in the case of cuttle-fish and rabbits the injection was made intravenously; in that of the other animals into the body-cavity.
For crabs the lethal dose was from 20 to 30 gemmiform pedicellariæ of Strongylocentrotus lividus.
The holothurians, star-fish, and frogs proved immune.
In the case of rabbits weighing 1½ kilogrammes, 40 pedicellariæ of Sphærechinus granularis, pounded up in 1 c.c. of water, produce death by asphyxia and general paralysis in from two to three minutes. The heart continues to beat after respiration has ceased.
For lizards and fishes the toxic dose is the same as for the crab. The cuttle-fish is paralysed and killed in two hours by 50 pedicellariæ. 274
This venom resists ebullition for fifteen minutes.
V. Henri and Mdlle. Kayalof made experiments in immunisation. Rabbits that receive every third day increasing doses of gemmiform pedicellariæ of Sphærechinus granularis tolerate well, after four injections, the toxin of 40 pedicellariæ, a lethal dose. The serum of these rabbits is not protective for either rabbit, crab, or fishes.
Frog serum (1 c.c.) injected into the body cavity of a crab, protects this animal against the pulp of pedicellariæ injected immediately afterwards.
The pedicellariæ easily become detached from sea-urchins. They remain fixed to objects which come into contact with them, and the urchin abandons them like poisoned arrows.
On touching a point on the surface of the body of an urchin, the spines are seen to incline towards the spot touched, and the pedicellariæ stretch themselves out and lean with their valves open towards the seat of the stimulus. In Sphærechinus granularis the heads of the gemmiform pedicellariæ are covered with sticky mucus forming a tiny drop, visible under the lens. A specimen of this species possesses more than 450 pedicellariæ.
Almost all Arachnids possess poison-glands, which are connected, in some cases with the buccal apparatus, in others with a special inoculatory organ situated at the posterior extremity of the body. The spiders and scorpions belong to this group, and their venom is particularly active.
On each side of the mouth of spiders is found an appendage ending in a fang (chelicera), at the extremity of which opens the excretory duct of a more or less developed poison-gland. The venom produced by these glands is instantly fatal to all small 275 animals upon which spiders feed. In man and large mammals their bite produces sensations of pain accompanied by swelling and muscular contractions as though caused by localised tetanus.
The venom of certain species of spiders sometimes causes very serious and even fatal results. Latrodectus malmignattus (the malmignatte of the South of France and Italy), and especially Latrodectus mactans, of Chile (fig. 96), are greatly dreaded.111 The area of distribution of the latter includes the whole of Tropical and Sub-tropical America. It is said that it frequently causes the death of milch cows, and that in man its bite produces tetanic effects, which last for several days, but are in most cases amenable to treatment.

Fig. 96.—Lactrodectus
mactans (formidabilis olim).
1, Female, twice natural size;
1a, its eyes, greatly enlarged.
Another dangerous spider is the Katipo (Latrodectus scelio), of New Zealand. This creature is confined to the sea-shore, and the natives are often bitten when collecting shell-fish or sea-weed. The Maoris are so much afraid of the bite of the Katipo that, when one of them has been bitten in his hut, and the animal cannot be found, they do not hesitate to burn the dwelling to the ground. Moreover, they are convinced that the death of the spider is absolutely necessary for the recovery of the patient.112 276
Kobert113 has made an experimental study of the venom of species of Latrodectus and Epeira. That of Latrodectus erebus (the Karakurte of South Russia) is particularly toxic.
If a dry extract of these spiders be prepared and injected into the veins of dogs or cats, it is found that a few milligrammes per kilogramme are sufficient to cause death, with phenomena of dyspnœa, convulsions, and progressive paralysis of respiration and the heart. Rabbits, rats, birds, frogs, and leeches are also sensitive to this poison, though the hedgehog is almost refractory. The young spiders, and even the eggs, are more toxic than the adults.
Spider-venom is destroyed by heating for forty minutes at + 70° C., and is precipitated by alcohol. When absorbed by ingestion it has no effect: it is hæmolytic and coagulates blood.
The study of arachnolysin by Ehrlich’s methods has been undertaken afresh by Hans Sachs,114 who has shown that rat’s and rabbit’s blood are most rapidly dissolved. Twenty-eight milligrammes of extract of Epeira are capable of completely dissolving 0·05 c.c. of blood.
By immunising guinea-pigs and rabbits, Sachs succeeded in obtaining a strongly antitoxic serum, which entirely prevents the hæmolysis of the sensitive red corpuscles.
The poison-apparatus of the scorpion is constituted by the last abdominal segment (telson), which is swollen and globular, and terminated by a hard, curved spine, with a very sharp point, near which can be distinguished, under the lens, two small oval orifices by which the poison is enabled to escape (fig. 97).
The poison-glands are two in number, symmetrically placed in 277 cavities, each of which is completely filled by the gland. They are separated from each other by a muscular septum formed of striated fibres inserted in the chitinous skeleton; by the contraction of this septum the animal is enabled to eject its venom.

Fig. 97.—Scorpio occitanus. (After Joyeux-Laffuie.)
1, Scorpion seizing a spider, and piercing it with its sting (natural size); 2, extremity of the abdomen (telson) enlarged, showing the poison-apparatus; 3, poison-apparatus detached from the abdomen, showing an isolated poison-gland.
The scorpion never stings backwards, but always in front of itself. It delivers stabs with its sting in two distinct ways. For the purpose of defending itself from attack it elevates its abdomen into a bow, and then regains its former position by suddenly unbending it. To strike an animal, such as a spider, which serves it for food, the scorpion seizes it with its pincers and holds it as in a vice. Then it raises its abdomen, brings the end of it close to its captive, 278 and, with a lever-like movement, drives the sting into its body. The victim immediately becomes paralysed and motionless.115
The poison-glands of a Scorpio occitanus from the South of France contain about 1 to 10 centigrammes of a toxic liquid, capable of furnishing 10 to 15 per cent. of dry extract. This liquid is decidedly acid; it reddens litmus paper and is miscible with water.
Its physiological effects are especially intense in the case of the arthropods upon which the scorpion habitually feeds, and in that of vertebrates in general. Batrachians, fishes, birds, and mammals are extremely susceptible to this poison. Half a milligramme of dry extract injected subcutaneously is sufficient to kill a guinea-pig, and 1 milligramme is lethal to the rabbit.
In poisoned animals there is first observed a period of violent excitement, accompanied by very acute pains; these are followed by muscular contractions, and finally by paralysis of the respiratory muscles, as in the case of intoxication by cobra-venom.
The effects of scorpion-poison, which clearly indicate the presence of a neurotoxin, have been very well described by Valentin,116 Paul Bert117 and Joyeux-Laffuie. Kyes118 has prepared a lecithide from scorpion-venom, which hæmolyses red corpuscles as do the lecithides of cobra-venom, and I myself119 have established the fact that the antivenomous serum of a horse vaccinated against cobra-venom effectively protects mice and guinea-pigs against intoxication by the venom of Scorpio occitanus; this has been verified by Metchnikoff. There is, therefore, a close affinity between this venom and that of Colubrine snakes. 279
On the other hand, it has been shown by the investigations of C. Nicolle and G. Catouillard that the same antivenomous serum has no effect upon the much weaker venom of the scorpion of Tunis (Heterometrus maurus), which, in the case of man and mammals in general, scarcely does more than produce a transient œdema at the point of inoculation.
The venom of Heterometrus maurus is, however, toxic enough to the sparrow. When one of these little birds is inoculated in the pectoral muscles with the contents of the poison-glands of a single scorpion belonging to this species, the following symptoms are observed: Immediate rigidity, doubtless connected with the pain, then, after a few seconds, depression and relaxation of the muscles. The bird remains upright, but its body sinks down more and more until it comes into contact with the ground; if on a perch, it soon becomes unsteady and drops off. There is dyspnœa, which any effort increases, and death supervenes suddenly; all at once the sparrow falls on its side, stiffens, sometimes has a few convulsions, and then finally becomes still. The time occupied by these phenomena is always short, although it varies from two minutes to half an hour.
Scorpion-venom is a strong irritant to the mucous membranes. When dropped into the eye of a rabbit it produces acute ophthalmia.
It has often been asserted that scorpions kill themselves with their own venom if enclosed in a circle of fire. This is an absolute myth, for it is easy to prove by experiment, as was done by Bourne at Madras,120 that these animals cannot be intoxicated by their own poisonous secretion, nor by that of other individuals of the same species. Moreover, it has been established by Metchnikoff,121 in very definite fashion, that the blood of the scorpion is antitoxic. If 0·1 c.c. of this blood be added to a dose of venom lethal to mice in half an hour, a mouse injected with this mixture resists indefinitely. This antitoxic power is exhibited both by Scorpio afer and the Algerian Androctonus. 280
It has been shown by Phisalix and Bertrand that certain species of Myriopods, including those of the genus Julus (Order Chilognatha, e.g., Julus terrestris), secrete throughout the entire extent of their body a volatile venom, which these authors compared to quinone.
The species of the genus Scolopendra (Order Chilopoda; Scolopendra cingulata, found in the South of France, Spain and Italy; S. gigantea and other forms, common in Africa, India, Indo-China and Equatorial America), have the second pair of post maxillary appendages transformed into formidable poison-claws, with which they can inflict bites which are very painful to human beings.

Fig. 98.—Scolopendra morsitans (S. Europe).
(After Claus.)
The tropical species may attain a length of 10 or even 15 cm. Their bodies are composed of 21 segments, each provided with a pair of jointed legs. They live in shady places, such as woods, hidden under stones, dead leaves, or the bark of old trees. They feed upon small insects, spiders, and larvæ, which they kill with their venom. The latter is secreted by a racemose gland situated at the base of the poison-claws; it escapes by a duct which opens at the apex.
This venom, the physiological study of which was commenced by Dubosq, is an acid, opalescent liquid, hardly miscible with water.
More complete experiments on this subject have been made by A. Briot,122 who prepared a solution by sectioning the labium and poison-claws, and crushing the whole in physiological salt solution. When injected intravenously into rabbits, it produces immediate 281 paralysis, with coagulation of the blood; subcutaneously it leads to the formation of enormous abscesses, with necrosis of the tissues. Small animals, such as spiders, species of Scutigera, beetles, &c., are very sensitive to it.
The bite of Scolopendridæ is very painful to human beings. In the Tropics such bites often cause somewhat serious results: insomnia, accelerated and intermittent pulse, and local œdema, which usually disappears after twenty-four hours. Well-authenticated fatal accidents have never been recorded (Bachelier,123 Saulie124).
A very large number of insects produce acrid or irritant secretions, which serve them as a means of defence, but cannot be considered as true venoms; the species of Meloë (oil-beetles) and Cantharis (blister-beetles), are the most remarkable in this respect.
The Order Hymenoptera is the only one that includes a multitude of species really provided with poison-glands and an inoculatory apparatus.

Fig. 99.—Poison-apparatus of the Bee.
gl.ac, Acid gland and its two
branches; V, poison-sac; gl.al,
alkaline gland; gor, gorget.
(After Carlet: figure borrowed
from Hommel.)
The poison-organs, which have been well studied, especially by Leuckart,125 Leydig,126 Carlet,127 and more especially by L. Bordas,128 282 Janet,129 and Seurat,130 always include two and sometimes three kinds of glands: the acid gland, the alkaline gland or gland of Dufour, and the accessory poison-gland (fig. 99).
The acid gland comprises a glandular portion (which sometimes takes the shape of a long flexuous tube, always bifid at its extremity, sometimes that of two tubes, simple or ramified, or again is composed of a bundle of cylindrical, simple or multifid canals), a poison-sac or reservoir, ovoid or spherical in shape, and an excretory duct, which is usually short.
The alkaline gland, or gland of Dufour, exists in all Hymenoptera, and presents the appearance of an irregular tube, with a striated surface and a spherical or conical upper extremity. Its excretory duct opens, beside that of the acid gland, at the enlarged base of the gorget of the sting (fig. 100).

Fig. 100.—Interior of the Gorget of the Bee, seen from its Posterior Aspect.
cv, Poison chamber; gor, gorget; st, stylet; ca, piston. Between the two stylets is seen the cleft fa, by which the air is able to enter into the air-chamber cai.
(After Carlet: figure borrowed from Hommel.)
The accessory poison-gland, which is lanceolate or ovoid in shape, consists of a small, granular mass, the extremely narrow excretory duct of which opens at almost the same point as that of the alkaline gland. It does not exist in all Hymenoptera.
The stings of hive bees (Apis mellifica), wasps (Vespa vulgaris), violet carpenter bees (Xylocopa violacea), and humble bees (Bombus lapidarius) cause considerable discomfort. The venom of the carpenter bee, which is of some strength, has been studied by P. Bert, and I have myself made experiments with that of the hive bee (A. mellifica). The venom extracted 283 from a couple of bees, by crushing the posterior extremity of the body in 1 c.c. of water, is sufficient to kill a mouse or a sparrow.
Death supervenes in a few minutes, from respiratory asphyxia, as in the case of intoxication by the venom of Colubrine snakes (Cobra). In the blood-vessels and in the heart the blood is black and remains fluid. It therefore appears that this venom contains a very active neurotoxin.
The phenomena of intoxication caused by the venom of these insects are, as a rule, slight, being limited to an acute pain, accompanied by a zone of œdema and burning itching. Sometimes however, when the stings are in the eyelids, lips, or tongue, they produce alarming and even fatal results, as shown by the following incident:—
On September 26, 1890, a young girl of Ville-d’Avray was eating grapes in the woods of Fausse-Repose, when she inadvertently swallowed a wasp. The unfortunate girl was stung in the back of the throat, and the wound became so rapidly inflamed that, in spite of the attentions of a doctor, she died in an hour from suffocation, in the arms of her friends.
Phisalix131 has studied the physiological action of bee-venom on sparrows inoculated either by the sting of the insect, or with an aqueous solution obtained by crushing the glands. In both cases a local effect, paralysis of the part inoculated, is first produced; this is followed by convulsions, which may last for several hours; the final stage is marked by coma and respiratory trouble, which ends in death.
After being heated for fifteen minutes at 100° C. the venom has no further local action; the general phenomena are merely diminished. If heated at 100° C. for thirty minutes, the venom ceases to cause convulsions, but remains stupefactive. Exposure for fifteen minutes to a temperature of 150° C. renders it completely inert. 284
This venom therefore comprises: (1) A phlogogenic substance, destroyed by ebullition, contained in the acid gland of the bee; (2) a poison causing convulsions, which does not resist a temperature of 100° C., if prolonged, and is probably produced by the alkaline gland; (3) a stupefactive poison, which is secreted by the acid gland, and is not entirely destroyed until a temperature of 150° C. is reached.
The poison-glands can easily be extracted by gently pulling at the stings of bees anæsthetised by chloroform.
The eggs of bees, like those of the toad and the viper, contain the specific venom. The amount, however, is small, since in order to produce lethal results in the sparrow it was found necessary to inoculate an emulsion obtained by crushing 926 eggs.
Phisalix132 makes the approximate calculation that, in the egg the weight of the toxic substances amounts to the one hundred and fiftieth part of the whole. Their effects are similar to those produced by the venom itself, but the convulsions are not so severe. The predominant poison in the egg appears to be that causing paralysis.
I have easily succeeded in vaccinating mice against doses of bee-venom certainly lethal, by repeatedly inoculating them with very small doses. Moreover, we find the same thing in the case of human beings, for we know that those who are in the habit of handling hives become quite accustomed to bee-stings, and finally feel not the slightest effect from them.
It has been shown by J. Morgenroth and U. Carpi,133 in a paper recently published, that the venom of bees, like that of the scorpion, possesses the property of hæmolysing the red corpuscles of several species of animals (the rabbit, guinea-pig, and goat), and that it is capable of combining with the lecithin to form a lecithide analogous to cobra-lecithide, the curious properties of which we have studied in detail. 285
This lecithide of bee-venom is from 200 to 500 times more hæmolysing than the venom itself, and resists ebullition like that of the cobra. In order to isolate it Morgenroth and Carpi employed the method recommended by P. Kyes: 1½ c.c. of a solution of pure venom is mixed with 1½ c.c. of a 5 per cent. solution of lecithin in methylic alcohol. After being kept for twenty-four hours at 37° C., 22 c.c. of absolute alcohol are added; the liquid is decanted, and the clear filtrate is mixed with 150 c.c. of ether. There is slowly formed a somewhat copious flocculent deposit, which is collected on a filter, washed several times with ether, and finally dried. The lecithide that remains on the filter dissolves completely in physiological salt solution.
It must be remarked that bee-venom, without the addition of lecithin, gives a scanty precipitate with ether. This precipitate, dissolved in physiological salt solution, possesses no hæmolysing power. The lecithide, on the contrary, dissolves red corpuscles almost instantaneously.
Normal horse-serum considerably inhibits hæmolysis by bee-venom + lecithin. This protective action of normal serums has already been observed by Langer; it is perhaps attributable to the cholesterin that they contain.
Among other Hymenoptera capable of inflicting very severe stings may be mentioned the species of Polistes and certain Pompilids, especially a species of Pompilus found in Natal, the painful stings of which have sometimes been experienced and described by travellers (P. Fabre, of Commentry).134
In the family Crabronidæ the females are provided with a sting and venom, which usually has little effect upon man, but is toxic to other insects. Thus, Cerceris bupresticida is remarkable for the stupefying effect of its venom upon the Buprestidæ destined for the food of its larvæ. It stings the beetles between the first and second segments of the thorax, with the result that the victim 286 is paralysed, though in other respects its bodily functions appear to continue; in fact, its intestine is seen to empty itself at long intervals. These effects are attributed by Mons. J. H. Fabre, of Avignon, to the direct action of the venom upon the ganglia of the thoracic nervous system.
Instances of Hymenoptera belonging to the tribe Entomophaga actually depositing their eggs beneath the skin of man are mentioned by Raphaël Blanchard.135
According to P. Fabre, the best treatment for wasp- or bee-stings would appear to consist in the application of strong saline solution, or a liniment of ammonia and olive oil. For my own part, I have tried hypochlorite of lime, in a 1 in 60 solution, or eau de Javel diluted to 1 per cent., and have always obtained such excellent results from these remedies that I do not hesitate to advise their use.
Certain Gastropodous Molluscs, chiefly Murex brandaris and M. trunculus, possess purple glands from which it is possible to extract a very active venom (Raphaël Dubois)136 by crushing them up with sand and alcohol. The alcoholic liquid, filtered and evaporated in a water-bath, yields a brown oily fluid. The subcutaneous injection of a few drops of this into a frog is sufficient to produce very decided toxic effects. Sluggishness and slowness of movement are seen to supervene fairly quickly; reflex actions are still exhibited, but the animal is no longer able to jump.
If the dose be not too strong, this condition of paresis lasts for several hours, and then disappears. In most cases, however, the paresis is succeeded by complete paralysis, and the animal appears as though suffering from curare. Yet the fact is that the venom is neither curare-like nor cardiac; the heart, muscles, motor endplates, 287 and motor and sensory nerves are spared; the nervous centres alone are attacked, especially the encephalon. The animal dies without convulsions.
Sea and fresh-water fishes (golden carp) are very sensitive to this venom; warm-blooded animals are refractory. It is therefore probable that, in the species of Murex, the purple gland is a poison-gland serving for defence, or for the capture of the prey upon which these molluscs feed.
Among the Cephalopods, the Octopods (Octopus vulgaris, common octopus, Eledone moschata, musky octopus, of the Mediterranean) possess two pairs of salivary glands, a small anterior pair, and a posterior pair of considerable size.
The Decapods (cuttle-fishes [Sepia], &c.), have only posterior salivary glands, of smaller dimensions in proportion to the size of the body.
On being crushed and macerated in water, the anterior glands yield a limpid and slightly acid juice; the posterior glands produce a viscid, ropy fluid, filterable with difficulty and neutral. The latter has an immediate paralysing effect upon Crustacea. It contains a substance of a diastasic nature, precipitable by alcohol, and destructible by heating for an hour at 58° C.
Owing to the poisonous properties of this juice, Octopods succeed in overpowering large prey, such as lobsters and crabs. Once they are seized by the tentacles of the octopus, or cuttle-fish, a bite inoculates these animals with venom that immediately destroys their power of movement, and the Cephalopod is able to continue its meal in perfect security, without having to fear the pincers of its prey.
An experimental study of this venom has been made by A. Briot,137 who found that crabs are very sensitive to it, while rats, frogs, rabbits, and fish do not appear to experience any inconvenience. 288
The means of defence in fishes are extremely varied. Some species (torpedoes or electric rays, electric eels) destroy their enemies by electric discharges; others are provided with true poison-glands and inoculatory organs, usually represented by opercular spines or by the fin-rays. The species of the genus Muræna, however, possess a poison-apparatus connected with the buccal teeth, as in the case of snakes.
It has been clearly established by Bottard138 that at least three very distinct types of venomous fishes exist, according as the venom-apparatus is:—
(1) Entirely closed (Synanceia type); (2) half closed (Thalassophryne type); (3) in more or less direct communication with the exterior (Trachinus and Scorpæna type).
The greater part of the following statements has been borrowed from the excellent work of the author referred to, from the writings of A. Corre,139 the fellowship thesis of Henry Coutière,140 and the magnificent atlas published at St. Petersburg in 1886 by P. Savtschenko, of the Russian Imperial Navy.
Except in the case of the species of Muræna, the venom of fishes 289 is generally found in one or more special glands, situate at the base of the dorsal or caudal fins, or beneath the opercular spines. When the animal defends itself it inflicts wounds with these rays, and ejects from its poison-glands a toxic or irritant liquid, which enters the sores.
The flesh of these fishes is not usually poisonous, whereas a fairly large number of other species, which do not inflict wounds, cause intoxicating effects when eaten. These latter do not come within the scope of this work; but the reader who may desire to obtain information with regard to them will find them well described in J. Pellegrin’s memoir,141 in that by Dupont, and especially in the papers of A. Corre.
Venomous fishes almost all belong to sedentary species, as in the case of the genera Trachinus, Cottus, Scorpæna, and Synanceia. This fact suggested to Dissard and Noë142 a very hazardous theory in order to explain the existence of a poison-apparatus in these animals. The venomous fishes being sedentary, say these authors, have no need of a poison-apparatus; their prey offers itself to them without effort on their part, and, on the other hand, they escape destruction by their enemies. If, therefore, they possess a poison-apparatus it is because the conditions under which they live entail the lowest value for the co-efficient of respiration, diminish the quantity of the ambient radiations and the oxygenation of the medium, and lead to diminished hæmatosis. For these reasons the activity of anaerobic life becomes greater, and the formation of venoms takes place.
This theory, derived from the conceptions of A. Gautier with regard to the formation of toxic leucomaines, appears scarcely tenable, for it is evident that the weever, for example, erects its first dorsal spine as soon as it is seized, and that Scorpæna and Synanceia likewise 290 protrude their venomous spines when conscious of danger. The poison-apparatus of these fishes is therefore of an eminently defensive character.
According to Bottard, the spawning season increases the activity of the poison-glands and at the same time the toxicity of the secreted product. Several species, such as those of the genus Cottus and the perch, possess no apparent secreting cells except at this period. Certain toxicophorous or poisonous fishes, such as the species of Tetrodon, are particularly noxious at the time when their genital glands are at their maximum activity.
The fishes of this family are all repulsively ugly. They have an elongate and but slightly compressed body, covered with ctenoid scales, and a large head in which the suborbital bones, which are broad, unite with the præopercular so as to form an osseous plate in the malar region. The pectoral fins are large, and provided with a few detached rays, which perform the function of tactile organs; the ventral fins are situate on the breast. These fishes are extremely voracious.
The most interesting type is the Synanceia termed by the Creoles of Réunion Crapaud de mer, and by those of Mauritius Laffe. In Java it is called Ikan-Satan (Devil-fish), and in Tahiti Nohu. It is distributed throughout almost all the warmer regions of the Indian and Pacific Oceans, and is found in Cochin-China and New Caledonia.
It is never taken in the open sea, but only among the fringing reefs, where it lives constantly concealed in holes or buried in the sand. It does not come out except to make a sudden dart at prey passing within its reach. When irritated it does not eject venom; for the latter to be expelled one has either to press hard upon the poison-sacs, after pushing back with the fingers the membranes covering the dorsal defensive armature, or the naked foot must be 291 placed on the back of the fish. The wound is very painful, and is accompanied by a series of alarming symptoms, which sometimes terminate fatally: fishermen are consequently much afraid of it.
There are a large number of species of this fish, peculiar to different regions. Synanceia brachio (fig. 101), the largest specimens of which attain the length of 45 cm., is the most common form in the Tropical Pacific.

Fig. 101.—Synanceia brachio, var. verrucosa. (After Savtschenko.)
The spiny rays of the dorsal fin of Synanceia are sharp-pointed, stout in the middle, and provided on each side with a small canal hollowed out in the thickness of the spine. Towards the middle of the latter there is attached a little double sac, or kind of closed pouch, which, on being compressed, allows the venom to escape in a thin jet which flows into the grooves of the spine. The expulsion of the venom is therefore not a voluntary act on the part of the fish; in order that it shall take place, pressure must be applied to the sacs in which it is contained.
This venom, when extracted from the glands, is limpid, bluish, and slightly acid. When introduced into the tissues, it produces very acute local pain, which extends throughout the affected limb. The 292 pain is excruciating, and sufferers have been observed to become actually delirious, striking and biting those around them, throwing themselves from side to side, and beseeching that the limb should be cut off; some of them have amputated the injured part themselves.
This condition is accompanied by considerable anxiety, and by attacks of leipothymia and sometimes of syncope. In some cases syncope has been followed by death; in others serious phlegmons, complicated by septicæmia, supervene. The inoculated spot becomes bluish, and then sphacelates over a larger or smaller area. These gangrenous wounds heal very slowly, more especially since they are usually produced on the sole of the foot (Bottard).
A single drop of the venom is sufficient to kill frogs in about three hours.

Fig. 102.—Cottus scorpius (Sea Scorpion). (After Savtschenko.)
The genus Cottus, which also belongs to the family Triglidæ, includes some forty venomous species found in the seas of the northern hemisphere, in Europe, Asia, and America.
In France the species of Cottus are generally called chabots (bullheads or miller’s thumbs), chaboisseaux (sea-scorpions), or caramassons. They are abundant on the coast of Normandy, and some of them (river bullheads) live in fresh water; they do not exceed 25 cm. in length. They have a liking for holes 293 in rocks, and fishermen are afraid of being stung by them (fig. 102).
Their poison-apparatus resembles that of the Weevers, but is less developed. It is situated in the culs-de-sac formed by the opercular spines. The culs-de-sac are lined with cells which produce a toxic secretion only during the spawning season, from November to the end of January. This fact explains how it is that the species of Cottus are declared by certain fishermen to be very venomous, while others say that they are absolutely harmless.
The genera Scorpæna, Pterois and Pelor also belong to the same group.

Fig. 103.—Scorpæna grandicornis (Caribbean Sea). (After Savtschenko.)
In Scorpæna the body is clothed with scales, and the head is large, slightly compressed, armed with spines, and has a bare pit behind; the single dorsal fin is provided with eleven spiny rays, and there are seven branchiostegal rays. Scorpæna grandicornis (fig. 103), found in the Caribbean Sea, is from 30 to 50 cm. in length, and has the back red and the eyes and belly yellow; Scorpæna diabolus (fig. 104), which occurs in the Indian Ocean 294 and Tropical Pacific, is red and brown, obliquely striped with white and brown; a third species, Scorpæna porcus (Scorpène truie), of smaller size, is met with in the Mediterranean. The venom of the latter has been studied by A. Briot,143 who sectioned the dorsal and opercular spines, and macerated them either in physiological saline solution, or in glycerine; he then tested the toxicity of these macerations on certain animals—frogs, rabbits, and rats.

Fig. 104.—Scorpæna diabolus (Indian and Pacific Oceans). (After Savtschenko.)
The frogs alone exhibited, as the result of subcutaneous injection into a limb, slight transient paralysis. No effect was found to be produced by the venom when injected intravenously into the rabbit, or subcutaneously into the rat.
The poison-apparatus of Scorpæna is situated in the spiny rays of the dorsal and anal fins. These rays are enveloped in the inter-radial membrane, which forms a sheath for them, and are scored with a double cannelure. At the bottom of these grooves are the 295 secreting cells, which are elongate, pressed one against the other, and supported at the base by a highly vascular substratum of connective tissue. The venom flows out between the layer of cells and the ensheathing membrane, which is capable of being pushed slightly back as the result of the penetration of the spine into the tissues, and then exerts pressure upon the reservoir. The latter is formed by the distension of the sheath under the pressure of the secreted liquid.

Fig. 105.—Pterois artemata (East Coast of Africa, Indian and Tropical Pacific Oceans). (After Savtschenko.)
There are twelve pairs of dorsal and three pairs of anal glands. 296 The pairs attached to the second anal spine are, as the direct result of the size of the latter, more developed than those of the other spines.
In the Rascasse, the opercular spines of which are greatly developed, there is a rudiment of a poison-apparatus at the bottom of the sheath formed by the skin of the gills.
The species of Pterois (fig. 105) are distinguished from those of Scorpæna by their dorsal fins, the rays of which are very long and curved backwards, above the membrane by which they are united. They are found in the Indian and Equatorial Pacific Oceans, and are very beautiful in colour, varying from reddish-brown to bright rose.
The poison-apparatus of these fishes is situated in the dorsal fin, and is precisely similar to that of Scorpæna.
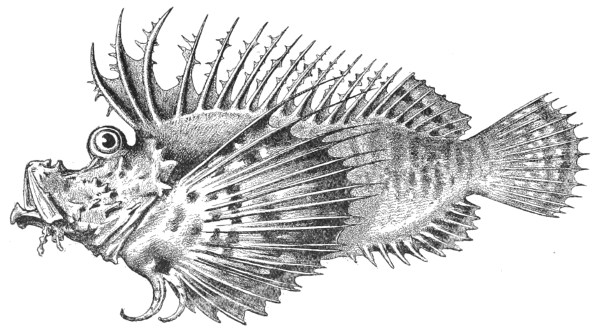
Fig. 106.—Pelor filamentosum (Family Triglidæ, Mauritius).
The species of Pelor (fig. 106) present greater resemblance to those of Synanceia, owing to their heads being crushed in in front. Their eyes stand up above the head and are very close together, which helps to give them an extremely ugly appearance. The skin is soft and spongy, and bristles with jagged fleshy shreds. 297
Their poison-apparatus is placed in the dorsal fins, as in the case of Scorpæna and Pterois.
Genus Trachinus (Weevers).—Four species of Weevers are found in European seas: the Greater Weever (Trachinus draco), the Lesser Weever (T. vipera), the Striped-headed Weever (T. radiatus), and the Mediterranean Spider Weever (T. araneus); other species are met with on the coast of Chile.
Weevers possess two sets of poison-apparatus, one of which is situated on the operculum, the other at the base of the spines of the dorsal fin (fig. 107).

Fig. 107.—Trachinus vipera (Lesser Weever).
The spine surmounting the operculum exhibits a double cannelure connected with a conical cavity excavated in the thickness of the base of the opercular bone. This spine is covered with a sheath, beneath which lie the secreting cells. The gland is an offshoot from the skin, and appears as a simple follicle invaginated in the opercular bone (fig. 108).
The dorsal apparatus is composed of from five to seven spines, to which the inter-radial membrane forms an adherent sheath which extends almost to the end of the rays. Each spine exhibits a deep double cannelure. The venom flows between the layer of cells clothing 298 the cannelures and the skin, which is distended to allow it to pass.
Towards the base of the spine, the edges of the cannelure are united, and form a hollow, bony cone, the walls of which are lined with the cells that secrete the toxic fluid.

Fig. 108.—A, Operculum and opercular spine of the Lesser Weever (Trachinus vipera); ar, articular surface of the operculum; c. op, body of the opercular spine; c. an, canal of the spine; z, space occupied by the poison-gland. B, Spine belonging to the first dorsal fin; c. an, efferent poison-canal in the spine.
Greater Weevers are usually from 12 to 30 cm. in length, and of a reddish or yellowish-grey colour, with blue or violet spots. They are caught in trawls and are fairly common on sandy bottoms. In the month of June they approach the shore for the purpose of spawning.
The venom of the Weever has formed the subject of interesting studies by Günther, Gressin,144 Bottard, Phisalix,145 and more recently by Kobert146 and A. Briot.147
In order to procure sufficient quantities of it for experimental purposes, Briot cuts off the venomous spines and the surrounding tissue with a pair of scissors; he then pounds the whole in a mortar, and mixes the pulp with pure glycerine. After filtration through paper, a toxic solution is obtained, which does not deteriorate by keeping, and is neutral to litmus. 299
A few drops of this liquid are sufficient to kill guinea-pigs, which, immediately after receiving an injection in the thigh, exhibit paralysis of the leg with tetanic convulsions; twenty-four hours later an eschar is formed, and death supervenes on the second or third day.
Two or three drops, introduced into the marginal vein of the ear of a rabbit, cause death from asphyxia in from four to ten minutes. The heart continues to beat for a fairly long time after respiration has entirely ceased; the blood is not coagulated.
The toxicity of this venom is completely destroyed by heating it to 100° C., by chloride of lime, and by chloride of gold. Antivenomous serum prepared from horses vaccinated against cobra-venom has absolutely no effect upon it in vitro. There is therefore no affinity between this venom and that of snakes.
Weever-venom dissolves the red corpuscles of the horse in the presence of normal heated horse-serum, but does not dissolve them in the presence of fresh serum. The non-heated serum, therefore, as I have shown with reference to the action of cobra-venom on the blood, contains a natural antihæmolysin.
Briot succeeded in vaccinating rabbits by accustoming them to the venom, and in obtaining from them a serum capable of neutralising the latter in vitro, and of immunising fresh rabbits against doses several times lethal, even when injected intravenously.
According to Gressin, the following phenomena are produced in man as the result of Weever-stings:—
“At first there is felt an excruciating, shooting, paralysing pain, which, in the case of nervous persons, may cause attacks of leipothymia ending in syncope. A kind of painful formication next pervades the injured limb, which becomes swollen and inflamed, and may even, if treatment be neglected, form the starting point of a gangrenous phlegmon.
“This condition is frequently accompanied by certain general phenomena—such as fever, delirium, and bilious vomiting, the duration of which is variable, since they may only last for two or three hours, or may continue for several days. Fishermen rightly 300 consider this variability to depend upon the amount of venom that has penetrated into the wound, and especially upon the season at which the accident takes place. The most serious results are recorded during the spawning season, and fishermen regard the Lesser Weever as being the more poisonous.”
In the fishes belonging to this family the body is elongated and depressed, while the spines in the anterior dorsal fin and in the ventral fins are slender, flexible, and seldom very solid. The ventral fins are inserted on the breast or on the throat, and are either separated or united together in the shape of a funnel. The skin is naked or covered with large scales, and the mouth is furnished with teeth. The males are distinguished by the presence of a long genital papilla. These fishes are carnivorous.

Fig. 109.—Callionymus lyra (Dragonet or Skulpin. Family Gobiidæ).
Several species of venomous Gobiidæ are met with on the shores 301 of France and in the tropical zone. The most important of these belong to the genus Callionymus (C. belennus, C. lacertus, C. vulsus, and C. lyra—fig. 109).
The Dragonet or Skulpin (Callionymus lyra), which is common on the coast of Calvados, may attain the length of 30 cm. In France it is popularly known as the Doucet, Dragonnet, Lavandière, Cornaud, or Capouri. Its colours are very vivid, orange and deep lilac.
In this fish the præopercular bone ends in three strong, conical, and very sharp points, diverging like the prongs of a trident. The upper margin of the opercular bone bears another point, which is directed upwards.
The skin of the gills forms a common sheath for this defensive armature, and the base of the sheath is prolonged into two culs-de-sac, the surface of which is clothed, during the spawning season, with cylindrical cells, the secretion of which is poisonous.
This venom, which is small in amount, does not appear to have any marked effect upon man (Bottard).
This family of Acanthopterygii includes several species of brilliantly coloured fishes with elongated and laterally compressed bodies, provided with a long dorsal fin, and having, on each side of the tail, a sharp spine placed in front of the anal fin. They are herbivorous, and are confined to the tropical seas.
The principal genera are: Teuthis (India), Acanthurus (Tropical Atlantic), Prionurus (Japan), and Naseus (Red Sea and Indian Ocean). The fishermen of Réunion are much afraid of the wounds inflicted by Acanthurus luridus, which they call Marguerite Porc or Grande Marguerite. A sting from this fish causes a very acute smarting pain, which may last for several hours, but usually has no serious consequences.
The poison-apparatus of these fishes is situated in the dorsal and anal fins, as in Scorpæna. 302
The venomous species belonging to this family are few in number. They are found in all tropical seas, but have no representatives in Europe. The best-known species are Batrachus tau (shores of Central America), and B. grunniens, or Grunting Batrachus (fig. 110).

Fig. 110.—Batrachus grunniens (West Indies).

Fig. 111.—Thalassophryne reticulata (Panama; Tropical Pacific). (After Savtschenko.)
The Grunting Batrachus, which does not exceed 30 cm. in length, is especially common in West Indian waters. When taken from the water it makes a peculiar grunting sound, whence its name is derived. The pectoral fins are reddish, the back is brown, and the sides are yellow, marbled with black. It has three spines in the anterior dorsal fin, and a fourth spine on the top of the operculum, with a small poison-sac at the base of each. 303
Next to this genus come the species of Thalassophryne, T. reticulata (fig. 111), found on the shores of Panama, and T. maculosa, of Bahia (Brazil), which are provided with a precisely similar poison-apparatus.
The physiological action of the venom of these two species has not yet been studied, but it is probable that it does not differ from that of the venom of the Weevers and the species of Synanceia.

Fig. 112.—Lophius setigerus (China Sea and Sea of Japan). (After Savtschenko.)
The fishes belonging to this family are of large size and compact shape, with the anterior part of the body greatly expanded. The head, which is broad, bears venomous spines, and the mouth is furnished with large teeth. These fishes are voracious, and lie 304 in wait for their prey at the bottom of the water in the mud of the shore. In order to attract it, they make use of cutaneous appendages attached to their spines, which they are able to elevate, and of filaments situated near the mouth.
The principal genus is Lophius, one species of which, L. setigerus (fig. 112), is found in the seas of China and Japan. Another species, L. piscatorius (the Sea Devil or Angler), occurs in the temperate climates of Europe, North America, Asia and Africa.
Certain other Acanthopterygii are capable of inflicting wounds, but, although fishermen often believe them to be venomous, or such properties are frequently attributed to them in stories, it is doubtful whether they possess poison-glands. The accidents produced by them are due rather to the fact that the spines in their fins are extremely sharp, and that their flesh is toxic. Those belonging to the Percidæ (the Perch family), especially the genus Serranus and S. ouatabili (fig. 113) in particular, are above all remarkable in this respect. The last-mentioned fish has two or three spines on its operculum.

Fig. 113.—Serranus ouatabili. (After Savtschenko.)
The same may be said of certain Squamipinnes, another family of Acanthopterygii, whose stout bodies are brightly coloured, and have very sharp, spiny rays in their dorsal and anal fins. The 305 most curious genus among the fishes belonging to this family is Holacanthus, in which the præoperculum is provided with an enormous spine like that of the Weever. Holocanthus imperator (fig. 114) is met with fairly often in the Indian Ocean and Malay Archipelago.

Fig. 114.—Holacanthus imperator (Indian Ocean and Malay Archipelago). (After Savtschenko.)
The Order Plectognathi (Family Gymnodontes) includes the genera Diodon, Tetrodon and Triodon, globular fishes, in which the jaw is transformed into a beak and furnished with a sharp dentary plate. Their œsophagus is dilated into a resonant air-pouch. When removed from the water they swallow air and dilate the pouch, and the expulsion of this air is accompanied by a loud noise.
Several species of Tetrodon are armed with spines, which produce 306 very painful wounds. Their flesh is toxic, but it has not been proved that poison-glands exist at the base of the spines.
On the shores of the Cape of Good Hope, Brazil, China, and Japan these fishes are much feared. The principal species are Tetrodon stellatus (Indian and Pacific Oceans; fig. 115) and T. rubripes (Japan; fig. 116).

Fig. 115.—Tetrodon stellatus (Indian and Pacific Oceans). (After Savtschenko.)

Fig. 116.—Tetrodon rubripes (Japan). (After Savtschenko.)
Closely allied to Diodon, and feared like the foregoing on account 307 of their spines, which are sometimes scattered all over the body, are the species of the genus Chilomycterus, the most important of which are C. orbicularis (fig. 117), and C. tigrinus (fig. 118), both of which are found in the Indian Ocean.

Fig. 117.—Chilomycterus orbicularis (Indian Ocean). (After Savtschenko.)

Fig. 118.—Chilomycterus tigrinus (Indian Ocean). (After Savtschenko.)
This Order is characterised by the presence of a pneumatic duct to the air-bladder. It consists of a large, number of families, only two of which, the Siluridæ and Murænidæ, include venomous species. 308
The majority of the very large number of species belonging to this family live in fresh water, and have the free margin of the lips almost always furnished with barbules (Silurus glanis; fig. 119). A few of them possess a poison-apparatus, which, however, attains its greatest development in Plotosus, the only genus of Siluridæ found exclusively in the sea.
The species of Plotosus frequent the shores of the Indian Ocean, and are met with in the Seychelles, Réunion, and Mauritius. In shape they resemble eels, and they bury themselves in the sand or mud, a habit which renders them very dangerous to fishermen.

Fig. 119.—Silurus glanis (Rivers of Central and Eastern Europe).
Plotosus lineatus, which is of a greenish-brown colour, striped with from four to six longitudinal whitish bands, is the most common. By the Creoles of Mauritius and Réunion it is called Machoiran, by the Malays Sambilang, and by the Abyssinians Koomat.
Its poison-apparatus is situated at the base of the dorsal and pectoral spines. These spines are strong, sharp, slightly incurved, 309 and furnished with hooked denticulations, which cause them to remain in the wound, in which they break off. Near their extremity there opens a small canal, which communicates with the culs-de-sac situated at the base of the spiny rays, which produce a venomous secretion. The dorsal spine has only a single cul-de-sac, while the pectoral spines have two.
The contraction of the local muscles, by compressing these culs-de-sac, can cause the venom to make its way into the canal of the spine, but the fluid does not spurt forth in a jet as in the case of Synanceia. The poison-apparatus is therefore passively defensive in character. Plotosus is capable of wounding only when the hand or foot is placed on its dorsal or pectoral spines.
Fishermen who are stung immediately feel an excruciating pain, which is soon accompanied by fever, and lasts for several days. Accidents caused by this fish are of fairly common occurrence in Réunion.
Of the fishes belonging to this family, the species of the genus Muræna alone concern us. They have an elongated body, without pectoral fins, and a naked skin, covered with a thick layer of viscid slime, as in the case of the eels. Their dentition is powerful, formed of long, recurved fangs, arranged in one or more rows. These fishes may attain a large size, exceeding 2 metres in length. More than one hundred species are known, all of which live in tropical or subtropical seas. Muræna helena is common in the Mediterranean in the vicinity of Nice and Toulon; M. moringa (fig. 120) is found in the Tropical Atlantic.
The species of Muræna live in deep water, and feed upon fishes or crustaceans. In hot countries they frequently venture into fresh water. Their skins are adorned with brightly coloured markings, which vary very greatly according to the species.
The poison-apparatus in Muræna consists of a pouch situated 310 above the membrane of the palate, which may contain ½ c.c. of venom, and three or four conical, curved teeth, with the convex surface in front, as in the fangs of snakes. The teeth are not pierced by a central canal, and the venom flows between them and the mucous membrane of the palate, which forms a sheath. The latter is withdrawn to the base of the teeth, while they are penetrating the tissues. The teeth are mobile; they are articulated with the palatine bone, in which they are inserted in small depressions, and a resistant fibrous tissue serves as the means of union. They can be deflexed backwards against the mucous membrane of the palate; in this position the first, second and fourth tooth (when the latter exists) disappear completely between the folds of the membrane. The third tooth normally remains erect, and it is this by which wounds must in most cases be inflicted. None of these teeth can be protruded beyond the vertical.

Fig. 120.—Muræna moringa (Tropical Atlantic). (After Savtschenko.)
In addition to the palatine teeth there are, among the groups of maxillary teeth, several mobile teeth, which are connected with the poison-reservoir.
Besides its toxic action the venom of Muræna has manifest digestive properties, and, in the case of a fish which has been dead for some little time the gland is no longer to be found, since its walls have undergone a rapid autodigestion. 311
The venoms of all the fishes of which I have just given a brief description, as regards their physiological action, present a fairly close resemblance to the venom of the Weever, and show scarcely any variation except in the intensity of their effects. They have been but little studied hitherto, and it is desirable that they should be better understood. 312
By the ancients the venom of salamanders and toads was dreaded as much as the most terrible poisons. These animals, however, are not very formidable, since they are devoid of inoculatory organs; their poison-apparatus is localised exclusively in the parotids and the skin. It is represented simply by more or less confluent glands in the form of sacs, secreting a viscid mucus, which has a nauseous odour and is highly toxic, even to animals of large size.
The salamander belongs to the Order Urodela, which is characterised by the persistence of the tail. Its body is heavy and thickset, and the flanks and the sides of the tail exhibit a series of glandular crypts, which secrete venom.
“The mucus which flows from the mouth, and resembles milk, eats away human hair,” wrote Pliny; “the spot moistened by it loses its colour, which subsequently returns. Of all venomous animals the salamander is the most terrible; it is capable of annihilating whole nations by poisoning the vegetation over a vast area. When the salamander climbs a tree all its fruit is poisoned, and those who eat of it die as surely as if they had taken aconite. Moreover, if bread be baked with wood touched by the animal, it is dangerous, and may occasion serious disorders. If the naked foot be defiled with the saliva of this creature, the beard and hair soon fall out. Sextius says that a salamander, preserved in honey, after the 313 removal of the entrails, head, and limbs, acts as a stimulant if taken internally.”
In ancient Rome, and also in Mediæval France, it was believed that the most furious fire could be extinguished simply by contact with one of these animals; charlatans sold the inoffensive salamander, which, if cast into the most terrible conflagration, was bound, they declared, to arrest its disastrous progress!
The explanation of this superstition is furnished by Duméril, who writes: “On being placed in the middle of burning charcoal, these victims of so cruel a curiosity, when put to the test, instantly allowed to exude from the many pores with which their skins are riddled a slimy humour, sufficiently abundant to form a viscid layer over that part of the glowing charcoal with which the animals were in contact. Since this surface, being no longer exposed to the air, immediately became quite black, it was supposed to be extinguished; but the salamanders sustained such severe burns that they soon succumbed.”148
The principal species of salamanders are:—
Salamandra atra (Black Salamander), which is found in the Alps and the mountains of Central Europe, close to the snow-line, and up to an altitude of 3,000 metres.
Salamandra maculosa (Spotted Salamander, fig. 121), distributed throughout almost the whole of Europe, and also found in North Africa.
Triton cristatus (Crested Newt), likewise common all over Europe.
Triton marmoratus (Marbled Newt, fig. 122), which is met with in damp and dark places, in Portugal, Spain, South and Central France, and as far north as the Forest of Fontainebleau.
Cryptobranchus japonicus (Great Japanese Salamander, fig. 123), which often exceeds 1 metre in length, and has a clumsy 314 body covered with large warts, and an enormous head, broad behind and flattened in front.
This giant salamander is now confined to a few provinces in the centre of Japan, between long. 34° and 36°, in damp, shady places, from 200 to 800 metres above sea-level. It is eaten by the Japanese, who also use it as a remedy for, or prophylactic against, contagious disorders. By nature it is extremely sluggish, but tries to bite when irritated, and then covers itself copiously with slime.

Fig. 121.—Salamandra maculosa (Europe and North Africa).

Fig. 122.—Triton marmoratus (male). (Europe.)
The venom secreted by salamanders evidently serves to protect these creatures against their enemies. So long ago as 1866, 315 316 Zaleski149 isolated from it a substance soluble in alcohol, insoluble in ether, and with a very strong alkaline reaction, to which he gave the name salamandarin. This substance, which is better known to-day as salamandrine, has been studied afresh by A. Dutartre,150 Phisalix and Langlois,151 and subsequently by Edwin and S. Faust.152

Fig. 123.—Cryptobranchus japonicus (Great Japanese Salamander).
The action of this poison on the frog is characterised by a period of violent convulsions, with general tetanic crises, followed by a period of paralysis, with arrest of respiration and complete muscular relaxation. According to the quantity of poison absorbed, this paralytic period may be followed by death, with arrest of the heart in diastole, or else by return to life, with more or less acute recurrence of convulsions.
S. Faust prepares salamandrine by pounding up whole salamanders in a small quantity of physiological saline solution. The thick pulp obtained in this way is filtered. One cubic centimetre of the filtrate, taken as a unit, contains about 5 decimilligrammes of active substance, which can be purified by treating the filtrate with alcohol, which dissolves the salamandrine and precipitates all the proteic substances that give biuret reaction. The salamandrine thus freed from proteins is saturated with sulphuric or phosphoric acid, when there is formed a crystallisable salt, which is washed and dried. This salt is soluble in alcohol and in water. Its chemical composition is as follows:—
C52H80Az4O2 + H2SO4.
The toxicity of this substance is such that from 7 to 9 decimilligrammes per kilogramme represent the lethal dose for dogs, when injected subcutaneously. The lethal dose for the rabbit is 317 still smaller. It produces convulsive phenomena, followed by arrest of respiration. The administration of chloral to the subjects of the experiment, either preventively or immediately after the poison, prevents the latter from taking effect. Besides salamandrine, S. Faust has isolated a second alkaloid, salamandridine, which, as a sulphate, corresponds to the formula (C20H31AzO)2 + H2SO4, crystallises in rhombic prisms, and is soluble with difficulty in water. The only difference between the two alkaloids is formed by a methylpyridic group, and both are derivatives of quinoline. They must therefore be considered as identical with the exclusively vegetable alkaloids.
S. Faust concludes from his physiological investigations that salamandrine takes effect upon the central nervous system, especially upon the respiratory centres. It is a convulsion-producing poison, comparable to picrotoxin, but its effects differ from those of the latter substance in that the convulsions are accompanied by tetanic spasms.
The venom of the Japanese Salamander (Cryptobranchus japonicus) has formed the subject of studies by Phisalix.153 This investigator has shown that this venom, which is highly soluble in water and in glycerine, is very unstable; alcohol and heating for twenty minutes at 60° C. are sufficient to destroy it. When inoculated into frogs it produces œdema and hæmorrhage; if injected into warm-blooded animals it causes necrosis. In sufficiently strong doses it kills by arresting respiration. Its effects strongly resemble those produced by Viperine venoms. This venom, if attenuated by being heated at 50° C. and injected into mammals, vaccinates them and leads to the formation in their blood of antitoxic substances, which are capable of preventing intoxication by salamander-venom, and, curiously enough, also confer immunity against viper-venom and the serum of the common snake. 318
Toads are easy to distinguish from frogs owing to their squat and clumsy shape, and to the mass of glands with which each side of the neck and a more or less extensive portion of the body is furnished in these animals. According to G. A. Boulenger, the number of known species amounts to seventy-six, which are found in the Old and New Worlds, but have no representatives in Australia. The species that are the most common, and most interesting from the point of view of their venoms, are:—
The Common Toad (Bufo vulgaris), in which the skin, which is very thick and rugose, is covered on the back with large rounded tubercles with reddish summits. This species is a great destroyer of insects, and, as such, is very useful to agriculturists.
The Natter-Jack (Bufo calamita), in which the digits are palmate at the base. When irritated it contracts its skin and covers itself with a white frothy exudation, which gives off an odour of burnt powder.
The Green Toad (Bufo viridis), which is especially abundant in Southern Europe, the Levant, and North Africa.
The Musical Toad (Bufo musicus), a species distributed throughout North America as far south as Mexico, and in which the back is covered with pointed conical tubercles resembling spines.
The Brown Pelobates (Pelobates fuscus), common in the neighbourhood of Paris, the skin of which is almost entirely smooth. Although it appears to be nearly destitute of glands, this animal secretes a very active venom, which has a penetrating odour and kills mice in a few minutes, producing vomiting, convulsions, and tetanic spasms of the muscles.
The toxicity of the venom of toads was long ago demonstrated by the experiments of Gratiolet and Cloëz.154 It is manifest only in the case of small animals, and in man merely produces slight inflammation of the mucous membranes, especially of the conjunctiva. 319
That this venom preserves its toxic properties for more than a year in the dry state was shown by Vulpian, and satisfactory studies of its composition and physiological action have been made by Fornara,155 G. Calmels,156 Phisalix and Bertrand,157 Schultz,158 Pröscher,159 and S. Faust.160
Toad-venom was prepared by Phisalix and Bertrand in the following manner: Holding the head of one of these batrachians under water, they expressed the contents of the parotid glands with the fingers or with a pair of forceps. They repeated the same operation with a second, and then with a third toad, until they had sufficiently impregnated the water, which serves to dissolve the venom. In this way they obtained an opalescent, acid liquid, which they filtered with a Chamberland candle under a pressure of from four to five atmospheres. There remained on the filter a yellowish substance, with a highly acid reaction and partly soluble in ether and chloroform, while there passed through the pores a clear, reddish, and slightly acid liquid, which on being evaporated left behind a greyish-white precipitate. This precipitate was separated by filtration, washed in water, and redissolved in absolute alcohol or chloroform. The albuminoid matters were thus separated, and the liquid, after being rendered limpid by filtration, was evaporated away. The substance obtained in this way represents one of the two active principles of the venom. It acts on the heart of the frog, and arrests it in systole. It assumes the appearance of a transparent resin, the composition of which roughly corresponds to the formula C119H117O25. It is the bufotalin of Phisalix and Bertrand, and is probably identical with that obtained by S. Faust, 320 the formula of which, according to the latter author, is said to be C11H23O5.
Bufotalin is readily soluble in alcohol, chloroform, acetone, acetate of ethyl, and acetic acid. When water is added to a solution of it in alcohol it is precipitated, giving a white emulsion, which has a very bitter taste.
From the aqueous extract whence the bufotalin has been separated, it is possible to separate a second poison, which acts on the nervous system and causes paralysis. In order to obtain it in a pure state, the extract is treated with alcohol at 96° C., filtered and distilled; the residue dissolved in water is defæcated with subacetate of lead and sulphuretted hydrogen. The solution thus obtained is successively exhausted with chloroform to extract the cardiac poison, and with ether, which removes almost the whole of the acetic acid. The second neurotoxic principle, called bufotenin, remains in the residue of the solution after being evaporated in vacuo.
Toad-venom, therefore, contains two principal toxic substances: bufotalin, which is of a resinoid nature, soluble in alcohol, but scarcely soluble in water, and is the cardiac poison; and bufotenin, which is readily soluble in those two solvents, and is the neurotoxic poison.161
Pröscher, on the other hand, has extracted from the skins of toads a hæmolytic substance, termed by him phrynolysin, which possesses all the properties of a true toxin and is not dialysable. It is obtained by pounding the skins with glass powder in physiological serum.
Phrynolysin dissolves the red corpuscles of the sheep very rapidly, and (in order of sensitiveness) those of the goat, rabbit, dog, ox, fowl, and guinea-pig. The red corpuscles of the pigeon, frog, and toad are scarcely affected. When heated at 56° C. it 321 loses its properties. By the ordinary methods of immunisation it is possible to obtain a very active antilysin.
There is, therefore, a very close analogy between the venoms of toads and salamanders. These highly complex substances are composed of mixtures of poisons, some of which are in all respects analogous to the vegetable alkaloids, while others are closely related to the microbic toxins and snake-venoms.
In the spawning season the cutaneous glands of the male toad are gorged with venom, while those of the female are empty. Phisalix162 has shown that at this period the venom of the female is accumulated in the eggs, which, if extracted from the abdomen at the moment of oviposition and dried in vacuo, give off in chloroform a product that has all the toxic properties of cutaneous venom (bufotalin and bufotenin). No trace of this poison is to be found in the tadpoles.
The Order Lacertilia includes only a single venomous species, which belongs to the family Lacertidæ, and is known as the Heloderm (Heloderma horridum, fig. 124). It is a kind of large lizard, with the head and body covered with small yellow tubercles on a chestnut-brown ground. It sometimes exceeds a metre in length, and its habitat is confined to the warm belt extending from the western slope of the Cordilleras of the Andes to the Pacific. It is met with especially in the vicinity of Tehuantepec, where it inspires the natives with very great dread. It is a slow-moving animal, and lives in dry places on the edges of woods. Its body exhales a strong, nauseous odour; when it is irritated, there escapes from its jaws a whitish, sticky slime, secreted by its highly developed salivary glands. Its food consists of small animals. Its bite is popularly supposed to be extremely noxious, but, as a rule, the wound, though painful at first, heals rapidly. 322 Sumichrast caused a fowl to be bitten in the wing by a young individual, which had not taken any food for a long time. After a few minutes the parts adjacent to the wound assumed a violet hue; the bird’s feathers were ruffled; a convulsive trembling seized its entire body, and it soon sank to the ground. At the end of about half an hour it lay stretched out as though dead, and from its half-open beak there flowed a sanguinolent saliva. There was no movement to give any sign of life, except that from time to time a slight shiver passed through the hinder part of its body. After two hours, life seemed gradually to return, and the bird picked itself up and crouched on the ground, without, however, standing upright, and still keeping its eyes closed. It remained thus for nearly twelve hours, at the end of which time it once more collapsed, and expired.

Fig. 124.—Heloderma horridum.
A large cat which Sumichrast caused to be bitten in the hind leg did not die, but immediately after being bitten the leg swelled considerably, and for several hours the cat continued to mew in 323 a way that showed that it was suffering acute pain. It was unable to stand, and remained stretched out on the same spot for a whole day, unable to get up, and completely stupefied.
Interesting observations on the Heloderm have been made by J. Van Denburgh and O. B. Wight. The saliva of this lizard was found to be highly toxic at certain times, and harmless at others. When injected subcutaneously it produces various effects, such as miction, defæcation, and abundant salivation, with accelerated respiration followed by vomiting. The animal drinks with avidity, and remains lying down, in a very depressed condition. Death finally supervenes, from arrest of respiration and also of the heart’s action. The poison likewise acts upon the arterial tension, which falls very rapidly and very markedly. The sensory nerves are also attacked; irritability is at first increased, then diminished, and at last entirely lost. These changes take place from behind forwards, and from the periphery to the centre. The coagulability of the blood is at first intensified and then lessened, as when acted upon by Viperine venom (H. Coupin).163
The only mammal that can be considered to be provided with a poison-apparatus belongs to the Order Monotremata, and is known as the Duck-billed Platypus (Ornithorhynchus paradoxus or O. anatinus, fig. 125). The head of this animal is furnished with a kind of flat duck’s bill, armed with two horny teeth in the upper jaw, while the body, which is covered with dense fur, resembles that of a beaver. The tail is broad and flat; the legs are short, and the feet are provided with five toes, armed with strong claws and webbed.
This singular animal is found only in Australia and Tasmania. It lives in burrows near watercourses, entered by holes which it digs in the bank, one above, the other on the water-level. It spends 324 much of its time in the water, and feeds upon worms and small fishes.
In the males the hind feet are armed with a spur, having an orifice at the extremity. At the will of the animal, there is discharged from this spur a venomous liquid secreted by a gland, which lies along the thigh, and is in communication with the spur by means of a wide subcutaneous duct (Patrick Hill).164
It has often been proved in Australia that this liquid, when inoculated by the puncture of the spur, may give rise to œdema and more or less intense general malaise. Interesting details with reference to the effects produced by this secretion have been published by C. J. Martin, in collaboration with Frank Tidswell.165

Fig. 125.—Ornithorhynchus paradoxus. (After Claus.)
When a dose greater than 2 centigrammes of dry extract of the venom of Ornithorhynchus is injected intravenously into the rabbit, it produces phenomena of intoxication analogous to those observed after inoculation with Viperine venoms.166 Death supervenes in from twenty-five to thirty minutes, and at the autopsy hæmorrhagic patches are found beneath the endocardium of the left ventricle.
This venom has been studied afresh in my laboratory by Noc, 325 thanks to the acquisition of a small supply kindly forwarded to me by C. J. Martin. Noc proved that it possesses in vitro certain properties of snake-venoms; like the venom of Lachesis lanceolatus, it induces coagulation in citrate-, oxalate-, chloridate-, and fluorate-plasmas. Heating at 80° C. destroys this coagulant power.
Contrary, however, to what is found in the case of the venoms of Vipera and Lachesis, the secretion of Ornithorhynchus is devoid of hæmolytic and proteolytic properties.
Lastly, its toxicity is very slight, at least five thousand times less than that of the venoms of Australian snakes. A mouse is not even killed by 5 centigrammes of dry extract, and in the case of the guinea-pig 10 centigrammes only produce a slight painful œdema.
It has been remarked that the volume and structure of the poison-gland exhibit variations according to the season of the year at which it is observed. It is therefore possible that these variations also affect the toxicity of the secretion (Spicer).167
By certain authors the poison of Ornithorhynchus is considered to be a defensive secretion of the males, which becomes especially active in the breeding season, and this hypothesis is plausible. In any case it would seem that as a venom the secretion is but very slightly nocuous.
It will have been seen from the papers quoted above that the chemical nature and physiology of the various venoms, other than those of snakes, are as yet little understood and need further investigation.
The main outlines of this vast subject have scarcely been traced, and the study offers a field of interesting investigations, in which the workers of the future will be able to reap an ample harvest of discoveries, pregnant with results for biological science. 326
I.—Case published by A. Beveridge, M.B., C.M., Surgeon S. Coorg Medical Fund (British Medical Journal, December 23, 1899, p. 1732).
“A strong coolie, aged 26, was bitten by a cobra on the right ankle, just above the internal malleolus. He was brought to the surgery about one hour after being bitten, in a state of comatose collapse. The pulse was rapid, and the surface of the body cold. He was given an injection of 10 c.c. of Calmette’s antivenene deeply into the right flank. He was kept under observation: the paresis and insensibility were very marked. On visiting him some hours afterwards I found he could walk without assistance, but staggered, and complained of weakness and pains in both legs. Next morning he was much improved, the paresis gradually wore off, and the pulse steadily gained strength. The patient returned to work four days later, quite recovered.
“A few days previously a coolie had died after being bitten by a snake under the same conditions, but without having been treated. Occurrences like these point to the necessity that every Government or private dispensary should be supplied with antivenene, which is certainly the best remedy for snake-bite available.” 327
II.—Case reported by Robert J. Ashton, M.B., Kaschwa Medical Mission, Mirzapur (N.W.P.).
“A coolie, aged 27, was bitten in the right foot by a cobra at 5.30 a.m., on September 16, 1900. Half an hour later 10 c.c. of antivenomous serum were injected subcutaneously into the left forearm. The patient experienced great pain in the foot, torpor, and great weakness. Recovery, without complications.”
III.—Case reported by Dr. Simond (Saigon).
“Nguyen-Van-Tranc, an Annamese, aged 25, employed in the Botanical Gardens at Saigon, was bitten at 10.30 a.m., on March 11, 1899, by a cobra which had escaped from its cage. The bite was inflicted on the palmar surface of the index finger of the right hand, and the fangs had penetrated deeply.
“This native, to whom a sensible comrade had applied a ligature round the wrist, was brought to the Pasteur Institute three hours later. He was drowsy, with drooping eyelids; his speech was difficult and almost unintelligible. Deglutition was impossible, and ingurgitated liquids caused vomiting. The hand was greatly swollen at the seat of the bite, and the œdema extended to the forearm. There was partial anæsthesia of the skin. As soon as the patient arrived, I gave a single injection, beneath the skin of the flank, of three doses of serum, that is, 30 c.c. In the evening I again injected 10 c.c. of serum. At 10 p.m. the general condition of the patient seemed to be improving. Next morning he was less depressed, spoke more easily, and was able to swallow. Convalescence began from this moment; the œdema and numbness of the hand and arm, however, persisted for several days.
“Recovery was complete on March 20. I have no doubt that in this very serious case the antivenomous serum preserved the life of the patient, since his condition was desperate when I saw him.
“This is the second instance within four months of the successful treatment of snake-bites at Saigon by Calmette’s serum. In the former case two natives were bitten by the same animal. One of them, who permitted the injection of serum, which was performed 328 by Dr. Sartre, recovered; the other, who refused it, died within twenty-four hours.”
IV.—Case reported by the Fathers of the Khurda-Mariapur Mission (India).
“At 1 p.m., on October 31, 1905, a woman, aged 35, who had been bitten by a cobra, was brought to us from Khurda. After being at our dispensary for about an hour she became drowsy; she paid no attention to anything that was said to her, and merely replied that she felt sick. We thereupon injected 10 c.c. of serum. The woman did not even appear to feel the prick when the needle was driven into her calf. Immediately after this was done she dozed and went to sleep. The pulse was feeble, and the entire body cold. We were disposed to give a second injection, but, since we had only two bottles left, we hesitated to sacrifice one of them. At last, after sleeping for about half an hour, the woman awoke of her own accord, sat up, and began to recover her senses. Bodily heat returned almost immediately, and a few moments later the patient asked to be allowed to go home; she was, however, kept at the dispensary. In the evening she continued to complain of headache, but on the following day she was able to walk, and was quite well.”
V.—Case reported by Dr. Brau (Saigon).
“Nhuong, an Annamese agriculturist, on passing through a piece of waste ground beside the barracks, at about 5.30 a.m. on Sunday, September 11, felt himself suddenly bitten behind the right knee. He caught a glimpse of a large blackish snake, with all the characteristics of a cobra, including the raised head and dilated hood, gliding hurriedly away, but was unable to overtake it.
“The seat of the bite merely showed two small blackish punctures. The part soon became painfully swollen, and the patient began to feel giddy. Other natives came to his help; he was lifted into a Malabar cart and brought to the Military Hospital, whence he was sent to my house, where he arrived about a quarter past six.
“I entered the vehicle, and immediately drove with the patient to the Pasteur Institute. The only treatment that he had received 329 was a ligature round the middle of the right thigh. The lower leg was enormously swollen, and the swelling was not stopped by the slight barrier formed by the ligature, but had already extended to the base of the limb.
“The patient lay stretched out between the two seats of the vehicle, with head thrown back and eye-balls turned up and ghastly. His skin and extremities were cold, and his pulse was scarcely perceptible. In order not to lose time, he was not even taken up to the first floor of the Institute, but was carried to an inoculating table. He was then made to swallow black coffee and rum, and was given an injection of as much as six doses of antivenomous serum, which had just been received from the Pasteur Institute at Lille.
“Under the stimulus of this injection, somewhat drastic I admit, an absolute resurrection took place in the sick man. The pulse became strong and bounding, bodily heat returned, and, although the swelling did not at once diminish, its progressive extension seemed to be sharply arrested, while the pain was also greatly lessened. The patient was able to sit up without assistance, and relate the incidents of his misadventure.
“In a few minutes time I thought it possible to have him taken to the Choquan Hospital, the Director of which Institution, First-class Surgeon-Major Angier, has been good enough to furnish me with a note of the subsequent history of this case.
“’The Annamese Nhuong, who entered the Choquan Hospital on September 11, suffering from snake-bite, was discharged on September 20.
“’On admission, heat and puffiness were observed in the calf and thigh. Slight dyspnœa, severe fever, tendency to coma. September 12, temperature 38°, 39·2° C. September 13, temperature 37·3°, 37·6° C. September 17, temperature 36·8°, 37° C.
“’On discharge, slight œdema and puffiness in the region of the bite. General condition good.’”
VI.—Case recorded by Dr. Robert Miller, Bengal-Nagpur Railway Company (Advocate of India, Bombay, January 15, 1902). 330
“On the evening of October 23 I was called to a coolie woman, who had been bitten by a large cobra about 7 o’clock; some two hours had already elapsed since the accident. The woman was, so to speak, moribund, unconscious, and suffering from paralysis of the throat, after having exhibited all the characteristic symptoms of poisoning by cobra-venom. I immediately injected 10 c.c. of Calmette’s serum, without any hope of a successful result, however, so desperate did the condition of the patient appear. The effect of the serum was marvellous; fifteen minutes later she regained consciousness. I gave a fresh injection of 10 c.c., and three hours after the first the patient was out of danger. Dr. Sen, my assistant-surgeon, was present. I have forwarded a note of this case to Dr. L. Rogers, Professor of Pathology at the Calcutta Medical College.”
VII.—Case recorded by Captain H. A. L. Howell, R.A.M.C. (British Medical Journal, January 25, 1902).
“Shortly before 4 p.m. on November 17, 1901, Lance-Corporal G., Royal Scots, was bitten on the right forefinger by a snake. On being brought to hospital, Assistant-Surgeon Raymond tied a tight ligature round the finger, scarified the wound, and applied a strong solution of calcium chloride. On my arrival I found the patient apparently quite well, and not at all alarmed. As I could get no information as to the nature of the snake, I injected into the patient’s flank at 4.30 p.m. 3 c.c. of Calmette’s serum, and sent for the snake, which was the property of one of the men in barracks. The snake was brought to me just before 6 p.m., and I found it to be a cobra about 3½ feet long, of the pale-coloured variety that natives call Brahmini cobra. I at once injected 7 c.c. of Calmette’s serum into the other flank. The patient thus received one full dose of serum. The ligature was removed from the finger, which was swollen and very painful.
“Up to half an hour after the bite the patient, a healthy and powerful man, presented no abnormal symptoms: pulse, respiration, pupils, temperature, and general appearance, all were normal. 331 His pulse and respiration began to increase in frequency, and the pulse became very compressible, but quite regular. After the first injection of serum his temperature was 98° F., pulse full, high tension, regular, 88, and respirations greatly increased in frequency. He now became very drowsy, and had to be roused when the second injection of serum was given. Soon after this the patient’s general condition and pulse improved.... He had complete loss of sensation in the bitten finger, in the part terminal to the site of the puncture, for some days.... The injection of Calmette’s serum gave rise to no local reaction, and caused no pain. It did not affect the temperature, but was followed in half an hour by perspiration, which was very profuse four hours after the injection.... The patient made a complete recovery.... The serum used in this case was fresh, having been prepared at Lille in July, 1901.”
VIII.—Note of case treated by Major Rennie, R.A.M.C., transmitted by M. Klobukowski, French Consul-General at Calcutta, September 5, 1899:—
“A remarkable cure effected by Major Rennie, by means of Calmette’s method, has just taken place at Meerut. Since the introduction of this remedy three years ago, its efficacy has been abundantly proved, but the present case is especially interesting, since it seems to show that the serum can be successfully employed even in cases apparently desperate. The well-known symptoms of poisoning by cobra-venom were already so advanced that the patient, who was insensible, was kept alive by artificial respiration in order to give time for the serum to be absorbed and to take effect.
“The truth of the above statements is attested by six doctors, and is also vouched for by the Commissioner and Magistrate of the military cantonment, who, although not medical men, have, nevertheless, had long experience of Indian matters.”
IX.—Case recorded by Binode Bihari Ghosal, Assistant-Surgeon, Jangipur (“A Case of Snake-bite [Cobra?].—Recovery,” Indian Medical Gazette, January, 1905, p. 18). 332
“While fastening her door about 10 o’clock one night a Hindu woman was bitten by a cobra in the left foot, about 1 inch above the metatarso-phalangeal joints of the second and third toes. About ten minutes after the bite natives applied three strong ligatures, one above the ankle, one below, and one above the knee-joint. Four hours later ‘Fowl’ treatment was applied, which it appears gives marvellous results. The author arrived about nine hours after the accident, during the ‘Fowl’ treatment, for which nineteen chickens had already been sacrificed. In spite of this the patient was pulseless (no radial pulse—the brachial pulse was thready and flickering); respiration about six per minute. An injection of strychnine improved her condition for a few minutes. When the incision, which had been made over the bite, was crucially enlarged, large quantities of dark blood were withdrawn by cupping. In spite of this the patient’s condition grew worse, and her respiration fell to three a minute; she then received an injection of 10 c.c. of Calmette’s serum in the left buttock. The pulse immediately became stronger, and respiration increased to ten per minute. About half an hour after the first, a fresh injection of 10 c.c. of serum was given in the same place. Within five minutes the appearance of the patient, who had seemed to be dying, became normal. The pulse grew stronger, and respiration was about fifteen per minute. One hour after the injections the patient was practically cured.
“The ‘Fowl’ treatment consists in applying directly to the wound, after the latter has been slightly enlarged by means of an incision, the anal apertures of living fowls, from which the surrounding feathers have been removed. The fowl immediately becomes drowsy, its eyes blink, and its head falls on its breast with the beak open, after which the bird rapidly succumbs. Twenty fowls had been employed in the present case, but in vain.” (The author does not appear to have troubled himself to ascertain whether the fowls were really dead, or had merely fallen into a hypnotic condition.) 333
X.—Case reported by Major G. Lamb, I.M.S., Plague Research Laboratory, Parel, Bombay, October 18, 1900.
“Ten days ago I was bitten by a large cobra, from which I was collecting venom. I had only some very old serum in the laboratory, but I immediately gave myself an injection of 18 c.c. Three hours after being bitten I felt faint, my legs became paralysed, and I was seized with vomiting. In the meantime, fresh serum had been obtained at a chemist’s, and I received an injection of 10 c.c. The symptoms improved very rapidly, and an hour later I felt perfectly well. I applied no local treatment, relying altogether upon the serum.”
XI.—Case reported by Dr. Angier, of Pnom-Penh (Cambodia).
“At 11.30 one night in April, 1901, His Majesty, the second King of Cambodia brought to me in a carriage one of his wives who, when crossing the courtyard of the palace at about 8 o’clock, was bitten by a snake, which she said was a cobra (in Cambodian Povek).
“The bite was situated in the lower third of the leg, in front of the internal malleolus. The patient complained continually; she was suffering greatly from the leg, which was swollen as high as the knee. Great lassitude. An injection of 10 c.c. of antivenomous serum was given, half in the leg and half in the flank. The wound was washed, squeezed and dressed. Twenty minutes later the pain had ceased, and the patient went away, feeling nothing more than a slight dulness in the injured limb.”
XII.—Cases reported by Dr. P. Lamy, of the Houdaille Expedition.
“Lamina, a Senegalese, bitten on the outside of the left thigh, on February 18, 1898. Treated with serum. Recovery.
“Momo Bolabine, bitten in the heel on April 20, 1898. Ten c.c. of serum. Recovery.” 334
XIII.—Case reported by Dr. Deschamps, of Thiès (Senegal).
“In the month of October, 1898, I was called to a native, a local constable, who had just been bitten by a Naja. The Ouoloffs of Senegal are much afraid of the bites of this reptile, since they are generally fatal. In this case the man had been bitten in the forehead by a snake, which was coiled up in his bed, as he was placing his head on the pillow. Being in the dark, he got up greatly frightened, lit a candle, and saw the snake glide from his bed and escape through the half-open door. I arrived a few minutes after the accident; the constable already felt very weak, and complained of nausea and of pains in the head and back of the neck. In the middle region of the forehead I found two adjacent wounds, around which the tissues were œdematous. I washed the wounds with a solution of permanganate of potash, and had a telegram sent to St. Louis asking for antivenomous serum. Half an hour after the bite, the patient was seized with vomiting and cold sweats. At 6 a.m. on the following day there was considerable œdema of the face and dyspnœa, while the pulse was small and intermittent. The patient, who had not slept, was dull and depressed. He vomited a little milk which I tried to make him take. Forty hours after the bite the patient, who was already paralysed, became comatose; the face and neck were enormously swollen. The dyspnœa had increased; it was difficult to hear the respiratory murmur; the pulse was thready, slow, and intermittent; the skin was cold; the temperature, taken in the axilla, was 35·8° C. At this moment the serum asked for arrived from St. Louis. I injected into the buttock the only dose that I possessed, 10 c.c. The coma persisted throughout the evening and during part of the night; at 6 a.m. on the following day, fourteen hours after the injection, the patient awoke and said that he felt quite well. The œdema of the face and neck had diminished, that of the eyelids had disappeared. Three days later the constable returned to duty.”
XIV.—Case reported by Professors H. P. Keatenje and A. Ruffer (Cairo). 335
“A girl named Hamida, aged 13, while picking cotton on October 7, 1896, at Ghizeh, near Cairo, was bitten in the left forearm by a large Egyptian cobra, which measured 3 feet in length. She cried out, and her brother and others who were working with her ran up. She was brought to hospital by the police at 7 p.m. in a state of complete collapse. She was almost cold, with upturned eyeballs and imperceptible pulse. The forearm had been bandaged with a dirty cloth, and the entire arm was covered with a thick layer of Nile mud (a favourite remedy among the Fellahîn). Above the wrist two deep punctures were clearly visible, evidently corresponding to the fangs of the reptile. The patient, whose condition seemed absolutely desperate, had no longer any reflexes; she was completely insensible; the moderately dilated pupils scarcely reacted at all to luminous impressions. Dr. Ruffer injected, with the customary antiseptic precautions, 20 c.c. of Calmette’s antivenomous serum beneath the skin of the abdomen. The child gave a groan while this was being done; this was at 7.30 p.m. At 11 o’clock at night her condition improved; the pulse was 140, and bodily heat returned; the patient replied to questions that were put to her. A second injection of 10 c.c. of serum was given in the flank. She slept for the remainder of the night, and passed her water four times under her. At 8 a.m. on October 8 she appeared to be out of danger. She took food, and dozed throughout the day. On the 9th she was convalescent. There were no complications resulting from the injection, neither eruptions nor pains in the joints.”
XV.-Case reported by Dr. Maclaud, of Konakry (French Guinea).
“At 7.30 p.m., on June 22, 1896, there was brought to the Konakry Hospital a native soldier, named Demba, who had just been bitten by a snake. This man, who was employed in the bakery, was stacking firewood, when he felt an extremely acute pain in the left foot; simultaneously he saw a large snake making off; he succeeded in killing it, and found it to be a black Naja. 336 After having applied a stout ligature to the limb, the injured man hastened to the hospital, where, immediately afterwards, he fell into a condition verging on coma. The body was bathed with cold sweat; the temperature was subnormal; the pulse, which was small and thready, was 140. There was difficulty in breathing, and severe vomiting. At intervals the patient was aroused by spasms, and excruciating pains in the injured limb, which exhibited considerable œdema above and below the ligature. Tendency to asphyxia. I washed the wounds with 1 per cent. solution of permanganate of potash, and injected a dose of antivenomous serum into the subcutaneous cellular tissue of the left flank. In view of the severity of the symptoms I gave two other injections of serum, an injection of 3 c.c., followed by one of 2 c.c. The patient dozed all night. Next day the general symptoms had entirely disappeared. Two days later Demba returned to duty.”
XVI.—Case reported by Surgeon-Captain Jay Gould (Nowgong, Central India, British Medical Journal, October 10, 1896, p. 1025).
“On June 11, 1896, a punkah coolie was bitten on the dorsum of the left foot, between the second and third toes. He had only the distinct mark of an incisor, a very slight prick, with a stain of blood which marked the spot. Within ten minutes we had injected 20 c.c. of Calmette’s serum into the abdominal wall, after which we made a local injection of a 1 in 60 solution of hyperchlorite of calcium. Two hours after the injection the temperature was subnormal, the pulse full and slow. Twelve hours later the patient was perfectly well and walking about.
“The snake was a Bungarus, full grown, measuring 28 inches. Unfortunately the syces killed it; it died the very moment I arrived, so that I was unable to test its virulence.” 337
XVII.—Case reported by Major S. J. Rennie, R.A.M.C., Meerut, N.W.P., India.
“A twelve-year old Hindu boy, named Moraddy, was brought to me at 6 p.m., on July 10, in a semi-comatose condition, with commencing paralysis of the respiratory muscles. I was told that the child was sleeping on the ground, when he was bitten in the left hand. He immediately felt very great pain and giddiness, and his arm began to swell. Two small wounds were clearly visible, corresponding to the marks of the fangs of a krait, or Bungarus cæruleus.
“The child had salivation, and ptosis of both eyelids. Respiration was difficult, and deglutition impossible; the pulse was 110 and dicrotic. The patient’s breathing was of an abdominal character; the surface of the body was covered with cold sweat. The child soon became lethargic and collapsed; his condition appeared absolutely desperate. I gave a subcutaneous injection of 12 c.c. of antivenomous serum, and commenced artificial respiration, which I continued for half an hour in order to give the serum time to take effect. In forty-eight hours the symptoms gradually disappeared, and the child became quite well. Diplopia of the left eye persisted for a few days, but this also entirely passed away.
“This case shows that, in Calmette’s antivenomous serum, we have a very powerful remedy against snake-bites, which may take effect even in desperate cases. It further proves that the serum will keep for a very long time, even when exposed to all the vicissitudes of the Indian climate, for the serum employed by me had been in my possession for nearly four years.”
XVIII.—Case reported by Mr. W. A. G. Fox, Table Mountain, Cape of Good Hope.
“On February 9, 1898, I was summoned to the Town Council’s 338 Camp to treat a native who had been bitten by a berg-adder in the left leg, just below the knee. I immediately injected a dose of Calmette’s antivenomous serum in the left flank, and the wounds were washed. The injection was given two and a quarter hours after the accident. The patient was already very ill when I saw him, and I have no doubt that, without the antivenomous serum, he would have died.
“On the following day he had recovered, and I saw him again three months later; since then he has not experienced any functional trouble.”
XIX.—Case recorded by Mr. H. W. Peal, Indian Museum, Calcutta (Indian Medical Gazette, July, 1903, p. 276).
“On April 1, 1903, at 7.30 p.m., a man was bitten at Dhamra, in Orissa, by a sea-snake which had been caught in a fishing net. He was not brought to me until 2.30 the next day, when he was in a state of collapse, semi-unconscious, and unable to speak, with eyes dull and almost closed. The bite was on the third finger of the left hand, just above the first joint. The finger was swollen, tense, and stiff. I gave the man an injection of 5 c.c. of antivenene ten minutes after he was brought to me. Three or four minutes after the injection the man with some assistance was able to sit up, and said he felt much better. He complained of great pain at the back of the neck and also in the lumbar region. He was able to speak fairly coherently after a little time. His eyes were brighter and he seemed to be aroused from his lethargy.
“I had about one hundred living sea-snakes with me, belonging to the three genera Enhydrina, Hydrus, and Distira. He identified Enhydrina valakadien as being the snake which bit him; so did the men who were with him. The snake was said to be about 3½ to 4 feet long.
“The antivenene did the man so much good, that he himself 339 asked me to give him a second injection. This I gave him at 2.25 p.m. (5 c.c.).
“Date on bottle used, May 8, 1900.
“The pains in the joints had disappeared on the second injection (which was given in opposite flank). At 5 o’clock the man walked away with assistance. He was quite well a couple of hours after the second injection, and when I saw him again on May 8 he was in perfect health.”
XX.—Case published by Dr. Marchand, of des Montils, Loir-et-Cher (Anjou médical, August, 1897).
“About 11 a.m., on Friday, July 23, Jules Bellier, aged 26, was mowing in a damp spot, when he was bitten in the heel by a large viper (Vipera berus). The bite, which was deep, was situated on the outside of the foot, 1 cm. behind the malleolus and 3 cm. above the plantar margin; at this point there were two punctures in the skin, 1 cm. apart. Directly after the accident the patient left his work, tied his hankerchief tightly round the lower third of his leg, made the wound bleed, and came to me with all speed, hopping on one foot for about a kilometre. When I saw him scarcely twenty minutes had elapsed since the accident; his general appearance was altered, and his pulse rapid. The patient had vomited twice; he complained of pains in the head, and of general weakness, and ’was afraid,’ he said, ‘of fainting.’ The foot and leg were painful under pressure; a slight tumefaction was visible in the peri-malleolar region, around the bites, which bled a little. Forthwith, after washing the wound freely with a solution of permanganate of potash, I injected 10 c.c. of Calmette’s serum into the antero-external region of the middle part of the thigh; then I enveloped the leg in a damp antiseptic dressing as high as the knee. The patient breathed more freely and plucked up his spirits. After lying down for quarter of an hour he went home on foot (he lives a hundred yards from my house). 340
“In the evening I saw my patient again. He was in bed, with a temperature of 37·2° C.; pulse 60; no malaise, no headache, no further vomiting; he had taken a little soup, and a small quantity of alcoholic infusion of lime-tree flowers. He complained of his leg, which was swollen as high as the knee; the pain was greater in the calf than at the malleolus. I applied a damp bandage. The patient had a good night, and slept for several hours, but still had pain in the leg. On the following morning, July 24, I found him cheerful, with no fever, and hungry. Around the bite the œdema had become considerable, and had extended to an equal degree as high as the instep; the calf and thigh were swollen, but to a much less extent. I gave a second injection of 10 c.c. of antivenomous serum in the cellular tissue of the abdominal wall. The day was good; indeed, the patient had no fever at any time; the spots at which the injections were made were but very slightly sensitive on pressure. In the evening the general condition of the patient was satisfactory; he complained most of his calf. Thinking that a contraction was possible, due to his having hopped along quickly on one leg after the accident, I ordered him a bath.
“On July 25, the second day after he was bitten, the only symptom still exhibited by the patient was a somewhat considerable amount of œdema in the peri-malleolar region and lower third of the leg. This œdema was slowly and gradually absorbed on the following days.
“Remarks.—(1) At this season of the year viper-bites are both frequent and dangerous in this district of the Loir-et-Cher. A year never passes without several cases occurring, and it has very often happened that deaths have had to be recorded in spite of the most careful treatment.
“(2) The therapeutic effect of Calmette’s serum was rapid and efficacious; the injections did not cause any pain or febrile reaction.
“(3) The œdema resulting from the bite was a long time in being absorbed; this, indeed, was the only remarkable symptom after the injection of the serum.” 341
XXI.—Case recorded by Dr. D. Paterne, of Blois (Anjou médical, September, 1897).
“My confrère and friend Dr. Marchand (des Montils) published in last month’s Anjou médical an interesting case of viper-bite, cured by Calmette’s serum. May I send you particulars of another case, which can only increase the interest of the one that you have already published? The facts are as follows:—
“Léon Bertre, aged 55, living at 17, Rue du Puits-Châtel, Blois, professes to be a snake-charmer, and really catches and destroys large numbers of dangerous reptiles in the vicinity of Blois.
“On Sunday, the 30th of last May, he went among the rocks of the Chaussée Saint-Victor on his favourite quest, and soon returned with ten large female vipers, and amused himself by exhibiting them to a group of interested spectators in an inn. A dog came up and began to bark. Bertre, whose attention was momentarily distracted, ceased to fix his gaze on the vipers, one of which, being no longer under the influence of its fascination, bit him on the back of the right hand, between the metacarpals of the thumb and index-finger. (I here reproduce the account of the occurrence as I received it from the snake-charmer’s own lips.) Bertre immediately felt an acute pain; his hand swelled up almost suddenly, and, since he was perfectly aware of the seriousness of what had happened, he ran with all speed in the direction of my consulting-room. The unfortunate man, however, had hardly gone 200 metres, when he fell insensible on the highway. He was brought to me, and Dr. Moreau, of Paris, locum tenens for Dr. Ferrand, of Blois, who was away, rendered first aid. He washed the wound, dressed it with perchloride of mercury, and injected 10 c.c. of Calmette’s serum into the right flank. The accident took place a little before 5 p.m., and the injection was given about 6 o’clock.
“Dr. Moreau, who was interested in the case, asked me to take charge of it, which I gladly consented to do. The patient’s general condition was very grave, since he remained two days and two nights without regaining consciousness. 342
“On Tuesday, June 1, about 11 o’clock, Dr. Moreau gave a second injection of 20 c.c. Considering the condition of the patient, we hardly hoped for a successful result. To our great surprise, however, the patient regained consciousness about 3 p.m., and the improvement progressed rapidly.”
XXII.—Case recorded by Dr. Thuau, of Baugé (Anjou médical, September, 1897).
“X., a young man of Volandry, a parish 10 kilometres from Baugé, was bitten in the heel at 10 a.m. on the 6th of last August, by an aspic, about 50 cm. in length, while engaged in harvesting. He at once had himself taken to Baugé, knowing that there was an antivenomous serum dispensary there, and about noon he arrived at the house of my confrère and friend Dr. Boell. The latter, in view of the grave symptoms exhibited by the patient (nausea, vertigo almost amounting to syncope, pain in the chest, profuse sweating, &c.), gave him, with all the customary precautions, a first injection of 10 c.c. of Calmette’s serum in the flank. After about half an hour, since the alarming symptoms did not appear to diminish, he did not hesitate to give a second injection of 10 c.c., and then had him sent to the Baugé Civil Hospital, where he came under my care about 3 p.m. I then found that this young man had been bitten in the left foot, a little below the external malleolus, midway between the latter and the plantar margin. The snake’s fangs had penetrated rather deeply; the two little wounds were about a centimetre apart. About this time the patient experienced great relief, and his general condition continued rapidly to improve. The axillary temperature was 37·8° C.
“Locally the patient complained of somewhat acute pain in the entire foot; the latter was purple and greatly swollen, and the swelling had affected the whole of the lower leg and extended to a little above the knee. I made a slight incision in the region of the two wounds caused by the bite, made the place bleed a little, and washed it with a solution of permanganate of potash, advising 343 that the dressing should be changed several times a day. In the evening the temperature was 37° C., and never varied again from the normal until recovery was complete on August 25.
“The two injections of antivenomous serum did not produce any painful or inflammatory reaction.”
XXIII.—Case reported by Dr. Clamouse, of Saint-Epain, Indre-et-Loire.
“Léonie C., a servant at a farm, aged 19, bitten on June 1, 1900, by a red viper on the dorsal face of the left ring-finger. Somewhat serious symptoms of intoxication. In default of serum, injection of Labarraque’s fluid, 1 in 12. Serum obtained from Tours was injected at 11 p.m. on June 2, thirty-eight hours after the accident.
“On the morning of June 3, very marked improvement. On June 7, general condition excellent. Recovery.”
XXIV.—Case reported by Dr. G. Moreau, of Neung-sur-Beuvron, Loir-et-Cher.
“A. B., aged 12, living at Villeny, in the canton of Neung-sur-Beuvron (Loir-et-Cher), was bitten on June 23, 1900, on the left external malleolus. The parents contented themselves with applying a ligature above the wound, and did not bring the child to me until 12.15 p.m.
“Tumefaction of the entire foot. Ecchymosis of the skin extending half-way up the leg. General condition excellent. I gave antiseptically an injection of 10 c.c. of antivenomous serum in the right flank, followed by a draught of acetate of ammonia and syrup of ether. Damp bandage applied to wound and swollen part.
“I saw the child again on the following day. Generalised œdema and tumefaction of the bitten limb. Heart excellent; no vomiting, no fever. I again gave an injection of 20 c.c. of serum, and ordered a continuance of damp phenic dressings to be applied to the entire limb. Condition very good.
“On June 25, no fever at the time of my visit. Pulse irregular. Ordered treatment to be continued. 344
“I did not see the patient for four days, when I was summoned by telegram. I found that the child had fever, 39° C. Complete tumefaction of the left leg, abdomen, and trunk, with ecchymosed patches. Prescribed quinine. Arhythmia of pulse and heart. Prescribed digitalis and Jaccoud’s tonic. The febrile condition was due to congestion of the base of the right lung. I ordered cupping and sinapisms alternately.
“I saw the child again two days later. The congestion still continued. Temperature 39° C., but the general tumefaction showed a tendency to diminish.
“On July 4 I again saw the child. Now only slight œdema. General condition very satisfactory. Temperature normal. The child was making rapid strides towards recovery. The leg was doing well, and the wound was almost healed.
“Summary: A very serious bite and, above all, great delay in injecting serum (injection not given until four hours after the accident); unforeseen complications in the lung, by which recovery was delayed.”
XXV.—Case reported by Mons. H. Moindrot, Assistant to Dr. Martel, of Saint-Étienne (Loire).
“Claude L., aged 8, living at Ricamarie, was brought, on May 26, 1904, to the Bellevue Hospital. The parents stated that about 10 o’clock the same morning, while playing near a stack of faggots, the child was bitten by a snake in the third finger of the right hand. Since the wound caused by the bite seemed to them of little importance, they contented themselves with squeezing the injured finger in order to make it bleed a little. A few moments later, however, the child began to complain of a feeling of distension in the region of the bite, caused by œdema, which soon increased to an alarming extent. A doctor, who was called in, carefully washed the wound, applied an aseptic dressing, and at once sent the little sufferer to the Hospital.
“On admission, enormous œdema, including fingers, hand, entire right arm, cervical region on the same side, and the anterior 345 face of the thorax, nearly as far as the inner margin of the false ribs. This œdema was not very painful, though fairly tense, yielding but slightly to pressure. In the affected region the skin was cold, of a dull livid colour, with a few ecchymosed patches. In the bitten finger, a small wound with no special characteristics.
“General condition bad; the child was unable to stand. He was indifferent to what was passing around him, merely groaning a little when examined. The pulse was feeble, thin, and easily compressible; it was also very irregular. The extremities were cold. Lungs: nothing abnormal on auscultation, rapidly performed, it is true. Respiration, however, was distinctly accelerated, 30 per minute. Temperature not taken on admission. No urine passed since the accident.
“The patient’s condition being so alarming, not to say desperate, we thought it almost useless to have recourse to Calmette’s method, more especially since at least seven hours had already elapsed since the child was bitten. Nevertheless we gave a hypodermic injection of 20 c.c. of Calmette’s serum. At the same time the wound was crucially incised, and bathed with a 1 in 1,000 solution of permanganate of potash, after which a damp dressing was applied to the whole of the swollen limb. The patient was put to bed, and kept warm. He was given an injection of 50 centigrammes of caffeine, and 300 grammes of artificial serum. In the evening the temperature was 36·8° C.
“May 27.—General condition more satisfactory; pulse still weak, but less irregular. Persistence of dyspnœa, explained by a series of small râles at the bases of both lungs. This morning the little patient passed his urine, about 200 grammes. He is more lively, and replies better to any questions addressed to him.
“May 28.—The improvement continues; the dyspnœa has almost entirely disappeared; only a few râles are still heard at the extreme base. The pulse is stronger and remains regular. The secretion of urine gradually reappears. The temperature of the extremities has become normal. 346
“On May 29 and following days the œdema continued to diminish, and had totally disappeared eight or ten days later.
“Recovery was complete by about June 15. The child was discharged on June 23, 1904.
“It seemed to us worth while to report this case, in order to emphasise the conclusion that forces itself upon us, namely that in all cases of bites from poisonous snakes an injection of Calmette’s serum should be given, without considering the efficacy of this therapeutic agent as being rendered doubtful by the length of time that may have elapsed since the bite was inflicted.
“In the present case, as we have seen, there was extensive intoxication, which had seriously affected the functions of the various organs, since we found cardiac arhythmia and pulmonary œdema, and that the patient was threatened with collapse, algidity, hypothermia, and anuria. Impregnation by the virus having continued for seven hours, we might have felt ourselves justified, on the one hand in merely employing the proper means for the relief of the general condition, on the other hand in treating the local condition, without having recourse to the serotherapeutic method, that seems to us in this case, in so far as it is permissible to make such a statement, to have been the determining factor in the recovery.”
XXVI.—Case recorded by Dr. Lapeyre, of Fontainebleau (from L’Abeille de Fontainebleau of June 27, 1902).
“M. X., who arrived at Fontainebleau on Sunday morning with a friend, keeps grass snakes at home, in Paris; he finds his hobby as good a means as any other to remind him of the forest and its charms. Human nature includes all kinds of tastes, so that this particular one need not be further discussed.
“The journey, therefore, had a twofold object: firstly to spend a whole day in sunshine and in the open air, and secondly to catch grass snakes to add to the collection.
“On leaving the train, our Parisian walked up the Amélie Road, and saw a snake under a rock. Never doubting that it was one 347 of the kind that he knew so well, to kneel down, pass his left arm into the hole, and seize the snake, was the work of a moment; he quickly succeeded, even better than he intended, for instead of his seizing a grass snake, the viper bit him so hard in the left forefinger, that he could only make it let go by pulling it off with his other hand. Well knowing that he had been dangerously bitten, he went down to the Station Road to get the wound dressed, after which, thinking that all necessary precautions had been taken he returned to the forest, but soon felt uncomfortable. His arm and then his body swelled up, and he was seized with vomiting. It was time to go to Fontainebleau to seek medical assistance, for he had acute pain in the abdomen and stomach, his tongue was swollen, and his body was turning black.
“Accompanied by his friend he reached the town. His condition becoming more serious every moment; the injured man was carried into a hotel, where Dr. Lapeyre administered injections of antivenomous serum. After three hours—the same period as had elapsed between the accident and the first treatment—the general condition of the patient, which had never ceased to be alarming, showed marked improvement. By the end of the day he appeared to be out of danger, and left for Paris on Tuesday evening, delighted at having got off so cheaply.”
XXVII.—Case recorded by Lieutenant C. C. Murison, I.M.S. (Indian Medical Gazette, May, 1902, p. 171).
“G. W. R., a Mahomedan, aged about 12, was admitted into hospital on March 10, 1902, at 9.30 p.m., having been bitten by a snake on the dorsum of the right foot an hour and a half previously. The snake was killed by his sister, and was subsequently identified at the Research Laboratory, Bombay, as an Echis carinata (Phoorsa).
“I saw the patient at about 9.45. The dorsum of the foot was 348 swollen, and the swelling extended above the ankle-joint. The knee reflexes were very exaggerated, and the boy was somewhat drowsy. Since he was gradually getting worse, I decided to inject 5 c.c. of Calmette’s antivenene. I got the hospital assistant under my supervision to inject it into the right calf, to cauterise the bite with silver nitrate, and to apply a 1 in 40 carbolic poultice. Very soon (fifteen minutes) after this the pain in the thigh, which had reached to the right groin, began to disappear. During the night the patient was very sleepy, and the attendants had great difficulty in keeping him awake.
“March 12.—This morning the patient is much better; there is still considerable swelling of the foot, but the pain is much less. All other symptoms are gone.”
XXVIII.—Case reported by Surgeon-Captain Sutherland I.M.S., Saugor, C.P., India.
Case of a woman bitten on the finger on July 22, 1898, by an Echis carinata. Treated six hours later with 10 c.c. of serum. Recovery.
XXIX.—Case reported by Dr. Moudon, of Konakry, French Guinea.
“On December 9, 1898, a Foulah woman, eight months pregnant, was collecting wood when she was bitten in the heel, behind the internal malleolus of the right foot, by a snake which, from the description given, must have been a Horned Viper. When I saw her, four hours after the accident, the whole of the lower leg was swollen and painful. The swelling extended to the groin, and the patient complained of vertigo and nausea. I immediately gave her an injection of 10 c.c. of antivenomous serum in the right flank, followed by a second injection at 10 p.m. Ten days later, with the Commandant of the Fulton, I saw her again at her village; she had no symptom of malaise, and the pregnancy was taking its normal course.” 349
XXX.—Case of a bite from a Horned Viper reported by Dr. Mons, in charge of the Military Hospital of Laghouat, Algeria.
“Mohamed ben Naouri, a day labourer, aged 26, during the summer catches Horned Vipers, which he stuffs and sells.
“On August 3 a Cerastes, which he was holding down on the sand with a forked stick, disengaged itself and fastened on his hand. The snake was a large one, about 50 cm. in length.
“The accident happened at 6.30 a.m., 6 kilometres from Laghouat, and the man was bitten on the joint between the second and third phalanges of the third finger of the right hand. He applied a ligature to his wrist, and started to run as fast as he could towards the Military Hospital, where he arrived an hour later.
“He was immediately given an injection of antivenomous serum, in accordance with the instructions, and, around the bite, five or six injections of permanganate of potash, 1 in 20. On the next and following days, tense œdema of the arm and left side of the chest. Extensive purplish ecchymosis of the inner face of the arm; no fever. The phenomena gradually diminished, and, on August 17, there was merely a trifling wound where the bite had been inflicted. The patient was discharged at his own request.
“Like Dr. Marchand (des Montils), we can certify that the action of Calmette’s serum was rapid and efficacious. The injection did not cause any pain or febrile reaction.”
XXXI.—Case reported by Dr. Blin, of Dahomey.
“On March 5, 1906, native hospital attendant C., while gathering vegetables in the hospital garden, was bitten in the right hand by a Cerastes. The bite was inflicted in the tip of the index finger. Ten minutes later a ligature was applied to the base of the finger and another to the upper arm, and as soon as we saw the man, which was after the lapse of about an hour, he was given an injection of 10 c.c. of antivenomous serum. The patient complained of feeling cold and vomited. The axillary temperature was 36·1° C.; the pulse was weak, irregular, and rapid. The 350 finger and hand were swollen. A few minutes later we gave a second injection of serum. Until evening (the accident took place at 11 a.m.) the patient suffered from nausea, but sweating set in, and at 7 o’clock the temperature had risen to 36·7° C. The feeling of depression was much less. On the following day the symptoms had disappeared, and forty-eight hours afterwards the patient returned to duty.”
XXXII.—Case reported by Dr. P. M. Travers, Chilubula Mission, North-eastern Rhodesia.
“On Thursday, September 6, 1906, information was brought to me that a child, aged 7 or 8, in inserting his hand into a mole’s hole had been bitten by a lifwafwa (’Death-Death,’ i.e., Puff Adder). I set off in all haste on my bicycle. An accident obliged me to leave the road when half-way, and, to complete the series of mishaps, I went to a village with a similar name, a good half-hour distant from that where the patient lived. The result was that by the time I arrived I should say that about two hours had elapsed since the child had been bitten. The snake had been killed, and was, indeed, a puff adder. It had bitten the child in the middle finger of the right hand, and half the arm was greatly swollen, and as hard as stone. As quickly as possible I gave an injection of 10 c.c. of antivenomous serum, and then vainly endeavoured to make the wound bleed. In a very short time the serum was absorbed. On the following morning the child was still ill, with wild eyes resembling those of an epileptic. He yawned continually, and did not seem altogether conscious; the inflammation, however, had greatly diminished. A few days later recovery was complete, but a large abscess formed on the forearm, and the hand became necrosed. I was obliged to amputate all the phalanges. The natives said the child was going to die during the night. In my opinion the serum saved the child’s life, and recovery would have been more rapid had I not been so late in arriving.” 351
XXXIII.—Case reported by Dr. Lhomme.
“In May, 1898, A., aged 48, a European convict undergoing sentence, was admitted to the Penitentiary Infirmary, of Roches de Kouvous (French Guiana), suffering from a poisonous bite.
“The man had been bitten while engaged in felling timber, at the place called Passouva. The locality is one that is infested with snakes, especially at the end of the wet season. Two venomous species in particular are found there in considerable numbers, the rattle-snake and another called the Grage by the blacks, which appears to be a Lachesis.
“The patient arrived at the Infirmary in the evening, about twelve hours after the accident. We endeavoured to obtain precise details, but, owing to special circumstances, A., who was alone, had been unable to see what animal had bitten him. The clinical signs, however, pointed to a venomous snake; inflammatory phenomena and pain set in soon after the wound was inflicted, and in a very short time became acute.
“Clinical Signs.—On examining the patient we found that the whole of the right arm was swollen. The skin, which was of a dark red colour, was acutely inflamed. The slightest touch or the least movement caused the patient to cry out. The hand showed traces of the bite, in the shape of two small red marks, each surrounded by a bluish areola. The general condition was good. The thermometer, however, indicated a slight rise of temperature, and the pulse seemed a little soft. The urine on being examined on the day after the accident contained a small quantity of albumin. Organs normal. General health before the accident excellent. Nothing worth mentioning in the previous history.
“Progress.—The conditions that we have just described disappeared very quickly, once the treatment was applied. The pain soon ceased, the temperature fell, and the patient was able to get a few hours sleep. By the following day the inflammatory phenomena had noticeably diminished. The œdema of the forearm 352 and hand, however, persisted for some time, though there was no formation of pus. On the third day after the accident the albumin had completely disappeared from the urine. Finally, after the lapse of a fortnight, the condition became normal, and the convict, who had recovered the entire use of his arm, was able to resume work.
“Treatment.—On admission to the Infirmary, A. received a hypodermic injection in the thorax of the contents of a bottle of antivenomous serum. At the same time he was given tonics (alcoholised coffee). The injured limb was placed in a hot phenic arm-bath. These baths were continued on the following days, alternately with damp dressings. Milk diet, and daily aperients.
“We saw the patient again more than a year after the accident; his recovery had been complete; there was no loss of power in the arm whatsoever, and he had never suffered from the nervous troubles mentioned by some authors as a complication ensuing after a long interval, and attributed by them to the antitoxic serum.”
XXXIV.—Case reported by M. Jean, Veterinary Surgeon of Artillery in Martinique.
“C., a negro, aged 26, employed in the artillery quarters at the Rivière d’Or, was bitten in the right leg by a snake measuring about 1 metre in length, which he declared was a Trigonocephalus. The patient came to me twenty minutes after the accident. The marks of the bite were clearly visible a hand’s breadth above the external malleolus. The wounds were inflamed, and appeared as two small red spots 1·5 cm. apart, from which a few drops of serum were exuding. I did not notice any congestion. The patient, however, complained of a feeling of weight in the leg, and supported himself upon the sound one. After making the man lie down upon a bed, I applied a tight ligature above the bitten part, and, with a penknife passed through a flame, I endeavoured to incise the wounds. The instrument, however, was blunt and I obtained but little blood. 353
“The treatment prescribed in Dr. Calmette’s directions was then strictly followed. With the usual antiseptic precautions, I made several hypodermic injections of the solution of hypochlorite of calcium round the bite, and injected the dose of serum indicated in two places in the abdominal wall. The patient was then vigorously rubbed and covered with woollen blankets. He was made to take two cups of a strong infusion of black coffee. Since he could not be induced to go to hospital, he was carried half an hour later to his home, where he placed himself in the hands of a ‘dresser.’
“According to information furnished by Captain Martin, who lived on the spot and was able to follow the course of the case, the patient remained throughout the day in a state of profound prostration, and had several attacks of syncope. The injured limb was greatly swollen, and the swelling, which extended to above the knee, produced a mechanical difficulty in using the joint, leading to a belief that paralysis was setting in. During the first five days the condition of the patient was so alarming as to cause a fatal issue to be apprehended. By degrees these symptoms diminished, until they disappeared about the fifth day.
“On the twentieth day, C. returned to his work. I saw him again a month later, when he was in perfect health; his leg had returned to its normal size, and all that remained were two small fibrous nodules showing where the bite had been inflicted.”
XXXV.—Case reported by Dr. Gries, Fort-de-France, Martinique.
“On June 21, 1896, a young black, who had just been bitten in the foot by a Bothrops of large size, was brought to the Fort-de-France Hospital. The entire limb was swollen and benumbed.
“Two hours after the accident I gave an injection of 10 c.c. of serum in the abdomen, and the patient was taken back to his family. I saw him again ten days later, and found that he was quite cured. His friends stated that recovery had taken place much more quickly than could have been hoped after so serious a bite, and without the usual complications.” 354
XXXVI.—Case reported by Dr. Gries, Fort-de-France, Martinique.
“About 7 a.m., on November 25, 1896, G., aged 23, a fusilier belonging to the disciplinary battalion, was bitten by a Bothrops at Fort Desaix under the following circumstances. One of his comrades had just caught the snake, and was holding its head down on the ground by means of a forked stick applied to the neck. G. passed a running noose round the reptile’s neck, but, his comrade having withdrawn the fork too soon, the snake had time to dart at him and bite him in left thumb. At the moment when he was bitten the man was squatting, but he quickly stood up, carrying with him the snake, which remained for a few seconds suspended from the thumb by its fangs, and did not let go until its victim had struck it on the head with his fist. G. immediately ran to one of his officers, who applied a tight ligature to the base of his thumb, and sent him off to the hospital, where he arrived on foot and quite out of breath, ten or twelve minutes after the accident. He was at once given a hypodermic injection of 10 c.c. of antivenomous serum in the left flank; the thumb was washed with a 1 in 60 solution of hypochlorite of calcium, after which the ligature was removed. A few moments later, thinking the case a serious one, I caused a second injection of 10 c.c. of serum to be given in the right flank.
“Immediately after being bitten the patient experienced complete loss of sensation in the limb, as far as the middle of the arm. About 9 a.m. he complained of acute shooting pains in the hand. At 11 o’clock the limb was still benumbed, but by degrees sensation returned. Profuse sweating.
“On November 26 sensation was restored in the whole limb; no inflammatory phenomena. The patient was perfectly well.
“The Bothrops on being brought to the hospital measured 1 metre 47 cm. in length.”
XXXVII.—Case reported by Dr. Lavigne, Colonial-Surgeon at Fort-de-France. 355
“At 7 a.m. on January 19, 1897, G., aged 22, was going along a footpath near Trouvaillant, when he was bitten in the left external malleolus by a Trigonocephalus which was rutting (a circumstance which, according to the natives, aggravates the character of the bite).
“After killing one of the reptiles (the other having escaped), the young man made his way to the detachment of gendarmery stationed close by. The officer in command applied a ligature to the upper part of the leg, cupped the man a few times, and sent information to us at the Military Hospital. On reaching the spot at 9.15 we found, on the postero-inferior surface of the left external malleolus, two small wounds resembling those caused by the bite of a snake. The leg was swollen and painful, and the patient could hardly put his foot to the ground.
“At 9.30, after taking the usual antiseptic precautions, we gave an injection of Calmette’s antivenomous serum, from a bottle dated December 26, 1896. Not having any hypochlorite of calcium at our disposal, we washed the wound with a 1 in 60 solution of hyposulphite of soda, and applied a dressing of carbolic gauze. An hour later the patient was taken to Saint Pierre in a carriage. Temperature 37·2° C. No vomiting, or tetanic phenomena. In the afternoon the pain was less acute, and the œdema seemed to have diminished a little. Mercurial ointment rubbed in.
“Four days later the patient, being cured without having had the least rise of temperature, proceeded to the country.
“This case is interesting, since a single dose of antivenomous serum (20 grammes), injected two hours and a half after the accident, sufficed to cure a young man bitten by a Trigonocephalus measuring 1 metre 20 cm. in length.”
XXXVIII.—Case recorded by Dr. P. Renaux, of Piriapolis, Uruguay (La Tribuna popular, Piriapolis, December 14, 1898). 356
Silverita, aged 20, bitten in the ankle by a Crotalus, on December 7, 1898. Symptoms of serious intoxication. Treated with a dose of antivenomous serum, injected half in the right flank, half in the left. Recovery.
XXXIX.—Case reported by Dr. Maclaud, of Konakry, French Guinea.
“A hound belonging to the Governor of Konakry was bitten in the ear by a black Naja. A similar accident happened last year, and the animal died on the fifth day. In the present case, serious phenomena had already manifested themselves: depression, convulsions, and great swelling of the entire head and anterior portion of the trunk. A dose of 10 c.c. was injected at three different points: in the flank, neck, and cellular tissue of the injured ear. Improvement was almost immediate. On the following day the animal recovered its appetite, and two days later was completely cured.”
XL.—Case reported by Dr. Marotte, of Konakry.
“At 10 a.m. on November 1, 1898, a large dog (a German brach), weighing 33 kilogrammes, was running about in some tall grass. It returned to its master, looking unhappy, with its eyelids swollen. Thinking that his dog had been bitten by a Naja, a snake which swarms round Konakry, he took it to the hospital. The animal was unable to cover the distance, which was only 300 metres; it was dragged along, but collapsed, and had to be carried. Its head was swollen, it panted, and its breathing was rapid and irregular; there was profuse salivation. On the inner face of the right ear the marks of the two fangs of the reptile were distinctly visible. 357 A hypodermic injection of 10 c.c. of antivenomous serum was immediately given in the right flank: this was at 10.10 a.m. At 11 o’clock the symptoms appeared to become less acute; the animal was easier, and its breathing became less rapid and more regular. At 1 o’clock the animal succeeded in getting on to its feet; the œdema had somewhat diminished, and it was able to half open its eyes. At 6 o’clock the dog was taken back to its master’s house; it seemed just as lively as though nothing had happened.
“On the following morning there was still a small amount of œdema in the eyelids, but the appearance of the head had become normal.”
XLI.—Case reported by M. de Maupas, of Challay, by Trôô.
“About 1 p.m., on July 30, 1898, a fairly large pointer was brought to me, bitten in the right paw. On shaving off the hair the marks of the two fangs of the snake were clearly visible. The wound had bled a little, and the injured limb was painful and swollen. I ligatured the paw above the joint, and injected a bottle of antivenomous serum, which had been in my possession since September 6, 1897. The effect was very rapid; after a quarter of an hour the dog reopened its eyes, which until then had been half closed. Towards 5 o’clock I removed the ligature. On the next day but one the swelling had almost disappeared, and the dog took its food of its own accord; it ran about and seemed lively.”
XLII.—Case reported by M. de Villiers, Mayor of Villiers-le-Duc, Côte-d’Or.
“About 2 p.m., on May 23, 1898, the widow Veillard, of Villiers-le-Duc, while driving her herd of cows to pasture among the brushwood, saw a viper which she killed, and then, a few moments later, three others in succession, which she likewise killed. Madame Veillard thought of leaving this place, which seemed to her to be too much infested, when one of her cows, which was 358 quietly feeding, swerved violently and dashed off through the scrub. With the help of her dog she succeeded in recovering the cow, which appeared uneasy, ceased to feed, and had a swollen muzzle. Madame Veillard then decided to return to the village.
“Towards 6 p.m., that is to say about three hours after the accident, I was sent for. The head and tongue of the cow were swollen, there was foam on the mouth, and the animal had difficulty in breathing. I injected a dose of antivenomous serum, from our first-aid station, beneath the skin of the shoulder, and gave a second injection a few moments later. The cow was then led back to her shed.
“At 9 p.m. she was quiet. The swelling did not increase, and the cow, which had previously refused all food, ate a handful of hay.
“On the following morning there was nothing to be seen beyond a little swelling in the neck. The animal fed as usual, and gave her normal quantity of milk.”
XLIII.—Case reported by M. P. Rat, engineer, of Saint-Rambert-en-Bugey, Ain.
“About 2 p.m. on Sunday, September 7, 1902, while I was out shooting, my bitch was bitten in the lower lip by a very large viper. Since I was a long way from home, I was unable to give the injections until 6 p.m., by which time the poor beast was in a pitiable condition. Her head was as large as a saucepan; she had no strength left, trembled, and was unable to stand. I injected 15 c.c. of serum in the left flank. This was all that I did.
“At 7 o’clock on the Monday morning the bitch ate and drank a little. She began to walk about, and by the evening had completely recovered. There was nothing but a very little swelling left.”
XLIV.—Case reported by Dr. Boyé, of Kissidougou, French Soudan.
“At Kan-Kau, on December 19, 1896, a cow belonging to the 359 post, which had been bitten by a snake at 8 a.m., seemed about to die. The snake, which was killed by the herdsman, was a Cerastes (Horned Viper).
“On going to the cattle-shed I found the animal on her side and panting, with the limbs completely relaxed. A thick foam was dropping from the half-open mouth, and asphyxia seemed imminent. The cow had been bitten in the teats, which were enormously swollen; the œdema extended over the whole of the belly and inner face of the thighs. Two doses of serum were injected, one at the base of the teats, the other in the subcutaneous tissue of the flank.
“On the following morning the œdema had diminished, and the breathing was much easier; the animal seemed to be conscious of what was passing around it.
“Forty-eight hours later the cow was able to walk and went out to graze, having apparently entirely recovered from the accident.”
From the time of our arrival in India, in February, 1901, we turned our attention to ensuring the regular collection of snake-venoms, with a view to satisfying the desire for them that had been expressed by our friend Professor Calmette, Director of the Pasteur Institute at Lille.
To gain this end, the moral and financial support of the Administration was indispensable, and this did not fail us. Acting on our suggestion, as formulated by letter on May 25, 1901, Governor Rodier, on June 11, issued an order by the terms of which a sum of 200 rupees was placed at the disposal of the 360 Chief of the Sanitary Service, “with a view to encourage the catching of poisonous snakes, and to assist the collection of snake-venom by the granting of bounties to natives.”
On the issue of this order, we drew up instructions for the collection of venom, which we caused to be distributed to all posts in the Colony. At the same time an appeal in the vernacular was posted up in each of our sanitary institutions, inviting the Hindus to commence the campaign forthwith. The notice placarded on the door of the Pondicherry Hospital ran as follows:—
“For the public good the Governor requests the population to capture poisonous snakes in all places where they are found; to take them alive if possible; and to bring them to the Hospital without removing their fangs.
“One rupee will immediately be paid for each snake brought in.
“Those who read this notice are requested to communicate it to their acquaintances.”
The Indian snake-charmers at once responded to this appeal, and the harvest of snakes was soon abundant. The first provision of 200 rupees, allotted to us by the Administration in June, 1901, had to be successively renewed in May and October, 1902. Since then, on the proposition of our successor, Dr. Camail, this sum of 200 rupees has been included in the local budget, thus definitively sanctioning the principle of the collection of venom in our Indian Settlements.
The venom forwarded by us to France has been exclusively derived from the cobra, or Naja tripudians. Other venomous serpents are found in the French Settlements in India, especially the species of Hydrophis, or sea-snakes; but the cobra is by far the commonest species, and our trading stations teem with it. Yanaon is infested by it at all seasons, but especially at the time when the Godavari is in flood; the reptiles then make their way towards 361 spots spared by the inundation, and the natives frequently kill them in their houses. Thus M. Mariapregassam, the Sanitary Officer in charge of the the Medical Service of this dependency, was in a position to procure for us, at the very commencement of our operations, important doses of venom: it should be added that he acquitted himself of this task with a perseverance and devotion worthy of the fullest recognition. Of the 653 cobras captured between August 1, 1901, and February 23, 1903, 229, or more than one-third, were furnished by Yanaon. Moreover, the cost price of the cobra fell in proportion to its numbers, and Yanaon paid for its snakes on the average at the rate of 33 centimes apiece; while at Chandarnagar and Pondicherry the snake-charmers frequently received 1 rupee (1 franc 67 centimes) per reptile, though it is true that even this was an extremely poor remuneration, when we consider the risk of the calling.
Again, owing to the limited amount of our grants, we were obliged to restrict our expenditure, regulating the purchase-price of the snakes according to the quantity of venom collected. Thus at Pondicherry, having observed that each cobra yielded on an average twenty drops of venom, we fixed the value of two drops at one fanon (one-eighth of a rupee), never more. In this way it was decidedly to the snake-charmer’s interest to bring us fresh snakes, and not such as had been previously deprived of their venom.
In spite of this new regulation, snakes continued to pour into the hospital, and several were repeatedly brought to us at the same time; in October and November, 1902, the figure 9 appears three times in our statistics.
The combined results of the snake-harvest at our five settlements, from August 1, 1901, to February 23, 1903, are shown in the following table:—
| Number of cobras |
Sum expended |
Quantity of venom collected |
Averagen weight of dry venom per cobra |
Average price of the cobra |
Average price of 1 gramme of venom |
|---|---|---|---|---|---|
| 653 | R. 446 | 242 | 0·37 | R. 0.68 | R. 1.84 |
| grammes | grammes | (1.13 fr.) | (3.07 fr.) |
362
On an average, therefore, nearly three cobras are necessary to furnish 1 gramme of dry venom, since freshly collected venom weighs more, owing to the proportion of water contained in it, which is greater than half the total weight. Thus the quantity of fresh venom extracted from an average cobra may be estimated at about 80 centigrammes. It may be added that the product of the glands differs considerably in appearance, according as it is derived from a dead or living snake. In the former case it takes the form of extremely small, glistening lamellæ, of a golden-yellow colour, similar in appearance, when in bulk, to iodoform. Venom extracted from the living snake, on the other hand, is of an amber-brown colour, and forms much larger lamellæ, which are translucent and slightly elongate, resembling particles of gum arabic. When the product is impure and mixed with a small quantity of blood, it has a dull appearance, and is of a dark, dirty brown, almost black colour.
At the Pondicherry Hospital venom is collected in the following manner;168 The snake is brought by the snake-charmer in a chatty, a kind of earthern pot, covered with a rag, or half a calabash. Ensconced at the bottom of this receptacle, the reptile has a difficulty in making up its mind to emerge, which it does only after having been stirred up several times. Once it is outside the charmer forces the cobra to uncoil, and, while the animal is moving slowly along, fastens it to the ground by placing the end of a bamboo on its neck, quite close to the head. The Indian then cautiously seizes the head of the cobra with his forefinger and thumb, and, with a rapid movement, throws it into a jar prepared for the purpose, containing a few tampons of absorbent wool impregnated with chloroform. As soon as the snake is inside, an assistant quickly slips a metal plate over the opening of the jar and presses his hand firmly down upon it. In a few minutes the 363 animal is dead; it is then taken out of the jar and its mouth is swabbed out; next, the fangs having been raised by means of a probe, a saucer is placed between the jaws, and, by pressure exerted upon the poison-glands laterally and from behind forwards, the venom is made to spurt out. The fresh product obtained in this way is of a very pale yellow colour, and viscid. It is protected from the air and light until completely desiccated; then, when it is in sufficient quantity and distributed in flakes round the sides of the saucer, like the colours on a palette, it is cautiously detached with a spatula, taking care to protect the eyes against risk from flying particles of venom. After being placed in well-corked bottles, the product is despatched to France.
In spite of the precautions observed in the course of these proceedings, and although the venom has not been extracted until after the death of the animal, some regrettable accidents have taken place, as we shall show further on.
Cases of bites from poisonous snakes appear but seldom in the statistics of our settlements in India, and, contrary to what is the case in the neighbouring English possessions, hardly any deaths are officially recognised in the annual returns as being due to this cause. It is true that the death statistics are very badly authenticated, and that the natives frequently conceal the real cause of death.
We have been informed by Dr. Cordier, Surgeon-Major of the Sepoy Corps, that, during a previous tour of duty in Bengal, he had successfully treated two cases of cobra-bite with Calmette’s serum.
At the end of 1901 the following note was forwarded to us by Dr. Paramananda Mariadassou, Physician to the Karikal Hospital:—
“Case I.—In the month of November, 1901, a woman of robust habit was brought about midnight to the Karikal Hospital, in a semi-comatose condition. Her husband stated that an hour before, while lying on a mat stretched on the ground, she felt herself bitten 364 in the shoulder when in the act of placing her head on the pillow. With a start of surprise she half rose and then lay down again. It was only on being bitten for the third time that she made up her mind to seek for the cause; passing her hand beneath the pillow in the darkness she touched the body of a snake and cried out. Directly afterwards she pointed out to her husband, who had at once hastened to her, the snake coiled up against the wall; the man killed the reptile and burnt it on the spot. According to him the snake was about a metre in length, and as thick as all five fingers put together. The woman had barely time to tell her husband what had happened, for she speedily became unconscious, and was in this condition when brought to the hospital.
“On admission the following symptoms were observed: The patient did not reply to questions put to her, the eyelids remained closed, and the teeth clenched. On the right shoulder, a little on the inner side of the deltoid prominence, two or three punctures were distinguishable, marked by a small spot of coagulated blood. The respiration was normal, but the pulse was feeble and thready. When a pledget impregnated with ammonia was held under her nose the woman reacted, but immediately relapsed into the soporose condition.
“Two doses of antivenomous serum were at once injected, one in each flank: the patient seemed scarcely to feel the insertion of the needle. The wound on the shoulder was then washed with a fresh solution of hypochlorite of calcium, 1 in 60, and six injections, each of 1 gramme, of the same solution were given round the wound. The latter was covered with a pad of cotton-wool saturated with the solution, and two more doses of serum were injected into the flanks. As the result of this treatment, which altogether took nearly half an hour, the woman began to unclench her teeth, and to respond to her name. She was made to swallow a few mouthfuls of very hot black coffee. A little later she resisted when a fresh injection of serum was about to be given her. After having taken a second cup of coffee she sat up, opened 365 her eyes, and recognised those about her. She immediately asked to go home, but was detained for some time longer. After being carried from the operating table to a bed, she was wrapped in a blanket; a few minutes later profuse perspiration set in, and the patient felt so well that she was allowed to go.
“On the following day the husband came to thank us, and stated that his wife had quite recovered, and that, while she had been unable to sleep for the remainder of the night, it was rather from fear caused by what had happened than in consequence of the pain.”
Case II.—We feel it our duty to relate in detail the following case, having regard to the peculiar circumstances under which it took place.
Coupin, the son of Ponnin, aged 25, living at Carouvadicoupom, near Pondicherry, is a snake-charmer, and one of those who regularly supply us with venom. In the course of July, 1902, this man pointed out to us a shrub169 growing in one of the alleys of the hospital, to which he attributed so marvellous a power against the bites of poisonous snakes, that he offered to make a cobra bite him in our presence, little as we showed any desire to witness such an exhibition. We allowed him to talk, not wishing to encourage such an act of bravado.
However, on July 23 he returned to the charge, and, as we were leaving the hospital accompanied by our colleagues, he showed us a cobra which he had just caught, and declared himself ready to carry out the proposed experiment there and then. In the face of so much confidence and sang-froid, and, apart from this, being interested in learning the justification for the reputation for immunity possessed by Hindu snake-charmers, we thought we ought not to oppose this voluntary test.
Coupin then turned out from the chatty in which it was imprisoned a medium-sized cobra, and amused himself for a few 366 moments by teasing it. Irritated by this sport, the snake reared itself up, hissed, and struck at its aggressor several times, as though it would bite him; but, at each dart, Coupin stopped it with his hand and gaze, and the snake remained fascinated, with open mouth, hesitating to drive home its fangs. At one moment the snake even crawled up to the native’s outstretched hand, and appeared to lick it: it was evident that the animal’s only intention was to defend itself, not to attack.
Coupin, however, was bent on getting bitten, and, by dint of exciting the snake, the latter became so exasperated that, with hood dilated, it struck at the snake-charmer’s right hand and drove in its fangs. The man quietly raised his arm, to allow it to be clearly seen that the cobra had a firm hold; then, forcing the animal to let go, he came to us to prove that he was really bitten. There were two bleeding spots, 6 millimetres apart, in the centre of the fourth intermetacarpal space of the right hand. The time was exactly 4.40 p.m.
The snake, which was immediately killed, was still able to yield twelve drops of venom, when lateral pressure was applied to its glands.
Coupin walked towards a shrub of the kind mentioned above, which he had planted that very morning in the courtyard of the hospital,170 plucked some leaves from it and began to chew them, making rather a wry face, for he said that they were very bitter. He then seemed to meditate for a few moments before the shrub. We asked him whether he was afraid, but he stoutly denied it, declaring that this was not his first experience, and that indeed he had been bitten by cobras so often that he could not remember the first occasion. We endeavoured to discover whether he was not rendered immune, as Indian snake-charmers are reputed to be, by inoculating themselves subcutaneously with increasing doses of venom, according to a principle to a certain extent analagous 367 to that on which the preparation of antivenomous serum is based. He, however, assured us that he knew nothing about such practices, and that the properties of the plant in question had always sufficed to cure him without other treatment. Such was his confidence in his specific that, before the cobra was killed, he suggested to our hospital-warder that he should allow himself to be bitten in his turn, in order to render the experiment still more conclusive, adding that his own assistant would be the subject at the next demonstration.
Nevertheless, a slight œdema began to appear round the bite. The injured man seemed to be suffering a little, but the pain did not extend above the wrist; a slight trembling appeared in the other arm. From time to time Coupin gently massaged the affected limb with his left hand from above downwards, in order, he said, to lessen the pain, and perhaps also with a view to checking the diffusion of the venom. A slight perspiration broke out over the body; the pulse was regular, 92, twenty minutes after the bite.
Towards 6.15, or about an hour and a half after the experiment, the man, refusing an injection of serum, prepared to go home, but we insisted that he should remain at the hospital for at least two hours longer, so as to enable us to watch his condition. He consented to this, and asked for something to eat. At this time the back of the hand was somewhat more swollen, but Coupin seemed to be little disturbed by it; he even declared that the swelling would increase still further on the following day, and that, in two or three days, he would make an incision in order to let out the impure blood collected at the spot; also that the trouble was now localised, and would not extend higher. He merely complained of acute thirst, and of a certain difficulty in swallowing, which was due, he declared, to the extreme bitterness of the leaves that he had just masticated. Temperature 36·8° C.; pulse 36. After the brief reaction at the outset, there was a certain amount of hypothermia, but the general condition seemed satisfactory, 368 and the man remained perfectly calm. We therefore left him under the supervision of the resident student and the male attendants, giving orders that we should be sent for immediately in case of any serious symptoms arising.
At 7.15 we received a note from the student to the effect that Coupin had vomited, that his pulse was small, there was difficulty in breathing, and that he had just received an injection of antivenomous serum. We hastened to the hospital, where we were speedily joined by Drs. Cordier and Lhomme.
On our arrival we found Coupin in a rather prostrate condition. The pupils were fixed, he was foaming slightly at the mouth, respiration was regular, the pulse was strong and quite rhythmical at 96; the man was somewhat chilly. The patient was unable to speak, but had not lost consciousness, for, when we asked him how he was, he indicated by signs that he felt very ill. We subsequently learnt, from the student on duty, that towards 6.45 the condition of the patient became suddenly worse, after his companion had administered to him some medicine, the nature of which we have been unable to discover; on swallowing this drug Coupin was seized with vomiting, and became collapsed. We thought it more likely, however, that this was a mere coincidence, rather than the result of a poison adding its effects to those produced by the bite of the snake.
While waiting for our arrival a ligature had been applied to the injured arm, and near the bite there had been made two deep incisions, from which some dark blood escaped. At the same time the patient received hypodermically a first injection of 10 c.c. of Dr. Calmette’s antivenomous serum, and two injections of ether and caffeine. As the result of this treatment we found a marked increase in pulse-rate. After a subcutaneous injection of 500 grammes of artificial serum, there appeared to be a further distinct improvement until about 8.45 (four hours after the bite). From this moment, however, the patient gradually sank. A second dose of 369 antivenomous serum was injected beneath the skin,171 and, since the respiratory movements gradually became slower, artificial respiration by Sylvester’s method was resorted to, while the tongue was rhythmically drawn forward. From time to time these proceedings were interrupted for a few moments, in order to observe the condition of the respiration; at the base of the thorax and in the flanks abrupt, jerky, tetaniform undulations were observable, but there was no free respiratory movement. The abdominal aorta, the pulsations of which were visible behind the navel, raising the anterior wall of the abdomen, was somewhat misleading, simulating the rhythmical oscillations of the abdominal respiratory type. The heart, however, continued to beat regularly, without any appreciable weakness. The radial pulse persisted, although weak and slow: rate 48.
The patient was kept alive by repeated injections of ether and artificial serum (1¼ litres in three doses). About 8.30 there appeared to be a slight improvement; the patient was warmer, and showed some degree of sensation in the region of the bite. On pinching the vasculo-nervous bundle in the left axilla reflex movements were produced in the fingers, which closed convulsively. The pulse, which had momentarily disappeared in the radial artery, could again be felt, weak but regular. At the same time, when the epigastric region was examined, a few faint respiratory movements were just perceptible. A third injection of antivenomous serum was given and artificial respiration was continued.
This factitious amelioration, however, was not maintained, and after a few scarcely visible movements of the thorax the end soon came. The eyes were fixed and insensible, the pupils slightly dilated; no sweats, no urine passed. The body slowly grew cold; the pulse disappeared from the femoral and carotid arteries, and diminished in the aorta. The contractions of the heart were once or twice irregular, and its beats became weaker and gradually 370 lower. The reflexes had disappeared. There were still a few slight muscular contractions near the base of the chest, and the patient quietly passed away at 11.5 p.m., as the result of the progressive arrest of the heart’s action, respiration itself having virtually disappeared two hours before the heart had ceased to beat.
Case III.—A week after this sad occurrence, another snake-charmer, Kingilien by name, aged 25, was bitten in the first phalanx of the right forefinger, when taking hold of a cobra in the courtyard of the Pondicherry Hospital. Refusing an injection of antivenomous serum, the man ran off as fast as he could go, after having a simple ligature applied to his wrist. Scarcely had he reached his dwelling, when he fell into a deep coma, in which condition he was carried to Cottacoupom, to the abode of one Souraire Kramani, a kind of sorcerer, who administered to him a certain medicament in a betel leaf. After having vomited a large quantity of bile he was taken home. At this time, according to the summary investigation that we caused to be made, the patient was unable to utter a single word; he could only open his mouth with difficulty, and his eyelids remained closed. Kingilien, who had partially regained consciousness, seemed to be suffering from continuous attacks of vertigo; his head, if pushed to one side, drooped, and the man was incapable of voluntary movement. Respiration was fairly easy, swallowing painful. The entire hand was greatly swollen; poultices of leaves were applied to it, after a few incisions had been made with a knife in the back of the hand, in order to reduce the congestion. The arm was rubbed from above downwards with the very bitter leaves of the Vëmbou, or mango-tree, and prayers were recited. This is all the information that I have been able to obtain with reference to this man, who, after a prolonged convalescence, is said to have recovered (?).
Case IV.—One Latchoumanin, aged 25, also a snake-charmer, of Caradicoupom, was bitten at the Hospital at 10 a.m., on August 2, while handling a cobra. The bite was situated in the second joint of the right thumb. After it had bitten the man, ten drops of venom were extracted from the reptile’s glands. 371
A ligature was immediately applied, and the wound was made to bleed by hard squeezing. Refusing all other treatment, especially injection of serum, although we repeatedly urged it, the man made off home with all speed, but on arrival failed to find the specific on which he was relying. All that was then done was to recite a few prayers over him, and a Brahmin priest was called to bless him. About 11.30, after acute suffering, Latchoumanin sank into a comatose condition. At 12.30 respiration became stertorous, and the patient succumbed at 2 p.m., four hours after being bitten. Just as the medical officer, deputed by us, reached the dying man, two chatties containing live snakes were deposited at his bedside, to ward off ill-luck!
On the whole, the evidence collected in the last two cases is as confused as it is incomplete, and we can scarcely say how much we regret the obstinacy of these unfortunate victims in refusing to submit to our treatment, for the serum would undoubtedly have produced its maximum effect in them, since it would have been possible to make use of it in good time. These disastrous occurrences, however, will not cure natives of their exclusive reliance upon empirical practices; and as regards the inhabitants of the Tamil country, that is to say, Southern India, it may be foreseen that for a long time to come they will continue to remain refractory to the serotherapic treatment, submission to which the English have had less difficulty in securing from the natives of Bengal, whose intellectual development undoubtedly stands on a higher plane. 372
1 As regards the distinctive characters of these various snakes, and of the majority of those that we shall describe in this book, we have followed the statements in the “Catalogue of the Snakes in the British Museum (Natural History),” vol. iii., by George Albert Boulenger, F.R.S. (London: Printed by order of the Trustees, 1896.)
2 Acad. de médecine, March 19, 1889.
3 Erpétologie générale, t. vii.
4 “Le serpent cracheur de la côte occidentale d’Afrique,” Société Zool. de France, 1895, p. 210. Bavay thinks that the Spitting Snake is a Naja haje, but the description that he gives of the head of the reptile, which was sent to him by Le Naour, certainly agrees with the characters of Sepedon. Moreover, I have satisfied myself that the many specimens of Naja haje that I have kept in captivity in my laboratory never possessed the faculty of spitting their poison to a distance.
5 “The Snakes of Australia,” Sydney, 1869.
6 L. Lannoy, “Thèse de doctorat ès sciences,” Paris, 1903, No. 1,138, série A, 454.
7 “Snake Commission Report,” 1874.
8 Bulletin de l’Académie de Médecine, t. x., 1883, p. 947.
9 Zeitschrift für Biologie, xxiii.
10 Kühne and Chittenden, “Ueber Albumosen,” Zeitschrift für Biologie, 1884.
11 Comptes rendus de la Société de Biologie, 29 février, 1896.
12 Annales de l’Institut Pasteur, 1896, p. 489.
13 Province médicale, 21 Septembre, 1901.
14 Rockefeller Institute for Medical Research, New York, 1906.
15 Proc. Roy. Soc., London, lxxiv., 1904, pp. 108-109.
16 Ibid., pp. 104-108.
17 “Communication de l’Institut Sérothérapique de l’État danois,” tome i., Copenhagen, 1906.
18 Indian Annals, July, 1868.
19 Medical News, Philadelphia, 1884.
20 “Zur Pathologie des Schlangenbisses,” Forschungen der Medicin, Berlin, 1890.
21 Annales de l’Institut Pasteur, t. xii., 1898, p. 369.
22 Thèse Bordeaux, 1902.
23 Virchow’s Archiv für Pathologie, Anatomie, und Physiologie, Band 179, 1905.
24 Société de Médecine et de Chirurgie de São Paulo (Brazil), June 15, 1904.
25 Medical Record, September 15, 1900.
26 Lancet, January 2, August 20, October 22, 1904, and September 23, 1905.
27 Proceedings of the Royal Society, vol. lxxi., 1903.
28 “The Constitution of Snake-venom and Snake-sera,” University of Pennsylvania Medical Bulletin, vol. xv., 1902, p. 345.
29 Fontana, “On Poisons,” translated by J. Skinner: London, 1787.
30 Smithsonian Reports, 1854.
31 Smithsonian Contributions to Knowledge, 1860.
32 Medical Times and Gazette, vol. ii., 1873.
33 “On the Physiological Action of the Venom of the Australian Black Snake,” Proceedings of the Royal Society of New South Wales, July, 1895.
34 Indian Medical Gazette, December, 1901.
35 Comptes rendus de la Société de Biologie, November 4, 1899.
36 Ibid., October 28, 1899.
37 Annales de l’Institut Pasteur, June, 1904.
38 At the Serum-therapic Laboratory of São Paulo (Brazil), Dr. Vital, Brazil, is at the present time preparing serum specific against Lachesis-venom.
39 Medical Times and Gazette, vol. ii., 1873.
40 “The Thanatophidia of India.” Churchill, London, 1872.
41 “On the Physiological Action of the Venom of the Australian Black Snake,” Proceedings of the Royal Society of New South Wales, July 3, 1895.
42 Archives biologiques, 1897 and 1898; Comptes rendus de la Société de Biologie, October 28, 1899.
43 Comptes rendus de la Société de Biologie, October 28, 1899; November 4, 1899; July 26, 1902.
44 Annales de l’Institut Pasteur, June, 1904.
45 Journal of Pathology and Bacteriology, 1899-1900.
46 Journal of Experimental Medicine, March 17, 1902; University of Pennsylvania Medical Bulletin, November, 1902.
47 Comptes rendus de l’Académie des Sciences, June 16, 1902.
48 Comptes rendus de la Société de Biologie, No. 27, 1902.
49 Berliner klinische Wochenschrift, Nos. 38, 39, 1902; Nos. 2-4, 1903; Nos. 42-43, 1903.
50 Annales de l’Institut Pasteur, 1904, p. 387.
51 Journal of Experimental Medicine, 1905, No. 2, pp. 191-222.
52 Lancet, April 2, 1904, pp. 916-921.
53 “The Constitution of Snake-venoms and Snake-sera,” University of Pennsylvania Medical Bulletin, vol. xv., November, 1902, p. 345.
54 Comptes rendus de l’Académie des Sciences, August 11, 1902.
55 Annales de l’Institut Pasteur, June, 1904.
56 “Sur l’action protéolytique des venins,” Comptes rendus de l’Académie des Sciences, September, 1902, and Thèse Paris, No. 1,138, 1903.
57 “On the Plurality of Cytolysins in Snake-venom,” University of Pennsylvania Medical Bulletin, vol. xvi., 1903, p. 163.
58 Ann. Soc. Méd. de Gand, 1905, fasc. 3.
59 Annales de l’Institut Pasteur, April, 1905.
60 Annales de l’Institut Pasteur, 1898.
61 Thèse Paris, No. 1138, 1903.
62 Comptes rendus de l’Académie des Sciences, August 11, 1902.
63 Comptes rendus de la Société de Biologie, January 11, 1896.
64 Wehrmann, Annales de l’Institut Pasteur, 1897 and 1898.
65 Carrière, “Sur le sort des toxines et des antitoxines dans le tube digestif,” ibid., 1898, p. 435.
66 British Medical Journal, 1895 and 1897.
67 Chatenay, “Les réactions leucocytaires vis-à-vis de certaines toxines,” Thèse Paris, 1894.
68 Flexner and Noguchi, “Snake-venom in Relation to Hæmolysis, Bacteriolysis, and Toxicity,” Journal of Experimental Medicine, vol. vi., March 17, 1902.
69 “Trattado del veneno della vipera,” 1787.
70 Archiv für mikroskopische Anatomie, Bd. ix., 1873.
71 Morphologisches Jahrbuch, Bd. viii., 1883.
72 Comptes rendus de la Société de Biologie, January 20, 1894.
73 Archives de Physiologie, 1894.
74 Comptes rendus de l’Académie des Sciences, Paris, January 22, 1894.
75 Comptes rendus de la Société de Biologie, January 13, 1894.
76 Annales de l’Institut Pasteur, 1897.
77 Archives italiennes de Biologie, 1888 and 1889.
78 Archives de Physiologie, 1894.
79 Comptes rendus de la Société de Biologie, July 25, 1903.
80 Journal of Pathology and Bacteriology, May, 1903.
81 Journal of Pathology and Bacteriology, 1900, p. 273.
82 Comptes rendus de la Société de Biologie, July 1, 1905.
83 “The Jungle Book,” by Rudyard Kipling. London: Macmillan and Co., Ltd. Reprint of 1905, pp. 183, 184.
84 Deutsche medicinische Wochenschrift, 1898.
85 Comptes rendus de la Société de Biologie, 1895, p. 639, and 1899, p. 77; Bulletin du Muséum d’Histoire naturelle, 1895, t. i., p. 294, t. ii., p. 100.
86 “Aperçu général sur l’Egypte.” Paris, 1840.
87 Brehm’s “Thierleben”; French edition, “Les Reptiles,” p. 480.
88 Maclure’s Magazine, April, 1894.
89 British Medical Journal, August 17, 1895.
90 Archives de médecine navale, 1867, p. 390.
91 Revue scientifique, 1892, p. 254.
92 Comptes rendus de l’Académie des Sciences, February 24, 1896.
93 “The Pharsalia of Lucan, translated into blank verse by Edward Ridley, Q.C., sometime Fellow of All Souls’ College, Oxford.” (London: Longmans, Green and Co., 1896). Book ix., p. 296, lines 1,046 to 1,065.
94 Journal of Physiology, vol. viii., 1887, p. 203.
95 “Les Vipères de France,” p. 136 (1889).
96 Annales de l’Institut Pasteur, 1892, p. 181.
97 Comptes rendus de l’Académie des Sciences, t. 118, February, 1894, p. 356, March, 1894, p. 720; Comptes rendus de la Société de Biologie, February, 1894, pp. 111, 120; Archives de Physiologie, July, 1894; Annales de l’Institut Pasteur, May, 1894, p. 275, April, 1895, p. 225.
98 British Medical Journal, June 15, 1895.
99 Cf. papers by George Lamb, Scientific Memoirs by Officers of the Medical and Sanitary Departments of the Government of India, New Series, Nos. 1, 3, 4, 5, 7, 10, 16; L. Rogers, Proceedings of the Royal Society of London, vol. lxxii., and Lancet, February 6, 1904; C. J. Martin, Intercolonial Medical Journal of Australasia, August 20, 1897, April 20, 1898; Dr. Hunter, Lancet, January 2, 1904; Dr. Tidswell, Australasian Medical Gazette, April 21, 1902; A. Calmette, Comptes rendus de l’Académie des Sciences, May 2, 1904; Dr. Vital Brazil, “Contribution a l’étude de l’intoxication ophidienne” (separately published pamphlet), Paris, 1905; G. Bill, Intercolonial Medical Journal of Australasia, July 20, 1902.
100 Annales de l’Institut Pasteur, 1895, No. 4.
101 “The Nature of the Antagonism between Toxins and Antitoxins,” Proceedings of the Royal Society of London, vol. lxiii., 1898, p. 420.
102 Calmette, Comptes rendus de l’Académie des Sciences, 1902, No. 24; Preston Kyes, Berliner klinische Wochenschrift, 1904, No. 19.
103 Berliner klinische Wochenschrift, 1905, No. 50.
104 Comptes rendus de l’Académie des Sciences, Paris, September, 1881.
105 “Le venin de la vipère,” Paris, 1889.
106 Transactions of the Third Intercolonial Congress, 1892, p. 152.
107 “Les morsures de vipères chez les animaux,” Recueil de médecine vétérinaire d’Alfort, May 15, 1897.
108 Comptes rendus de la Société de Biologie, December 13, 1902; June 6, July 25, 1903; February 20, 1904.
109 Semaine médicale, June 24, 1903.
110 Comptes rendus de la Société de Biologie, May 19, 1906.
111 “El Latrodectus formidabilis de Chile,” by Fredérico Puga-Borne, Santiago, 1892; and “Biologia Centrali-Americana,” Arachnida, vol. ii., pl. 35.
112 Blackwell, “Experiments and Observations on the Poison of Araneida,” Transactions of the Linnean Society of London, 1855, p. 31. See also “Insect Life” (U.S. Department of Agriculture, 1889), vol. i., parts 7, 8, 9 and 10, and vol. ii., part 5.
113 “Beiträge zur Kenntniss der Giftspinnen.” Stuttgart, 1901.
114 “Zur Kenntniss des Kreuzspinnengiftes,” Hofm. Beiträge, ii., p. 125, 1902.
115 Joyeux-Laffuie, “Appareil venimeux et venin de scorpion” (Thesis for the degree of Doctor of Science), Paris, 1883; and Comptes rendus de l’Académie des Sciences, November 6, 1882.
116 “Ueber die Giftwirkung des Nordafrikanischen Skorpiones,” Zeitschrift für Biologie, Bd. xii., p. 170, 1876.
117 Comptes rendus de la Société de Biologie, 1885, p. 574.
118 Berliner klinische Wochenschrift, 1903, Nos. 42, 43.
119 Annales de l’Institut Pasteur, 1895, p. 232.
120 Proceedings of the Royal Society, vol. xlii., 1887, p. 17.
121 “L’immunité dans les maladies infectieuses,” Paris, 1901.
122 Comptes rendus de la Société de Biologie, November 15, 1904.
123 “La scolopendre et sa piqure,” Thèse Paris, 1887.
124 “Appareil venimeux et venin de la scolopendre,” Thèse Montpellier, 1885.
125 “Lehrbuch der Anatomie der Wirbellosen Tiere,” 1848.
126 Arch für anat. Wissensch., 1859.
127 Comptes rendus de l’Acad. des Sciences, 1884.
128 “Appareil glandulaire des hyménoptères,” Paris, 1894.
129 Comptes rendus de l’Académie des Sciences, 1898.
130 Annales sc. Anat. Zoologie, 8e série, t. x., 1898.
131 Comptes rendus de l’Académie des Sciences, July 25, 1890.
132 Comptes rendus de l’Académie des Sciences, July 24, 1905.
133 Berliner klinische Wochenschrift, 1906, No. 44.
134 Comptes rendus de l’Académie de Médecine, t. liii., 1905, p. 498.
135 “Traité de zoologie médicale,” t. ii. (Paris).
136 Comptes rendus de la Société de Biologie, January 17, 1908.
137 Comptes rendus de la Société de Biologie, February 25, 1905.
138 “Les poissons venimeux,” Thèse Paris, 1889.
139 “Poissons venimeux et poissons vénéneux,” Archives de Physiologie, May, 1872; Archives de médecine navale, February, 1865, and January, 1881.
140 “Poissons venimeux et poissons vénéneux,” Thèse Paris, 1899.
141 “Les poissons vénéneux,” Paris, 1900.
142 “Sédentarité des poissons venimeux,” Comptes rendus de la Société de Biologie, 1895, p. 86.
143 Comptes rendus de la Société de Biologie, 1904, p. 666.
144 Thèse Paris, 1884.
145 Bulletin du Muséum d’histoire naturelle, 1899.
146 “Giftfische und Fischgifte,” Vorträge im Rostocker Fischerverein, 1902, and Die medizinische Wochenschrift, 1902.
147 Comptes rendus de la Société de Biologie, October 25 and November 8, 1902, and June 21, 1904; Journal de Physiologie, March, 1903.
148 Brehm (Sauvage’s translation), “Les Merveilles de la Nature—Reptiles et Batraciens” (Paris, 1885).
149 Hoppe-Seyler’s “Med.-chem. Untersuchungen,” Berlin, 1866.
150 Comptes rendus de l’Académie des Sciences, April 1, 1889, and January 29, 1890.
151 Ibid., 1890.
152 “Beitrage zur Kenntniss des Salamandarins und Salamanderalkaloïde,” Archiv. f. experimentale Pathologie und Pharmakologie, Bd. xli., 1898, p. 219, and Bd. xliii., 1900, p. 84.
153 Comptes rendus de la Société de Biologie, 1897, pp. 723, 823.
154 Comptes rendus de l’Académie des Sciences, April 21, 1851, and May, 1852.
155 Journal de Thérapeutique, 1877, p. 929.
156 Comptes rendus de l’Académie des Sciences, February 25, 1884.
157 Archives de Physiologie, 1893, p. 511.
158 Archiv für mikroskopische Anatomie, Bd. ii., 1889, p. 57.
159 “Zur Kenntniss des Krotengiftes,” Hofm. Beiträge, Bd. i., 1901, p. 575.
160 “Ueber Bufonin und Bufotalin,” Leipzig, 1902; and Archiv für experimentale Pathologie und Pharmakologie, December, 1902.
161 “Rapports des venins avec la biologie générale,” Revue générale des Sciences, December 30, 1903.
162 Comptes rendus de l’Académie des Sciences, December 14, 1903.
163 H. Coupin, La Nature, September 19, 1903.
164 “On the Ornithorhynchus paradoxus, its Venomous Spur and General Structure,” Trans. Linn. Soc., 1822, p. 622.
165 “Observations on the Femoral Gland of Ornithorhynchus and its Secretion;” Proc. Linn. Soc. New South Wales, vol. ix., July, 1894.
166 “Note sur la sécrétion venimeuse de l’Ornithorhynchus paradoxus,” Comptés rendus de la Société de Biologie, March 12, 1904.
167 “On the Effects of Wounds Inflicted by the Spurs of the Platypus,” Proceedings of the Royal Society of Tasmania, 1876.
168 The photographs, by which this note was accompanied, are reproduced in Chapter IV. of this volume, pp. 155, 157, 158.
169 Polygala telophioides, Will.
170 We subsequently learnt that he had informed his friends of his intention, but had not said a word about it to his wife.
171 The thick and cloudy appearance of the contents of the bottles made us hesitate to have recourse to intravenous injection.
Inconsistent spelling and hyphenation are as in the original.